
Corrosion behavior of WE54 magnesium alloy in 3.5%NaCl solution
LI Zhuo-qun(李卓群), SHAN Da-yong(单大勇), CHEN Rong-shi(陈荣石),
KE Wei(柯 伟), HAN En-hou(韩恩厚)
Environmental Corrosion Center, Institute of Metal Research, Chinese Academy of Sciences, Shenyang 110016, China
Received 28 July 2006; accepted 15 September 2006
Abstract: The corrosion behavior of WE54 magnesium alloy was investigated in 3.5%NaCl aqueous solution. The electrochemical study shows that the value of corrosion potential of WE54 magnesium was -1.8V(vs SCE). In the initial stage of immersion, a kind of visible thin film, which has the appearance of artificial conversion coating by microscopy observation, formed on the surface of WE54 alloy. Through the comparison of corrosion behavior between WE54 and AZ91D, it is shown that the value of corrosion potential of WE54 is approximately 200 mV lower than that of AZ91D, and the corrosion rate of WE54 was one order smaller than that of AZ91D. After 72 h constant immersion and corrosion products removal, deeper grooves along grain boundaries was revealed but lighter corrosion was apparent on the matrix of WE54 alloy. As far as AZ91D alloy was concerned, severe localized corrosion was dominant and network-bone-like structure was remained.
Key words: magnesium alloys; WE54; corrosion; electrochemical behavior; microstructure
1 Introduction
Magnesium and its alloys have excellent physical and mechanical properties for a number of applications. In particular, its high strength/mass ratio makes it an ideal metal for automotive and aerospace applications[1]. There has been a rapid growth in interest in recent years in the development of high strength, lightmass magnesium alloys for elevated temperature applications[2-3].
The Mg-Y-Nd system, which usually contains Y, Nd and some other rare-earth elements, is the most successful magnesium alloys in this category at present[3]. However, poor corrosion resistance is one of the main challenges in the widespread use of magnesium particular for outdoor applications[1, 4]. Two new generations of Mg alloys have been identified, having corrosion rates comparable to those of Al-based alloys. One is a high purity Mg-Al alloy, such as AZ91 and the second type of new alloy is the My-Y-Nd system, which we mentioned above, with examples being WE54 and WE43[5]. Considerable research has been carried out in the corrosion behavior of AZ91D[6-7]. But studies on the corrosion behavior of magnesium alloy WE54 are limited.
In this study, the basal corrosion behavior of magnesium WE54 in 3.5%NaCl aqueous solution was investigated at ambient temperature. And the aim of this study is to develop a further study of this alloy and its corrosion performance.
2 Experimental
WE54 and AZ91D magnesium alloys used in this study were prepared according to industrial casting norm. Specimens, which have the dimension of 10 mm×10 mm×10 mm, were used for all the experiments. The components of these alloys are listed in Table 1.
For immersion testing, the specimens were ground with 4 000 grit SiC paper, washed with distilled water and dried by flowing air. The polished face were exposed to the 3.5%NaCl aqueous solution(>30 mL/cm2) for various time intervals. The corrosion products were cleaned in a solution of 200 g/L CrO3+10 g/L AgNO3+10 g/L BaCl2. Specimens were dipped into the solution for a few seconds, followed by an alcohol washing.
Electrochemical polarization experiments were carried out using potentiostat EG&G model 273, computer driven (EG&G M352 software). Electrodes for potentiodynamic polarization were embedded in an epoxy resin to provide insulation, leaving 1 cm2 alloy surface in contact with the electrolyte. The electrolyte was still 3.5%NaCl aqueous solution. Three electrodes system was adopted. The counter electrode was platinum and a saturated calomel electrode (SCE) was used as reference. The polarization started from a cathodic potential about -250 mV negative to the corrosion potential and stopped at an anodic potential +1 000 mV positive to the corrosion potential. The scanning rate was 0.5 mV/s.
