炭材料高温抗氧化莫来石/SiC复合涂层的制备及其1150 °C高温抗氧化性能
来源期刊:中国有色金属学报(英文版)2012年第12期
论文作者:杨 鑫 苏哲安 黄启忠 柴立元
文章页码:2997 - 3002
关键词:炭材料;抗氧化;涂层;SiC;莫来石
Key words:carbon materials; oxidation resistance; coating; SiC; mullite
摘 要:采用化学气相反应及料浆刷涂烧结复合工艺在石墨表面制备高温抗氧化莫来石/SiC复合涂层。XRD物相分析结果显示涂层外层由莫来石及微量SiO2相组成,涂层内层主要由β-SiC相组成。通过高温抗氧化试验研究涂层的高温抗氧化行为并测试涂层氧化后的洛氏硬度。结果表明:所制备的莫来石/SiC复合涂层具有良好的高温抗氧化及热震性能,经过1150 °C、109 h的高温氧化及12次1150 °C→室温的循环热震试验后,涂层试样的质量增加率为0.085%。硬度测试结果表明:所制备的莫来石/SiC复合涂层各层之间具有良好的结合性能。
Abstract: To protect carbon materials from oxidation, mullite/SiC coatings were prepared on graphite by chemical vapor reaction (CVR) and slurry sintering. The XRD analyses show that the phase of the outer-layer coating is composed of SiO2 and mullite, and the inner-layer coating is mainly composed of β-SiC. The anti-oxidation behavior of the coating and the Rockwell hardness (HRB) of the coating after oxidation were investigated. The oxidation test shows that the as-prepared multi-layer coating exhibits excellent anti- oxidation and thermal shock resistance at high temperature. After oxidation at 1150 °C for 109 h and thermal shock cycling between 1150 °C and room temperature for 12 times, the mass gain of the coated sample is 0.085%. Meanwhile, the indentation tests also demonstrate that the as-prepared coating has good bonding ability between the layers.
Trans. Nonferrous Met. Soc. China 22(2012) 2997-3002
YANG Xin1, 2, SU Zhe-an1, HUANG Qi-zhong1, CHAI Li-yuan2
1. State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China;
2. School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China
Received 11 November 2011; accepted 4 June 2012
Abstract: To protect carbon materials from oxidation, mullite/SiC coatings were prepared on graphite by chemical vapor reaction (CVR) and slurry sintering. The XRD analyses show that the phase of the outer-layer coating is composed of SiO2 and mullite, and the inner-layer coating is mainly composed of β-SiC. The anti-oxidation behavior of the coating and the Rockwell hardness (HRB) of the coating after oxidation were investigated. The oxidation test shows that the as-prepared multi-layer coating exhibits excellent anti- oxidation and thermal shock resistance at high temperature. After oxidation at 1150 °C for 109 h and thermal shock cycling between 1150 °C and room temperature for 12 times, the mass gain of the coated sample is 0.085%. Meanwhile, the indentation tests also demonstrate that the as-prepared coating has good bonding ability between the layers.
Key words: carbon materials; oxidation resistance; coating; SiC; mullite
1 Introduction
Carbon materials are ideal high-temperature structurals material for engineering and aerospace applications, due to their advantages of lightweight, high strength, excellent thermal shock and thermal erosion resistance, they are widely used in electrical contact, heating unit, fusion reactors, space shuttle, rocket nozzles, hypersonic vehicles and propulsion systems [1-3]. However, their defect of easy oxidation above 450 °C in an oxidizing environment limits their broad applications. To solve this problem, much effort has been conducted and it is widely acknowledged that coating method is an effective way that can extend their service life at high temperatures [4].
