
Atomic simulation on evolution of nano-crystallizaion in amorphous metals
WANG Yu(Õı ”Ó), WANG Xiu-xi(Õı–„œ≤), WANG Hai-long(Õı∫£¡˙)
Key Laboratory of Mechanical Behavior and Design of Materials, Chinese Academy of Sciences,
University of Science and Technology of China, Hefei 230026, China
Received 10 April 2006; accepted 25 April 2006
Abstract: The deformation-induced nano-crystallization behavior of amorphous pure Ni was investigated by using a molecular dynamics simulation. The microevolution mechanism of the nano-crystallization, the crystallization process in the multicomponent amorphous Ni-Pd alloys and the temperature effect on the nano-crystallization behavior in amorphous metals were studied. The results show that the small nano-crystalline grain will nucleate and grow during the compression deformation. The deformation induces the growth of the ordered clusters in the amorphous metals and the nano-crystalline grain grows under the shearing combination and shearing deposition. The nano-crystalline grain will nucleate in a lower strain under a higher temperature. The combining severe plastic deformation with thermal annealing treatments presents a new opportunity for developing bulk nano-crystalline materials with controlled microstructures.
Key words: atomic simulation; evolution; nano-crystallization; amorphous metals
1 Introduction
Because of the breakthrough in preparation of bulk metallic glasses (BMGs) in 1990s[1,2], BMGs have been largely investigated due to their unique physical, chemical and mechanical properties being of special interest for a wide range of potential applications. However, BMGs display little or no plastic deformation at room temperature because the plastic flow in a BMG is governed by the formation of localized shear bands[3, 4]. It has been observed that the precipitation of ductile or nano-scale crystalline phases may be used to improve the mechanical behaviors of BMGs[5-7], since nano-crystals may disrupt shear-band propagation. Thermal annealing has been a typical method to induce nano-crystalline grain in an amorphous matrix. However, the formation of brittle intermetallic compounds may strongly degrade the toughness and the plasticity of the material.
Recently, nano-crystallization induced by plastic deformation has received increased attention since the formation of nano-crystals in metallic glass has been observed during different deformation processes such as bending, tension, ball-milling, nano-indentation and cold-rolling[7-12]. CHEN et al[9], using transmission electron microscopy (TEM), reported the formation of nano-crystallites at shear bands of Al-based amorphous alloys (Al90Fe5Gd5, Al90Fe5Ce5 and Al87Ni8.7Y4.3) bent at room temperature. High energy ball milling was also observed to lead to the formation of nano-crystallites in Al-based amorphous alloys[8]. GAO et al[8] observed nano-crystal precipitation in amorphous Al90Fe5Gd5 within vein protrusion on tensile fracture surfaces and along crack propagation paths, as well as within shear bands resulting from bending. According to all these experiments, combining severe plastic deformation of vitreous quenching products with thermal annealing treatments presents new opportunities for developing bulk nano-crystalline materials with controlled microstructures.
Molecular dynamics (MD) simulation is a very useful tool in materials research and has been widely used in studying the crystallization and glass formation processes of alloys during rapid solidification [13, 14] to provide an atomic description of the structural development in these processes. However, little work has been done to study the crystallization process of amorphous alloys during mechanical deformation and the associated crystal nucleation, grain growth with MD simulation. The crystallization process of amorphous alloys during plastic deformation is very complex. It usually involves nucleation and growth. It is therefore of interest to study this process with MD simulation.
The purpose of the present study is to investigate the nano-crystallization process of amorphous materials under compression deformation, by using the molecular dynamics simulation, in order to get an atomic description of the crystallization process and analyze the nucleation and grain growth process. The target materials are pure nickel and Ni-Pd alloy based on the EAM interatomic potential[15-17]. Special attention is paid to microstructure evolution in the nano-crystallization process of the amorphous materials. And the effects of the component and the temperature on the crystallization were also studied.
2 Simulation methods
2.1 Molecular dynamics
An amorphous nickel sample with size of 3.56 nm°¡3.56 nm°¡10.7 nm (in x, y and z direction), involving 12 000 atoms, was prepared using molecular dynamics based on EAM potential with fourth-order prediction-correction algorithm, by holding a single crystalline sample for several tens of pico-seconds over melting temperature and rapidly quenching to 10 K. To simulate a compression deformation, the atoms in the top end of the sample, which had a thickness of 0.5 nm in z direction, were shifted downwards along the z direction by 6°¡10-6 nm in each load step, with a time step of 3.5°¡10-15 s in each load step. The other end of the sample was fixed during the simulation. The whole simulation process included 500 000 steps. All the simulations were performed at an adiabatic procedure. During simulation, a periodic boundary condition was given to the x and y directions, while free surfaces were give in the z direction of the sample. The sample configuration is shown in Fig.1.
