AZ31 þ�Ͻ����л�������ASP����MgAl-LDH��ZnAl-LDHĤ��ʴ�ԵıȽ�
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2020���9��
�������ߣ��¼��� ���� �ⷼ ���ܹ� ���� ���緼 ���� ����
����ҳ�룺2424 - 2434
�ؼ��ʣ�ˮ��ʯ��þ�Ͻ��춬�������ʴ����ʴ���Ƽ�
Key words��hydrotalcite; magnesium alloy; aspartic acid; corrosion resistance; corrosion inhibitor
ժ Ҫ��ʹ��һ����ˮ�ȷ��ֱ��Ʊ��춬����(ASP)����ZnAl��״˫��������(ZnAl-LDH)��MgAl-LDHͿ�㡣�����Ʊ�������Ϳ�����ò���ɷ֡��ṹ����ʴ�Խ��бȽ��о���������������������ܵIJ�״����Ƭ(NS)�ڻ����ϴ�ֱ����������MgAl-ASP-LDHͿ����ֳ��߿�϶�����ʵ���ά(3D)õ����Ƭ״�ṹ�����ݲ����У�MgAl-ASP-LDHͿ��չʾ����ZnAl-ASP-LDHͿ���������;��Ժ���ʴ�ԡ�ZnAl-ASP-LDH��MgAl- ASP-LDHͿ��ĸ�ʴ�����ܶȱ�Mg�Ͻ���ĵ�2~3��������������������л�ASP�����ӵ�ZnAl/MgAl-LDHĤ�����������þ�Ͻ����ʴ�ԡ�
Abstract: The aspartic acid (ASP), an environment-friendly corrosion inhibitor, was explored to intercalate into the ZnAl-layered double hydroxide (ZnAl-LDH) and MgAl-LDH coatings on AZ31 magnesium alloys by a facile one-step hydrothermal method. The morphology, composition, structure and corrosion resistance of the prepared coatings were comparatively investigated. It is found that the uniform and dense layered nanosheet (NS) vertically grows on the substrate, and the MgAl-ASP-LDH films exhibit a three-dimensional (3D) rose-like sheet structure with high ratio of pore coverage. The MgAl-ASP-LDH coating exhibits better corrosion resistance and durability of longtime immersion than ZnAl-ASP-LDH coating. The corrosion current density of two coatings is two or three orders of magnitude lower than that of bare Mg alloy, indicating that ZnAl/MgAl-LDH films intercalated with organic ASP anions can significantly improve the corrosion resistance of Mg alloy.
Trans. Nonferrous Met. Soc. China 30(2020) 2424-2434
Jia-ling CHEN1,2, Liang FANG1,2, Fang WU1, Xian-guang ZENG2,Jia HU3, Shu-fang ZHANG4, Bin JIANG3, Hai-jun LUO5
1. Chongqing Key Laboratory of Soft Condensed Matter Physics and Smart Materials, State Key Laboratory of Power Transmission Equipment & System Safety and New Technology, College of Physics, Chongqing University, Chongqing 400044, China;
2. Material Corrosion and Protection Key Laboratory of Sichuan Province, Institute of Materials Science and Engineering, Sichuan University of Science and Engineering, Zigong 643000, China;
3. National Engineering Research Center for Magnesium Alloys, College of Materials Science and Engineering, Chongqing University, Chongqing 400044, China;
4. College of Software, Chongqing College of Electronic Engineering, Chongqing 401331, China;
5. Chongqing Key Laboratory on Optoelectronic Functional Materials, College of Physics and Electronic Engineering, Chongqing Normal University, Chongqing 401331, China
Received 23 December 2019; accepted 16 July 2020
Abstract: The aspartic acid (ASP), an environment-friendly corrosion inhibitor, was explored to intercalate into the ZnAl-layered double hydroxide (ZnAl-LDH) and MgAl-LDH coatings on AZ31 magnesium alloys by a facile one-step hydrothermal method. The morphology, composition, structure and corrosion resistance of the prepared coatings were comparatively investigated. It is found that the uniform and dense layered nanosheet (NS) vertically grows on the substrate, and the MgAl-ASP-LDH films exhibit a three-dimensional (3D) rose-like sheet structure with high ratio of pore coverage. The MgAl-ASP-LDH coating exhibits better corrosion resistance and durability of longtime immersion than ZnAl-ASP-LDH coating. The corrosion current density of two coatings is two or three orders of magnitude lower than that of bare Mg alloy, indicating that ZnAl/MgAl-LDH films intercalated with organic ASP anions can significantly improve the corrosion resistance of Mg alloy.
Key words: hydrotalcite; magnesium alloy; aspartic acid; corrosion resistance; corrosion inhibitor
1 Introduction
As the lightest structural material, magnesium (Mg) alloys have been widely applied in auto- mobile, electronics, computer, aerospace and other industries [1,2]. But their poor corrosion resistance due to the low standard potential (-2.36 V) limits their extensive applications [3-6]. So, plenty of surface treatment measures have been developed to improve their corrosion resistance [7-10]. Among these protective coatings, layered double hydroxides (LDHs) have drawn increasing attention for their excellent anti-corrosion performance owing to the unique lamellar structure and excellent exchangeability of interlayer anions.
LDH is a class of anionic-intercalated clay material with a hierarchical structure [11-14], which can be expressed by a general formula: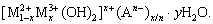 The hydrotalcite layer is composed of M2+ and M3+ cations locating in the octahedral pores, x is the molar ratio of M3+/(M2++M3+), An- is interlayer anions, and m is the number of water molecules in the interlayer [15-18]. The interlayer mainly consists of anion and water molecules with weak electrostatic force and hydrogen bonding, which allows the interlayer anions to easily exchange with other anions. The unique interlayer anion exchange ability allows LDHs to capture aggressive anions (such as Cl-) and delay corrosion reactions. Besides, the corrosion inhibitors encapsulated into the LDHs laminates may further protect the substrate from local corrosion [19,20]. Therefore, it is proposed to insert a corrosion inhibitor into the laminated LDH to increase corrosion resistance or obtain self-healing protection of the metal substrate. However, the appropriate LDHs and the corrosion inhibitor for the protection of Mg alloy still remains to be found out.