To apply coatings on the carbon materials, silicon-based ceramics such as SiC and Si3N4 are usually used as coating materials due to their good mechanical properties, excellent anti-oxidation property, and good physical-chemical compatibility with carbon materials [5]. However, the SiC coatings prepared by the reported methods [6-9] are difficult to obtain a dense structure. Moreover, these prepared coatings are prone to crack when they endure the severe environment with rapid heating and cooling. Therefore, multi-layer coating with different ceramic phases has been proposed. It is well known that mullite is the prime candidate for the coating of SiC and SiC/SiC composites due to its close coefficients of thermal expansion (CTEs) match (the CTEs of mullite and silicon carbide are 5.3×10-6 °C-1 and 4.7×10-6 °C-1, respectively) with SiC and chemical compatibility [10]. In addition, as a potential coating material, mullite also has the advantages of excellent anti-oxidation and corrosion resistance, stability at elevated temperatures, low thermal conductivity and so on [11-13].
The previous work reported that SiC-Al2O3- mullite multi-coatings prepared by the pack cementation technique had excellent oxidation resistance from 1300 to 1600 °C [14-17]. However, few literatures have been published about this anti-oxidation coating for long-term application at 1150 °C. Though pack cementation method is a simple and low cost technique that can be conducted in laboratory. However, the SiC-Al2O3- mullite multi-coatings obtained by this method represents disadvantages that will hinder their potential applications, such as the non-uniform coating thickness due to the gravity influence in the preparation process, the requirement of an extra procedure to remove the loose embedded pack material, and the difficulty of treating parts with complex shape and large size [18,19]. Compared with the pack cementation method, the slurry method is more suitable to prepare ceramic coatings for its advantages of low cost, easy operation, and no special requirements to the shape of sample [20]. Meanwhile, the chemical vapor reaction (CVR) has also been demonstrated to be a potential technology that can coat large complex components uniformly with low cost [21-23]. The combination of these two methods may offer a new way to prepare SiC/mullite coating for carbon material anti-oxidation application.
In the present work, the SiC inner coating was prepared by CVR, and then the mullite outer-layer coating was formed via the slurry sintering. The anti-oxidation property of the mullite/SiC coating was tested and the hardness (HRB) of the multi-layer coating after oxidation was investigated.
2 Experimental
The graphite with a density of 1.76 g/cm3 was used as the substrate for coating and then cut into small specimens. Before the CVR, the specimens were hand-polished using 600 grit SiC paper, then cleaned ultrasonically with ethanol and dried at 120 °C for 2 h. The materials used in the CVR were Si sheets (industrial reagent, >99.4%) and SiO2 powders (industrial reagent, >99.2%). After being washed by dilute hydrochloric acid to remove the impurities, the Si sheets and SiO2 powders were placed in a graphite crucible and heated to generate vapor at temperature above their melting points. Meanwhile, the as-prepared specimens were exposed to the mixed vapor to form the coatings. The whole process was conducted at the temperatures within the range of 1650-2100 °C for 1-3 h, followed by a natural cooling course.
The outer-layer mullite coating was prepared by slurry sintering. The staring materials used for slurry were Al2O3 and SiO2 powders, with the mass ratio of 3:1. The powders were blended with silica sol and then mixed thoroughly to produce a viscosity for brushing slurry on the specimen. After drying, the as-coated sample was sintered at 1600-1800 °C for 2-6 h to form the mullite coating.
The isothermal oxidation test of the coated sample was carried out at 1150 °C in an electrical furnace. The furnace was first heated up to 1150 °C in air, then the coated sample was placed into the furnace and maintained at that temperature for a specified time. Meanwhile, the coated specimen also endured the thermal shock cycling from high temperature to room temperature when taken out of the furnace directly to air. Cumulative mass changes of the coated sample after every thermal cycle were measured by the precision balance and reported as a function of time. The hardness (HRB) of the mullite/SiC coating after oxidation test was measured by Rockwell’s indentation with 980 N an as applied load for 10 s on the oxidized coating surface (500MRA Electric Rockwell Apparatus). Steel sphere with a diameter of 1.588 mm was used in the measurement, and the reported value was an average of five measurements. The surface morphologies of the coatings were observed with scanning electron microscopy (SEM). X-ray diffraction (XRD) and energy dispersive spectroscopy (EDS) were also used to identify crystalline structures and analyze element distribution in the coating, respectively.
