
Microstructure and properties of oxalate conversion coating on AZ91D magnesium alloy
JIANG Yong-feng(蒋永锋), ZHOU Hai-tao(周海涛), ZENG Su-min(曾苏民)
School of Materials Science and Engineering, Central South University, Changsha 410083, China
Received 10 August 2009; accepted 15 September 2009
Abstract: The oxalate coating formed on AZ91D magnesium alloy by chemical conversion treatment methods in oxalate salt solutions was investigated. The surface morphologies and chemical composition of coating were examined using scanning electron microscopy (SEM) equipped with energy dispersive analysis of X-ray (EDX). Electrochemical impedance spectroscopy (EIS), potentiodynamic polarization curves and salt spray tests were employed to evaluate corrosion protection of the coating to substrate in 5% NaCl solution. The mechanism of coating formations was also considered in details. The results indicate that a compact and dense surface morphology with fine particle clusters of the oxalate coating on magnesium alloy is presented, which mainly consists of oxide or/and organic of Mg, Al and Zn. And the anti-corrosion of the magnesium after oxalate conversion treatment is better than that of the magnesium substrate. The results of salt spray test for oxalate coating is evaluated as Grade 9 according to ASTM B117. The electric resistance of oxalate chemical conversion coating to substrate is below 0.1 Ω.
Key words: magnesium alloy; oxalate conversion coating; microstructure; corrosion
1 Introduction
Magnesium and magnesium alloys exhibit an attractive combination of low density and high specific strength, associated with good damping and electromagnetic shielding capacity, making them ideal candidates for light-weight engineering applications, especially in communication, computer and consumption (3C) products[1-3]. However, magnesium alloys suffer dissolved contaminants from solid, liquid and gas in a moist exposed atmosphere to form electrolyte, resulting in the risk of corrosion and cracking due to electrochemical reactions, which is a limitation in their application for 3C structural parts.
Surface modification treatment is one of methods to alter the corrosion resistance of magnesium alloy to a significant extent in applications. Among a variety of surface treatments, chemical conversion treatment, in which (di)chromate[1-3], permanganate[1-2], phosphate [4-8], cerium[9-11], phytic[12-13], stannate[14-16], vanadium[17], fluoride[18], organic baths[19-20] and molybdate/phosphate[21] are generally used, is commonly applied to magnesium alloys in 3C products for a paint base to improve the adhesion and protective qualities of organic coatings or for stand-alone to prevent corrosion during shipment and storage, and in a certain situation, to provide long-term stabilization of machined surfaces[2]. The most successful conversion treatment is based on the use of chromates and has been widely investigated in the past[2]. Such processes vary from the widely used chromate/manganese dip treatment to anodic treatment in fluoride-containing bath[2-3], which can give rise to contamination by hexavalent ion of chromium and is strictly controlled due to recent environmental legislation in worldwide application. Therefore, new systems, showing environmental friendliness, must be studied and developed. Among the possible alternatives, several formulations are proposed as aforementioned. However, these processes are not only unstable for storage but also difficult to replenish.
Therefore, many attempts to find environmentally acceptable alternatives with better performance in surface modification processes have led to the identification of oxalate salt as chemical conversion coating on magnesium alloys. These precipitates, in turn,give rise to a blocking effect and reduce the rate of the reduction reaction of magnesium alloys, associated with good adhesion and good conductivity to substrate, which is of great importance for application of magnesium alloys in 3C parts to release electrostatic charges. Therefore, microstructure and properties of the oxalate chemical conversion coating on AZ91D magnesium alloy are investigated, and the formation mechanism of coating is also considered.
2 Experimental
The nominal composition of the AZ91D used in the present study is Mg-(8.8%-9.5%)Al-(0.45%-0.90%)Zn- (0.17%-0.40%)Mn-(0-0.05%)Si-(0-0.004%)Fe (mass fraction), which was provided by Pou Yuen Technology Co., Ltd, China. The substrates were cut from die-cast alloy parts. The substrate surfaces were successively treated by following processes in oxalate conversion treatment procedure: alkaline degreasing, oxalate chemical conversion immersion, conductive treatment, adjusting treatment and hot drying. The procedures of conductive treatment and adjusting treatment were aimed to improve the conductivity and to neutralize alkalinity of the coatings, respectively. The compositions and operating conditions of baths are presented in Table 1. And the operation conditions for different procedures of specimens are listed in Table 2.
