


Trans. Nonferrous Met. Soc. China 22(2012) 129-133
Effects of synthesis temperature and raw materials composition on preparation of ��-Sialon based composites from fly ash
MA Bei-yue, LI Ying, YAN Chen, DING Yu-shi
School of Materials and Metallurgy, Northeastern University, Shenyang 110819, China
Received 30 October 2010; accepted 27 May 2011
Abstract: ��-Sialon based composites were successfully prepared from fly ash and carbon black under nitrogen atmosphere by carbothermal reduction-nitridation process. Effects of heating temperature and raw materials composition on synthesis process were investigated, and the formation process of the composites was also discussed. The phase composition and microstructure of the composites were characterized by X-ray diffraction and scanning electronic microscopy. The results show that increasing heating temperature or mass ratio of carbon black to fly ash can promote the formation of ��-Sialon. The ��-Sialon based composites can be synthesized at 1723 K for 6 h while heating the sample with mass ratio of carbon black to fly ash of 0.56. The as-received ��-Sialon in the composites exists as granular with an average particle size of 2-3 ��m. The preparation process of ��-Sialon based composites includes the formation of O��-Sialon, X-Sialon and ��-Sialon as well as the conversion processes of O��-Sialon and X-Sialon to ��-Sialon.
Key words: Sialon; composites; carbothermal reduction-nitridation process; fly ash; synthesis temperature; raw materials composition
1 Introduction
Sialon includes ��-Sialon (Si6-zAlzOzN8-z, 02-xAlxO1+xN2-x, 0xSi12-(m+n)Alm+nOnN16-n, 0 pyrophyllite [8], bauxite [9], zeolite [10], coal gangue [11, 12] and rice husks [13].
Fly ash emerges as a by-product from the combustion of raw coal in thermal power plants. Until 2020, the present accumulating amount of fly ash in China is approximately three billion tons. The mass waste causes serious environmental problems. Thus, the effective and comprehensive utilization of waste fly ash can provide an important route to solve these problems. Meanwhile, the high value-added products with low cost such as mullite and mullite-zirconia-corundum composites can be produced using fly ash [14, 15].
In the present work, the ��-Sialon based composites were synthesized by carbothermal reduction-nitridation process, with fly ash and carbon black as raw materials. The effects of heating temperature and raw materials composition on synthesis process were investigated, and the formation process of the composites was also discussed.
2 Experimental
2.1 Raw materials
Fly ash (mesh size��74 ��m) and carbon black (mesh size��30 ��m) were used as raw materials, and the chemical composition of fly ash (mass fraction) was Al2O3 41.20%, SiO2 48.49%, Fe2O3 3.37%, CaO 3.31%, TiO2 1.30% and MgO 0.20%. In Fig. 1 the crystalline phases of fly ash mainly include mullite as well as small amounts of corundum (Al2O3) and quartz (SiO2). In addition, the mass fraction of C in carbon black and the volume fraction of nitrogen gas (N2) were 98.0% and 99.99%, respectively.
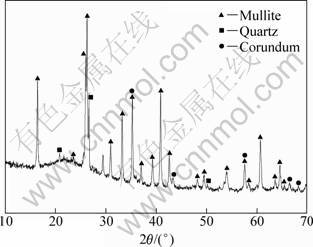
Fig. 1 XRD pattern of fly ash
2.2 Preparation of samples
The overall chemical reaction equation for synthesizing ��-Sialon from fly ash by carbothermal reduction-nitridation process can be expressed as
Al6Si2O13(s)+4SiO2(s)+15C(s)+5N2(g)=
2Si3Al3O3N5(s)+15CO(g) (1)
According to reaction (1), the mass ratio of carbon black to fly ash (mc/mf) was 0.25. Fly ash was mixed with carbon black in mc/mf of 0.28, 0.30, 0.42 and 0.56. This over-stoichiometry of carbon black was necessary to promote the carbothermal reduction-nitridation reaction. Mixing was performed in a ball mill with anhydrous ethanol for 12 h, and then the mixed powders were pressed to form the samples with 20 mm in diameter and 10 mm in thickness under a pressure of 20 MPa. Then the formed samples were dried fully at 393 K and put into a graphite crucible. The crucible was placed in an atmosphere-controlled tubular furnace and then heated up to 1673 K and 1723 K for 6 h, respectively. During the synthesis process, the N2 flow remained at 1.0 L/min. After the predetermined temperature and time reached, the system was cooled to room temperature.