In a similar way, the amorphous materials Ni9Pd1 and Ni7Pd3 were created by rapid cooling from a melting liquid composition of Ni-Pd system, which include 10% Pd atoms and 30% Pd atoms, respectively. The other parameters during the simulation process were the same as the pure amorphous nickel one.
2.2 Pair analysis technique
The pair analysis technique was mainly developed by HONEYCUTT et al[18] to study the melting and freezing of atoms clusters and it has been used to monitor the structural change during rapid solidification process[19].

Fig.1 Computational model of pure amorphous nickel
In this technique, a pair of atoms are taken as a bond pair if their distance is smaller than a specified cut-off distance, which is chosen to be equal to the position of the first minimum in the corresponding radial distribution function. HONEYCUTT et al[18] used a sequence of four integers i, j, k, and l to show various atom pairs. For any pair of atoms A-B (root pair), if the two atoms form a bond, i=1; otherwise, i=2. The second integer j represents the number of common atoms, which form bonds with both atom A and atom B. The third integer k represents the number of bonds formed among the common atoms. The fourth integer l represents the way that the bonds formed among the common atoms. If these bonds link in turn, l=1; otherwise l=2. For example, the 1551 atom pair means that the two root pair atoms form a bond and they have five bonded common atoms that form five bonds among the common atoms.
The various atom pairs characterize different local structures. The 1551, 1541 and 1431 pairs are relati- vely numerous in liquid and amorphous states. The 1421 and 1422 pairs are found mostly in FCC-like and HCP- like local structures, while the 1441 and 1661 pairs are found greatly in bcc-like local structures. By analyzing the variations of the atoms pairs, information on the structural change of the atomic system can be obtained.
3 Results and discussion
The initial pure amorphous nickel sample is shown in Fig.1. The sample is obtained by rapidly cooling a liquid sample using the MD methods. Radial distribution function (RDF) is a ratio between the average atom number density at a distance r from any given atom and the same quantity from an atom in an ideal gas with the same overall density. For comparison, RDF for a crystalline sample of FCC Ni and an amorphous sample of Ni at 10 K are shown in Fig.2.
The peaks on the RDF are shown most clearly for a crystal (Fig.2(a)), where individual peaks correspond to the first, second nearest neighbor atoms and so on. The split of the second peak (in the range of 0.4-0.5 nm) of the amorphous sample (Fig.2 (b)) is a well-known feature in the RDF of metallic glasses[20]. In addition, the second peak in the RDF of crystalline sample is absent in the amorphous one. This is also a difference between the crystalline sample and amorphous nickel sample.

Fig.2 Radial distribution function (RDF) of pure nickel: (a) Crystalline sample; (b) Amorphous sample
In order to understand the deformation effect on the crystallization, the atom configurations in the x-z section under different strains are shown in Fig.3. It can be seen that the partial crystallization is observed when the unixial strain is about 23.07%. Plotting the RDF under different strains in Fig. 4, the special characteristic of split of the second peak (in the range of 0.4-0.5 nm) is weakened gradually with the strain increasing. Meanwhile a peak appears around 0.35 nm, which

Fig.3 Atom configurations in x-z section under different strains: (a) 4.03%; (b) 23.07%
represents the second nearest neighbor in the crystalline grain. From Figs.3 and 4, it can be concluded that the deformation induces the crystallization in the amorphous pure Ni.
With the methods of inversion, Fig.5 shows the evolution process of a general crystallographic plane that is selected from a crystalline grain in the final deformed configuration. When the strain is small (Figs.5(a) and (b)), the configuration is still in a disorder status. In
Fig.5(c), the strain is about 9.31%. Partial long-distance order structure appears in the plane, which is regarded as the precursor structure reported by SUTTON et al[21]. With increasing strain, single atom diffuses and adheres to the precursor nucleus to form an initial crystalline plane (Fig.5(d)). Then the initial crystalline plane absorbs the atoms from the amorphous segment by shearing combination and shearing deposition and a large crystalline plane is formed (Figs.5(e), (f), (g) and (h)). During the next load procedure, only the grain rotation is found and no obvious distortion appears.
