��ұ��ɰ���̳��軯����IJ���
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2020���7��
�������ߣ����� ��˪�� ֣�Ž� �Ӻ��� ����־ ���о� ̷��
����ҳ�룺1964 - 1979
�ؼ��ʣ���ұ��ɰ���̳����軯��̼����
Key words��refractory gold concentrate; calcine; dust; cyanidation; carbonaceous matter
ժ Ҫ�����öԱȷ����о���ұ��ɰ���̳��軯����IJ��졣���������ֱ���軯ʱ��ɰ���̳��н�Ľ����ʷֱ�Ϊ85.31%��54.30%���顢̼�������������ʽ�ǵ������߽�����ʲ������Ҫԭ����NaOHԤ�������軯���𣬱�ɰ���̳��н���������ʷֱ�Ϊ87.70%��58.60%���к�Ԫ�ص��ѳ������Ԥ���������н����ʧ������������谭��ͬ�������Ԥ������ɰ���̳��н�Ľ����ʡ���H2SO4Ԥ������ɰ���̳��н���������ʷֱ�ﵽ94.96%��80.40%��̼�����Ӱ���DZ�ɰ���̳��н�����ʲ������Ҫԭ������Щ���죬����������˹��գ���ɰ���̳��н�Ľ����ʷֱ�ﵽ94.91%��91.90%��
Abstract: Differences of cyanide leaching between the calcine and the dust from a refractory gold concentrate were investigated by comparative method. Results showed that gold leaching efficiencies of the calcine and the dust were 85.31% and 54.30%, respectively, with direct cyanidation. Contents and existing forms of arsenic and carbon were the main reasons for those differences. The maximum gold leaching efficiencies of the calcine and the dust were 87.70% and 58.60%, respectively, with cyanidation after NaOH pre-leaching. Harmful elements removal, gold loss in NaOH pre-leaching and iron oxides hindrance codetermined gold leaching efficiencies of the calcine and the dust. After H2SO4 pre-leaching, the maximum gold leaching efficiencies of the calcine and the dust achieved 94.96% and 80.40%, respectively. The effect of carbonaceous matter was the main reason for differences for leaching efficiencies of the calcine and the dust. Based on those differences, two proper gold extraction processes were put forward, and gold leaching efficiencies for the calcine and the dust achieved 94.91% and 91.90%, respectively.
Trans. Nonferrous Met. Soc. China 30(2020) 1964-1979
Pan CAO1, Shang-hua ZHANG1, Ya-jie ZHENG1, Han-bing HE1, Shen-zhi LAI2, Xing-jun WANG2, Bing TAN2
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. Western Region Gold Yili Co., Ltd., Yining 835100, China
Received 10 November 2019; accepted 21 May 2020
Abstract: Differences of cyanide leaching between the calcine and the dust from a refractory gold concentrate were investigated by comparative method. Results showed that gold leaching efficiencies of the calcine and the dust were 85.31% and 54.30%, respectively, with direct cyanidation. Contents and existing forms of arsenic and carbon were the main reasons for those differences. The maximum gold leaching efficiencies of the calcine and the dust were 87.70% and 58.60%, respectively, with cyanidation after NaOH pre-leaching. Harmful elements removal, gold loss in NaOH pre-leaching and iron oxides hindrance codetermined gold leaching efficiencies of the calcine and the dust. After H2SO4 pre-leaching, the maximum gold leaching efficiencies of the calcine and the dust achieved 94.96% and 80.40%, respectively. The effect of carbonaceous matter was the main reason for differences for leaching efficiencies of the calcine and the dust. Based on those differences, two proper gold extraction processes were put forward, and gold leaching efficiencies for the calcine and the dust achieved 94.91% and 91.90%, respectively.
Key words: refractory gold concentrate; calcine; dust; cyanidation; carbonaceous matter
1 Introduction
With rapid depletion of free-milling gold ores, the proportion of gold extracted from refractory gold ore is increasing [1-3]. Refractory gold ores are defined as ores for which the leaching efficiency of gold by cyanidation is less than 80%, even after fine grinding [4,5]. Gold in refractory gold ores can be effectively leached and recovered only after pretreatment [6-11]. The main pretreatment methods of refractory gold ores include oxidation roasting, pressurized oxidation, ultrafine grinding and biological oxidation [8,12,13]. Oxidative roasting technology is used extensively for the desulfidation and dearsenization of refractory gold concentrates in China because this technology has certain advantages such as the modest investment, high efficiency, and technical maturity [14,15]. Its purpose is to remove harmful elements from ores and form porous calcine, so that gold can be effectively leached during cyanidation [16].
During the roasting process of refractory gold concentrates, calcine and dust are produced. In recent years, many scholars have carried out detailed researches on the chemical compositions, mineral compositions, structure and gold extraction processes of calcine. So, the technology for gold extraction from calcine was gradually matured, and gold leaching efficiency was generally more than 90% by cyanidation [3,7,8,16,17]. However, few researches on the mineral compositions, properties and gold extraction processes for dust have been done. Dust has usually been treated by the same process as calcine, which resulted in a high gold content (generally 5 usually 7 g/t) in cyanidation tailing [16,18,19].
In this work, the differences of cyanide leaching between the calcine and the dust from a refractory gold concentrate were studied, and the reasons for the differences were illustrated based on the mineralogy analysis of the calcine and the dust. And based on those differences, proper Au extraction processes for the calcine and the dust were put forward. Through the researches in this study, the differences of cyanide leaching between the calcine and the dust were illustrated, and the efficient leaching of gold from the calcine and the dust was realized. This study provided a reference to extract gold from gold-bearing calcine, dust and cyanide tailings.
2 Experimental
2.1 Materials
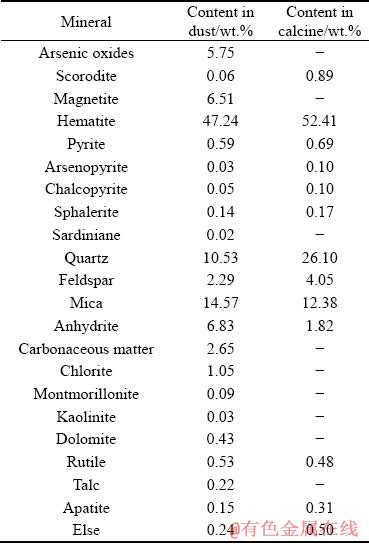
The calcine and the dust, produced in the two-stage roasting pretreatment process of a refractory gold concentrate, were provided by Axi Gold Mine, China. Major element compositions of the calcine and the dust are listed in Table 1. Mineral liberation analysis (MLA) was used to determine mineral compositions, and the results are shown in Table 2.
Table 1 Major element compositions of calcine and dust (wt.%)

