J. Cent. South Univ. Technol. (2008) 15: 515-519
DOI: 10.1007/s11771-008-0097-5
Adsorption of copper(Ⅱ) and chromium(Ⅵ) on diaspore
LIU Xiao-wen(刘晓文), WANG Jian-rong(王建绒), HU Yue-hua(胡岳华)
(School of Resources Processing and Bioengineering, Central South University, Changsha 410083, China)
Abstract: The adsorption of Cu(Ⅱ) and Cr(Ⅵ) on diaspore was studied with the help of X-ray diffraction analysis, BET measurement, zeta potential measurement and atomic adsorption spectrometry. The adsorption equilibrium almost reaches within 60 min. The adsorption isotherms of Cu(Ⅱ) and Cr(Ⅵ) could be well described by the Langmuir equation. The adsorption capacities of Cu(Ⅱ) and Cr(Ⅵ) are 1.944 and 1.292 mg/g, respectively. The adsorption percentage of Cr(Ⅱ) increases with the increment of solution pH, but the adsorption percentage of Cr(Ⅵ) decreases. This could be explained by zeta potential theoretical and electrostatic attraction between metal ions and diaspore surface.
Key words: diaspore; adsorption; copper(Ⅱ); chromium(Ⅵ); zeta potential
1 Introduction
Environmental contamination due to heavy metal is caused by mining, agricultures and so on[1]. Heavy metals, such as copper, lead, chromium and nickel, are toxic even in extremely minute quantities[2-3]. There are varieties of minerals that are capable of removing metal ions from aqueous solution in the nature. Adsorption is a common mechanism of metal removal that takes place on mineral-solution interface[4]. Aluminum oxides, abounding in nature, are good adsorbents in many environments, for their capability of being well dispersed and coated on other particles. Absorption of metal ions to oxides in soil sediments is significant because metal ions affects their mobility and bioavailability[5-6].
Research on various mineral adsorbents has been carried out. Diaspore is one of the most important aluminum oxide minerals in the nature. From the spectroscopic analysis, FROST and KLOPROGGE[7] found that diaspore was isostructural with goethite. The structure of diaspore is based on oxygen atoms in hexagonal close packing. Hydroxyls are found at the corners of octahedra and aluminum atoms in the center of octahedra. Octahedra are linked together by edge sharing to form double chains. The adsorption of metal ions on goethite has been studied in the recent years[5, 8], however, there are few studies made in adsorption of diaspore[9-10].
Like most minerals, metal ions are adsorbed by diaspore in a narrow pH range and the adsorption depends on the concentration of substrate, the presence of competing and complexing ions and temperature.
The aim of this work is to make an attempt to provide some information of metal ions (Cu(Ⅱ) and Cr(Ⅵ)) on diaspore surface. The amount of adsorption was measured by atomic adsorption spectrometer(AAS). The zeta potential theoretical and electrostatic attraction were used to explain the different adsorptions between Cu(Ⅱ) and Cr(Ⅵ).
2 Experimental
2.1 Materials and characterization
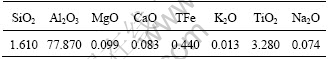
The diaspore sample was selected from Henan Province in China. The collected sample was ground by planetary ball milling. The chemical composition is listed in Table 1. The typical X-ray diffraction (Shimadzu) pattern of diaspore is shown in Fig.1, indicating that the sample is nearly pure diaspore. The BET surface area of sample was found to be 5.57 m2/g by nitrogen gas adsorption method at 77.4 K with a sorptionmeter (NOVA1000).
Table 1 Chemical composition of diaspore sample(mass fraction, %)
2.2 Zeta potential measurements
The zeta potential of diaspore was measured using a laser Doppler electrophoresis, Marvel Zetasizer 3000Hs instrument. The content of solid was about 0.001 5 % mg/L. The suspension was magnetically stirred for 3 min and transferred to a testing vessel, after which the zeta potential measurement was made. The pH value was adjusted by adding 0.1 mol/L HNO3 or NaOH solution.