From the evolution process of the microstructure of

Fig.4 Radial distribution function(RDF) of amorphous pure nickel under different strains

Fig.5 Evolution of a crystal plane in pure amorphous Ni under different strains: (a) 3.10%; (b) 6.20%; (c) 9.31%; (d) 12.41%; (e) 15.51%; (f) 18.61%; (g) 21.71%; (h) 24.82%; (i) 27.92%;(j) 31.02%
the crystalline plane, it can be thought that the deformation increases the energy of the atoms and overcomes the barrier of the crystallization in the amorphous metals. The short-distance order clusters in the amorphous structure increase gradually to form the initial crystalline nucleus with the atoms diffusing. And then, by shearing combination and shearing deposition, the nucleus grows to form a nano-crystalline grain. LU
et al[22] proposed a new approach on the micro-
mechanism for thermal crystallization of amorphous alloys, which is based on the experimental results of the in situ TEM observation of dynamic thermal crystallization in amorphous Ni-P alloy. They found that the crystallization is composed of two processes: the diffusion of single atoms from amorphous state to crystalline surface and the growth of ordered clusters and their shearing combination or shearing deposition, as shown in Fig.6. Our simulation results on the deformation

Fig.6 Schematic illustration of new developed crystallization micromechanism in one-dimension: (a) Growth process;
(b) Nucleation process
induced crystallization of amorphous metals also clearly describe the same microevolution process.
For comparison with the result of pure amorphous Ni sample, we also do simulation on the compression deformation of amorphous alloys Ni9Pd1 and Ni7Pd3 at 10 K. By using the pair analysis methods, we select the
1421 type and 1422 type atoms pairs, which are mostly found in FCC-like and HCP-like local structures, from three deformed samples when the unixial strain is 31.02%. As shown in Fig.7, it can be seen that there are several obvious nano-crystalline grains or sub-
nano-crystalline grains in the Ni sample. While in the alloys sample, few 1421 type and 1422 type atoms pairs appear. This indicates that it is easier to induce the nano-crystallization in the pure amorphous nickel by the external deformation than that in amorphous alloys. In fact, the number of the component increase, the barrier of the crystallization energy increases, the deformation induced crystallization would be more difficult. According to the molecular dynamics research on the melting and solidification of amorphous alloy, single component metal is difficult to form the amorphous materials by rapid cooling than the multicomponent alloy. This also means that the activation energy of crystallization of single-component amorphous metals is smaller than that of multicomponent amorphous alloys.
The temperature effect on the crystallization process of amorphous alloy, Ni9Pd1, under the unixial strain 31.02% is shown in Fig.8. It can be found that there are more 1421 type and 1422 type atom pairs in
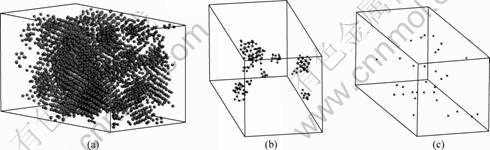
Fig.7 Crystallization status in three models under unixial strain of 31.02%: (a) Pure amorphous Ni; (b) Amorphous alloy Ni9Pd1;(c) Amorphous alloy Ni7Pd3
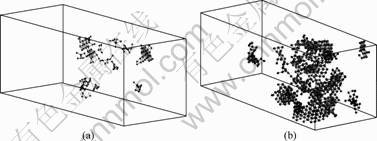
Fig.8 Temperature effect on crystallization in Ni9Pd1 samples under unixial strain of 31.02%: (a) 10 K; (b) 300 K
the 300 K samples than in the 10 K ones. Actually, with the increase of temperature, the movement of the atoms increases. This would speed up the diffusion and deposition in the crystalline/amorphous interface, which means there would be more crystalline structure in the sample at higher temperature than that at lower temperature. In the other aspects, the thermal energy would also help the nucleus pass through the energy barrier. More nuclei would be formed, which also enhances the crystallization in the amorphous materials. This means that there will be more crystalline grain at the higher temperature than at the lower temperature under the external deformation. Thus, if we combine severe plastic deformation with thermal annealing treatment methods, a new opportunity for developing bulk nano-crystalline materials with controlled microstructures will be present.
4 Conclusions
1) The small nano-crystalline grain will nucleate and grow during the compression deformation. The deformation induced the growth of the ordered clusters in the amorphous metals and the nano-crystalline grain grows under the shearing combination and shearing deposition.
2) There are fewer 1 421 and 1 422 type atom pairs in the multicomponent deformed sample, which means it°Øs more difficult to form a crystalline structure in the multicomponent amorphous materials by external deformation. The nucleus is easier to be formed at a higher temperature.
Acknowledgement
The authors are appreciative of the facilities offered by the Laboratory of Mechanical and Materials Science of USTC.
References
[1] INOUE A, ZHANG A T, KIM S G, MATSUMOTO T. Mg-Cu-Y amorphous alloys with high mechanical strengths produced by a metallic mold casting method[J]. Mater Trans JIM, 1991, 32: 609-616.
[2] INOUE A. High strength bulk amorphous alloys with low critical cooling rates[J]. Mater Trans JIM, 1995, 36: 866-875.