Table 2 shows that the calcine was mainly composed of hematite, quartz and mica, and the dust was mainly composed of hematite, magnetite, quartz, mica, anhydrite, arsenic oxides and carbonaceous matter.
2.2 Leaching experiments
2.2.1 Pre-leaching with sulfuric acid
The sample was leached with H2SO4 in a 1 L three-neck flask controlling the leaching temperature (90 ��C) within ��2 ��C by a heating sleeve. The liquid/solid ratio (L/S) and leaching time were 4:1 and 4 h, respectively. At the end of acid leaching period, solid and liquid phases were separated by filtration, and then H2SO4 leaching residues were washed thoroughly with distilled water and used as the feed of cyanidation.
Table 2 Major mineral compositions in calcine and dust
2.2.2 Pre-leaching with sodium hydroxide
The sample was leached with NaOH in a 500 mL three-neck flask controlling the leaching temperature (90 ��C) within ��2 ��C by a heating sleeve. The liquid/solid ratio and leaching time were 3:1 [20] and 2 h, respectively. At the end of leaching period, solid and liquid phases were separated by filtration, and then NaOH pre-leaching residues were washed thoroughly with distilled water (except for special instruction) and used as the feed of cyanide leaching.
2.2.3 Cyanide leaching
Cyanide leaching experiments were carried out in a 500 mL three-neck flask with a liquid/solid ratio of 4:1 at room temperature for a predetermined time. NaCN concentration was 2000 mg/L, and the pH of cyanide leaching was 11. In direct cyanidation, solution samples were taken at regular time intervals and analyzed for their Au and As contents.
2.3 Analysis methods
X-ray diffraction (XRD) patterns of the calcine and the dust were obtained using a Rigaku D/max- TTR III X-ray diffractometer with Cu K�� radiation (��=1.54056  , 40 kV, 250 mA) in the 2�� range of 10��-70�� at a scanning speed of 10 (��)/min. The mineral compositions and occurrence state of gold in the calcine and the dust were determined using the MLA system. The MLA system consists of a special software package and an FEI Quanta 600F scanning electron microscope (SEM) equipped with an energy dispersive spectrometry (EDS). CELEP and SERBEST [21] have given the detailed test principle of the MLA system.
, 40 kV, 250 mA) in the 2�� range of 10��-70�� at a scanning speed of 10 (��)/min. The mineral compositions and occurrence state of gold in the calcine and the dust were determined using the MLA system. The MLA system consists of a special software package and an FEI Quanta 600F scanning electron microscope (SEM) equipped with an energy dispersive spectrometry (EDS). CELEP and SERBEST [21] have given the detailed test principle of the MLA system.
The contents of gold and silver were determined by fire assay gravimetric measurement method (GB/T 20899.1��2007 and 20899.2��2007). The contents of C and S in the calcine, the dust and leaching residues were analyzed using a high-frequency infrared carbon-sulfur analytical instrument (HCS, HCS-800, Kaide Instruments Co.). Other elemental analysis in the calcine and the dust was performed by means of inductively coupled plasma mass spectrometry (ICP-MS, 7000 series, Agilent) after X-ray fluorescence (XRF) analysis. Iodine leaching method was used to determine dissociation degree of gold [3]. Phase compositions of carbon were determined using the method of non-aqueous titration, and the arsenic phase compositions were determined according to the method in Ref. [22]. The determination of As, Fe and S contents in leaching solution was performed using an inductively coupled plasma optical emission spectrometry (ICP-OES, iCAP- 7000 series, Thermo Fisher Scientific). The morphology of alkaline pre-leaching residues was observed using a scanning electron microscopy (SEM, JSM-I T300, JEOL).
3 Results and discussion
3.1 Differences of direct cyanidation between calcine and dust
3.1.1 Difference of gold leaching efficiency between calcine and dust
Gold leaching efficiencies of the calcine and the dust at different cyanidation time are presented in Fig. 1.

Fig. 1 Gold leaching efficiencies of calcine and dust at different cyanidation time
Figure 1 illustrates that gold leaching efficiency of the calcine increased from 0 to 85.31% and that of the dust increased from 0 to 54.30% with increasing cyanidation time from 0 to 48 h.
3.1.2 Effects of arsenic on cyanidation
Arsenic concentrations in cyanide leaching solution at different cyanidation time (gold-bearing pregnant solution) during direct cyanidation of the calcine and the dust are shown in Fig. 2.
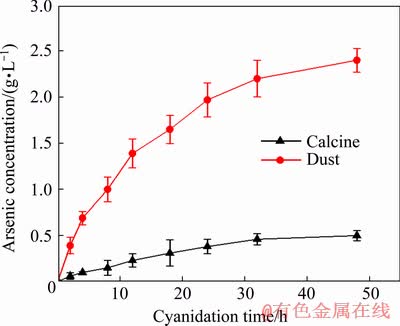
Fig. 2 Arsenic concentrations in cyanide leaching solution at different cyanidation time during direct cyanidation of calcine and dust
Figure 2 shows that arsenic concentration in cyanide leaching solution for the calcine increased from 0 to 0.50 g/L and that for the dust increased from 0 to 2.40 g/L with increasing cyanidation time from 0 to 48 h. Arsenic concentration for the dust was always higher than that for the calcine. In addition, the difference of arsenic concentration gradually increased with a prolongation of cyanidation time.
Phase compositions of the arsenic in the calcine and the dust are presented in Table 3.
Table 3 Phase compositions of arsenic in calcine and dust (mass fraction, %)

Since the pH of cyanide leaching solution was maintained at 11 with NaOH solution during cyanidation, the major reactions of arsenic oxides during cyanidation are illustrated in Eqs. (1) and (2) [23,24].
As2O3+2NaOH=2NaAsO2+H2O
��rG��=-50.26 kJ/mol (1)
As2O5+6NaOH=2Na3AsO4+3H2O
��rG��=-282.35 kJ/mol (2)
The standard equilibrium constant K can be calculated as Eq. (3) [25].
K=exp[-��rG��/(RT)] (3)
According to the ��rG�� values of the above reactions, the standard equilibrium constants of Eqs. (1) and (2) are 108.81 and 1049.46, respectively. Thermodynamic calculation showed that As2O3 and As2O5 could react with sodium hydroxide. As a result, the arsenic existing in As2O3 and As2O5 entered into cyanide leaching solution.
Arsenopyrite is difficult to dissolve during cyanidation and its detrimental effect on gold extraction mainly comes from its encapsulation to gold [23]. Fortunately, the effect of arsenopyrite in the calcine and the dust on gold leaching could be neglected because the contents of arsenopyrite in both the calcine and the dust were very low.
Ferric arsenate could react with sodium hydroxide in alkaline cyanide solution, and as a result, arsenic in ferric arsenate could enter into cyanide leaching solution, and the reaction was illustrated in Eq. (4) [14]:
FeAsO4+3NaOH+H2O=NaFe(OH)4+Na2HAsO4 (4)
Both arsenic oxides and ferric arsenate could react with sodium hydroxide, and arsenic concentration in cyanide leaching solution increased with a prolongation of cyanidation time. Furthermore, arsenic content in the dust was higher than that in the calcine, and the higher the arsenic content was, the more the reactants reacting with NaOH were. Therefore, arsenic concentration in cyanide leaching solution for the dust was always higher than that for the calcine.
The distribution coefficient diagrams of As(V) and As(III) in aqueous solution are shown in Fig. 3 [26,27].