2.3 Characterization of samples
The samples with different mc/mf values synthesized at 1673 and 1723 K were oxidized in air at 973 K for 2 h to remove residual carbon. The phase composition and microstructure of the products were characterized by X-ray diffractometer (XRD, Cu K�� radiation, 30 kV and 30 mA), scanning electronic microscopy (SEM) and energy dispersive spectrum (EDS).
3 Results and discussion
3.1 Phase composition and microstructure
Figure 2 shows the XRD patterns of the samples with different mc/mf values synthesized at 1673 K for 6 h. With increasing mc/mf from 0.28 to 0.30 (Figs. 2(a) and (b)), the diffraction intensities of mullite and O��-Sialon phases weaken gradually, and the diffraction intensities of ��-Sialon and corundum phase strengthen. This indicates that increasing carbon content in a sample can promote the carbothermal reduction-nitridation reaction. The samples with mc/mf of 0.28 and 0.30 all consist of mullite and O��-Sialon. In Fig. 2(c), ��-Sialon can be detected in the sample with mc/mf of 0.42, and the sample is composed of mullite, O��-Sialon and ��-Sialon. In Fig. 2(d), corundum can be detected in the higher carbon content sample (mc/mf=0.56), and the sample mainly includes mullite, corundum, O��-Sialon and ��-Sialon.
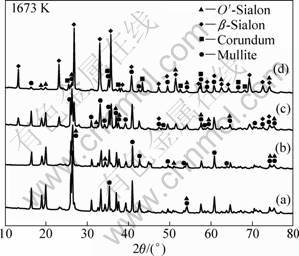
Fig. 2 XRD patterns of samples with different mc/mf values synthesized at 1673 K for 6 h: (a) 0.28; (b) 0.30; (c) 0.42; (d) 0.56
Figure 3 shows the XRD patterns of the samples with different mc/mf values synthesized at 1723 K for 6 h. With increasing mc/mf from 0.28 to 0.56, the diffraction intensities of mullite and X-Sialon phases weaken gradually, and the diffraction intensities of ��-Sialon and corundum phase strengthen. In Figs. 3(a) and (b) the samples with mc/mf of 0.28 and 0.30 all consist of mullite, corundum and X-Sialon. ��-Sialon can be detected in the sample with mc/mf of 0.42 (Fig. 3(c)), and the mullite phase vanishes completely, showing that the sample is composed of X-Sialon, ��-Sialon and corundum. In Fig. 3(d) the diffraction intensity of X-Sialon in the sample with mc/mf of 0.56 weakens remarkably, and the main crystalline phase is ��-Sialon. Thus, increasing carbon content in a sample can promote the decomposition of mullite and the formation of ��-Sialon.
Figures 4(a) and (b) show the SEM images of the samples with mc/mf of 0.56 synthesized at 1673 K and 1723 K for 6 h, respectively. The particles in the composites exist as granular. The average particle size synthesized at 1673 K and 1723 K reaches 1-2 ��m (Fig. 4(a)) and 2-3 ��m (Fig. 4(b)), respectively. EDS analysis (Fig. 4(c)) indicates that the particle (point 1) as shown in Fig. 4(b) is composed of Si, Al, O and N elements. By combing with XRD pattern (Fig. 3(d)), point 1 is ��-Sialon.
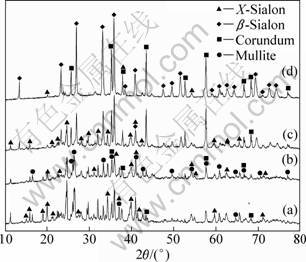
Fig. 3 XRD patterns of samples with different mc/mf values synthesized at 1723 K for 6 h: (a) 0.28; (b) 0.30; (c) 0.42; (d) 0.56

Fig. 4 SEM images of samples with mc/mf of 0.56 synthesized at 1673 K (a) and 1723 K (b), and EDS pattern of point 1 in Fig. 4(b)
3.2 Analysis of formation process
The carbothermal reduction-nitridation reaction process for synthesizing ��-Sialon based composites from fly ash is very complex and involves many chemical reactions. During the synthesis process, CO, SiO, Si2N2O, Si3N4 and AlN are regarded as important intermediate products. With changing heating temperature and raw materials composition, O��-Sialon, X-Sialon and ��-Sialon may be formed.