The hydrotalcite layer is composed of M2+ and M3+ cations locating in the octahedral pores, x is the molar ratio of M3+/(M2++M3+), An- is interlayer anions, and m is the number of water molecules in the interlayer [15-18]. The interlayer mainly consists of anion and water molecules with weak electrostatic force and hydrogen bonding, which allows the interlayer anions to easily exchange with other anions. The unique interlayer anion exchange ability allows LDHs to capture aggressive anions (such as Cl-) and delay corrosion reactions. Besides, the corrosion inhibitors encapsulated into the LDHs laminates may further protect the substrate from local corrosion [19,20]. Therefore, it is proposed to insert a corrosion inhibitor into the laminated LDH to increase corrosion resistance or obtain self-healing protection of the metal substrate. However, the appropriate LDHs and the corrosion inhibitor for the protection of Mg alloy still remains to be found out.
Two kinds of typical LDH, MgAl-LDH and ZnAl-LDH, have been explored. For instance, WANG et al [21] prepared the Mg-Al LDH films by a hydrothermal method and found their LDHs have a graded nanosheet (NS) structure, which uniformly cover the surface of substrate and effectively improve the corrosion resistance of Mg alloys. But the graded structure of the LDH films was destroyed and the protection ability was reduced after immersion in a 3.5 wt.% NaCl solution for 6 h. Based on the structural memory effect of LDH during its rehydration, MONTEMOR et al [16] fabricated MgAl-MBT- LDH by intercalating the calcined MgAl-CO3- LDH in which 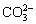 was volatilized with mercaptobenzothiazole (MBT) and got an enhanced corrosion resistance to chloride-containing solution. CHEN et al [17] deposited MgAl-PA-LDH by embedding phytic acid (PA), an organic compound with metal chelating ability, into the synthesized MgAl-CO3-LDH laminate and achieved the synergistic protection of LDH and corrosion inhibitor (PA).
was volatilized with mercaptobenzothiazole (MBT) and got an enhanced corrosion resistance to chloride-containing solution. CHEN et al [17] deposited MgAl-PA-LDH by embedding phytic acid (PA), an organic compound with metal chelating ability, into the synthesized MgAl-CO3-LDH laminate and achieved the synergistic protection of LDH and corrosion inhibitor (PA).
A few work based on ZnAl-LDH has also been carried out. For example, GUO et al [22] synthesized a dense Zn-Al LDH film to act as a ��fence�� between the Al alloy substrate and the corrosion solution to effectively prevent the transfer of charge and inhibit the corrosion process. ZHANG et al [23] found that ZnAl-LDH-lauric acid (LA) films can improve the corrosion resistance of Al alloys. BUCHHEIT et al [15] reported that the 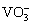 can be released out from the ZnAl-VO3-LDH coating to avoid further corrosion in a corrosive environment.
can be released out from the ZnAl-VO3-LDH coating to avoid further corrosion in a corrosive environment.
However, the corrosion inhibitors adopted on MgAl-LDH and ZnAl-LDH are mainly inorganic anions. Owing to special functional groups of nitrogen, oxygen, sulfur and phosphorus, and multiple bonds or aromatic rings in the structure, the organic compounds are usually excellent inhibitors and organic protective coating is one of the commercial ways to protect metal surface against corrosion. Aspartic acid (ASP), a natural and environment-friendly organic compound, was found to have the ability to protect Al alloys from corrosion [24]. As far as we know, their application in the protection of Mg alloy has been less reported.
Meanwhile, most of the LDH-based anti- corrosion coatings were synthesized by a complicated two-step approach (the corrosion inhibitors are usually inserted into the LDH laminates by ion-exchange after the formation of LDH), so a simple and more cost-effective way, especially one-step method is urgent to develop. In our previous work, ZnAl-LDHs intercalated with different inorganic anions (NO3-, Cl-, 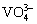 ,
, 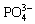 , and
, and  ) [18] and rose-like MgAl-LDH coating encapsulated with organic ASP [25] were produced on AZ31 Mg alloys with a facile hydrothermal method, and their corrosion resistance or self-healing was studied.
) [18] and rose-like MgAl-LDH coating encapsulated with organic ASP [25] were produced on AZ31 Mg alloys with a facile hydrothermal method, and their corrosion resistance or self-healing was studied.
Therefore, in this work, the ASP was chosen as the corrosion inhibitor and inserted into the MgAl-LDH and ZnAl-LDH laminate to form MgAl-ASP-LDH and ZnAl-ASP-LDH films on AZ31 Mg alloy by a simple hydrothermal method, respectively. The morphology, structure and the corrosion resistance of these two types of LDHs were comparatively investigated by XRD, FTIR, SEM and electrochemical test systems.
2 Experimental
2.1 Pretreatment of Mg alloy substrate
AZ31 Mg alloy sheets (2.5-3.5 wt.% Al, 0.6-1.4 wt.% Zn, 0.2-1.0 wt.% Mn, <0.08 wt.% Si, <0.04 wt.% Ca, <0.004 wt.% Fe, <0.01 wt.% Cu, <0.001 wt.% Ni and balanced Mg) with a size of 20 mm �� 25 mm �� 2.0 mm were employed as the substrates. All the chemical reagents used in this experiment, including Mg(NO3)2��6H2O, Zn(NO3)2�� 6H2O, Al(NO3)3��9H2O, C4H7NO4(ASP), NaCl and NaOH were of analytical grade.
AZ31 samples were polished with 400, 800, 1500 and 2000 grit SiC abrasive papers, followed by ultrasonic cleaning in ethanol and dried at 60 ��C. In order to remove impurities and oxides on the substrate surface, the Mg sheets were immersed in 0.5 wt.% NaOH solution for ultrasonic cleaning for 1 min. Then, they were sonicated in alcohol for 10 min and then dried for use.