Table 1 Compositions and operating conditions of baths

Table 2 Operation conditions of different processes of specimens

The corrosion resistance of conversion coatings is evaluated with salt spray tests according to ASTM B117. Open circuit potentials (OCP), potentiodynamic polarization and electrochemical impedance spectroscopy (EIS) of the magnesium alloys were measured by an electrometer coupled with an amplifier in which the specimens acted as working electrode using a PS-268A electrochemical measurement system. A saturated calomel electrode which acted as reference electrode and a platinum counter electrode were also included in the electrochemical cell. A 5% NaCl solution was used and the testing was carried out at ambient temperature to obtain polarized data.
The surface morphologies and chemical composition of coating were examined by FEI-Quanta 200 (PHILIPS) scanning electron microscope (SEM) equipped with energy dispersive analysis of X-rays (EDX). The electrical conductivity was also determined with a DF2823 mini-Ohm meter. The adhesion to substrate of coating on magnesium alloy was evaluated using a reciprocal wiping test by alcohol-cotton under a load of 50 N until appearance of dirty in cotton, where 50 cycles at least are the desire of applications.
3 Results and discussion
3.1 Morphologies and compositions
It is shown in Fig.1(a) that a compact and dense surface morphology with fine particles cluster of the oxalate coating is presented on magnesium alloy, whereas cerium conversion treatments appear to consist of a thin and cracked coating with ‘dry-mud’ morphology[11] and stannate coating has some areas that are not covered with hemisphere particles[15]. It is apparent that the particles of the coatings are homogeneously distributed on the substrate surface. By comparing Fig.1(a) with Fig.1(b), it is indicated that the particle size tends to decrease with the chemical conversion treatment time from 120 s to 240 s, implying that the immersion in conversion bath for 240 s is better than that for 120 s to specimen with combination with immersion in adjusting bath for 120 s. Therefore, the best surface morphology is presented in Fig.1(b).
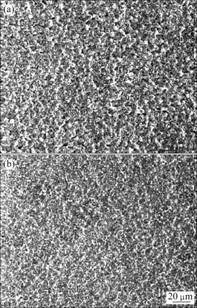
Fig.1 Surface morphologies of oxalate conversion coating on magnesium alloy: (a) Specimen No.3; (b) Specimen No.5
Figs.2(a) and 2(b) show EDX spectra of oxalate conversion coating on magnesium alloy in conversion bath for 120 s and conductive bath for 240 s (all with conductive treatment time of 120 s), respectively. As can be seen, compound layer mainly consists of oxide or/and organic of Mg, Al and Zn elements, which is in concordance with the results of oxalate chemical conversion treatment. Moreover, it is worth mentioning that these EDX spectra also come from carbon and nitrogen, and there is very small amount (negligible) of these elements. It is clear that the layer contains 49% Mg, 4% Al, 1% Zn and 36% oxygen (molar fraction) in average, which is not consistent with composition of matrix for Mg, Al and Zn. In addition, it can be seen that much magnesium element is dissolved to electrolyte during chemical conversion process, which is demonstrated by XRD pattern that the substrate near interface between coating and substrate consists mainly of Mg17Al12 and Mg0.97Zn. It should be noted that Mg17Al12 is a main phase in the vicinity of interface for substrate, whereas a small amount of Mg is detected.