Fig. 3 Distribution coefficient (��) diagrams of As in aqueous solution
Figure 3(a) shows that As(V) exists mainly in the forms of  and
and 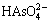 in aqueous solution at pH 11, and the distribution coefficients of
in aqueous solution at pH 11, and the distribution coefficients of  and
and 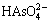 are 24.24% and 75.75%, respectively. Figure 3(b) shows that As(III) mainly exists in the form of
are 24.24% and 75.75%, respectively. Figure 3(b) shows that As(III) mainly exists in the form of  in aqueous solution at pH 11, and its distribution coefficient is 98.44%. The arsenic entering cyanide leaching solution (pH=11) mainly existed in the forms of
in aqueous solution at pH 11, and its distribution coefficient is 98.44%. The arsenic entering cyanide leaching solution (pH=11) mainly existed in the forms of  ,
, 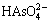 and
and  . When
. When  ,
, 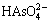 and
and  in solution contacted with gold mineral particles, they formed passivation films on gold particles. As a result, the contact between gold mineral particles and cyanide was hindered, and gold leaching efficiency decreased. In addition, the higher the arsenic concentration in the cyanide leaching solution is, the more the passivation films are, and the more serious the detrimental effect of arsenic on gold leaching is [28]. Therefore, compared with the calcine, the detrimental effect of arsenic on gold leaching efficiency of the dust was more serious.
in solution contacted with gold mineral particles, they formed passivation films on gold particles. As a result, the contact between gold mineral particles and cyanide was hindered, and gold leaching efficiency decreased. In addition, the higher the arsenic concentration in the cyanide leaching solution is, the more the passivation films are, and the more serious the detrimental effect of arsenic on gold leaching is [28]. Therefore, compared with the calcine, the detrimental effect of arsenic on gold leaching efficiency of the dust was more serious.
3.1.3 Effects of carbon on cyanidation
Phase compositions of the carbon in the calcine and the dust are shown in Table 4. Polarization microscope and SEM images of carbonaceous matter in the dust are shown in Fig. 4.
Table 4 Phase compositions of carbon in calcine and dust (wt.%)

Figure 4 shows that carbonaceous matter was fine flake, leaf or needle-like with unidirectional extension, and had obvious double-reflection under reflected light, which indicated that some carbonaceous matters had crystallized to graphite. In addition, the disseminated association of carbonaceous matter with gangue minerals (iron minerals, arsenic oxides and other non-metallic minerals) was not close, and the particle size of carbonaceous matter ranged from 0.002 to 0.005 mm.
Carbonaceous matter existing in the forms of graphite and organic carbon had detrimental effects on gold leaching [29-31]. The detrimental effects were mainly manifested in the preg-robbing effect of those carbonaceous matters in the cyanide leaching process, and the degree of the preg-robbing effect depended on the content of carbonaceous matter and its existence forms [29-31]. Due to the preg-robbing effect, the gold in the form of gold-cyanide complex adsorbed on those carbonaceous matters eventually lost in cyanidation tailing, thus gold leaching efficiency decreased [30,32]. The contents of graphite and organic carbon in the dust were 0.22% and 0.25%, respectively, while no carbonaceous matter existed in the calcine as shown in Table 4. Therefore, the preg-robbing effect of carbonaceous matter on gold leaching for the dust was more serious than that for the calcine.

Fig. 4 Images of carbonaceous matter in dust
3.1.4 Effects of gold occurrence state on cyanidation
In order to further clarify the effects of gold occurrence state on cyanidation of the calcine and the dust, the types and chemical compositions, particle size, output forms and dissociation degree of gold minerals in the calcine and the dust were analyzed.
The types and distribution rates of gold minerals in the calcine and the dust are illustrated in Fig. 5.

Fig. 5 Types and distribution rates of gold minerals in calcine and dust
It can be seen from Fig. 5 that gold minerals in the calcine and the dust were composed of native gold and electrum. The distribution rates of native gold in the calcine and the dust were 85% and 86%, respectively, and those of electrum were 15% and 14%, respectively. The results of EDS (Energy dispersive X-ray spectroscopy) showed that the average contents of gold in gold minerals for the dust and the calcine were 90.27% and 90.51%, respectively. The types of gold minerals and the average content of gold in gold minerals were basically the same. Therefore, the effects of the types of gold minerals and the content of gold in gold minerals on cyanidation could be ignored.
The size distribution of gold mineral particles in the calcine and the dust is shown in Table 5.
Table 5 Size distribution of gold mineral particles in calcine and dust

As shown in Table 5, the distribution rate of gold mineral particles in the dust with a particle size less than 0.005 mm accounted for 100%. However, the distribution rate of gold mineral particles in the calcine with a particle size less than 0.005 mm accounted for only 6.08%. Compared with the calcine, the particle size of the gold minerals in the dust was finer, thus the specific surface area of gold mineral particles in the dust was larger [33,34]. In addition, the gold in the dust mainly existed in the forms of the monomer and exposed gold (Table 6). And a large specific surface area of gold mineral particles was beneficial to the contact of gold mineral particles with cyanide. Therefore, from the point of view of the particle size of gold minerals, it could be seen that gold mineral particles in the dust were easier to be extracted than those in the calcine during cyanide leaching process.
The output forms and distribution rate of gold minerals in the calcine and the dust are shown in Fig. 6 and Table 6, respectively.
Figure 6 presents that disseminated association of gold mineral particles with matrix minerals in the calcine was closer than that in the dust. And the closer the disseminated association was, the smaller the contact area between gold mineral particles and cyanide was, and the lower the gold leaching efficiency was in the same cyanidation time [3]. Table 6 shows that the distribution rate of the monomer and exposed gold in the calcine was 89.25%, and the distribution rates of the gold encapsulated in sulfide minerals, iron minerals and gangue minerals were 1.00%, 8.45% and 1.30%, respectively. The distribution rate of the monomer and exposed gold in the dust was 91.22%, and the distribution rates of the gold encapsulated in sulfide minerals, iron minerals and gangue minerals were 1.10%, 7.35% and 0.33%, respectively.
The above analysis showed that the particle size of gold minerals in the dust was finer than that in the calcine, and the disseminated association of gold mineral particles with matrix minerals in the dust was looser than that in the calcine. At the same time, the dissociation degree of gold minerals in the dust was higher than that in the calcine. Therefore, in terms of occurrence state of gold, gold leaching efficiency of the dust should be higher than that of the calcine. However, in an actual cyanide leaching process, gold leaching efficiency of the dust was significantly lower than that of the calcine. These results indicated that the particle size, output forms and dissociation degree of gold minerals in the calcine and the dust were not the major factors determining gold extraction.