3 Results and discussion
Figure 1(a) shows the XRD pattern of the as-prepared inner coating after the CVR. The phase analysis demonstrates that the observed strong peaks are corresponding to β-SiC, indicating that the phase composition of the inner coating is β-SiC. Figure 1(b) displays the XRD pattern of the mullite outer layer, and it is obvious that the outer layer mainly consists of SiO2 and mullite phases.
Typical macro-morphology of the SiC inner layer and mullite outer layer is displayed in Fig. 2. From Fig. 2(a), it can be seen that the as-prepared SiC inner coating is composed of many bright crystals and it reveals as a gray outer layer. After slurry sintering, a smooth and white mullite outer layer is covered uniformly on the inner SiC coating (Fig. 2(b)).
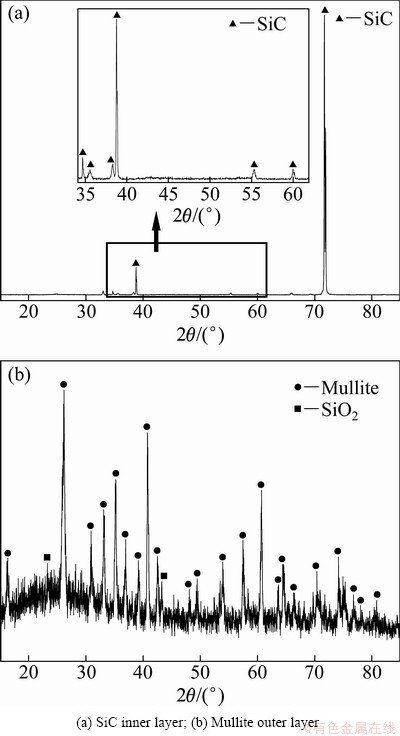
Fig. 1 XRD patterns of as-prepared coatings on graphite

Fig. 2 Macro-morphology of SiC coating and mullite/SiC coating on graphite
Figure 3 shows the surface and cross-section morphologies of the SiC inner coating. It is clear that the coating surface is composed of SiC micro-crystals together with large SiC grains which are homogenously distributed and randomly oriented (Fig. 3(a)). The cross- section morphology of the SiC inner coating is shown in Fig. 3(b). It can be seen that the obtained coating has an uneven thickness distribution (about 100 μm), and there is no obvious interface between the coating and the substrate, which reveals the good compatibility and adherence between them.
The SEM surface morphologies of the as-prepared mullite outer coating after slurry sintering are shown in Fig. 4. It is observed that the coating is dense and there are no visible cracks or holes formed on the coating surface (Fig. 4(a)), indicating the formation of a dense structure for mullite outer coating. Further observation shows that the formed mullite particles distribute randomly around the SiO2 galss (Fig. 4(b)).

Fig. 3 SEM images of CVR SiC coating on graphite

Fig. 4 Surface morphologies of as-prepared mullite outer coating
Figure 5 presents the isothermal oxidation curve of the mullite/SiC coated sample in air at 1150 °C. As shown in Fig. 5, it is obvious that the coated sample exhibits excellent oxidation resistance in air at 1150 °C, and it gains mass continuously in the whole oxidation process. After 109 h oxidation, the mass gain of the coated sample is 0.085%, which confirms that the as-prepared coating has excellent anti-oxidation property at high temperature and it can provide long-term protection for carbon materials. Meanwhile, the thermal shock resistance of the coated sample was also investigated in the oxidation test. After enduring 12 thermal cycles from 1150 °C to room temperature, no spallation or peeling was found. Therefore, it can be inferred that the multi-layer coating has an excellent thermal shock resistance, which may be attributed to the good bonding ability among the carbon substrate, the SiC inner layer and mullite outer layer.