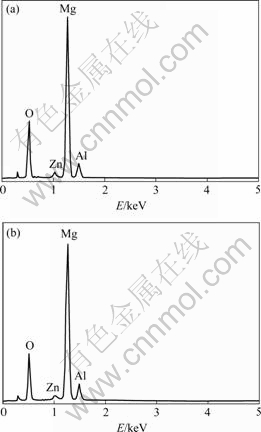
Fig.2 EDX spectra of oxalate conversion coating on magnesium alloys: (a) Specimen No.3; (b) Specimen No.5
3.2 Salt spray test
Fig.3(a) shows the morphology for sample after 24 h in salt spray test. It can be found that fewer corrosion products in salt spray test are presented as pitting corrosion, and can be classified as Grade 9 according to ASTM B117. In these corrosion processes, the particle size decreases while the pore size hardly changes, indicating that the matrix is wholly covered by the oxalate coating. The flakes stripping (Fig.3(b)) appears in partial specimen, which can give rise to pitting corrosion and show as white powder rust on magnesium alloy. The specimen obtained by repeated immersion shows a better corrosion resistance because an increase passive current is detected. Probably, the conversion coating obtained by repeated immersion enables to obtain a denser and less porous layer, and the passive layer becomes more compact during this step, which makes the subsequent depositions more efficient[4].

Fig.3 Morphologies of specimens after 24 h in salt spray test: (a) Specimen No.5; (b) Specimen No.3
3.3 Open circuit potential (OCP)
The superimposed curves of AZ91D alloy for 1 200 s on open circuit potential with treatment in different processes of degreasing, conversion treatment, conductive treatment and adjusting treatment with except of maintaining operation parameters of degreasing and adjusting treatment for 720 s at 70 ℃ and 900 s at 80 ℃, respectively, as a function of immersion time in 5% NaCl aqueous solution are shown in Fig.4. The OCP remains a constant at a value of -1 570 mV and -1 590 mV, respectively, for specimens No.1 and 2. However, during initial immersion, the OCP of specimens No.3-7 increases with time; and with time going on, the OCP tends to achieve a constant value. These illustrate that a variation of the procedure of conversion treatment can result in changes of the OCP, which is varied with increasing immersion time in conductive treatment bath. There is the formation of corrosion films distributed uniformly on specimens with careful visual examination, which reveals that no pitting corrosion occurs.
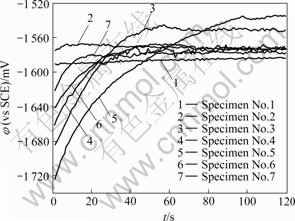
Fig.4 Variation of open circuit potential of AZ91D magnesium alloy with different procedures after immersion in 5% NaCl solution
3.4 Potentiodynamic curves
The corrosion resistance of specimens can be given on the basis of the corrosion potential (φcorr), the corrosion current density (Jcorr), and the anodic/cathodic Tafel slopes, which are derived from the measured polarization curves.
The polarization of magnesium alloy substrate is measured by PS268A electrochemical measurement system. It is clear shown in Fig.5 that corrosion potential of substrate is -1 581.3 mV, and the corrosion current density is 182.99 μA/cm2. The anode polarization curve is relatively smooth, but the cathodic polarization curve is relatively sharp, indicating that the substrate is susceptible to corrosion. Fig.5 also shows the polarization curves of magnesium alloy treated with different processing parameters. As can be seen, the corrosion potential of oxalate coated magnesium alloy is -1 471.0 mV, and the corrosion current density is 30.43 μA/cm2 after immersion in conversion bath for 240 s and in conductive bath for 120 s (Specimen No.5). These results are in agreement with the morphologies of the coatings formed on the surface of the magnesium alloy. The similar results can be concluded with other processing parameters (Specimens No.4, 6 and 7). Therefore, the specimen obtained by repeated conversion immersion shows a better corrosion resistance because an increased passive current is detected, although the φcorr is approximately the same for the specimens No.2 or 3. Probably, the conversion coating obtained by repeated conversion immersion enables to obtain a denser and less porous layer. The passive layer becomes more compact during the step of conversion treatment and this makes the subsequent depositions more efficient[4-8]. This difference may be attributed to the effects of Al on the reacting products, such as MgAl2O4 and Al2O3, in conversion-coated layer, which influence the corrosion potential of the conversion-coated layer. The phosphate conversion treatment indeed offers an equivalent corrosion resistance to the Cr6+-based conversion treatment for Mg-Al-Zn alloy[4-6]. The porosity of the coating decreases as the phosphating time increases up to 10 min[7-8].