SiO2(s)+C(s)=SiO(g)+CO(g) (2)
2SiO(g)+C(s)+N2(g)=Si2N2O(s)+CO(g) (3)
2SiO2(s)+3C(s)+N2(g)=Si2N2O(s)+3CO(g) (4)
3SiO(g)+3C(s)+2N2(g)=Si3N4(s)+3CO(g) (5)
3SiO2(s)+6C(s)+2N2(g)=Si3N4(s)+6CO(g) (6)
3Si2N2O(s)+3C(s)+N2(g)=2Si3N4(s)+3CO(g) (7)
Al2O3(s)+3C(s)+N2(g)=2AlN(s)+3CO(g) (8)
1.83Si2N2O(s)+0.17Al2O3(s)=2Si1.83Al0.17O1.17N1.83(s)(9)
Al2O3(s)+Si3N4(s)+AlN(s)=Si3Al3O3N5(s) (10)
1/2Al6Si2O13(s)+2SiO2(s)+15/2C(s)+5/2N2(g)=
Si3Al3O3N5(s)+15/2CO(g) (11)
0.17/6Al6Si2O13(s)+5.32/3SiO2(s)+5.49/2C(s)+
1.83/2N2(g)=Si1.83Al0.17O1.17N1.83(s)+5.49/2CO(g)(12)
Al6Si2O13(s)+SiO2(s)+3C(s)+N2(g)=
Si3Al6O12N2(s)+3CO(g) (13)
The Gibbs free energies of formation of Al6Si2O13, SiO2, Al2O3, CO, O��-Sialon and ��-Sialon are listed in Table 1 [16, 17]. When the partial pressure of N2 is equal to pQ(0.1 MPa), the Gibbs free energies for reactions (11) and (12) as well as the relationship between partial pressure of CO gas (pCO)and temperature can be further obtained. It is noted that the Gibbs formation free energy of O��-Sialon (x=0.17) is lack, so it is approximately equal to that of O��-Sialon (x=0.2) during the process of calculating relational thermodynamics data.
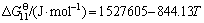 ,
,
lg(pCO/pQ)=5.88-10637.65/T (14)
 ,
,
lg(pCO/pQ)=5.59-6446.46/T (15)
Table 1 Gibbs free energies of formation of some compounds in Al2O3-SiO2-C-N2 system
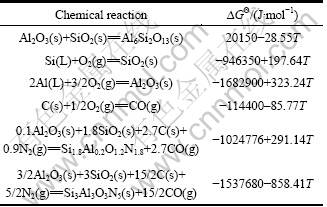
Figure 5 shows the predomination region diagram for Al2O3-SiO2-C-N2 system plotted by the thermodynamic data as shown in Eqs. (14) and (15). It indicates that when the partial pressure of CO gas (pCO) in the reacting furnace remains constant, with increasing heating temperature, C in a sample can react with SiO2, Al2O3 and N2 to form O��-Sialon (reactions (2)-(4), (9), (12)) and further produce ��-Sialon (reactions (2), (5)-(8), (10), (11)). The predomination regions change from Al2O3(s) + SiO2(s) + C(s) to Al2O3(s) + O��-Sialon(s) + C(s) and Al2O3(s) + ��-Sialon(s) + C(s), respectively. When the temperature remains constant, with decreasing pCO, the predomination regions change from Al2O3(s) + SiO2(s) + C(s) to Al2O3(s) + O��-Sialon(s) + C(s) and Al2O3(s) + ��-Sialon(s) + C(s), respectively. Thus, proper partial pressure of CO gas and temperature are key factors for the synthesis of ��-Sialon based composites.
