


Trans. Nonferrous Met. Soc. China 22(2012) 1146-1150
Effect of electrolytic MnO2 pretreatment on performance of as-prepared LiMn2O4
ZHAO Yu-qian1, JIANG Qing-lai2, WANG Wei-gang3, DU Ke3, HU Guo-rong3
1. School of Geosciences and Info-Physics, Central South University, Changsha 410083, China;
2. Powder Metallurgy Research Institute, Central South University, Changsha 410083, China;
3. School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China
Received 12 December 2011; accepted 23 February 2012
Abstract: To investigate the effect of electrolytic MnO2 (EMD) on the performance of LiMn2O4, several pretreatment methods, such as acid treating, presintering and impregnating with chromic salt, were used. The pretreated EMD and prepared LiMn2O4 were characterized by X-ray diffraction and inductively coupled plasma emission spectrometry. Charge and discharge tests of Li/LiMn2O4 batteries were also employed to evaluate electrochemical performance. The experimental results show that inorganic impurity contents in EMD decrease remarkably after acid treating; presintering EMD can remove adsorbent water and organic impurity, enlarge pore space and increase active reaction sites; pre-doping chromium in EMD can form more homogenous compound substance LiCr0.05Mn1.95O4, which shows better structural stability and capacity retention.
Key words: lithium-ion batteries; cathode material; LiMn2O4; MnO2
1 Introduction
Lithium-ion batteries, owing to their high energy and power density, have been used in a wide variety of portable electric devices, hybrid electric vehicles and many power supplies. The capacity of lithium-ion batteries is usually limited by cathode, and the cathode materials used in Li-ion batteries are mainly LiCoO2, LiNiO2, and LiMnO2, with layered structure, and their ramifications, spinel LiMn2O4, olivine LiFePO4, etc [1-3]. Among them, spinel LiMn2O4 is considered the preferred cathode material for lithium ion batteries because of its merits, such as low cost, high voltage platform, high-rate charge-discharge capacity, easy processing, and environmental friendliness [4-8]. However, its application is limited by its instability caused by Jahn-Teller effect and dissolution of manganese into electrolyte during the charge-discharge cycle [9,10]. To date, a variety of doping and coating techniques have been explored to control the structure and hence improve the electrochemical properties of LiMn2O4 [11].
Preparation methods and raw materials have great influence on the structure and performance of cathode materials. There are two methods for spinel LiMn2O4 preparation: solid-state method and wet-chemical method. Solid-state method has been adopted widely because of its simple process, easily controllable condition, and being suit for industrialization. In a typical solid-state method to produce LiMn2O4, EMD and Li2CO3 are chosen as the raw materials. Here, MnO2 can be looked as the frame of LiMn2O4 and its properties, such as structure, chemical composition, grain size, morphology, have great influence on the structure and performance of as-prepared spinel LiMn2O4 [12]. EMD is mostly produced to satisfy the demands for its role in disposable alkaline manganese battery. There are now few literatures for the researches on EMD pretreatment for the synthesis of LiMn2O4. In this work, the effect of EMD pretreatment on the performance of as-prepared LiMn2O4 is studied.
2 Experimental
2.1 Preparation of materials
In acid treating, EMD was immersed in 1 mol/L H2SO4 solution for 24 h and washed with purified water copiously, then dried under 120 °C. The obtained EMD was named as A-EMD.
In presintering, EMD was heated under 400 °C for 6 h. The obtained EMD was named as H-EMD.
In impregnating with chromic salt, EMD was immersed in 50% (CH3COO)3Cr solution for 10 d to make Cr3+ ions infuse into the EMD pores, dried, and then heated under 400 °C for 6 h. The obtained EMD was named as C-EMD.
Spinel LiMn2O4 was prepared by a solid-state reaction from a stoichiometric mixture of Li2CO3 (99.9% in mass fraction) and electrolytic MnO2 (Mn content of 59.6%) at 770 °C for 12 h. The as-prepared samples were named as A-EMD-LMO, H-EMD-LMO, and C-EMD-LMO from A-EMD, H-EMD, and C-EMD, correspondingly.
2.2 Characterization of materials
The phase identification of samples was carried out by powder X-ray diffraction (XRD, D/max 2550VB, Cu Kα, 40 kV, 250 mA, 10°-80°, Japan). Elementary content was analyzed by inductively coupled plasma emission spectrometry (ICP, Baird Corp. America).
The cathode materials were assembled into 2025-coin type cells to evaluate the electrochemical performance. The cathodes were prepared with slurry consisting of 80% (mass fraction) active materials, 10% conductive acetylene black and 10% polyvinylidene fluoride (PVDF) binder in N-methyl-2-pyrrolidone (NMP) solvent. This mixture was then tape-cast by a doctor blade on an aluminum foil current collector and dried at 120 °C for 12 h in a vacuum oven. The coated cathode foil was then cut into circular discs of 10 mm in diameter. The coin cells were assembled using 2025-stainless steel coin-type containers in an argon-filled glove box. Lithium foils were served as the anode and reference electrode, 1 mol/L LiPF6 dissolved in 1:1 in volume ratio ratio of ethylene carbonate and dimethyl carbonate (EC: DMC) was used as the electrolyte, and a thin polypropylene film (Celgard 2400) acted as the separator. The charge-discharge cycles were performed using LAND battery test system with a current density of 29.6 mA/g (0.2C) in the voltage range of 4.3-3.2 V at room temperature.
3 Results and discussion
3.1 Effect of pretreatment method on structure and chemical composition of EMD
Figure 1 shows the X-ray diffraction patterns of EMD, A-EMD and H-EMD. For the diffraction pattern of EMD without any treatment, some wide and symmetrical diffraction peaks of 2θ at about 22.1°, 37.2°, 42.6°, 56.3°, 67.4°, etc. can be observed. Compared with the diffraction peaks of ideal ramsdellite-MnO2, EMD shows a wider (130) peak with hiding in (021) peak at 2θ≈37°, which shows typical character of micro-twins defect. This structure of EMD belongs to γ-MnO2 type, which features in the disordered alternate growth of the unit cells of pyrolusite and ramsdellite [13] (shown in Fig. 2).
The structure of A-EMD in Fig. 1 shows no change after acid treating and belongs to typical γ-MnO2 type. But the structure of H-EMD, indicated as pyrolusite- MnO2, shows great change. The 2θ=22.1° peak shifts from low angle to high angle, and the peaks of 55°-59°and 64°-71° split obviously, which indicate that micro-twins defect decreased [14]. After being treated under 400 °C for 6 h, the space arrangement of the unit cells is more order and the void area is smaller, γ-MnO2 (pyrolusite and ramsdellite) is completely transformed into β-MnO2 (pyrolusite, shown in Fig. 2). Compared with EMD (γ-MnO2), it may be easier for H-EMD (β-MnO2) to transform into LiMn2O4. PISTOIA et al [15] believed that the force to propel Li-MnO2 transforming into spinel LiMn2O4 is the electrostatic interaction between Li ions and its adjacent octahedral Mn ions. Pyrolusite (β-MnO2) possesses a narrower void tunnel structure and shorter spacing distance between Li+ and Mn4-x, which is beneficial for the shift of Mn ions to void spaces and for the transformation into spinel structure.