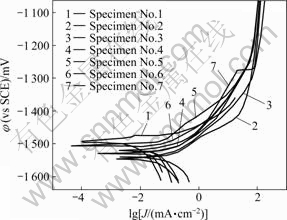
Fig.5 Polarization curves of magnesium alloy with different procedures after immersion in 5% NaCl solutions
3.5 Electrochemical impendence spectroscopy
The electrochemical impendence spectra of AZ91D with different processes of chemical conversion of degreasing, conversion treatment, conductive treatment, adjusting treatment in addition to retaining degreasing and adjusting treatment for 720 s at 70 ℃ and 900 s at 80 ℃, respectively, are similar except for the difference in diameters for loop, as shown in Fig.6. This possibly reveals that the corrosion mechanisms for specimens after conversion treatments are consistent and corrosion rates tend to be different. Due to small dimension of curves in EIS plots for the magnesium alloy, the Nyquist plots, which exhibit two loops, are difficult to discern in comparison with those of the coatings in different treating conditions. This perhaps illustrates, as previous results indicates, that a porous layer of corrosion products covers the surface. Sublayer is a thin insulating film with some small free areas. All the capacity values of specimens in EIS plots imply the formation of a thick and protective film on the substrate, which is in agreement with the results of salt spray test.

Fig.6 EIS plots of AZ91D with different procedures after immersion in 5% NaCl solution
3.6 Electric resistance
The electric resistance of coatings to substrate was determined using a DF2823 mini-ohm meter. It is demonstrated that the electric resistance is below 0.1 Ωto some extent, which can attain the standard about the structural parts of portable computer.
3.7 Adhesion
The adhesion to substrate of coating on magnesium alloy was evaluated by reciprocally wiping by alcohol- cotton under a load of 50 N until appearance of dirty. There are above 50 cycles for reciprocal wiping by alcohol-cotton without change of color or appearance of adhesive ash, indicating that it can satisfy the requirements for the adhesion of coating.
3.8 Mechanisms of formation
The reactions (1-6) occur when the formation of conversion coatings on AZ91D magnesium alloy with the immersion of specimens is carried out in the solution containing oxalate and hexamethylenetetramine:
Mg-2e→Mg2+ (1)
Al-3e→Al3+ (2)
C2H2O2+Mg2+→Mg(C2O2)+H2↑ (3)
3(C2O2)2-+2Al3+→Al2(C2O2)3 (4)
C6H12N4+Mg2+→Mg6(C6H12N4) (5)
C6H12N4+Al3+→Al4(C6H12N4) (6)
This may be a formation by mutual promotion between oxalate or hexamethylenetetramine and substrate, resulting in nucleation and growth of particles for compounds on substrate surface as shown in Fig.1. It is used as inhibitor in surface treatment for the hexamethylenetetramine in space structure which is similar to a cage. When the conversion coating combines with oxalate salt, it will hold back corrosion ions such as chloride ions to attack.
Following the conversion coating treatment, the processing of conductive treatment is continued. The reactions in this step occur in the solution containing organic acid such as tartaric acid and citrate acid in the pH range of 2.0-2.5. The reactions are carried out between acid and substrate, at the same time, acid and acid also mutual polymerize in previous treatments, which results in the formation of electron conductive polymer, due to organic acid containing carboxyl group, hydroxyl group and metal ions. The typical formation reactions of electron conductive polymers are presented as follows (reactions 7-9):
HOOC―COH―CHOH―COOH+Mg→
C4H2O6+H2↑ (7)
HOOC―CH2―COH(COOH)―CH2―COOH+Mg→
C6H6O7+H2↑ (8)
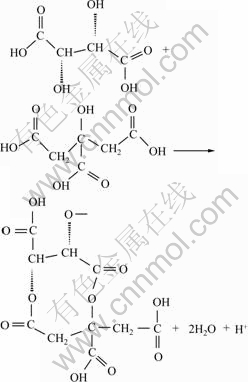 (9)
(9)
Previous studies[22-23] revealed that a variation of H+ can easily switch between conducting and insulating states of electron accompanied with the change of other properties. There is a unique property in comparison with the majority of electron conducting materials. Therefore, following this operation, the adjusting treatment is carried out in alkaline solution for neutralization of surface on magnesium alloy substrate in order to improve adhesion and protective properties of coatings.