2.2 Preparation of MgAl-ASP-LDH, ZnAl-ASP- LDH and ASP coating
The preparation process is shown in Fig. 1. The detail synthesis process of MgAl-ASP-LDH on AZ31 alloy is as follows. 0.769 g Mg(NO3)2��
6H2O, 0.375g Al(NO3)3��9H2O and 0.044 g ASP were homogeneously dissolved in 50 mL deionized (DI) water and magnetically stirred. Then the pH of the solution was adjusted to 10 by the dropwise addition of NaOH (2 mol/L), which was carried out in a glove box with nitrogen gas to prevent the carbonate ions due to CO2 in the air from intercalating the LDH. The mixed solution and the treated Mg alloy were transferred to a hydrothermal kettle (denoted as MgAl-ASP-LDH coating), hydrothermally reacted at 393 K for 12 h and taken out after cooling in the air. Finally, they were washed with DI water by ultrasonication for 15 min and then dried.
The fabrication process of ZnAl-ASP-LDH coating is similar with that of MgAl-ASP-LDH coating. In typical synthesis, 0.04 mol/L Zn(NO3)2��
6H2O and 0.02 mol/L Al(NO3)3��9H2O were used as reactants. Mg alloy was immersed in ASP solution under same environment, and then the ASP coating was successfully prepared.
2.3 Characterization and electrochemical measurement
The morphology was observed by field emission scanning electron microscopy (FE-SEM, Mira3 LMH). The crystallographic structure was detected by X-ray diffractometry (XRD, Bruker D8 Advance Cu K��). The composition was confirmed by Fourier transform infrared spectroscopy (FTIR Nicolet iS50).
The polarization curves and electrochemical impedance spectroscopy (EIS) were used to judge the corrosion resistance of the coating, which were measured with an electrochemical workstation (CHI760E, Shanghai Chenhua, China) at room temperature (RT) using 3.5 wt.% NaCl solution. The AZ31 Mg alloy substrate and synthesized sample were employed as working electrode (1 cm �� 1 cm), while the saturated calomel electrode (SCE) and platinum electrode (3 cm �� 3 cm) as reference electrode and counter electrode, respectively. Prior to the measurement, the working electrode was immersed in 3.5 wt.% NaCl solution until the open circuit voltage was stable. The polarization curves were recorded from -2.0 to 0 V (vs SCE) at a sweep rate of 1 mV/s. The EIS measurements were carried out in a vibration frequency from 100 kHz to 100 MHz with an amplitude disturbance of 1 mV and 2 s of rest time. All the spectra were recorded at the open circuit potential.
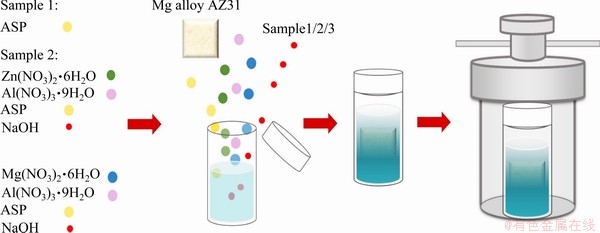
Fig. 1 Schematic preparation process of ZnAl-ASP-LDH and MgAl-ASP-LDH coatings
3 Results and discussion
3.1 Morphology and structure
The morphologies of the surface and cross- section of ASP, ZnAl-ASP-LDH and MgAl-ASP- LDH coatings are displayed in Fig. 2. From Figs. 2(a, b), it is obvious that the surface of the ASP coating is covered with a layer of dense particles, which compactly coats the substrate. For the ZnAl-ASP-LDHs films (Figs. 2(d, e)), it is shown that the LDH NS films vertically grow on Mg alloy and exhibit a uniform and dense NS structure. There are some pores on the film surface, and they cause the corrosive substance penetrating into the barrier layer, which is not conducive to the protection of Mg alloys.
Compared with the ZnAl-ASP-LDH coating, the MgAl-ASP-LDH coating grows very uniformly and retains the lamellar structure of LDH with only a few holes (Figs. 2(g, h)), coming into being a three-dimensional porous rose-like sheet structure, which can effectively prevent the penetration of corrosion solution. And the LDH NSs can increase the specific surface area contacting with the corrosive solution, thereby improving the capture efficiency of corrosive ion Cl-.
The cross-sectional morphologies of ASP, ZnAl-ASP-LDH and MgAl-ASP-LDH coating on the AZ31 Mg substrate are shown in Figs. 2(c, f, i). It can be clearly seen that all the films have strong adhesion to the substrate and grow uniformly and densely on the surface of the Mg alloy. The thickness values of ASP, ZnAl-ASP-LDH and MgAl-ASP-LDH films are about 3.95, 9.49 and 17.01 ��m, respectively, and the MgAl-ASP-LDH film is the thickest.
The XRD patterns and FT-IR spectra of ASP, ZnAl-ASP-LDH and MgAl-ASP-LDH coating are shown in Fig. 3. From Fig. 3(a), it is observed that ZnAl-ASP-LDH and MgAl-ASP-LDH exhibit (003) and (006) characteristic diffraction peaks of LDH (standard alignment card 22-700 [26]). The (003) peaks of MgAl-NO3-LDH, MgAl-ASP-LDH, ZnAl-NO3-LDH and ZnAl-ASP-LDH locate at 2��=11.25��, 10.9��, 11.22�� and 10.69��, respectively, indicating that the peaks of the LDH intercalated with ASP shift toward low angle. This shift is mainly ascribed to the difference in the size of intercalated nitrate anions and ASP anions.

Fig. 2 SEM images of surface (a, b, d, e, g, h) and cross-section (c, f, i) of coating on Mg alloys
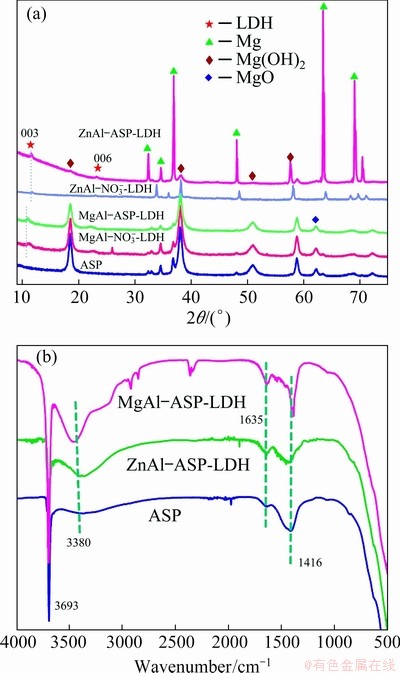
Fig. 3 XRD patterns (a) and FT-IR spectra (b) of ASP, ZnAl-ASP-LDH and MgAl-ASP-LDH
The spacing of (003) planes, d(003), calculated by Bragg equation (d=��/(2sin��)) for MgAl-NO3-- LDH, MgAl-ASP-LDH, ZnAl-NO3--LDH and ZnAl-ASP-LDH are 0.911, 0.941, 0.785 and 0.824 nm, respectively, meaning that spacing of (003) planes of ASP-inserted LDH samples is larger than that of NO-3-inserted LDH samples.
