J. Cent. South Univ. Technol. (2008) 15: 606-611
DOI: 10.1007/s11771-008-0113-9
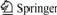
Separation of baicalin from Scutellaria Baicalensis Georgi with polyamide
CHI Ru-an(≥ÿ»Í∞≤)1, 2, ZHOU Fang(÷Ð ∑º)1, 2, HUANG Kun(ª∆ Á˚)1, ZHANG Yue-fei(’≈‘Ω∑«)1
(1. School of Chemical Engineering and Pharmacy, Wuhan Institute of Technology, Wuhan 430073, China;
2. Key Laboratory for Green Chemical Process of Ministry of Education, Wuhan Institute of Technology,
Wuhan 430073, China)
Abstract: Separation of baicalin from Scutellaria Baicalensis Georgi with polyamide was studied. The adsorption isotherm, kinetic equation and desorption law were investigated by static and dynamic adsorption methods. The results show that the kinetic behavior is mainly controlled by the liquid film diffusion process and obeys the Boyd film diffusion equation. Equilibrium data for the adsorption of baicalin are correlated with Freundlich isotherm equation, i.e. q=3.8ce2.057, suggesting that the relative capacity of polyamide to baicalin is somewhat small. The desorption results indicate that the baicalin with mass fraction of 33.86% and the least impurities can be obtained by chromatography using 60% ethanol as the eluant at room temperature.
Key words: baicalin; Scutellaria Baicalensis Georgi; adsorption; separation
1 Introduction
Scutellaria Baicalensis Georgi is a traditional Chinese herbal medicine, whose biological activity has been well known, for example, anti-inflammation, anti-cancer, treating bacterial and viral infections of the respiratory and the gastrointestinal tract, detoxifying toxicosis, reducing the total cholesterol level and decreasing blood pressures[1]. There are flavonoids including baicalin, baicalein, wogonin and ¶¬-sitosterol, baicalin in the plant. Baicalin, which is a flavonoid compound purified from Scutellaria Baicalensis Georgi, has been demonstrated to possess anti-toxin, anti-fever, anti- oxidant and anti-pyretic[2-3].
Baicalin is mainly extracted from Scutellaria Baicalensis Georgi with water and base solution and then precipitated by acid solution at present[4]. However, it has the disadvantage of low recovery. So it is essential to develop an effective and new method for separation of baicalin in herb. Polyamide is a kind of high molecular compound polymerized by carboxamide-group. As an adsorbent, it is widely applied to separating and purifying organic compounds due to the merits such as good stability of physical and chemical properties, excellent adsorbing selectivity, hardly being affected by inorganic compound, being regenerated and desorbed easily, long service life, easy formation of the closed circuit and saving cost[5]. In this work, based on the ethanol extraction, the processing parameters of purifying baicalin with polyamide were investigated to provide a theoretical basis for the industrial production.
2 Adsorption theory
The action between polyamide and baicalin is shown in Fig.1.
Polyamide needs to be pretreated by boiling in absolute ethanol for 2 h, because there are small amount alcohol-soluble compounds in polyamide powder and the mobile phase is organic solvent in the next experiment.
The separation of baicalin with polyamide constitutes of two steps of adsorption and de-adsorption, which is a typical adsorption-desorption mass transfer procedure. The procedure of baicalin in the leachate adsorbed in polyamide particle is subdivided into the following two stages: 1) adsorption of leachate from the liquid film to the surface of polyamide; 2) diffusion of baicalin from the exterior surface to the interior of polyamide.
The desorption is the reverse procedure. The separation procedure is shown in Fig.2.
The flavonoid compounds are adsorbed on the exterior surface when arriving at the surface of polyamide particle. Part of flavonoid molecules adsorbed diffuse to the interior along the pore wall of particles, which is called surface diffusion. Other molecules further diffuse to the inner pore of particles, which is called pore diffusion. And the flavonoid molecules may collide with the surface of pore wall to be adsorbed. Therefore, the adsorption procedure is composed of external diffusion, inner diffusion and surface adsorption[6-7]. The procedure of adsorbent to adsorb molecules from the fluid can be subdivided into the following three steps: 1) traveling of the flavonoid molecules in the fluid film around adsorbent; 2) diffusion of molecules adsorbed in the particle; 3) adsorption reaction in the interior surface of adsorbent. Surely, the slowest one is the controlling step.