Fig.7 shows that current density is closely linearly proportional to potential of electrode containing organic acid such as tartaric acid and citrate acid, which indicates a carrier generation mechanism dependent of potential, implying that electrostatic charge can be released when potential of surface attains a certain value. This further demonstrates the mechanism of conductive treatment.
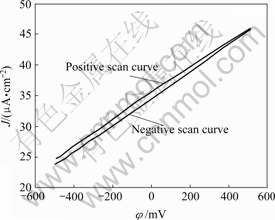
Fig.7 Dependence of current density on potential of electrodes in conductive treatment bath containing tartaric acid and citrate acid
Therefore, the increase of thickness for the oxalate coatings with chemical conversion time continues to some extent. The formation of hydroxides is replenished and reciprocates another circle of reactions.
4 Conclusions
1) A compact and dense surface morphology of oxalate coating with fine particle clusters is presented on magnesium alloy.
2) The polarization curves reveal that the corrosion resistance of the magnesium after oxalate conversion treatment is better than that of the magnesium substrate.
3) The electric resistance of oxalate conversion coating to substrate is below 0.1 Ω.
4) The appearance of dirty ash in alcohol-cotton is above 50 cycles in reciprocally wiping on the oxalate chemical conversion coating.
5) The current density is closely linearly proportional to potential of electrode containing organic acid such as tartaric acid and citrate acid.
References
[1] CHEN Zhen-hua. Magnesium alloys [M]. Beijing: Chemical Industry Press, 2004: 385. (in Chinese)
[2] AVEDESIAN M M, BAKER H S. Magnesium and magnesium alloys [M]. New York: ASM International, 1999: 194.
[3] GRAY J E, LUAN B. Protective coating on magnesium and its alloys―A critical review [J]. Journal of Alloys and Compounds, 2002, 336: 88-113.
[4] ZHOU Wan-qiu, SHAN Da-yong, HAN En-hou, KE Wei. Structure and formation mechanism of phosphate conversion coating on die-cast AZ91D magnesium alloy [J]. Corrosion Science, 2008, 50: 329-337.
[5] ZHANG Hua, YAO Guang-chun, WANG Shu-lan, LIU Yi-han, LUO Hong-jie. A chrome-free conversion coating for magnesium-lithium alloy by a phosphate-permanganate solution [J]. Surface & Coatings Technology,2008, 202: 1825-1830.
[6] LIU L Y, JIANG Z H, LI G Y. A study and application of zinc phosphate coating on AZ91D magnesium alloy [J]. Surface & Coating Technology, 2006, 200: 3021-3026.
[7] KOUISNI L, AZZI M, DALARD F. Phosphate coatings on magnesium alloy AM60 (Part 1): Study of the formation and the growth of zinc phosphate films [J]. Surface & Coating Technology, 2004, 185: 58-67.
[8] KOUISNI L, AZZI M, DALARD F. Phosphate coatings on magnesium alloy AM60 (Part 2): Electrochemical behaviour in borate buffer solution [J]. Surface & Coating Technology, 2005, 192: 239-246.
[9] DABALA M, BRUNELLIA K, NAPOLITANIB E, MAGRINIA M. Cerium-based chemical conversion coating on AZ63 magnesium alloy [J]. Surface & Coatings Technology, 2003, 172: 227-232.