From FT-IR spectra (Fig. 3(b)), the absorption band at 3380 cm-1 is associated with N��H vibration, and 1635 and 1416 cm-1 are assigned to ��COOH vibration of ASP molecules [27]. The occurrence of these three peaks indicates that ASP actually exist in the corresponding ZnAl-LDH and MgAl-LDH samples. The 3693 cm-1 absorption is attributed to inter-layer water molecules and water molecules absorbed on the surface. The XRD and FT-IR patterns reveal that ASP ions are successfully intercalated into ZnAl-LDH and MgAl-LDH laminates, respectively.
In order to further verify the presence of intercalated aspartate ions, sensitive XPS is measured for MgAl-NO3--LDH, MgAl-ASP-LDH, ZnAl-NO3--LDH and ZnAl-ASP-LDH. The XPS results are shown in Fig. 4. Figure 4(a) shows the survey spectra of MgAl-LDH before and after intercalation of ASP. The high-resolution O 1s spectra of MgAl-NO3--LDH and MgAl-ASP-LDH are shown in Figs. 4(b, c), respectively. The peaks centered at 530.8, 531.6 and 531.3 eV correspond to oxygen peaks in hydroxyl bonding with Mg or Al and N��O bonding in NO3-, respectively [28]. From Fig. 4(c), in addition to these three peaks, another peak at 532.2 eV obviously appears which is related to the O��C=O bond [29], indicating that the organic aspartic acid ion is successfully inserted into the MgAl-LDH laminate. The survey of ZnAl-NO3--LDH and ZnAl-ASP-LDH, and the O 1s spectra of ZnAl-NO3--LDH and ZnAl- ASP-LDH are shown in Figs. 4(d, e, f), respectively. Similarly, by comparing the O 1s spectra of Figs. 4(e, f), it can be found that ASP ion is also successfully intercalated into the ZnAl-LDH laminate.
3.2 Corrosion resistance
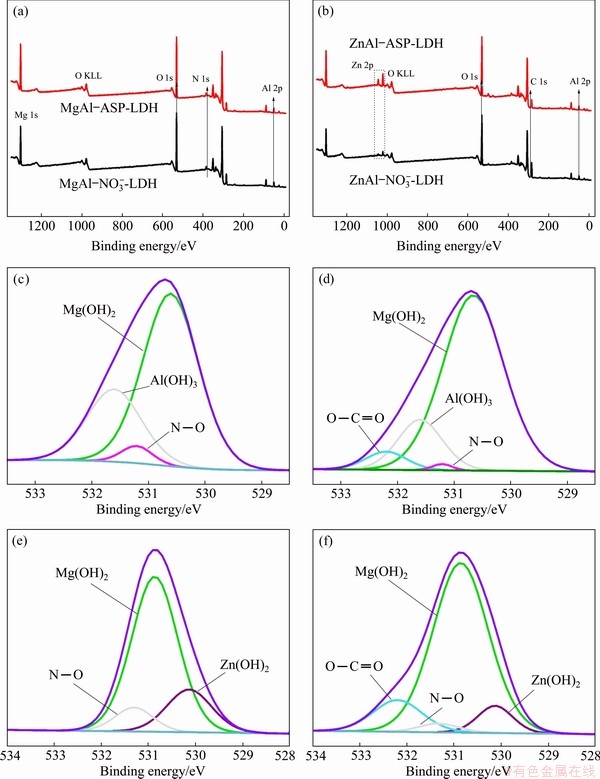
Fig. 4 Survey of MgAl-NO3--LDH, MgAl-ASP-LDH (a) and ZnAl-NO3-LDH, ZnAl-ASP-LDH (b), and O 1s spectra of MgAl- NO3--LDH (c), MgAl-ASP-LDH (d), ZnAl-NO3--LDH (e), ZnAl-ASP-LDH (f)
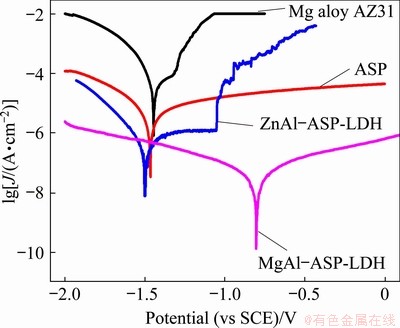
Fig. 5 Polarization curves of Mg alloy AZ31, ASP, ZnAl-ASP-LDH and MgAl-ASP-LDH coatings
The polarization curves of ASP, MgAl-ASP- LDH, ZnAl-ASP-LDH coatings are shown in Fig. 5. For comparison, the polarization curve of the Mg substrate is also included. The corrosion parameters of the polarization curves are listed in Table 1. It can be seen from Table 1 that the MgAl-ASP-LDH coating has the largest corrosion potential (��corr), and the corrosion current density (Jcorr) of AZ31 alloy, ASP, ZnAl-ASP-LDH and MgAl-ASP-LDHs coatings decreases from 6.984��10-5 to 3.351��10-6, 3.933��10-7 and 2.769��10-8 A/cm2. Typically, the corrosion current density of ASP, ZnAl-ASP-LDH and MgAl-ASP- LDH coatings is less than that of the Mg alloy substrate by about one, two and three orders of magnitude, respectively. It is known that the larger the corrosion potential is and the lower the corrosion current density is, the better the corrosion resistance is. So, the MgAl-ASP-LDHs coating exhibits the best corrosion resistance among four samples studied. Meanwhile, the anti-corrosion properties of the fabricated samples are compared with those of ZnAl-NO3--LDH and MgAl-NO3--LDH films reported in our previous work [18,25], and it is found that the corrosion resistance of the ZnAl-LDH and MgAl-LDH films intercalated with organic ASP anion is much better than that of the corresponding LDH intercalated with inorganic NO3- anion.