The microstructure and corrosion morphology of specimens were characterized under environmental scanning electron microscope(ESEM, Philips-XL30). Specimens for microstructure observation were first polished with 4000 grit SiC paper and polishing clothes, the rinsed with de-ionized water and dried in air at room temperature.
Table 1 Chemical composition of WE54 and AZ91D alloys (mass fraction, %)

3 Results and discussion
3.1 Microstructure
Fig.1 shows the microstructure of the WE54 ingots. The alloy exhibits a typical cast microstructure with equiaxed grains, which are separated by a grain boundary eutectic consisting of α-Mg matrix and the second phases. Some transmission electron microscopy reveals that the microstructure of the initial cast alloy consists of homogeneously distributed intermetallic β″ precipitates with a DO19 ordered hexagonal crystal structure and β′ precipitates with a base-centered orthorhombic crystal structure. Some heterogeneously distributed β precipi- tates with a face-cantered cubic crystal structure are also observed on matrix dislocation[8]. The microstructure of the AZ91D ingots, which is quite similar to that of the WE54, has been shown in many studies[6]. But the main difference between them is the compositions of the second phases. The composition of the second phase in the AZ91D is the Mg17Al12, while in the WE54 the second phases probably have the compositions Mg14Nd2Y for β and Mg12NdY for β′ [2]. The corrosion resistance of the magnesium alloy is strongly determined by the characteristics of the second phases, which caused internal galvanic corrosion.

Fig.1 Microstructure of WE54 ingots
3.2 Immersion studies
Fig.2 shows the surface features of specimens, which were exposed to 3.5%NaCl aqueous solution for 1 h and 2 h. In the initial stage of immersion, a kind of thin film, which has the appearance of some artificial conversion coatings in microscopy observation (and has a microscopically yellow color) formed on the surface of the alloy. It seems that the thin film only covered the α-Mg matrix. But this thin film could not stand more than 3 h. Some weak parts of the film broke quickly and localized corrosion features could be observed as shown is Fig.3. Fig.4 shows that after 72 h constant immersion and corrosion products removal, deeper groove along grain boundaries were revealed but lighter corrosion was apparent on the matrix of WE54 alloy. As far as the AZ91D alloy, another kind of thin film formed on the surface of specimens in the initial stage of immersion, but it could not be observed as clearly as WE54 alloy. After 72 h constant immersion and corrosion products removal, severe localized corrosion was dominant with several β particles remained on the surface unaffected while the entire matrix was dissolved (Fig.5).
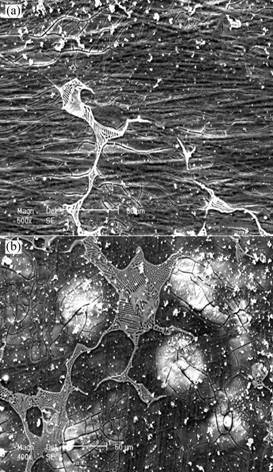
Fig.2 Micro corrosion morphologies for WE54 ingots in 3.5%NaCl aqueous solution: (a) 1 h; (b) 2 h
Several factors, such as the role of the second phases and the quasi-passive hydroxide, might contribute to these different corrosion morphologies between WE54 and AZ91D. The second phase Mg17Al12, whose corrosion potential is 490 mV, 420 mV higher than pure Mg and AZ91D, is highly cathodic to the α matrix. When AZ91D exposed to aggressive electrolyte containing Cl-, severe galvanic corrosion occurred between the Mg17Al12 phase and the matrix along the grain boundaries. The Mg17Al12 phase was so resistant that it remained intact when the matrix around it was corroded out. To the authors’ knowledge, the absolute corrosion potential of the second phases in the WE54 alloy, has not been figured out, however, it can be estimated from the potentio-dynamic polarization curves. The second phases in the WE54 were not as effective as that in the AZ91D when act as cathode, so the galvanic corrosion in WE54 were lighter than that in AZ91D. The grooved corrosion morphology may be caused by the weaker second phases and their loose connection. Corrosion occurred not only in the matrix but also in the second phases. When matrix around the second phases was corroded out, the loose connected second phases also fall off. The reduction of the second phases caused the reduction of the cathodic fraction on the surface, finally leading to the reduction of corrosion rate. The thin film formed on the WE54 in the initial stage of immersion did not show excellent protection to the alloy.