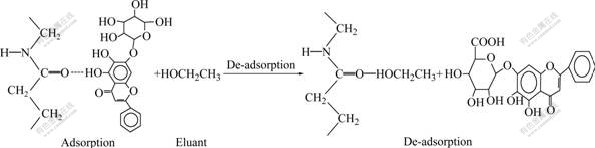
Fig.1 Desorption procedure of baicalin
Fig.2 Adsorption procedure of flavoinoids with polyamide: 1°™Exterior diffusion; 2°™Exterior surface diffusion; 3°™Surface diffusion; 4°™Pore diffusion; 5°™Interior surface diffusion
The velocity of the third step is so fast that its effect can be ignored. The adsorption velocity of the total procedure is thus determined by the film diffusion and the intraparticle diffusion. Both the Boyd film diffusion equation and the intraparticle diffusion equation are employed to exam the behavior of polyamide adsorbing baicalin.
Boyd film diffusion equation[8]:
-ln(1-F)=kt (1)
Intraparticle diffusion equation[9]:
qt=Kit0.5 (2)
where F=qt/qe (qt is the adsorption capacity, qe is the equilibrium adsorption capacity of specific polyamide); t is the adsorption time; k is the rate constant of liquid film diffusion; Ki is the adsorption velocity constant of polyamide to flavonoid.
3 Experimental
3.1 Materials and chemicals
The Scutellaria Baicalensis Georgi was provided by the Sanjiu Pharmacy, China. Baicalin, a standard sample (No. 110715-200514), was purchased from the National Institute for the Control of Pharmaceutical and Biological Products, China. Polyamide (0.075-0.150 mm) was purchased from the Sinopharm Chemical Reagent Co., Ltd. Deionized water and reagent of chemical grade were used in all experiments.
3.2 Analytical method
The baicalin content in samples was determined by the UV spectrophotometer using standard baicalin as check sample[10]. The standard curve regression equation is y=0.069 35x+0.013 4 (R=0.999 65).
3.3 Preparation of leachate with baicalin
A sample of 50 g Scutellaria Baicalensis Georgi was dispersed in 500 mL of 60% ethanol solution and leached at 80 °Ê for 2 h twice. The leachate with baicalin was then collected for the next experiment.
3.4 Adsorption isotherm
Six samples of polyamide with different masses were respectively added in six Erlenmeyer flasks with 25 mL of leachate prepared, and adsorbed in the picking table at 25 °Ê for 2 h. The equilibrium concentration of baicalin (ce) was measured with a spectrophotometer. The adsorption isotherm ce vs qe was then drawn, which was calculated by the equation: qe=(c0-ce)V/m, where V is the volume of leachate, 25 mL, m is the mass of polyamide.
3.5 Static adsorption kinetics
Three samples of 250 mL leachate with different baicalin contents were respectively put into three Erlenmeyer flasks with 0.5 g of polyamide, and stirred for different given times. The concentration of baicalin was determined with a spectrophotometer to calculate the adsorption capacity (qe) and the exchange rate (F) at this time[11-12], then the adsorption rate curve was drawn.
3.6 Dynamic de-adsorption experiment
5 mL of leachate was added on the column wet-packed with 7.5 g of polyamide, and then eluted with different concentrations of ethanol solution (100%, 80%, 60%, and 40%) at a velocity of 1.2 mL/min. The eluent was collected and the content of baicalin was determined by UV spectrum, then the elution graphs under different concentrations were drawn[13-14].
4 Results and discussion
4.1 Adsorption isotherm
At room temperature (25 °Ê), the adsorption iso- therm curve of baicalin adsorbed onto polyamide is shown in Fig.3.
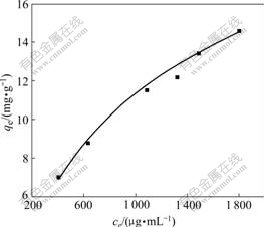
Fig.3 Adsorption isotherm curve of baicalin with polyamide
Fig.3 shows that the adsorption capacity gradually increases with increasing equilibrium concentration (ce) of baicaliin in the leachate. Relative data were introduced into the Freundlich equation to investigate whether the adsorption isotherm of baicalin adsorbed onto polyamide is in agreement with the Freundlich equation[13], whose logarithm form is shown as follows:
ln qe=ln k+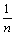 ln ce (3)
ln ce (3)
where k and n are the characteristic constants, k represents the relative adsorptive capacity, and n describes the heterogeneity of adsorbent surface and the relative adsorptive strength. n°ð1 stands for the preferential adsorption procedure, which means the adsorption is easy to conduct. The Freundlich linear regression curve was obtained by plotting ln qe with ln ce (shown in Fig.4), which reveals nicer linear correlation, and the correlation constant is higher than 0.99, suggesting that the adsorption isotherm of baicalin obeys the Freundlich equation. The characteristic constants were calculated to be k=3.8 and n=2.057. Here n=2.057£æ1 shows that the adsorption of baicalin by polyamide is preferential adsorption procedure.