During the carbothermal reduction-nitridation reaction process, X-Sialon can be formed (reaction (13)). With increasing synthesis temperature and carbon content, X-Sialon and O��-Sialon can be nitridized to produce ��-Sialon (reactions (16) and (17)). This can be confirmed by the XRD patterns as shown in Figs. 2 and 3.
Si3Al6O12N2(s)+3SiO2(s)+12C(s)+4N2(g)=
2Si3Al3O3N5(s)+12CO(g) (16)
Si1.83Al0.17O1.17N1.83(s)+0.83Al2O3(s)+1.83C(s)+ 0.61N2(g)
=0.61Si3Al3O3N5(s)+1.83CO(g) (17)
Additionally, Al2O3 and SiO2 in fly ash can also react with C and N2 to form O��-Sialon, X-Sialon and ��-Sialon (reactions (18)-(20)):
0.17Al2O3(s)+3.66SiO2(s)+5.49C(s)+1.83N2(g)=
2Si1.83Al0.17O1.17N1.83(s)+5.49CO(g) (18)
3Al2O3(s)+3SiO2(s)+3C(s)+N2(g)=
Si3Al6O12N2(s)+3CO(g) (19)
3Al2O3(s)+6SiO2(s)+15C(s)+5N2(g)=
2Si3Al3O3N5(s)+15CO(g) (20)

Fig. 5 Predomination region diagram for Al2O3-SiO2-C-N2 system ( =0.1 MPa)
=0.1 MPa)
The formation process of ��-Sialon based composites can be summarized as follows.
1) In a sample with lower carbon content (mc/mf= 0.28-0.42), O��-Sialon and X-Sialon can be fabricated at 1673 K and 1723 K, respectively. And they can be further converted into ��-Sialon by increasing synthesis temperature and carbon content.
2) In a sample with higher carbon content (mc/mf= 0.56), ��-Sialon can be directly produced at 1673 K and 1723 K.
4 Conclusions
1) The ��-Sialon based composites can be successfully prepared from fly ash by carbothermal reduction-nitridation process, and the proper technological parameters are mc/mf of 0.56 and heating temperature of 1723 K.
2) The as-received ��-Sialon exists as granular, and the average particle size of ��-Sialon is 2-3 ��m.
3) The preparation processes of ��-Sialon based composites includes the formation of O��-Sialon, X-Sialon and ��-Sialon as well as the conversion process of O��-Sialon and X-Sialon to ��-Sialon.
References
[1] ESER O, KURAMA S, GUNKAYA G. The production of ��-Sialon ceramics with low amounts of additive at low sintering temperature [J]. J Eur Ceram Soc, 2010, 30(14): 2985-2990.
[2] MA Bei-yue, YU Jing-kun, LIU Tao, YAN Zheng-guo. Preparation of ��-sialon/ZrN bonded corundum composites from zircon by nitridation reaction sintering process [J]. J Cent South Univ Technol, 2009, 16(5): 725-729.
[3] QIU Xiong-er, PAN Wei, ZHOU Yi-chun, PENG Hu, LI Jun, TAN Cong-bin. Microwave Synthesis of ��-Sialon [J]. Rare Metal Materials and Engineering, 2007, 36(2): 354-358. (in Chinese)
[4] PRADEILLES N, RECORDM C, GRANIER D, MARIN-AYRAL R M. synthesis of ��-SiAlON: A combined method using sol�Cgel and SHS processes [J]. Ceram Int, 2008, 34(5): 1189-1194.
[5] SHAHIEN M, RADWAN M, KIRIHARA S, MIYAMOTO Y, SAKURAI T. Combustion synthesis of single-phase ��-sialons (z=2�C4) [J]. J Eur Ceram Soc, 2010, 30(9): 1925-1930.
[6] LI J H, MA H W, FANG Q F. Synthesis of prismatic ��-sialon from the precursor of SBA-15 incorporated with Al(NO3)3 via carbothermal reduction nitridation [J]. Ceram Int, 2008, 34(8): 1791-1795.
[7] XU L H, LIAN F, ZHANG H, BI Y B, CHENG K, QIAN Y B. Optimal design and preparation of beta-Sialon multiphase materials from natural clay [J]. Mater Design, 2006, 27(7): 595-600.