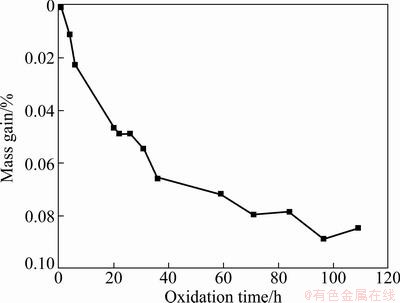
Fig. 5 Isothermal oxidation curve of mullite/SiC coated sample in air at 1150 °C
The XRD analysis of the coated sample after oxidation verifies that the oxidized coating is composed of mullite and SiO2 (Fig. 6). It can be seen from Fig. 6 that the SiO2 peaks become stronger compared with Fig. 1(b), this may be attributed to the oxidation of the inner SiC coating. Figure 7 displays the surface image of the coated sample after oxidation at 1150 °C. It can be seen that a smooth glass-like layer is formed on the coating surface, and the EDS analyses reveal that the component of this glass-like layer is mainly composed of O, Al and Si elements, which is consistent with the XRD result (Fig. 6).

Fig. 6 XRD pattern of mullite/SiC coating after oxidation

Fig. 7 SEM images (a,b) and EDS result of mullite/SiC coating after oxidation in air at 1150 °C

Fig. 8 SEM images of mullite/SiC coating after isothermal oxidation and indentation test
In addition, the hardness of the multi-layer coating after oxidation was also investigated. After the indentation tests for five times, the average Rockwell hardness of the oxidized coating is HRB 51.7, and the typical SEM images of the coating after isothermal oxidation and indentation tests are shown in Fig. 8. As shown in Fig. 8(a), sphere indentation shape and cracks formed around the edge of the indenter can be clearly seen. Meanwhile, it should be noted that the surface cracks formed during the indentation are very short (Fig. 8(b)), and no peeling off or delamination of the multi-layers is observed, indicating the good bonding ability between them. To further understand the bonding ability between the multi-coating layers, the cross- section of the mullite/SiC coating after isothermal oxidation and indentation test was investigated and the results are displayed in Fig. 9. From Fig. 9(a), it is evident that the multi-coating reveals a typical double layer structure. By EDS analysis, the inner SiC layer and outer mullite layer can be distinguished easily (Figs. 9(b) and (c)). Meanwhile, it can be seen that there are no penetrating cracks or holes formed in the coating and the graphite substrate does not show any sign of oxidation, which suggests that the coating is dense and the excellent anti-oxidation and thermal shock resistance of the coated sample is mainly contributed to the dense structure of the multi-layer coating. More importantly, the cross-section micrograph (Fig. 9(a)) clearly shows that the coating is still integrate and adheres well with each other even after long time isothermal oxidation, thermal shock cycling and indentation tests, indicating the good bonding ability between the layers.
4 Conclusions
1) Oxidation protective mullite/SiC coating can be prepared on graphite by CVR and slurry sintering.
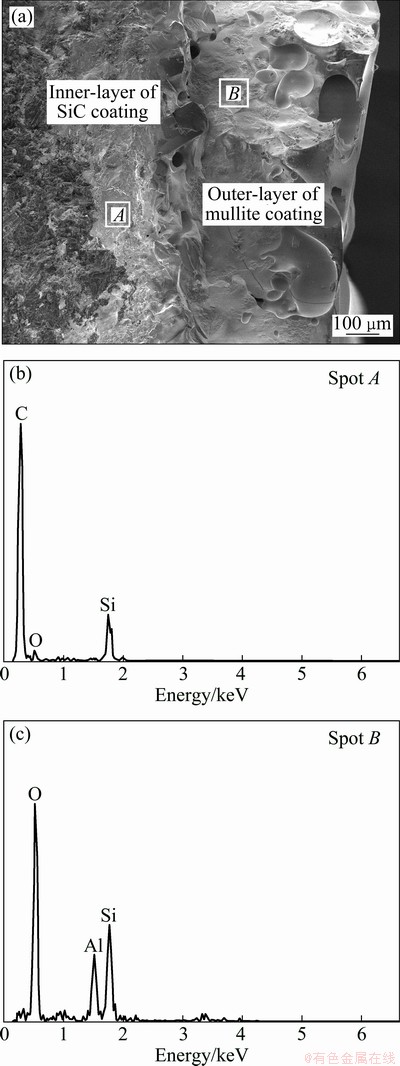
Fig. 9 Cross-section morphology (a) and EDS analyses (b,c) of mullite/SiC coating after isothermal oxidation and indentation test
2) Oxidation test carried out at 1150 °C shows that the coated graphite gains mass continuously in the whole oxidation process. After 109 h oxidation, the mass gain of the coated sample is 0.085%, which confirms that the as-prepared coating has excellent anti-oxidation property at high temperature and it can provide long-term protection for carbon materials.