Fig. 6 Output forms (determined by BSE) of gold minerals in calcine (a-d) and dust (e-h) (Au, H and Q represents gold, hematite and quartz, respectively)
Table 6 Dissociation degree of gold in calcine and dust

3.2 Difference of cyanide leaching between calcine and dust after sodium hydroxide pre-leaching
Alkaline pre-leaching can effectively remove arsenic and antimony, thus reducing the influences of harmful elements on cyanidation [3,20,35-37]. Therefore, NaOH leaching was used as a pretreatment method for the calcine and the dust, and the difference of cyanide leaching between the calcine and the dust after NaOH pre-leaching was studied comparatively in this study. The results are shown in Fig. 7.
As shown in Fig. 7(a), the removal efficiency of arsenic in the calcine increased from 0 to 51.65% and that of arsenic in the dust increased from 0 to 99.66% with increasing NaOH concentration from 0 to 6 mol/L. The arsenic in the calcine mainly existed in the form of iron arsenate, and the arsenic in the dust mainly existed in the form of arsenic oxides. Both arsenic oxides and iron arsenate could react with sodium hydroxide, and thus arsenic was removed.
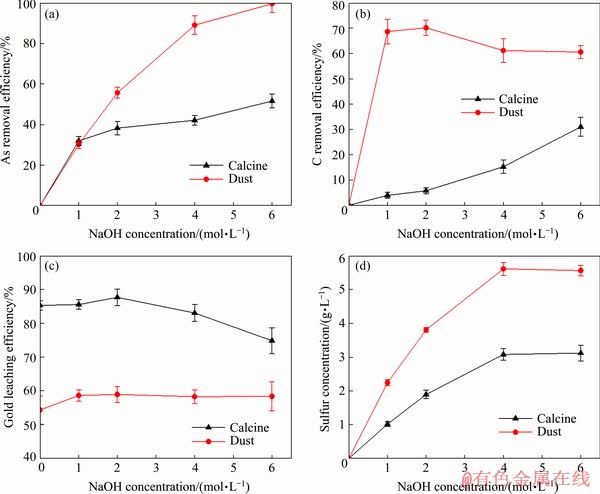
Fig. 7 Effects of NaOH concentration on removal efficiencies of arsenic and carbon (a, b), gold leaching efficiencies of calcine and dust after NaOH pre-leaching (c) and sulfur concentration in NaOH leaching solution (d)
Figure 7(b) shows that the removal efficiency of carbon in the calcine increased from 0 to 31.02% with increasing NaOH concentration from 0 to 6 mol/L. However, for the dust, the removal efficiency of carbon increased from 0 to 70.23% with increasing NaOH concentration from 0 to 2 mol/L, and then decreased with increasing NaOH concentration from 2 to 6 mol/L. Certain carbonates and humic acids could react with NaOH and enter into the solution [38,39], which led to the increase of carbon removal efficiency. However, the slurry viscosity increased under the high concentration of NaOH, therefore the removal efficiency of carbon decreased when NaOH concentration was higher than 2 mol/L.
As shown in Fig. 7(c), when NaOH concentration increased from 0 to 2 mol/L, gold leaching efficiency of the calcine after NaOH pre-leaching increased from 85.31% to 87.70%. When NaOH concentration exceeded 2 mol/L, gold leaching efficiency decreased. Studies showed that sulfide was decomposed into thiosulfate and polysulfide during alkaline pre-leaching process, and both thiosulfate and polysulfide could form strong soluble complexes with gold, which led to the loss of gold into alkaline pre-leaching solution [4]. As shown in Fig. 7(d), for the calcine, the sulfur concentration in NaOH leaching solution increased from 0 to 3120.20 mg/L with the increasing NaOH concentration from 0 to 6 mol/L. The higher the sulfur concentration was, the higher the concentrations of thiosulfate and polysulfide were, and the more the amount of gold lost in NaOH leaching solution was. When NaOH concentration was less than 2 mol/L, gold leaching efficiency increased because of the less loss of gold and the removal of harmful elements such as arsenic. However, when NaOH concentration was higher than 2 mol/L, the loss of gold in NaOH pre-leaching process was more, which led to the decrease of gold leaching efficiency. For the dust, when NaOH concentration increased from 0 to 1 mol/L, gold leaching efficiency of the dust after NaOH pre-leaching increased from 54.30% to 58.60%, and that did not change significantly with a continuous increase in NaOH concentration. NaOH pre-leaching could effectively remove arsenic and humic acids from the dust, thus improving gold leaching efficiency to a certain extent. But the carbon in the form of graphite was almost insoluble in NaOH solution [37,40]. As a result, NaOH pre-leaching could not eliminate the preg-robbing effect of graphite in the dust. In the same way, as shown in Fig. 7(d), for the dust, the sulfur concentration in NaOH leaching solution increased from 0 to 5672.90 mg/L with the increase of NaOH concentration from 0 to 6 mol/L, and the decomposition of sulfide in the dust during NaOH pre-leaching also caused the loss of gold. In addition, researches showed that iron oxide film or covering was easily to form on the surface of gold grains in the calcine and the dust during roasting [41], and NaOH pre-leaching could not effectively dissolve the iron oxide film coating on gold grains [20,37]. As a result, iron oxide film would hinder the improvement of gold leaching efficiency.
To sum up, NaOH pre-leaching could not significantly improve gold leaching efficiency for both the calcine and the dust. In order to improve gold leaching efficiency, not only the influences of harmful elements should be eliminated, but also the iron oxide coating on the surface of gold grains should be destroyed to fully expose the surface of gold grains.
3.3 Difference of cyanide leaching between calcine and dust after sulfuric acid pre- leaching
Acid pre-leaching can not only effectively dissociate the gold covered and wrapped by iron oxides, but also remove part of arsenic, so it is widely used as a method of pretreatment for calcine before cyanide leaching to improve gold leaching efficiency [3,37]. Therefore, the difference of cyanide leaching between the calcine and the dust after H2SO4 pre-leaching was studied comparatively. The results are shown in Fig. 8.