[8] YU Jing-kun, DAI Wen-bin, DAI Shu-ping. Synthesis of Sialon-SiC composites and its application [J]. Journal of Northeast University: Natural Science, 2003, 24(9): 824-827. (in Chinese)
[9] ZHANG H J, HAN B, LIU Z J. Preparation and oxidation of bauxite-based ��-Sialon bonded SiC composite[J]. Mater Res Bull, 2006, 41(9): 1681-1689.
[10] LI F J, WAKIHARA T, TATAMI J, KOMEYA K, MEGURO T. Synthesis of ��-Sialon powder by carbothermal reduction-nitridation of zeolites with different compositions [J]. J Eur Ceram Soc, 2007, 27(5): 2535-2540.
[11] HAO Hong-shun, XU Li-hua, ZHAI Wei, ZHANG Zuo-shun, ZHANG Xiao-meng, XIE Zhi-peng. Development of Sialon ecomaterials drived from solid waste of containing silicon and aluminum [J]. J Inorg Mater, 2010, 25(11): 1121-1127. (in Chinese)
[12] LUO X Y, SUN J L, DENG C J, HONG Y R. Synthesis of ��-Sialon from coal gangue [J]. J Mater Sci Technol, 2003, 19(1): 93-96.
[13] RAHMAN L A, SALEH M I. The formation of ��-Sialon in the carbothermal reduction of digested rice husks [J]. Mater Lett, 1995, 23(1-3): 157-160.
[14] DONG Y C, FENG X Y, FENGX F, DING Y W, LIU X Q, MENG G Y. Preparation of low-cost mullite ceramics from natural bauxite and industrial waste fly ash [J]. J Alloys Compd, 2008, 460(1-2): 599-606.
[15] MA Bei-yue, LI Ying, CUI Shao-gang, ZHAI Yu-chun. Preparation and sintering properties of zirconia-mullite-corundum composites using fly ash and zircon [J]. Transactions of Nonferrous Metals Society of China, 2010, 20(12): 2331-2335.
[16] LIANG Ying-jiao, CHE Yin-chang. Handbook of thermodynamic data in inorganic [M]. Shenyang: Northeastern University Press, 1993. (in Chinese)
[17] CHEN Zhao-you. Chemical thermodynamics of refractories[M]. Beijing: Metallurgical Industry Press, 2005. (in Chinese)
�ϳ��¶Ⱥ�������ɶԷ�ú���Ʊ���-Sialon�����ϲ��ϵ�Ӱ��
����Խ���� Ӣ���� ��������ʯ
������ѧ ������ұ��ѧԺ������ 110819
ժ Ҫ���Է�ú�Һ�̿��Ϊԭ�ϣ�����̼�Ȼ�ԭ�������ɹ��Ʊ�����-Sialon�����ϲ��ϡ��о��˼����¶Ⱥ�������ɶԺϳɹ��̵�Ӱ�죬�����˲��ϵ����ɹ��̡�����XRD��SEM�ֶα����˺ϳɲ��ϵ�����ɺ����ṹ������������������¶ȣ�����̿�����ú�ҵ������Ⱦ����Դٽ���-Sialon�����ɣ���̿�����ú��������Ϊ0.56������������1723 K������6 h�����Ժϳɦ�-Sialon�����ϲ��ϣ��ϳɲ����Ц�-Sialon������״��ʽ���ڣ�ƽ������Ϊ2~3 ��m����-Sialon�����ϲ��ϵ����ɹ��̰���O��-Sialon��X-Sialon�ͦ�-Sialon�����ɼ�O��-Sialon��X-Sialon���-Sialon��ת�����̡�
�ؼ��ʣ���¡�����ϲ��ϣ�̼�Ȼ�ԭ����������ú�ң��ϳ��¶ȣ��������
(Edited by LI Xiang-qun)
Foundation item: Project (51074038) supported by the National Natural Science Foundation of China; Project (N100302002) supported by the Fundamental Research Funds for the Central Universities, China
Corresponding author: LI Ying; Tel: +86-24-83688995; E-mail: liying@mail.neu.edu.cn; beiyue_ma@yahoo.com.cn
DOI: 10.1016/S1003-6326(11)61151-5