3) Rockwell indentation tests were performed on the oxidized coating surface, and the average hardness of the oxidized coating is HRB 51.7. After the indentation test, no peeling off or delamination of the multi layers are found, indicating the good bonding ability between them.
References
[1] Shimada S, Sato T. Preparation and high temperature oxidation of SiC compositionally graded graphite coated with HfO2 [J]. Carbon, 2002, 40: 2469-2475.
[2] Zhu Y C, Ohtani S, Sato Y, Iwamoto N. The improvement in oxidation resistance of CVD-SiC coated C/C composites by silicon infiltration pretreatment [J]. Carbon, 1998, 36: 929-935.
[3] Kim J I, Kim W J, Choi D J, Park J Y, Ryu W S. Design of a C/SiC functionally graded coating for the oxidation protection of C/C composites [J]. Carbon, 2005, 43: 1749-1757.
[4] Fu Qian-gang, Li He-jun, Shi Xiao-hong, Li Ke-zhi, Sun Guo-dong. Silicon carbide coating to protect carbon/carbon composites against oxidation [J]. Scripta Materialia, 2005, 52: 923-927.
[5] Yang Xin, Zou Yan-hong, Huang Qi-zhong, Su Zhe-an, Chang Xin, Zhang Ming-yu, Xiao Yong. Improved oxidation resistance of chemical vapor reaction SiC coating modified with silica for carbon/carbon composites [J]. J Cent South Univ T, 2010, 17(1): 1-6.
[6] Zhao Juan, Wang Gui, Guo Quan-gui, Liu Lang. Microstructure and property of SiC coating for carbon materials [J]. Fusion Engineering and Design, 2007, 82: 363-368.
[7] Cheng Lai-fei, Xu Yong-dong, Zhang Li-tong, Yin Xiao-wei. Preparation of an oxidation protection coating for C/C composites by low pressure chemical vapor deposition [J]. Carbon, 2000, 38: 1493-1498.
[8] Huang Jian-feng, Zeng Xie-rong, Li He-jun, Xiong Xin-bo, Fu Ye-wei. Influence of the preparation temperature on the phase, microstructure and anti-oxidation property of a SiC coating for C/C composites [J]. Carbon, 2004, 42: 1517-1521.
[9] Jacobson N S, Roth D J, Rauser R W, Cawley J D, Curry D M. Oxidation through coating cracks of SiC-protected carbon/carbon [J]. Surface & Coatings Technology, 2008, 203: 372-383.
[10] Li Shi-bin, Lu Zhen-lin, Gao Ji-qiang, Jin Zhi-hao. A study on the cycling oxidation behavior of mullite-coated silicon carbide [J]. Materials Chemistry and Physics, 2003, 78: 655-659.
[11] Auger M L, Sarin V K. A kinetic investigation of CVD mullite coatings on Si-based ceramics [J]. International Journal of Refractory Metals & Hard Materials, 2001, 19: 479-494.
[12] Hamidouche M, Bouaouadja N, Olagnon C, Fantozzi G. Thermal shock behaviour of mullite ceramic [J]. Ceramics International, 2003, 29: 599-609.
[13] Schneider H, Schreuer J, Hildmann B. Structure and properties of mullite―A review [J]. Journal of the European Ceramic Society,2008, 28: 329-344.
[14] Huang Jian-feng, Zeng Xie-rong, Li He-jun, Xiong Xin-bo, Huang Min. Mullite-Al2O3-SiC oxidation protective coating for carbon/carbon composites [J]. Carbon, 2003, 41: 2825-2829.
[15] Huang Jian-feng, Zeng Xie-rong, Li He-jun, Xiong Xin-bo, Huang Min. Al2O3-mullite-SiC-Al4SiC4 multi-composition coating for carbon/carbon composites [J]. Materials Letters, 2004, 58: 2627-2630.