[10] MONTEMOR M F, SIMOES A M, FERREIRA M G S, CARMEZIM M J. Composition and corrosion resistance of cerium conversion films on the AZ31 magnesium alloy and its relation to the salt anion [J]. Applied Surface Science, 2008, 254: 1806-1814.
[11] LI Ling-jie, LEI Jing-lei, YU Sheng-hai, TIAN Yu-jing, JIANG Qi-quan, PAN Fu-sheng. Formation and characterization of cerium conversion coatings on magnesium alloy [J]. Journal of Rare Earths, 2008, 26(3): 383-384.
[12] CUI Hu-fang, LI Qing-fen, LI Ying, WANG Fu-hui, JIN Guo, DING Ming-hui. Microstructure and corrosion resistance of phytic acid conversion coatings for magnesium alloy [J]. Applied Surface Science, 2008, 255: 2098-2103.
[13] GAO Li-li, ZHANG Chun-hong, ZHANG Mi-lin, HUANG Xiao-mei, JIANG Xi. Phytic acid conversion coating on Mg-Li alloy [J]. Journal of Alloys and Compounds, 2009, 485: 789-793.
[14] HANDY A S. The effect of surface modification and stannate concentration on the corrosion protection performance of magnesium alloys [J]. Surface & Coatings Technology, 2008, 203: 240-249.
[15] ELSENTRIECY H H, AZUMI K, KONNO H. Improvement in stannate chemical conversion coatings on AZ91D magnesium alloy using the potentiostatic technique [J]. Electrochimica Acta,2007, 53: 1006-1012.
[16] ZUCCHI F, FRIGNANI A, GRASSI V, TRABANELLI G, MONTICELLI C. Stannate and permanganate conversion coatings on AZ31 magnesium alloy [J]. Corrosion Science,2007, 49: 4542-4552.
[17] YANG K H, GER M D, HWU W H, SUNG Y, LIU Y C. Study of vanadium-based chemical conversion coating on the corrosion resistance of magnesium alloy [J]. Materials Chemistry and Physics,2007, 101: 480-485.
[18] LI Jian-zhong, HUANG Jiu-gui, TIAN Yan-wen, LIU Chang-sheng. Corrosion action and passivation mechanism of magnesium alloy in fluoride solution [J]. Trans Nonferrous Met Soc China, 2009, 19(1): 50-54.
[19] ZHAO Ming, WU Shu-sen, AN Ping, FUKUDA Y, NAKAE H. Growth of multi-elements complex coating on AZ91D magnesium alloy through conversion treatment [J]. Journal of Alloys and Compounds,2007, 427: 310-315.
[20] CHEN Xiao-ming, LI Guang-yu, LIAN Jian-she, JIANG Qing. An organic chromium-free conversion coating on AZ91D magnesium alloy [J]. Applied Surface Science, 2008, 255: 2322-2328.
[21] YONG Zhi-yi, ZHU Jin, QIU Cheng, LIU Ya-li. Molybdate/ phosphate composite conversion coating on magnesium alloy surface for corrosion protection [J]. Applied Surface Science, 2008, 255: 1672-1680.
[22] INZELT G, PINERI M, SCHULTZE J W, VOROTYNTSEV M A. Electron and proton conducting polymers: Recent developments and prospects [J]. Electrochimica Acta, 2000, 45: 2403-2421.
[23] MOSESA D, OKUMOTOA H, COMORETTO D, LEEA C H, HEEGE A J, OHNISHIB T, NOGUCHIB T. Mechanism of carrier generation in conducting polymers [J]. Synthetic Metals, 1997, 84: 539-544.
Foundation item: Project(20070420821) supported by the China Postdoctoral Science Foundation; Project supported by the Special Project for Postdoctoral Scientific Research of Hunan Provincial Scientific and Technology Department of China; Project supported by the Postdoctoral Foundation of Central South University, China
Corresponding author: JIANG Yong-feng; Tel: +86-13813557936; E-mail address: sjtujyf001@sohu.com
DOI: 10.1016/S1003-6326(09)60043-1
(Edited by YANG Bing)