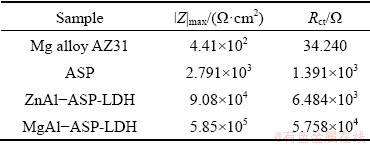
EIS measurements are employed to further characterize the corrosion inhibition effect of the coatings on the Mg alloy AZ31. The |Z|max of bare Mg alloy is only 446 ����cm2, whereas the |Z|max of MgAl-ASP-LDH coatings is 5.85��105 ����cm2, which is increased by three orders of magnitude in comparison with the Mg alloy matrix (Fig. 6(a)). MgAl-ASP-LDH coating shows the highest |Z|max value, followed by the ZnAl-ASP-LDH (9.08��104 ����cm2) and ASP (2.79��103 ����cm2) coatings. The equivalent circuit (EC) models for Mg alloy and coatings are given in Fig. 6(d) based on the Bode phase plots (Fig. 6(b)). As known, the material with a higher Z modulus at lower frequencies exhibits better corrosion resistance on
the metal substrate [30,31], Thus, the samples coated with MgAl-ASP-LDH possesses better corrosion resistance. From Fig. 6(c), it is found that the arc radius of the MgAl-ASP-LDH, ZnAl-ASP- LDH and ASP coatings decreases in turn, meaning that the MgAl-ASP-LDH with the largest arc radius has the best corrosion resistance. From Fig. 6(d), charge transfer resistor (Rct) is connected in parallel with the electric double layer capacitor (Cedl) and in series with the coating resistor (Rcoat), where CPEcoat and Rs are the coating capacitance and solution resistance, respectively. Here, because the Nyquist diagram deviates from the ideal behavior, the constant phase element (Q) is superior to the pure capacitor. The value of the pseudo capacitor (CPEcoat) can be calculated by the following expression [32]:
C=(Q0��R)1/n/R (1)
where C (F/cm2) is a pseudo capacitance, Q0 (S��sn/cm2) is a constant phase element, n is a frequency factor and R (��) is a resistance. The transfer resistance values are calculated by the fitting circuit and given in Table 2. It is found that the transfer resistances of the ASP, ZnAl-ASP- LDH and MgAl-ASP-LDH coating are 1.391��103, 6.484��103 and 5.758��104 ��, respectively, which are much larger than the transfer resistance of AZ31 alloy (34.240 ��), indicating that all three coatings can improve the corrosion resistance of Mg alloy and MgAl-ASP-LDH coating is the best one. These results illustrate that the ZnAl-LDH or MgAl-LDH films intercalated with ASP prepared by one-step in situ method can effectively improve the corrosion resistance of the AZ31 Mg alloy.
Table 1 Corrosion potential and corrosion current density of Mg alloy AZ31, ASP, MgAl-ASP-LDH, ZnAl-ASP-LDH coatings in 3.5 wt.% NaCl solution
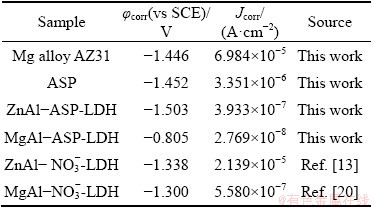
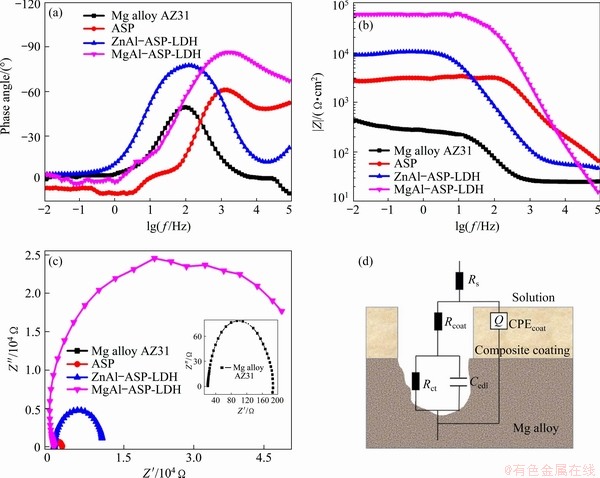
Fig. 6 Bode magnitude plots (a), Bode phase plots (b), Nyquist diagram (c) and equivalent electrical circuit (d) of Mg alloy AZ31, ASP, ZnAl-ASP-LDH and MgAl-ASP-LDH
Table 2 Maximum impedance value and transfer resistance of Mg alloy AZ31, ASP, MgAl-ASP-LDH, ZnAl-ASP-LDH coatings in 3.5 wt.% NaCl solution
3.3 Morphology and corrosion resistance after immersion

Fig. 7 SEM images of ZnAl-ASP-LDH (168 h) (a) and MgAl-ASP-LDH (480 h) (b) after immersion in 3.5 wt.% NaCl solution
In order to investigate the electrochemical properties of the coating with long immersion time, the ZnAl-ASP-LDH and MgAl-ASP-LDH coatings were immersed in 3.5 wt.% NaCl solution for 7 days (168 h) and 20 days (480 h), respectively. The morphologies of the samples after the immersion experiments are shown in Fig. 7. It can be seen from Fig. 7(a) that the one week��s immersion into the corrosive NaCl solution causes the nanosheets of the ZnAl-ASP-LDHs film to be completely dissolved, and the insoluble precipitates evenly and smoothly cover the surface of the Mg alloy, which can effectively slow down the corrosion of Mg alloy to some extent. Astonishingly, the MgAl-ASP-LDH coating still maintains the typical LDH nanosheet structure after soaking in NaCl solution for near three weeks (Fig. 7(b)). Compared with the morphology before corrosion, even though some LDH nanosheets have been dissolved and peeled off, the surface of the Mg alloy is still covered by the dense three- dimensional flaky LDH, which effectively prevents the invasion of the corrosion solution.