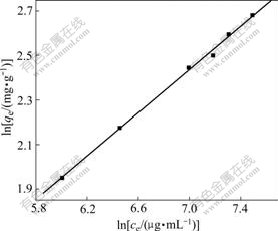
Fig.4 Linear regression of Freudlich equation
Consequently, the adsorption isotherm corresponds to the Freundlich equation, and is the preferential adsorption procedure at room temperature. The isothermal adsorption equation is
qe=3.8ce2.057 (4)
In Eqn.(4), k, describing the adsorption capacity, is 3.8, which suggests that the relative adsorption capacity of polyamide to baicalin is somewhat small.
4.2 Adsorption mechanism
The IR spectra of polyamide and polyamide adsorbed with baicalin are shown in Fig.5.
In Fig.5, the spectral bands at about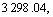
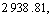
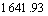 and
and 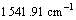 belong to the stretching vibration peaks of dimer and paraformal N°™H, the stretching vibration peak of C=O, the couple of N°™H flexural vibration peak and C°™N stretching vibration peak, respectively. It is suggested that many amido and carbonyl polar groups are contained in polyamide molecules, and these groups are all the adsorbing active sites. From Fig.5, it can be seen that IR spectrum of the polyamide has little difference with that of the polyamide adsorbed with baicalin, and the site and peak shape of main characteristic peaks are almost the same, which eliminate the possibility of chemical reaction between baicalin and amido. Considering all the above experimental results, the adsorption process of baicalin by polyamide is a rapid adsorbing one, and is mainly controlled by the physical adsorption. The weak hydrogen bond force and van der Waals force are the major adsorbing acting forces[15].
belong to the stretching vibration peaks of dimer and paraformal N°™H, the stretching vibration peak of C=O, the couple of N°™H flexural vibration peak and C°™N stretching vibration peak, respectively. It is suggested that many amido and carbonyl polar groups are contained in polyamide molecules, and these groups are all the adsorbing active sites. From Fig.5, it can be seen that IR spectrum of the polyamide has little difference with that of the polyamide adsorbed with baicalin, and the site and peak shape of main characteristic peaks are almost the same, which eliminate the possibility of chemical reaction between baicalin and amido. Considering all the above experimental results, the adsorption process of baicalin by polyamide is a rapid adsorbing one, and is mainly controlled by the physical adsorption. The weak hydrogen bond force and van der Waals force are the major adsorbing acting forces[15].
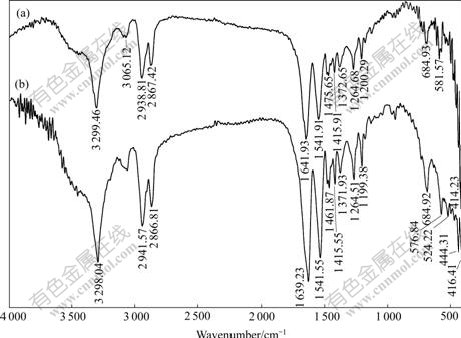
Fig.5 Comparison between IR spectra of polyamide (a) and polyamide adsorbed with baicalin (b)
4.3 Adsorption kinetics
The variation of exchange rate (F) with increasing time (t) at different initial concentrations of baicalin and room temperature is shown in Fig.6.
As shown in Fig.6, the adsorption of polyamide to baicalin is basically completed in 20 min. The exchange rate (F) is up to 95% under different initial concentrations. Therefore, the analysis of adsorption rate of polyamide should be focused on the initial 20 min leaching, which plays an important role in the investigation of adsorption rate. Both Boyd film diffusion equation and intraparticle diffusion equation were employed to simulate the adsorption procedure. The results are shown in Figs.7 and 8, respectively.