Fig.3 Localized corrosion morphology for WE54 ingots in 3.5%NaCl aqueous solution
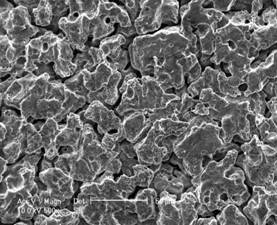
Fig.4 Micro corrosion morphology for WE54 ingot in 3.5%NaCl aqueous solution after immersion for 72 h and corrosion products removal

Fig.5 Micro corrosion morphologies for AZ91D ingots in 3.5%NaCl aqueous solution after immersion for 72 h and corrosion products removal
3.3 Potentio-dynamic polarization
The potentio-dynamic polarization curves for WE54 and AZ91D in 3.5%NaCl aqueous solution are shown in Fig.6. The AZ91D alloy do not show any passivity in this solution while the WE54 alloy shows a very small current plateau. The Ecorr for WE54 is about 200 mV lower than that of AZ91D, and the corrosion rate of WE54 is one order smaller than that of AZ91D. The second phases in the WE54 are composed of some rare earth elements. These compositions show more active than the β phase in AZ91D. So the value of corrosion potential of WE54 is more negative than AZ91D.
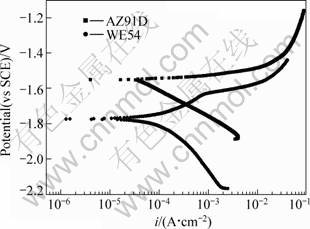
Fig.6 Potentiodynamic polarization curves for WE54 and AZ91D ingots in 3.5%NaCl aqueous solution
4 Conclusions
1) Corrosion behavior of WE54 is different from that of AZ91D. The main reason is the different characteristics of the second phases in the two alloys.
2) The value of corrosion potential of WE54 is -1.8 V(vs SCE), approximately 200 mV lower than that of AZ91D, and the corrosion rate of WE54 is one order smaller than that of AZ91D.
3) The corrosion morphology of WE54 after long time immersion is relative flat with deeper grooves along grain boundaries, which is different from that of AZ91D.
References
[1] FRAY J E, LUAN B. Protective coatings on magnesium and its alloys-a critical review[J]. J Alloys Compd, 2002, 336: 88-113.
[2] NIE J F, MUDDLE B C. Characterization of strengthening precipitate phases in a Mg-Y-Nd alloys[J]. Acta Mater, 2000, 48: 1691-1703
[3] NIE J F, MUDDLE B C Precipitation in magnesium alloys WE54 during isothermal ageing at 250℃ [J]. Scripta Mater, 1999, 40(10): 1089-1094.
[4] MAKER G L, KRUGER J. Corrosion studies of rapidly solidified magnesium alloys[J]. J Electrochem Soc, 1990, 137(2): 414-421.
[5] BIRSS V, XIA S, YUE R, RATERICK G, Richard J R. Characterization of oxide films formed on magnesium-based WE43 alloy using AC/DC anodization in silicate solution[J]. J Electrochem Soc, 2004, 151(1): B1-B10.
[6] SONG Guang-ling, ATRENS A. Corrosion mechanisms of magnesium alloys[J]. Adv Eng Mater, 1999, 1: 11-31.
[7] MATHIEU S, RAPIN C, HAZAN J, STEINMETZ P. Corrosion behavior of high pressure die-cast and semi-solid cast AZ91D alloys[J]. Corr Sci, 2002, 44: 2737-2756.
(Edited by YANG Hua)
Foundation item: Project(50574086) supported by the National Natural Science Foundation of China
Corresponding author: SHAN Da-yong; Tel: +86-24-23893115; E-mail: dyshan@imr.ac.cn