Fig. 8 Effects of H2SO4 concentration on gold leaching efficiency from acid leaching residues (a), iron (b) and arsenic (c) removal efficiencies during pre-leaching, and carbon content in acid leaching residues and residue rate (d)
Figure 8(a) shows that gold leaching efficiency of the calcine after H2SO4 pre-leaching increased from 85.31% to 94.96% with increasing H2SO4 concentration from 0 to 45% (mass fraction). Gold leaching efficiency of the dust after H2SO4 pre-leaching increased from 54.30% to 80.40% with increasing H2SO4 concentration from 0 to 15%. However, gold leaching efficiency decreased from 80.40% to 73.52% with increasing H2SO4 concentration from 15% to 45%. These results indicated that there was a significant difference between the gold leaching efficiencies of the calcine and the dust after H2SO4 pre-leaching.
Figure 8(b) illustrates that when H2SO4 concentration increased from 0 to 45%, iron removal efficiency in the calcine increased from 0 to 98.73%, and that in the dust increased from 0 to 77.79%.
The reactions of iron oxides with sulfuric acid are illustrated in Eqs. (5) and (6) [42,43]:
Fe2O3+3H2SO4=Fe2(SO4)3+3H2O
��rG��=-261.21 kJ/mol (5)
Fe3O4+4H2SO4=Fe2(SO4)3+FeSO4+4H2O
��rG��=-362.79 kJ/mol (6)
According to the ��rG�� of the above reactions, the standard equilibrium constants of Eq. (5) and Eq. (6) calculated with Eq. (3) are 1045.76 and 1063.55, respectively. The thermodynamic calculations showed that the reaction tendency of Fe2O3 and Fe3O4 with sulfuric acid was very large. With an increase in H2SO4 concentration, the iron leaching efficiency increased gradually. As a result, the iron removal efficiency increased with an increase in H2SO4 concentration. The removal of iron could not only further dissociate gold wrapped in iron minerals, but also remove iron-bearing impurities on the surface of dissociated gold mineral particles, thus the contact area and activity of interaction between gold mineral particles and cyanide increased, and gold leaching efficiency was significantly improved.
According to Fig. 8(c), the arsenic removal efficiency in the calcine increased from 0 to 88.50% with increasing H2SO4 concentration from 0 to 45%. For the dust, the arsenic removal efficiency increased from 0 to 89.60% with increasing H2SO4 concentration from 0 to 25%, while that decreased to 84.60% when H2SO4 concentration increased to 45%.
The arsenic in the calcine mainly existed in the form of iron arsenate. The reaction of ferric arsenate with sulfuric acid is presented in Eq. (7) [24]:
4FeAsO4+6H2SO4=4H3AsO4+2Fe2(SO4)3
��rG��=-341.01 kJ/mol (7)
According to the ��rG�� of Eq. (7), the standard equilibrium constant of Eq. (7) calculated by Eq. (3) is 1059.73. This indicated that the reaction tendency of iron arsenate with sulfuric acid was very large. And the reaction would proceed more thoroughly as acid concentration increased. The analysis results were consistent with the variation of arsenic removal efficiency shown in Fig. 8(c).
For the dust, during H2SO4 pre-leaching process, As2O5 was easily soluble in sulfuric acid solution. While the solubility of As2O3 in H2SO4 solution decreased with an increase in H2SO4 concentration [26]. When H2SO4 concentration was higher than 25%, the decrease of arsenic removal efficiency might be caused by the decrease in solubility of As2O3 (Fig. 8(c)).
For the calcine and the dust, the removal of arsenic by H2SO4 pre-leaching could reduce the amount of arsenic entering cyanide leaching solution, which was beneficial to the improvement of gold leaching efficiency.
Figure 8(d) presents that for the calcine, when H2SO4 concentration increased from 0 to 45%, the residue rate decreased from 100.00% to 45.60%, and the carbon content in the acid leaching residues had few change. For dust, the residue rate decreased from 100.00% to 53.50% with increasing H2SO4 concentration from 0 to 45%, and the carbon content in the acid leaching residues increased from 2.89% to 4.06%. As the residue rate decreased and carbon in the dust dissolved a little during pre-leaching, the carbon content in the acid leaching residues increased. When the same mass of the dust and the acid leaching residues were cyanided under the same conditions, the carbon content in the acid leaching residues was higher than that in the dust, thus the total carbon content in the acid leaching residues was higher than that in the dust. Therefore, the preg-robbing effect in cyanidation process of the acid leaching residues was enhanced, which made more gold-cyanide complex in cyanide leaching solution be adsorbed on the carbonaceous matter, and finally lose in cyanidation tailing. Especially when H2SO4 concentration was higher than 15%, the carbon content in the acid leaching residues had an obvious increase, thus the preg-robbing effect of carbonaceous matter was significantly enhanced and gold leaching efficiency decreased dramatically.
In summary, for the calcine, H2SO4 pre-leaching could effectively remove iron and arsenic. The effective removal of iron and arsenic could significantly improve gold leaching efficiency, and the higher the removal efficiencies of arsenic and iron were, the higher gold leaching efficiency was. For the dust, when H2SO4 concentration was less than 15%, the removal of iron and arsenic could effectively improve gold leaching efficiency. However, the preg-robbing effect of carbonaceous matter was significantly enhanced and gold leaching efficiency decreased when H2SO4 concentration was higher than 15%.
3.4 Proper extraction processes of gold from calcine and dust
According to the studies on the differences of cyanide leaching between the calcine and the dust, the proper extraction process of gold from the calcine is presented in Fig. 9, by which the gold leaching efficiency is 94.91%.

Fig. 9 Flowsheet of proper extraction process of gold from calcine
For the dust, after H2SO4 pre-leaching, the maximum gold leaching efficiency was only 80.40%. Major element compositions and dissociation degree of gold in the cyanidation tailing are shown in Tables 7 and 8, respectively.
Table 7 Major element compositions of cyanidation tailing (wt.%)

Table 7 shows that the content of gold was 7.74 g/t, and the contents of As, C and S were 0.42%, 3.01% and 0.78%, respectively, in the cyanidation tailing. Table 8 shows that the distribution rate of the monomer and exposed gold was 66.92%, and the contents of the gold encapsulated in sulfide, iron and gangue minerals were 5.43%, 14.21% and 13.44%, respectively, in cyanidation tailing. The results indicated that the effects of harmful components (such as arsenic, carbonaceous matter) on cyanide leaching could not be completely eliminated by H2SO4 pre-leaching.
Table 8 Content and distribution rate of gold in cyanidation tailing