It can be noted that the curves of -ln(1-F) vs t
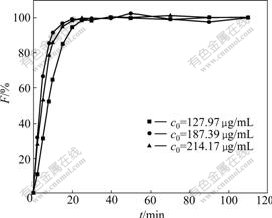
Fig.6 Adsorption curves of baicalin with polyamide at different initial concentrations of baicalin
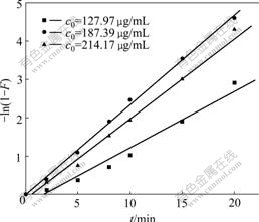
Fig.7 Curves of ®Cln(1-F) vs t at different initial concentrations of baicalin
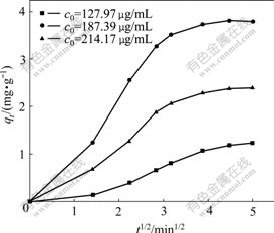
Fig.8 Curves of qt vs t1/2 at different initial concentrations of baicalin
reveal better linear relationship in Fig.7, and the curves of qt vs t1/2 show inferior linear relationship in Fig.8. The adsorption kinetics of polyamide to baicalin follows the Boyd film diffusion equation, which suggests that the film diffusion is the control step. The film diffusion rate constant (k) under different initial concentrations of baicalin were calculated by the slopes of lines in Fig.7 and shown in Table 1.
Table 1 Adsorption rate constants of baicalin with polyamide under different initial concentrations of baicalin
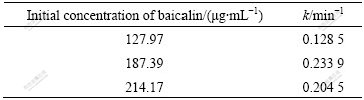
As shown in Table 1, the diffusion rate constant of film decreases firstly, and then increases with increasing initial concentration of baicalin. When the concentration of baicalin increases persistently, the baicalin molecules resembled on the surface of polyamide reach saturation gradually and the increasing trend of adsorption velocity weakens slowly. When the concentration of baicalin gets supersaturation, the increased collision probability among baicalin molecules begins to inhibit the liquid film diffusion surrounding polyamide, and make the diffusion rate constant decrease. In this work, polyamide adsorbs baicalin molecule quickly due to the proper concentration of the leachate selected.
4.4 Desorption results
The leachate was eluted by 100%, 80%, 60% and 40% of ethanol eluent, respectively. The elution curves are shown in Fig.9.
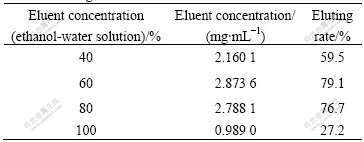
Fig.9 suggests that the baicalin adsorbed by polyamide can be eluted by different concentrations of ethanol-water solution, and the eluted baicalin distributes intensively in a short period. Therefore, each elution curve has a peak under different ethanol solutions. The eluent was collected and the baicalin content was measured to calculate the elution rate. The results are shown in Table 2.

Fig.9 Elution curves of leachate by single eluent
Table 2 Eluting results at different eluent concentrations
The eluent eluting from 12 to 32 min by 60% ethanol was collected, concentrated and dried. The content of baicalin is 33.86%. At the same time, the HPLC graphs of baicalin standard, leachate liquid and eluting products are compared and shown in Fig.10.
From Table 2 and Fig.10, the elution peak, the product content and the impurities contents contained are the larger, higher and lower, respectively, when using 60% of ethanol solution as the eluent.
5 Conclusions
1) Using polyamide as an adsorbent, the adsorption mechanism, adsorption kinetics and desorption laws of baicalin are investigated.
2) The adsorption procedure of polyamide to baicalin in Scutellaria Baicalensis Georgi agrees with the Freundlich equation, q=3.8ce2.057, which is a kind of preferential adsorption; the control step of adsorption is the liquid film diffusion, which satisfies with Boyd equation. The adsorption rate equation is -ln(1-F)= 0.128 5 t, and the diffusion rate constant (k) increases
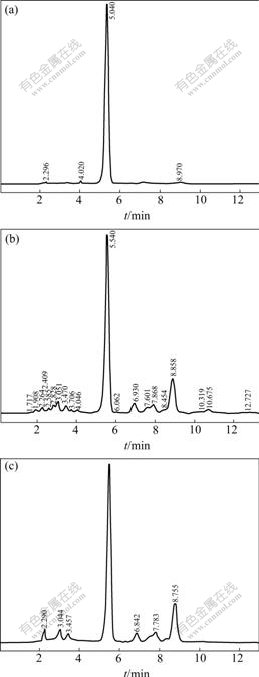
Fig.10 HPLC spectra of different samples (Chromatography column: InertsilODS-3C18 (150 mm°¡4.6 mm, 5 ¶Ãm); Elution: methanol-water (0.4% phosphorous acid); Monitor wave length: 278 nm; Column temperature: room temperature; Current velocity: 1 mL/min; Injection dose: 10 ¶ÃL): (a) Standard of baicalin; (b)Leachate liquid; (c)Product eluted
firstly and then decreases with the increase of initial concentration of baicalin.