[3] XI X K, ZHAO D Q, PAN M X, WANG W H, WU Y, LEWANDOWSKI J J. Fracture of brittle metallic glasses: brittleness or plasticity[J]. Phys Rev Lett, 2005, 94: 125510-125513.
[4] ZHANG Z F, HE G, ECKERT J, SCHULTZ L. Fracture mechanisms in bulk metallic glassy materials[J]. Phys Rev Lett, 2003, 91: 045505-045508.
[5] HE G, ZHANG Z F, LOSER W, ECKERT J, SCHULTZ L. Effect of Ta on glass formation, thermal stability and mechanical properties of a Zr52.25Cu28.5Ni4.75Al9.5Ta5 bulk metallic glass[J]. Acta Materialia, 2003, 51: 2383-2395.
[6] LEE J G, PARK S S, LEE D G, LEE S, KIM N J. Mechanical property and fracture behavior of strip cast Zr-base BMG alloy containing crystalline phase[J]. Intermetallics, 2004, 12: 1125-1131.
[7] JIANG W H, ATZMON M. The effect of compression and tension on shear-band structure and nanocrystallization in amorphous Al90Fe5Gd5: a high-resolution transmission electron microscopy study[J]. Acta Materialia, 2003, 51: 4095-4105.
[8] GAO M C, HACKENBERG R E, SHIFLET G J. Deformation-induced nanocrystal precipitation in Al-base metallic glasses[J]. Materals Transactions, 2001, 42(8): 1741-1747.
[9] CHEN H, HE Y, SHIFLET G J, POON S J. Deformation-induced nano crystal formation in shear bands of amorphous alloys[J]. Nature, 1994, 367(10): 541-543.
[10] KIM J J, CHOI Y, SURESH S, ARGON A S. Nano crystallization during nano indentation of a bulk amorphous metal alloy at room temperature[J]. Science, 2002, 295: 654-657.
[11] HEBERT R J, PEREPEZKO J H. Effect of cold-rolling on the crystallization behavior of amorphous Al88Y7Fe5 alloy[J]. Mater Sci Eng A, 2004, 375-377: 728-732.
[12] BOUCHARAT N, HEBERT R, ROSNER H, VALIEV R, WILDE G. Nano crystallization of amorphous Al88Y7Fe5 alloy induced by plastic deformation[J]. Scripta Materialia, 2005, 53: 823-828.
[13] QI L, ZHANG H F, HU Z Q. Molecular dynamic simulation of glass formation in binary liquid metal: Cu-Ag using EAM[J]. Intermetallics, 2004, 12: 1191-1195.
[14] QI Y, CAGIN T, KIMURA Y, GODDARD W A III. Molecular dynamics simulations of glass formation and crystallization in binary liquid metals: Cu-Ag and Cu-Ni[J]. Phys Rev B, 1999, 59: 3527-3533.
[15] BASKES M I. Modified embedded atom potentials for cubic materials and impurities[J]. Phys Rev B, 1992, 46: 2727-2742.
[16] DAW M S, BASKES M I. Semiempirical, quantum mechanical calculation of hydrogen embrittlement in metals[J]. Phys Rev Lett, 1983, 50: 1285-1288.
[17] DAW M S, BASKES M I. Embedded-atom method: Derivation and application to impurities, surfaces, and other defects in metals[J]. Phys Rev B, 1984, 29: 6443-6453.
[18] HONEYCUTT J D, ANDERSEN H C. Molecular dynamics study of melting and freezing of small lennard-jones clusters[J]. J Phys Chem, 1987, 91: 4950-4963.
[19] PEI Q X, LU C, LEE H P. Crystallization of amorphous alloy during isothermal annealing: a molecular dynamics study[J]. J Phys: Condens Matter, 2005, 17: 1493-1504.
[20] LI Q, ZHANG H F, HU Z Q. Molecular dynamics simulation of melting and solidification in binary liquid metal: Cu-Ag[J]. Acta Metall Sinica, 2004, 40(7): 736-740.(in Chinese)
[21] SUTTON M, YANG Y S, MAINVILLE J, JORDAN-SWEET J L, LUDWIG K F, STEPHENSON G B. Observation of a precursor during the crystallization of amorphous NiZr2[J]. Phys Rev Lett, 1989, 62: 288-291.
[22] LU K, WANG J T. An approach to new micro-mechanism for crystallization of amorphous alloys[J]. Acta Metall Sinica, 1991, 27(1): B38-B43.(in Chinese)
(Edited by YANG You-ping)
Foundation item: Project(10502047) supported by the National Natural Science Foundation of China; Project (2005038166) supported by China Postdoctoral Science Foundation
Corresponding author: WANG Yu; Tel: +86-551-3601245; Fax: +86-551-3606459; E-mail: wyu@ustc.edu.cn