NaOH pre-leaching can effectively remove arsenic oxides and arsenate films formed on the surface of gold mineral particles during cyanidation process, and effectively dissolve sulfides to further dissociate gold encapsulated in sulfide minerals, thereby significantly improve gold leaching efficiency [4,14,20]. At the same time, a large number of studies have shown that stage cyanidation can remove gold-cyanide complex ions in time, thus increase the activity of cyanide to dissolve gold, and reduce the adsorption of carbonaceous matter to gold [28,44,45]. Therefore, in order to improve gold leaching efficiency of the dust, the effect of NaOH pre-leaching on cyanidation of cyanidation residue, obtained from cyanidation of the dust after pre-leaching with 15% H2SO4 solution, was studied. It was found that after NaOH pre-leaching the filtering performance of pulp was poor, the filtering speed was slow especially under the condition of high NaOH concentration, and alkali leaching residues need to be washed many times to reach the neutral state. Therefore, in order to simplify the process and reduce wastewater production, direct cyanidation of unwashed alkali leaching residues was adopted.
The cyanidation time was shortened to 24 h for removing gold-cyanide complex ions from cyanide leaching solution in time and reducing the adsorption of carbonaceous matter to gold. Gold leaching efficiency was 62.00% when the dust was cyanided for 24 h after pre-leaching with 15% H2SO4 solution. Under the conditions of liquid- solid ratio of 3:1, leaching temperature of 90 ��C, leaching time of 2 h and stirring speed of 300 r/min, the effect of NaOH concentration on the removal efficiencies of arsenic and carbon in the first cyanidation residue and the gold leaching efficiency of NaOH pre-leaching residues are shown in Fig. 10.

Fig. 10 Effects of NaOH concentration on removal efficiencies of arsenic and carbon in the first cyanidation residue (a) and gold leaching efficiency from NaOH pre-leaching residues (b)
Figure 10 shows that with increasing NaOH concentration from 0 to 2 mol/L, the carbon removal efficiency increased from 0 to 70.50%, the arsenic removal efficiency increased from 0 to 47.45%, and the gold leaching efficiency of NaOH pre-leaching residues increased from 50.96% to 64.62%. Through pre-leaching with 2 mol/L NaOH solution, the amount of gold extracted by cyanidation from the NaOH pre-leaching residue accounted for 24.55% of the mass of gold in the dust. However, with increasing NaOH concentration from 2 to 6 mol/L, the carbon and arsenic removal efficiencies and gold leaching efficiency decreased. The reason was that the filtration of pulp was very poor, the moisture content of filter cake was high, and the entrainment of harmful elements to gold leaching was serious at high alkali concentration. When NaOH concentration was 6 mol/L, the removal efficiencies of arsenic and carbon and gold leaching efficiency were 36.42%, 60.74% and 52.78%, respectively. The optimum NaOH concentration during pre- leaching was 2 mol/L.
SME images of NaOH pre-leaching residues obtained under different NaOH concentrations are presented in Fig. 11.
Figure 11 shows that the porosity of particles in the NaOH pre-leaching residues declined with an increase in NaOH concentration. Especially, when NaOH concentration was higher than 2 mol/L, the agglomeration phenomenon of particles in the NaOH pre-leaching residues aggravated, which resulted in an obvious decrease in the porosity, and thus the contact between gold mineral particles and cyanide was hindered. As a result, gold leaching efficiency decreased with increasing NaOH concentration from 2 to 6 mol/L. And the other reason for the decrease of gold leaching efficiency was the reduction of removal efficiencies of arsenic and carbon.
After the dust was successively leached by 15% H2SO4 solution, cyanide solution, 2 mol/L NaOH solution and cyanide solution, the total gold leaching efficiency achieved 86.55%, the total removal efficiencies of arsenic and carbon were 91.38% and 74.10%, respectively. The gold content in the secondary-stage cyanidation residue was 5.31 g/t. These results indicated that after H2SO4 pre-leaching, NaOH pre-leaching could continue to remove arsenic and carbon from cyanidation residues, and the dissolving activity of cyanide to gold was improved after the removal of impurities, and thus gold leaching efficiency was improved.
In order to further reduce the content of gold in the secondary cyanidation residue, the secondary cyanidation residue was pretreated again using NaOH solution of 2 mol/L (the second NaOH pre-leaching), and then cyanidation (the third cyanidation) was conducted. The experimental results are shown in Table 9.

Fig. 11 SME images of NaOH pre-leaching residues obtained under different NaOH concentrations
Table 9 Results of the second NaOH pre-leaching and the third cyanidation (wt.%)

Table 9 shows that removal efficiencies of arsenic and carbon in the second NaOH pre-leaching process were 40.01% and 17.23%, respectively. And the total removal efficiencies of arsenic and carbon are 95.63% and 79.60%, respectively. Gold leaching efficiency of the third cyanidation was 37.67%, and the mass of gold extracted by cyanidation from the second NaOH pre-leaching residue accounted for 5.35% of the mass of gold in the dust. The total gold leaching efficiency was 91.90% through three-stage cyanidation, and the gold content in the final cyanidation tailing was 3.31 g/t.
The content and dissociation degree of gold in the final cyanidation tailing are shown in Table 10.
Table 10 shows that the content of gold was 3.31 g/t, and the content of the monomer and exposed gold was 1.18 g/t, and the contents of gold encapsulated in sulfide, iron and gangue minerals in the final cyanidation tailing were 0.09, 1.12 and 0.92 g/t, respectively. Compared with the results shown in Table 8, the contents of the monomer and exposed gold and the gold encapsulated in sulfide and gangue minerals were decreased. These results indicated that the unfavorable factors to cyanide leaching could be further eliminated or reduced through NaOH pre-leaching and stage-cyanidation after the dust was leached by H2SO4 solution.
Table 10 Content and distribution rate of gold in final cyanidation tailing

Based on the above analysis, the proper extraction process of gold from the dust is shown in Fig. 12.