3) The results of de-adsorption experiments show that the eluting effect is the best and the eluting product contains the least impurities, when the leachate liquid is eluted by 60% ethanol solution (100 mL) at room temperature and the eluent velocity of 1.2 mL/min. The baicalin content of product reaches 33.86%.
References
[1] LI Hua-bin, JIANG Yue, CHEN Feng. Separation methods used for Scutellaria Baicalensis active components [J]. Journal of Chromatography B, 2004, 812: 277-290.
[2] SHI H L, ZHAO B L, XIN W J. Scavenging effects of baicalin on free radicals and its protection on erythrocyte membrane from free radical injury [J]. Biochem Mol Biol Interact, 1995, 35: 981-984.
[3] LIU Er-bao, WEI Hong-qing, ZHAO Xiu-li, LI Xiao-xia, JIANG Feng-xian. Determination of baicalin in traditional Chinese preparation by high performance liquid chromatography with chemiluminescence detection [J]. Chinese Chemical Letters, 2004, 15(9): 1067-1070.
[4] AZIZAH A M, RUSLAWATI N M N, SWEE T T. Extraction and characterization of antioxidant from cocoa by-products [J]. Food Chemistry, 1999, 64: 199-202.
[5] ZHU Li-li, XU Xiao-jie. Selective separation of active inhibitors of epidermal growth factor receptor from Caragana Jubata by molecularly imprinted solid-phase extraction [J]. Journal of Chromatography A, 2003, 991: 151-158.
[6] YANG R T. Gas separation by adsorption processes [M]. Boston: Butterworth Publishers, 1987.
[7] HO Y S, MCKAY G. A kinetic study of dye sorption by biosorbent waste product pith [J]. Resources, Concentration and Recycling, 1999, 25: 171-193.
[8] LI Ming-yu, ZENG Qing-xuan, FENG Chang-gen. Study on performances of ion exchange fiber adsorbing and desorbing ferulicacid [J]. China Pharmacy Journal, 2005, 40(1): 40-43. (in Chinese)
[9] WANG Xue-jiang, ZHANG Quan-yin, LI An-min, CHEN Jin-long. Adsorption of salicylic acid from aqueous solution by NDA-100 macroreticular resin [J]. Acta Scientiae Circumstantiae, 2002, 22(5): 658. (in Chinese)
[10] LEI Ning-fei, PENG Shu-ming, ZHOU Jia-yu, HU Guo-ku, LIU Rong, CHEN Fang. Study on the extraction technology of baicalin from Scutellaria Baicalensis Georgi [J]. Lishizhen Medicine and Meteria Medica Research, 2007, 18(11): 2664-2666.
[11] DU Jie, ZHANG Li-ping, WANG Fan, PU Jun-wen. Study on adsorption of baicalin on immobilized tannins macroporous adsorption resin [J]. Biomass Chemical Engineering, 2007, 41(2): 23-27. (in Chinese)
[12] XIAO Wen-jun, FU Dong-he, HU Xiang-wen, YAN Zhi-hui, XIAO Li-zheng. Study on technology of separation and purification for flavones in lotus leaf by HP-20 [J]. Natural Product Research and Development, 2007, 27, 19: 692-695.
[13] LI Xin, ZENG Qing-xuan, FENG Chang-gen, ZHOU Shao-ji. Adsorption properties of cation ion-exchange fiber for UDMH [J]. The Chinese Journal of Process Engineering, 2006, 6(1): 23-27. (in Chinese)
[14] SHI Lei, JIANG Qin, WANG Su-qing, CANG Ji-yong, ZHANG Hua, HE Jian-gong. Study on purification of mycophenolic acid with macroprous adsorption resin [J]. Chinese Pharmaceutical Journal, 2007, 42(6): 454-457.
[15] LI Yun-xia, SUO Quan-ling, HE Wen-zhi, LI Chun-ping, HUANG Yan-chun. Separation and structural characterization of baicalin in Scutellaria Baicalensis Georgi [J]. Chinese Traditional Patent Medicine, 2007, 29(11): 1648-1651.
(Edited by CHEN Wei-ping)
Foundation item: Project(2006ABC014) supported by the Natural Science Foundation of Hubei Province, China
Received date: 2008-01-16; Accepted date: 2008-03-08
Corresponding author: CHI Ru-an, Professor, PhD; Tel: +86-27-87194500; E-mail: rac@mail.wit.edu.cn