Usually, there are three roles for the LDH films with ASP anions in enhancing the anti- corrosion performance: (1) the high-density nanosheets of LDHs films behave as a barrier coating to protect the aggressive Cl- anions from directly contacting the Mg substrate, which will avoid the invasion of the corrosion solution and delay the initiation of the corrosion; (2) owing to the anion-exchange of LDHs, the intercalated ASP anions exchange with the Cl- anions, meaning that the concentration of harmful Cl- will decrease after its entrapment; (3) the released ASP anions in the aggressive environment can form a corrosion inhibitor protective layer (usually called as self-healing coating) on the surface of Mg alloy. Shortly speaking, the corrosion resistance of ASP-intercalated LDHs films can be improved by the barrier coating, the entrapment of harmful chlorides and the inhibition protective layer.
Table 3 Comparison of corrosion resistance of ZnAl-LDH or MgAl-LDH films intercalated with different organic anions (corrosion solution: 3.5 wt.% NaCl)
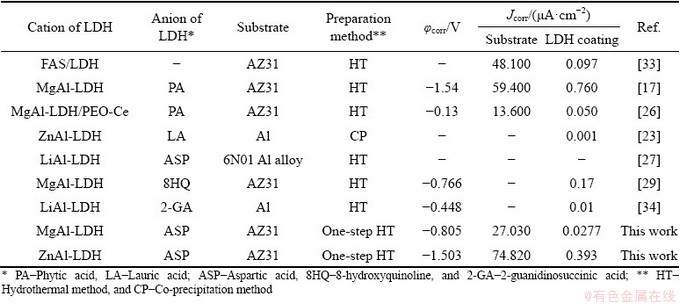
As far as ZnAl-ASP-LDH and MgAl-ASP- LDH are concerned, the thicker coating and the rose-like sheet structure make it more advantageous for the MgAl-ASP-LDH to form barrier layer, and the larger d(003) spacing (0.941 nm vs 0.824 nm of ZnAl-ASP-LDH) causes it more conducive to absorb Cl- and release interlayer anions. Meanwhile, the Mg2+ ions corroded from Mg alloys are more likely to form an inhibition protective MgAl-LDH layer than the ZnAl-LDH.
Therefore, due to the comprehensive effect of the above three factors, the MgAl-ASP-LDH films demonstrate better anti-corrosion property than the ZnAl-ASP-LDH films.
3.4 Comparison of corrosion resistance of MgAl- LDH or ZnAl-LDH films with different organic anions
The corrosion resistances of ZnAl-LDH or MgAl-LDH films intercalated with different organic anions are briefly compared and the results are given in Table 3. It is found that the corrosion potential of LDH coating is in the range from -1.50 to 0.120 V. The corrosion current density of the metal substrate is at 10-5 A/cm2 order of magnitude, while that of the LDH coating is usually at 10-7-10-9 A/cm2 order of magnitude, indicating LDH coating can greatly increase the corrosion resistance. Among the LDHs intercalated with organic anions, the LA-intercalated ZnAl-LDH coatings fabricated by ZHANG et al [23] got the lowest corrosion current density (~1��10-9 A/cm2). The corrosion current density of the MgAl-ASP- LDH prepared in this work is lower than that of the MgAl-PA-LDH deposited by CHEN et al [17] and ZHANG et al [26], implying that ASP-intercalated MgAl-LDH has better corrosion resistance than the PA-intercalated one.
4 Conclusions
(1) ASP is successfully inserted into the ZnAl-LDH and MgAl-LDH laminate with a simple one-step hydrothermal method instead of the conventional two-step anion exchange method.
(2) The ZnAl-ASP-LDH films exhibit a porous NS structure, while the MgAl-ASP-LDH coatings display a three-dimensional rose-like sheet structure with high ratio of pore coverage.
(3) In the immersion test, ZnAl-ASP-LDH coating is dissolved and disappears after soaking in 3.5 wt.% NaCl solution for 168 h. On the contrary, the MgAl-ASP-LDH coating still maintains a dense 3D sheet-like structure after immersing for 480 h. The coating of MgAl-ASP-LDH shows better durability in corrosion solution.
(4) Typically, the corrosion current density of ZnAl-ASP-LDH and MgAl-ASP-LDH coating is two or three orders of magnitude lower than that of bare Mg alloy, respectively, indicating that the ZnAl-LDH or MgAl-LDH films intercalated with organic ASP anions can effectively improve the corrosion resistance of Mg alloy.
References
[1] GUO Lian, WU Wei, ZHOU Yong-feng, ZHANG Fen, ZENG Rong-chang, ZENG Jian-min. Layered double hydroxide coatings on magnesium alloys: A review [J]. Journal of Materials Science & Technology, 2018, 9: 1455-1466.
[2] IQBAL N, IQBAL S, IQBAL T, BAKHSHESHI-RAD H R, ALSAKKAF A, KAMIL A, RAFIQ M, KADIR A, IDRIS M H, RAGHAV H B. Zinc-doped hydroxyapatite-zeolite/ polycaprolactone composites coating on magnesium substrate for enhancing in-vitro corrosion and antibacterial performance [J]. Transactions of Nonferrous Metals Society of China, 2020, 30: 123-133.
[3] HEAKAL, El-TAIB F, FEKRY A M, ABD El-BARR JIBRIL M. Electrochemical behaviour of the Mg alloy AZ91D in borate solutions [J]. Corrosion Science, 2011, 4: 1174-1185.
[4] LIU Dan, SONG Ying-wei, SHAN Da-yong, HAN En-hou. Self-healing coatings for magnesium alloys: A review [J]. Surface Technology. 2016, 45: 28-35 (in Chinese).
[5] WANG Wei, WANG Xin, LIU Xiao-jie, FAN Wei-jie, LI Wei-hua. Research progress of self-healing coatings in marine environment [J]. Equipment Environmental Engineering, 2018, 15: 89-97. (in Chinese)
[6] YANG H Y, CHEN X B, GUO X W, WU G H, DING W J, BIRNILIS N. Coating pretreatment for Mg alloy AZ91D [J]. Applied Surface Science, 2012, 14: 5472-5481.
[7] SHI Liu-ting, HU Jia, LIN Xu-dong, FANG Liang, WU Fang, XIE Jiao, MENG Fan-ming. A robust superhydrophobic PPS-PTFE/SiO2 composite coating on AZ31 Mg alloy with excellent wear and corrosion resistance properties [J]. Journal of Alloys and Compounds, 2017, 721: 157-163.