Fig. 12 Flowsheet of proper extraction process of gold from dust
4 Conclusions
(1) Gold leaching efficiency of the calcine was 85.31%, which was 31.01% higher than that in dust by direct cyanidation. The contents and existing forms of arsenic and carbon were the main reasons for the difference of gold leaching efficiency between the calcine and the dust with direct cyanidation.
(2) When NaOH concentration increased from 0 to 2 mol/L, gold leaching efficiency of the calcine after NaOH pre-leaching increased from 85.31% to 87.70%. When NaOH concentration exceeds 2 mol/L, gold leaching efficiency decreased. When NaOH concentration increased from 0 to 1 mol/L, gold leaching efficiency of the dust after NaOH pre-leaching increased from 54.30% to 58.60%, and gold leaching efficiency did not change significantly with a continuous increase in NaOH concentration. In order to improve the gold leaching efficiencies of the calcine and the dust, not only the influences of harmful elements should be eliminated, but also the iron oxide coating on the surface of gold grains should be destroyed to fully expose the surface of gold grains.
(3) H2SO4 pre-leaching could effectively remove iron and arsenic from the calcine and significantly improve gold leaching efficiency. For the dust, the removal of iron and arsenic could effectively improve gold leaching efficiency when H2SO4 concentration was less than 15%. However, the preg-robbing effect of carbonaceous matter was significantly enhanced and gold leaching efficiency decreased when H2SO4 concentration was higher than 15%.
(4) For the calcine, after pre-leaching with 30% H2SO4 solution, gold leaching efficiency was 94.91%, which was 9.60% higher than that in direct cyanidation with a cyanidation time of 48 h. For the dust, through once cyanidation after pre-leaching with 15% H2SO4 solution and twice 2 mol/L NaOH solution pre-leaching and cyanidation, the total gold leaching efficiency achieved 91.90%, and the content of gold in final cyanidation tailing was 3.31 g/t.
References
[1] YANG Yong-bin, XIE Zhao-hui, XU Bin, LI Qian, JIANG Tao. Gold extraction from a high carbon low-grade refractory gold ore by flotation-roasting-leaching process [C]// NEELAMEGGHAM N R, SHAFIA A, OOSTERHOF H, ANIMESH J, DREISINGER D, WANG S J. Proceedings of the 144th Annual Meeting & Exhibition. Orlando, FL: TMS, 2015: 63-70.
[2] BAS A D, SAFIZADEH F, ZHANG W, GHALI E, CHOI Y. Active and passive behaviors of gold in cyanide solutions [J]. Transaction of Nonferrous Metals Society of China, 2015, 25: 3442-3453.
[3] ZHANG Shuang-hua, ZHENG Ya-jie, CAO Pan, LI Chao-hui, LAI Shen-zhi, WANG Xing-jun. Process mineralogy characteristics of acid leaching residue produced in low-temperature roasting-acid leaching pretreatment process of refractory gold concentrates [J]. International Journal of Minerals, Metallurgy and Materials, 2018, 25: 1132-1139.
[4] SNYDERS C A, AKDOGAN G, BRADSHAW S M, van VREDEN J H, SMITH R. The development of a caustic pre-leaching step for the recovery of Au from a refractory ore tailings heap [J]. Minerals Engineering, 2018, 121: 23-30.
[5] YANG Tian-zu, RAO Shuai, LIU Wei-feng, ZHANG Du-chao, CHEN Lin. A selective process for extracting antimony from refractory gold ore [J]. Hydrometallurgy, 2017, 169: 571-575.
[6] LI Qian, JI Fang-zhou, XU Bin, HU Jian-jian, YANG Yong-bin, JIANG Tao. Consolidation mechanism of gold concentrates containing sulfur and carbon during oxygen- enriched air roasting [J]. International Journal of Minerals, Metallurgy and Materials, 2017, 24: 386-392.
[7] DANG Xiao-e, KE Wen-shuai, TANG Chen, LV Jun, LIU Cheng-peng. Increasing leaching rate of gold cyanide of two-stage calcination generated from refractory ore containing arsenopyrite and pyrrhotite [J]. Rare Metals, 2016, 35: 804-810.
[8] de MICHELIS I, OLIVIERI A, UBALDINI S, FERELLA F, BEOLCHINI F, VEGLIO F. Roasting and chlorine leaching of gold-bearing refractory concentrate: Experimental and process analysis [J]. International Journal of Mining Science and Technology, 2013, 23: 709-715.
[9] GUO Xue-yi, XIN Yun-tao, WANG Hao, TIAN Qing-hua. Mineralogical characterization and pretreatment for antimony extraction by ozone of antimony-bearing refractory gold concentrates [J]. Transactions of Nonferrous Metals Society of China, 2017, 27: 1888-1895.
[10] ZHANG Xu, FENG Ya-li, LI Hao-ren. Enhancement of bio-oxidation of refractory arsenopyritic gold ore by adding pyrolusite in bioleaching system [J]. Transactions of Nonferrous Metals Society of China, 2016, 26: 2479-2484.
[11] KONYRATBEKOVA S S, BAIKONUROVA A, USSOLTSEVA G A, ERUST C, AKCIL A. Thermodynamic and kinetic of iodine�Ciodide leaching in gold hydrometallurgy [J]. Transactions of Nonferrous Metals Society of China, 2015, 25: 3774-3783.
[12] FOMCHENKO N V, KONDRAT'EVA T F, MURAVYOV M I. A new concept of the biohydrometallurgical technology for gold recovery from refractory sulfide concentrates [J]. Hydrometallurgy, 2016, 164: 78-82.
[13] GAO Guo-long, LI Deng-xin, ZHOU Yong, SUN Xian-hao, SUN Wen. Kinetics of high-sulphur and high-arsenic refractory gold concentrate oxidation by dilute nitric acid under mild conditions [J]. Minerals Engineering, 2009, 22: 111-115.
[14] LI Qian, ZHANG Yan, LI Xi-shan, XU Bin, YANG Yong-bin, JIANG Tao, LI Hong-wei. Intensification of gold leaching from a multi-refractory gold concentrate by the two-stage roasting-alkaline sulfide washing-cyanidation process [C]//HWANG J Y, JIANG Tao, KENNEDY M W, YUCEL O, PISTORIUS P C, SESHADRI V, ZHAO B J, GREGUREK D, KESKINKILIC E. Proceedings of the 8th International Symposium on High-temperature Metallurgical Processing. San Diego, CL: TMS, 2017: 449-457.
[15] XU Bin, YANG Yong-bin, JIANG Tao, LI Qian, ZHANG Xi, WANG Dan. Improved thiosulfate leaching of a refractory gold concentrate calcine with additives [J]. Hydrometallurgy, 2015, 152: 214-222.
[16] THOMAS K G, COLE A P. Roasting developments��Especially oxygenated roasting [J]. Developments in Mineral Processing, 2005, 15: 403-432.
[17] MA S J, LUO W J, MO W, SU X J, LIU P, YANG J L. Removal of arsenic and sulfur from a refractory gold concentrate by microwave heating [J]. Minerals Engineering, 2010, 23: 61-63.