Fig.1 X-ray powder diffraction pattern of diaspore sample
2.3 Experimental procedures
Stock solutions were prepared by dissolving CuSO4 and K2CrO4 (AR) into distilled water. The initial solution pH value was adjusted with 0.1 mol/L HNO3 or NaOH solution.
In adsorption experiments, diaspore and aqueous solution were placed in 250 mL beaker, magnetically stirred for 1 h at 25 ℃ and a constant pH value. At the end of each reaction, the suspension was centrifuged, and the supernatant was collected with a syringe for spectrum analysis of metal ion. The Hitachi Z-8000 polarized Zeaman atomic adsorption spectrometer(AAS) was used to determine the ion concentration.
The adsorption percentage(Y) at equilibrium was calculated by
Y=(ρ0-ρe)/ρ0 (1)
where ρ0 and ρe are the initial and equilibrium solution concentrations, mg/L.
3 Results and discussion
3.1 Zeta potential of diaspore suspension
The zeta potentials of diaspore suspension are shown in Fig.2. As expected, the zeta potential decreases with the increase of solution pH value. The isoelectric point(IEP) of diaspore obtained by reading the intercept of trend line is about 7.10. This value is in agreement with previous results in range of 5.8-7.7[11-13].
3.2 Effect of time
The kinetic experiment indicates that the adsorption of Cu(Ⅱ) at pH=6 and Cr(Ⅵ) at pH=4 on the diaspore is fairly rapid and reaches equilibrium in about 60 min (Fig.3). Though the adsorption percentage of Cu(Ⅱ) increases a little, it is not a significant change compared with total adsorption. Other adsorption experiments were made within 60 min.
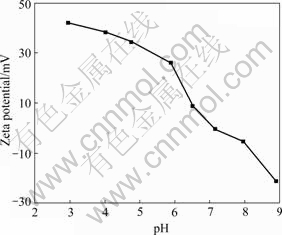
Fig.2 Zeta potentials of diaspore as function of pH value

Fig.3 Effect of time on adsorbing percentages of Cu (Ⅱ) (a) and Cr(Ⅵ) (b)
3.3 Adsorption isotherm
The adsorption isotherms were obtained at a constant temperature of 25 ℃ and a given pH value of 6±0.2 for Cu (Ⅱ) and of 3±0.2 for Cr(Ⅵ) to study the effect of initial metal ion concentration on adsorption. The concentration of diaspore was maintained at 10 mg/mL and the concentrations of metal ions were in the range of 5-50 mg/L. The concentrations of Cu(Ⅱ) and Cr(Ⅵ) used in this study were based on the concentrations in natural contaminated systems and instrument detection limits. The adsorption isotherms of diaspore are shown in Fig.4.

Fig.4 Adsorption isotherms of Cu(Ⅱ) (a) and Cr(Ⅵ) (b) on diaspore
The empirical modeling approach has been widely used for adsorption systems. According to the results shown in Fig.4, the two-parameter Langmuir equation was applied.
ρe/Qe=1/(KQt)+ρe/Qt (2)
where ρe is equilibrium solution concentration, mg/L; Qe is the amount of metal adsorbed, mg/g; K is the adsorption equilibrium constant and Qt is the adsorption capacity corresponding to monolayer coverage, mg/g[5-6].
The graphs of ρe/Qe vs ρe are given in Fig.5. The model fits the data well. Qt was calculated to be 1.944 mg/g for Cu(Ⅱ) and 1.292 mg/g for Cr(Ⅵ). The Cu(Ⅱ) adsorption capacity on diaspore is smaller than that on goethite. Diaspore and goethite have the similar crystal structure. However, diaspore sample has much less BET surface area in experiment[5, 8].

Fig.5 Langmuir adsorption isotherms of Cu(Ⅱ) (a) and Cr(Ⅵ) (b)
The equilibrium pH values after Cu(Ⅱ) and Cr(Ⅵ) adsorption at different initial concentrations are shown in Fig.6. The equilibrium pH value is higher than that of the solution before adsorption, at a low initial concentration. This may be due to the protonation of diaspore on the active surface. As the initial concentration increases, the equilibrium pH of solution decreases after Cu2+ adsorption. Cu2+ hydrolysis in solution forms the Cu(OH)+ and Cu2(OH)2+ which are adsorbed on diaspore surface. H+ is released during hydrolysis and adsorption. But the Cr(Ⅵ) adsorption is different from the Cu(Ⅱ) adsorption, which may be due to the CrO42+ hydrolysis to form HCrO4- and Cr2O72-, which reduces the H+ concentration. The equilibrium pH value of solution shows a positive correlation with the initial Cr(Ⅵ) concentration.

Fig.6 Equilibrium pH as function of initial concentration of Cu(Ⅱ) (a) and Cr(Ⅵ) (b)
3.4 Effect of pH value
The pH of solutions plays an important role in adsorbing metal ion from wastewater systems. It controls the surface properties of adsorbent. The experiments were carried out in the pH range from 3.0 to 8.0. The metal ions concentration was 10 mg/L.
Fig.7(a) shows the effect of pH value on the adsorption percentage of Cu(Ⅱ). Copper absorption by diaspore is highly pH-dependent. The amount of adsorbed copper shows a positive correlation with pH value. The adsorption percentage gets 99.6% at pH 6.95. This result supports the observation that the increase of pH value leads to the increase in negative charge on diaspore surface, which is advantageous to absorption of copper. These values are in excellent agreement with those reported in Refs.[5, 8]. Previous studies have shown that the adsorption of Cu(Ⅱ) on mineral surfaces is anomalous. The different isotherms of Cu(Ⅱ) adsorption on goethite at a high metal concentration were found[8]. It is explained that the competitive adsorption of Cu(OH)+ and Cu2(OH)2+ is one of the main reasons.