[8] CHENG Yuan-fen, DU Wen-bo, LIU Ke, FU Jun-jian, WANG Zhao-hui, LI Shu-bo, FU Jin-long. Mechanical properties and corrosion behaviors of Mg-4Zn-0.2Mn-0.2Ca alloy after long term in vitro degradation [J]. Transactions of Nonferrous Metals Society of China, 2020, 30: 363-372.
[9] WANG Shu-yan, SI Nai-chao, XIA Yong-ping, LIU Li. Influence of nano-SiC on microstructure and property of MAO coating formed on AZ91D magnesium alloy [J]. Transactions of Nonferrous Metals Society of China, 2015, 25: 1926-1934.
[10] XIE Jiao, HU Jia, LIN Xu-dong, FANG Liang, WU Fang, LIAO Xiao-ling, LUO Hai-jun, SHI Liu-ting. Robust and anti-corrosive PDMS/SiO2 superhydrophobic coatings fabricated on magnesium alloys with different-sized SiO2 nanoparticles [J]. Applied Surface Science, 2018, 457: 870-880.
[11] PAHALAGEDAR M N, SAMARAWEERA M, DHARMARATHNA S, KUO Chung-hao, PAHALAGEDARA L R, GASCO J A, SUIB S L. Removal of azo dyes: Intercalation into sonochemically synthesized NiAl layered double hydroxide [J]. The Journal of Physical Chemistry C, 2014, 31: 17801-17809.
[12] LU Yi, JIANG Bin, FANG Liang, LING Fa-ling, GAO Jie-mei, WU Fang, ZHANG Xi-hua. High performance NiFe layered double hydroxide for methyl orange dye and Cr(VI) adsorption [J]. Chemosphere, 2016, 152: 415-422.
[13] WANG Zhi-hu, ZHANG Ju-mei, LI YAN, BAI Li-jing, ZHANG Guo-jun. Enhanced corrosion resistance of micro- arc oxidation coated magnesium alloy by superhydrophobic Mg-Al layered double hydroxide coating [J]. Transactions of Nonferrous Metals Society of China, 2019, 29: 2066-2077.
[14] ZENG Rong-chang, LIU Zhen-guo, ZHANG Fen, Li Shuo-qi, HE Qing-kun, CUI Hong-zhi, HAN En-hou. Corrosion resistance of in-situ Mg�CAl hydrotalcite conversion film on AZ31 magnesium alloy by one-step formation [J]. Transactions of Nonferrous Metals Society of China, 2015, 25: 1917-1925.
[15] BUCHHEIT R G, GUAN Hong, MAHAJANAM S, WONG F. Active corrosion protection and corrosion sensing in chromate-free organic coatings [J]. Progress in Organic Coatings, 2003, 3-4: 174-182.
[16] MONTEMOR M F, SNIHIROVA D V, TARYBA M G, LAMAKA S V, KARTSONAKIS I A, BALASKAS A C, KORDAS G C, TEDIM J, KUZENTSOVA A, ZHELUDKEVICH M L. Evaluation of self-healing ability in protective coatings modified with combinations of layered double hydroxides and cerium molibdate nanocontainers filled with corrosion inhibitors [J]. Electrochimica Acta, 2012, 60: 31-40.
[17] CHEN Jun, SONG Ying-wei, SHAN Da-yong, Han En-hou. Modifications of the hydrotalcite film on AZ31 Mg alloy by phytic acid: The effects on morphology, composition and corrosion resistance [J]. Corrosion Science, 2013, 74: 130-138.
[18] TANG Yan, WU Fang, FANG Liang, GUAN Ting, HU Jia, ZHANG Shu-fang. A comparative study and optimization of corrosion resistance of ZnAl layered double hydroxides films intercalated with different anions on AZ31 Mg alloys [J]. Surface and Coatings Technology, 2019, 358: 594-603.
[19] POZNYAK S K, TEDIM J, RODRIGUES L M, SALAK A N, ZHELUDKEVICH M L, DICK L F P, FERREIRA M G S. Novel inorganic host layered double hydroxides intercalated with guest organic inhibitors for anticorrosion applications [J]. ACS Applied Materials & Interfaces, 2009, 10: 2353-2362.
[20] LIN J K, HSIA C L, UAN J Y. Characterization of Mg, Al-hydrotalcite conversion film on Mg alloy and Cl- and CO32- anion-exchangeability of the film in a corrosive environment [J]. Scripta Materialia, 2007, 11: 927-930.
[21] WANG Jun, LI Dan-dan, YU Xiang, JING Xiao-yan, ZHANG Mi-lin, JIANG Zhao-hua. Hydrotalcite conversion coating on Mg alloy and its corrosion resistance [J]. Journal of Alloys and Compounds, 2010, 1-2: 271-274.
[22] GUO Xiao-xiao, XU Sai-long, ZHAO Li-li, LU Wei, ZHANG Fa-zhi, EVANS D G, DUAN Xue. One-step hydrothermal crystallization of a layered double hydroxide/ alumina bilayer film on aluminum and its corrosion resistance properties [J]. Langmuir, 2009, 17: 9894-7.
[23] ZHANG Fa-zhi, ZHAO Li-li, CHEN Hong-yun, XU Sai-long, EVANS D G, DUAN Xue. Corrosion resistance of superhydrophobic layered double hydroxide films on aluminum [J]. Angewandte Chemie International Edition, 2008, 13: 2466-2469.
[24] BEREKET G, YURT A. The inhibition effect of amino acids and hydroxy carboxylic acids on pitting corrosion of aluminum alloy 7075 [J]. Corrosion Science, 2001, 6: 1179-1195.
[25] CHEN Jia-ling, FANG Liang, WU Fang, XIE Jiao, HU Jia, JIANG Bin, LUO Hai-jun. Corrosion resistance of a self-healing rose-like MgAl-LDH coating intercalated with aspartic acid on AZ31 Mg alloy [J]. Progress in Organic Coatings, 2019, 136: 105234.