[16] Huang Jian-feng, Zeng Xie-rong, Li He-jun, Li Ke-zhi, Xiong Xin-bo. Oxidation behavior of SiC-Al2O3-mullite multi-coating coated carbon/carbon composites at high temperature [J]. Carbon, 2005, 43: 1580-1583.
[17] Huang Jian-feng, Li He-jun, Zeng Xie-rong, Xiong Xin-bo, Li Ke-zhi. Influence of preparation technology on the microstructure and anti-oxidation property of SiC-Al2O3-mullite multi-coatings for carbon/carbon composites [J]. Applied Surface Science, 2006, 252: 4244-4249.
[18] Li He-jun, Jiao Geng-sheng, Li Ke-zhi, Wang Chuang. Multilayer oxidation resistant coating for SiC coated carbon/carbon composites at high temperature [J]. Materials Science and Engineering A, 2008, 475: 279-284.
[19] Yang Xin, Huang Qi-zhong, Zou Yan-hong, Chang Xin, Su Zhe-an, Zhang Ming-yu, Xie Zhi-yong. Anti-oxidation behavior of chemical vapor reaction SiC coatings on different carbon materials at high temperatures [J]. Transactions of Nonferrous Metals Sociecty of China, 2009, 19(5): 1044-1050.
[20] Fu Qian-gang, Li He-jun, Shi Xiao-hong, Li Ke-zhi, Wang Chuang, Huang Min. Double-layer oxidation protective SiC/glass coatings for carbon/carbon composites [J]. Surface & Coatings Technology, 2006, 200: 3473-3477.
[21] Kowbel W, Withers J C, Ransone P O. CVD and CVR silicon-based functionally gradient coatings on C-C composites [J]. Carbon, 1995, 33: 415-426.
[22] LIU Xing-fang, HUANG Qi-zhong, SU Zhe-an, JIANG Jian-xian. Preparation of SiC coating by chemical vapor reaction [J]. Journal of the Chinese Ceramic Society, 2004, 32, 906. (in Chinese)
[23] YANG Xin, HUANG Qi-zhong, ZOU Yan-hong, SU Zhe-an, XIE Zhi-yong, ZHANG Ming-yu, CHANG Xin. Analysis on the structure variation of SiC coatings on different carbon materials prepared with chemical vapor reaction [J]. Acta Chimica Sinica, 2008, 66, 2742. (in Chinese).
杨 鑫1, 2,苏哲安1,黄启忠1,柴立元2
1. 中南大学 粉末冶金国家重点实验室,长沙 410083;
2. 中南大学 冶金科学与工程学院,长沙 410083
摘 要:采用化学气相反应及料浆刷涂烧结复合工艺在石墨表面制备高温抗氧化莫来石/SiC复合涂层。XRD物相分析结果显示涂层外层由莫来石及微量SiO2相组成,涂层内层主要由β-SiC相组成。通过高温抗氧化试验研究涂层的高温抗氧化行为并测试涂层氧化后的洛氏硬度。结果表明:所制备的莫来石/SiC复合涂层具有良好的高温抗氧化及热震性能,经过1150 °C、109 h的高温氧化及12次1150 °C→室温的循环热震试验后,涂层试样的质量增加率为0.085%。硬度测试结果表明:所制备的莫来石/SiC复合涂层各层之间具有良好的结合性能。
关键词:炭材料;抗氧化;涂层;SiC;莫来石
(Edited by LI Xiang-qun)
Foundation item: Project (2012M511752) supported by China Postdoctoral Science Foundation; Project (2011CB605801) supported by the National Basical Research Program of China; Project (2012QNZT004) supported by the Fundamental Research Funds of the Central Universities, China; Project supported by the Freedom Explore Program of Central South University, China; Project (CSUZC2012026) supported by the Open-End Fund for the Valuable and Precision Instruments of Central South University, China; Project supported by the Postdoctoral Science Foundation of Central South University, China
Corresponding author: HUANG Qi-zhong; Tel: +86-731-88836078; Fax: +86-731-88836081; E-mail: qzhuang@csu.edu.cn
DOI: 10.1016/S1003-6326(11)61562-8