Fig.7 Effect of pH value on adsorption percentage of Cu(Ⅱ) (a) and Cr(Ⅵ) (b)
The adsorption of Cr(Ⅵ) on diaspore as a function of pH is shown in Fig.7(b). Compared with the case of copper, adsorption of Cr(Ⅵ) demonstrates a gradually decreasing trend with the increase of pH value. The adsorption percentage varies from 22.2% to 73.1%. It is in agreement with those reported in Ref.[14]. It was reported that Cr(Ⅵ) exists mainly in forms of CrO42-, HCrO4- and Cr2O72- in solution. The less amount of Cr(Ⅵ) adsorption at pH>pHIEP=7.10 is due to the negative surface electrical charge on diaspore.
4 Conclusions
1) Cu(Ⅱ) and Cr(Ⅵ) can be removed from aqueous solution using diaspore.
2) The adsorption isotherms of Cu(Ⅱ) and Cr(Ⅵ) on diaspore can be well described by Langmuir equation.
3) The adsorption capacities of Cu(Ⅱ) and Cr(Ⅵ) are 1.944 and 1.292 mg/g, respectively. The adsorption percentage of Cu(Ⅱ) on diaspore is positive correlated with pH value from 3.0 to 8.0, but that of Cr(Ⅵ) demonstrates a decreasing trend of pH value.
References
[1] Sitting M. Handbook of toxic and hazardous chemicals [M]. Bracknell: Noyes Publications, 1981.
[2] Das D D, Mahapatra R, Pradhan J, DAS S N, THAKUR R S. Removal of Cr(Ⅵ) from aqueous solution using activated cow dung carbon [J]. Journal of Colloid and Interface Science, 2000, 232(2): 235-240.
[3] Bereket G, Arog A Z, Ozel M Z. Removal of Pb(Ⅱ), Cd(Ⅱ), Cu(Ⅱ) and Zn(Ⅱ) from aqueous solutions [J]. Journal of Colloid and Interface Science, 1997, 187(2): 338-343.
[4] Hesham A, Philip R W. XPS study of the adsorption of lead on goethite (α-FeOOH) [J]. Applied Surface Science, 1998, 136(1/2): 46-54.
[5] Juang R S, Wu W L. Adsorption of sulfate and copper(Ⅱ) on goethite in relation to the change of zeta potentials [J]. Journal of Colloid and Interface Science, 2002, 249(1): 22-29.
[6] Trivedi P, Axe L, Dyer J. Adsorption of metal ions onto goethite: Single-adsorbate and competitive systems [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2001, 191(1/2): 107-121.
[7] Frost R L, Kloprogge J T. Dehydroxylation and the vibrational spectroscopy of aluminum oxyhydroxides using infrared emission spectroscopy (Part Ⅲ): Diaspore [J]. Applied Spectroscopy, 1999, 53(7): 829-835.
[8] Rodda D P, Wells J D, Johnson B B. Anomalous adsorption of copper(Ⅱ) on goethite [J]. Journal of Colloid and Interface Science, 1996, 184(2): 564-569.
[9] CHEN Xiang-qing, HU Yue-hua, WANG Yu-hua, XIONG Dao-ling. Effects of sodium hexmetaphosphate on flotation separation of diaspore and kaolinite [J]. Journal of Central South University of Technology, 2005, 12(4): 420-424.
[10] HU Yue-hua, LIU Xiao-wen, XU Zheng-he. Role of crystal structure in flotation separation of diaspore from kaolinite, pyrophyllite and illite [J]. Minerals Engineering, 2003, 16(3): 219-227.
[11] Parks G A. The isoelectric points of solids, solid hydroxides, and aqueous hydroxo complex systems [J]. Chemical Review, 1965, 65: 177-198.
[12] Yoon R H, Salman T, Donnay G. Predictive point zero charge of oxides and hydroxides [J]. Journal of Colloid and Interface Science, 1979, 70(3): 483-493.
[13] LIU Xiao-wen, HU Yue-hua, XU Zheng-he. Effect of chemical composition on electrokinetics of diaspore [J]. Journal of Colloid and Interface Science, 2003, 267(1): 211-216.
[14] Ko K R, Ryu S K, Park S J. Effect of ozone treatment on Cu(Ⅱ) and Cr(Ⅵ) adsorption behavious of activated carbon fibers[J]. Carbon, 2004, 42(8/9): 1864-1867.
Foundation item: Project(2005CB623701) supported by the Major State Basic Research Development Program of China
Received date: 2007-10-10; Accepted date: 2007-11-28
Corresponding author: HU Yue-hua, Professer; Tel: +86-731-8879815; E-mail: hyh@mail.csu.edu.cn
(Edited by YANG Hua)