[26] ZHANG Gen, WU Liang, TANG Ai-tao, MA Yan-long, SONG Guang-Ling, ZHENG Da-jiang, JIANG Bin, ATRENS A, PAN Fu-sheng. Active corrosion protection by a smart coating based on a MgAl-layered double hydroxide on a cerium-modified plasma electrolytic oxidation coating on Mg alloy AZ31 [J]. Corrosion Science, 2018, 139: 370-382.
[27] ZHANG Cai-xia, LUO Xiao-hu, PAN Xin-yu, LIAO Li-ying, WU Xiao-song, LIU Ya-li. Self-healing Li-Al layered double hydroxide conversion coating modified with aspartic acid for 6N01 Al alloy [J]. Applied Surface Science, 2017, 394: 275-281.
[28] PENG Feng, LI Hua, WANG Dong-hui, TIAN Peng, TIAN Ya-xin, YUAN Guang-yin, XU De-min, LIU Xuan-yong. Enhanced corrosion resistance and biocompatibility of magnesium alloy by Mg�CAl-layered double hydroxide [J]. ACS Applied Materials & Interfaces, 2016, 8: 35033-35044.
[29] MUHAMMAD J A, ZHAO Jing-mao, VAHDAT Z A, GHULAM Y, WANG Wei, WEI Shi-xiong, ZHAO Zhi-jie, WAHEED Q K. In-situ intercalation of 8-hydroxyquinoline in Mg-Al LDH coating to improve the corrosion resistance of AZ31 [J]. Corrosion Science, 2019, 157: 1-10.
[30] LI Ying-dong, LI Song-mei, ZHANG You, YU Mei, LIU Jian-hua. Fabrication of superhydrophobic layered double hydroxides films with different metal cations on anodized aluminum 2198 alloy [J]. Material Letter, 2015, 142: 137-140.
[31] GUO Xiao-xiao, XU Sai-long, ZHAO Li-li, LU Wei, ZHANG Fa-zhi, EVANS D G, DUAN Xue. One-step hydrothermal crystallization of a layered double hydroxide/ alumina bilayer film on aluminum and its corrosion resistance properties [J]. Langmuir, 2009, 17: 9894-9897.
[32] WU Liang, YANG Dan-ni, ZHANG Gen, ZHANG Zhi, ZHAN Sheng, TANG Ai-tao, PAN Fu-sheng. Fabrication and characterization of Mg-M layered double hydroxide films on anodized magnesium alloy AZ31 [J]. Applied Surface Science, 2018, 431: 177-186.
[33] GAO Rui, LIU Qi, WANG Jun, ZHAN Xiao-fei, YANG Wan-lu, LIU Jing-yuan, LIU Lian-he. Fabrication of fibrous szaibelyite with hierarchical structure superhydrophobic coating on AZ31 magnesium alloy for corrosion protection [J]. Chemical Engineering Journal, 2014, 241: 352-359.
[34] LI Ji, LIN Kai-dong, LUO Xiao-hu, ZHAN Hui-xia, CHENG F Y, LI Xiang-bo, LIU Ya-li. Enhanced corrosion protection property of Li-Al layered double hydroxides (LDHs) film modified by 2-guanidinosuccinic acid with excellent self-repairing and self-antibacterial properties [J]. Applied Surface Science, 2019, 480: 384-394.
�¼���1,2���� ��1,2���� ��1�����ܹ�2���� ��3�����緼4���� ��3������5
1. �����ѧ ����ѧԺ �����װ����ϵͳ��ȫ���¼��������ص�ʵ���ң�
������̬���������ܲ����о��������ص�ʵ���ң����� 400044��
2. �Ĵ��ữ����ѧ ���Ͽ�ѧ�빤��ѧԺ ���ϱ����븯ʴ�Ĵ�ʡ�ص�ʵ���ң��Թ� 643000��
3. �����ѧ ���Ͽ�ѧ�빤��ѧԺ þ�Ͻ���ҹ����о����ģ����� 400044��
4. ������ӹ���ѧԺ ����ѧԺ������ 401331��
5. ����ʦ����ѧ ��������ӹ���ѧԺ ��繦�ܲ����������ص�ʵ���ң����� 401331
ժ Ҫ��ʹ��һ����ˮ�ȷ��ֱ��Ʊ��춬����(ASP)����ZnAl��״˫��������(ZnAl-LDH)��MgAl-LDHͿ�㡣�����Ʊ�������Ϳ�����ò���ɷ֡��ṹ����ʴ�Խ��бȽ��о���������������������ܵIJ�״����Ƭ(NS)�ڻ����ϴ�ֱ����������MgAl-ASP-LDHͿ����ֳ��߿�϶�����ʵ���ά(3D)õ����Ƭ״�ṹ�����ݲ����У�MgAl-ASP-LDHͿ��չʾ����ZnAl-ASP-LDHͿ���������;��Ժ���ʴ�ԡ�ZnAl-ASP-LDH��MgAl- ASP-LDHͿ��ĸ�ʴ�����ܶȱ�Mg�Ͻ���ĵ�2~3��������������������л�ASP�����ӵ�ZnAl/MgAl-LDHĤ�����������þ�Ͻ����ʴ�ԡ�
�ؼ��ʣ�ˮ��ʯ��þ�Ͻ��춬�������ʴ����ʴ���Ƽ�
(Edited by Bing YANG)
Foundation item: Projects (cstc2019jcyj-msxmX0566, cstc2018jcyjAX0450, cstc2018jcyjA2923, cstc2017jcyjAX0393) supported by the Natural Science Foundation of Chongqing City, China; Project (2019CDXZWL002) supported by President Foundation of Chongqing University, China; Projects (2019CDXYWL0029, 2018CDJDWL0011) supported by Fundamental Research Funds for the Central Universities of China; Projects (KJKJQN201800102, KJQN201800619, KJ1703042) supported by Science and Technology Research Program of Chongqing Municipal Education Commission, China; Projects (202003150060, 202003150078) supported by Sharing Fund of Large-scale Equipment of Chongqing University, China
Corresponding author: Liang FANG, E-mail: lfang@cqu.edu.cn; Xiao-guang ZENG, E-mail: hnzxg1979@126.com
DOI: 10.1016/S1003-6326(20)65389-4