[18] LI Yun, WANG Yun, YUAN Chao-xin, SUN Jian-wei. Study on improving gold extraction of two-stage roasting calcine from arsenic-bearing gold concentrate [J]. Nonferrous Metals: Extractive Metallurgy, 2010, 6: 33-36. (in Chinese)
[19] ZHANG Yue-hong, LI Yun, LIU Hong-xiao, LUAN Dong-wu, HUANG Hai-hui, WANG Yun. Technical transformation of two-stage roasting for refractory gold concentrates [J]. Nonferrous Metals: Extractive Metallurgy, 2015, 11: 36-38. (in Chinese)
[20] ALP I, CELEP O, PAKTUNC D, THIBAULT Y. Influence of potassium hydroxide pretreatment on the extraction of gold and silver from a refractory ore [J]. Hydrometallurgy, 2014, 146: 64-71.
[21] CELEP O, SERBEST V. Characterization of an iron oxy/hydroxide (gossan type) bearing refractory gold and silver ore by diagnostic leaching [J]. Transactions of Nonferrous Metals Society of China, 2015, 25: 1286-1297.
[22] ZHANG Hui-bin. Chemical phase analysis of ores and industrial products [M]. Beijing: Metallurgical Industry Press, 1992. (in Chinese)
[23] LU Yi-yuan, BIN Wan-da. Metallurgy of precious metals [M]. Changsha: Central South University Press, 2004. (in Chinese)
[24] DEAN J A. Handbooks of LAN-Shi chemistry [M]. 15th ed. New York: McGraw-Hill, Inc., 1998. (in Chinese)
[25] MCNAUGHT A D, WILKINSON A. Compendium of chemical terminology [M]. 2nd ed. Oxford: Blackwell Scientific Publications, 1997.
[26] PENG Ying-lin. Technology of copper electrolyte purification by arsenic valence-adjustment and its fundamental theory [D]. Changsha: Central South University, 2013. (in Chinese)
[27] LONG Hua, ZHENG Ya-jie, PENG Ying-lin, HE Han-bin. Recovery of alkali, selenium and arsenic from antimony smelting arsenic-alkali residue [J]. Journal of Cleaner Production, 2020, 251. https:doiorg/10.1016/j.jclepro.2019. 119673.
[28] HUNG Li-huang. Extraction technology of gold and silver [M]. Beijing: Metallurgical Industry Press, 2005. (in Chinese)
[29] MUBAROK M Z, WINARKO R, CHAERUN S K, RIZKI I N, ICHLAS Z T. Improving gold recovery from refractory gold ores through biooxidation using iron-sulfur-oxidizing/ sulfur-oxidizing mixotrophic bacteria [J]. Hydrometallurgy, 2017, 168: 69-75.
[30] TAN H, FENG D, LUKEY G C, van DEVENTER J S J. The behaviour of carbonaceous matter in cyanide leaching of gold [J]. Hydrometallurgy, 2005, 78: 226-235.
[31] PYKE B L, JOHNSTON R F, BROOKS P. The characterisation and behaviour of carbonaceous material in a refractory gold bearing ore [J]. Minerals Engineering, 1999, 12: 851-862.
[32] OFORI-SARPONG G, OSSEO-ASARE K. Preg-robbing of gold from cyanide and non-cyanide complexes: Effect of fungi pretreatment of carbonaceous matter [J]. International Journal of Mineral Processing, 2013, 119: 27-33.
[33] FU Hai-lu, JIA Cai-yun, CHEN Qiao-shan, CAO Xue-ting, ZHANG Xiao-min. Effect of particle size on the transformation kinetics of flue gas desulfurization gypsum to ��-calcium sulfate hemihydrate under hydrothermal conditions [J]. Particuology, 2018, 40: 98-104.
[34] LIU Rui, ZHANG Tong-lai, LI Yang, ZHOU Zun-ning. Effect of particle size on thermal decomposition of alkali metal picrates [J]. Thermochimica Acta, 2014, 583: 78-85.
[35] XU Bin, YANG Yong-bin, LI Qian, JIANG Tao, LIU Shi-qian, LI Guang-hui. The development of an environmentally friendly leaching process of a high C, As and Sb bearing sulfide gold concentrate [J]. Minerals Engineering, 2016, 89: 138-147.
[36] CELEP O, ALP I, DEVECI H. Improved gold and silver extraction from a refractory antimony ore by pretreatment with alkaline sulphide leach [J]. Hydrometallurgy, 2011, 105: 234-239.
[37] LORENZEN L. Some guidelines to the design of a diagnostic leaching experiment [J]. Minerals Engineering, 1995, 8: 247-256.
[38] ZHOU Shun-gui, CHEN Shan-shan, YUAN Yong, LU Qin. Influence of humic acid complexation with metal ions on extracellular electron transfer activity [J]. Scientific Reports, 2015, 5: 1-9.
[39] HARRIS D C. The Mineralogy of gold and its relevance to gold recoveries [J]. Mineralium Deposita, 1990, 25(S): s3-s7.
[40] MENG Yu-qun. A new extraction process of carbonaceous refractory gold concentrate [J]. Transaction of Nonferrous Metals Society of China, 2005, 15: 1176-1184.
[41] WANG Yong. Process study on recovery gold from sulfur concentrate containing gold and arsenic [D]. Changsha: Central South University, 2012. (in Chinese)
[42] LIU Kui, CHEN Qi-yuan, YIN Zhou-an, HU Hui-ping, DIING Zhi-ying. Kinetics of leaching of a Chinese laterite containing maghemite and magnetite in sulfuric acid solutions [J]. Hydrometallurgy, 2012, 125-126: 125-136.
[43] ZHENG Ya-jie. Study on new technology and fundumental theory of polyferric sulfate and phosphorous polyferric sulfate prepared from pyrite cinders [D]. Changsha: Central South University, 2002. (in Chinese)
[44] ZHOU Chong-song. Studies on adsorption and desorption of gold on activated carbon [D]. Chengdu: Chengdu University of Technology, 2005. (in Chinese)
[45] XU Tian-yun. Improving total recovery rate of gold by stage cyanide [J]. China Mining Engineering, 1982, 6: 17-22. (in Chinese).
�� ��1����˪��1��֣�Ž�1���Ӻ���1������־2�����о�2��̷ ��2
1. ���ϴ�ѧ ұ���뻷��ѧԺ����ɳ 410083��
2. �����ƽ������������ι�˾������ 835100
ժ Ҫ�����öԱȷ����о���ұ��ɰ���̳��軯����IJ��졣���������ֱ���軯ʱ��ɰ���̳��н�Ľ����ʷֱ�Ϊ85.31%��54.30%���顢̼�������������ʽ�ǵ������߽�����ʲ������Ҫԭ����NaOHԤ�������軯���𣬱�ɰ���̳��н���������ʷֱ�Ϊ87.70%��58.60%���к�Ԫ�ص��ѳ������Ԥ���������н����ʧ������������谭��ͬ�������Ԥ������ɰ���̳��н�Ľ����ʡ���H2SO4Ԥ������ɰ���̳��н���������ʷֱ�ﵽ94.96%��80.40%��̼�����Ӱ���DZ�ɰ���̳��н�����ʲ������Ҫԭ������Щ���죬����������˹��գ���ɰ���̳��н�Ľ����ʷֱ�ﵽ94.91%��91.90%��
�ؼ��ʣ���ұ��ɰ���̳����軯��̼����
(Edited by Wei-ping CHEN)
Foundation item: Project (201552) supported by the Special Project on the Strategic Emerging Industries of Xinjiang Uygur Autonomous Region, China; Project (2017SK2254) supported by the Key Research and Development Project of Hunan Province, China
Corresponding author: Ya-jie ZHENG; Tel: +86-731-88836285; E-mail: zyj@csu.edu.cn
DOI: 10.1016/S1003-6326(20)65354-7