Fig. 1 XRD patterns of EMD, A-EMD and H-EMD

Fig. 2 Structural change scheme of MnO2
The structure of C-EMD is complex, which can be observed from Fig. 3. After being impregnated with chromic salt and pretreated under high temperature, the impregnated Cr2+ in EMD is oxidized to Cr2O3, whose diffraction peaks can be observed in Fig. 3. At the same time, Mn4+ is partly reduced to lower valence state. Not only β-MnO2 but also a new phase Mn3O4 appears in the XRD patterns of C-EMD.

Fig. 3 XRD pattern of C-EMD
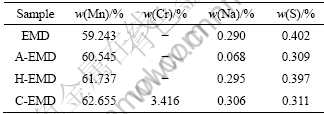
Table 1 shows the content analysis of major elements of EMD pretreated by different methods. Compared with the untreated EMD, the Na content in A-EMD decreases from 0.290% to 0.068%, with the reduced degree up to 76.66%. It owes to the exchange of Na+ and H+ and a mass of Na+ escaping from the pores of MnO2 during the acid treatment. Meanwhile, S content also decreases by 23.13%. And the decrease of impurities raises the Mn content from 59.243% to 60.545%. To H-EMD, the impurities contents, such as Na and S, remain almost unchanged, but the Mn content increases obviously. This is mainly due to the elimination of water and other organic impurities under high temperature. To C-EMD, the Mn content raises because MnO2 is partly reduced to Mn3O4.
Table 1 Elemental analyses of EMD pretreated by different methods
3.2 Effect of EMD pretreatment on structure and performance of LiMn2O4
The XRD patterns of LiMn2O4 samples prepared by the above different kinds of EMD are shown in Fig. 4. The diffraction peaks of all the samples belong to pure cubic spinel LiMn2O4 type, in which Li and Mn occupy the tetrahedral 8a and octahedral 16d sites, respectively. The calculated lattice parameters for EMD-LMO, A-EMD-LMO, H-EMD-LMO and C-EMD-LMO are 0.8226, 0.8231, 0.8226 and 0.8221 nm respectively.
The lattice parameters of EMD-LMO and H-EMD-LMO are very close. A-EMD-LMO shows the largest lattice parameters, which may be due to the decrease of impure Na content. HE [16] found that Na+ could substitute the tetrahedral Li+ in spinel structure and part of Li+ ions shift to the octahedral Mn3+, which forms [NadLi1-d]8a[LidMn2-d]16dO4. The impure Na+ decreases the Mn3+ content. So, the lattice shrinks and the parameters decrease. Reversely, the decrease of Na+ content in A-EMD-LMO increases the lattice parameters. No impurity phase of Cr is found in the pattern of C-EMD-LMO, which shows that solid solution LiCr0.05Mn1.95O4 is formed in the sample. Meanwhile, Cr3+ with a smaller ionic radius substitutes Mn3+ partly, which decreases the lattice parameters, and improves the stability of the structure [17].
Figure 5 shows the initial charge-discharge curves of LiMn2O4 obtained from EMD pretreated by different methods. As a reference, EMD-LMO has an initial discharge capacity of 118.7 mA・h/g. A-EMD-LMO has the highest discharge capacity of 125.5 mA・h/g because the Na+ content decreases after acid treating, which hence decreases cation mixing degree in spinel LiMn2O4 and increases the Li+ amount for deintercalation [18]. H-EMD-LMO has a capacity of 122.6 mA・h/g, which is a little higher than that of the untreated sample. And the capacity of C-EMD-LMO decreases to 110.9 mA・h/g mainly because Cr substitutes Mn sites partly, thus decreasing the Mn3+ amount and capacity of the spinel.

Fig. 4 XRD patterns of as-prepared samples
Figure 6 shows the cycleability curves of LiMn2O4 obtained from EMD pretreated by different methods. As for the LiMn2O4 samples from the untreated EMD, A-EMD, H-EMD and C-EMD, the capacity retention ratios after 40 cycles under room temperature keeps 82%, 74.9%, 84.5% and 93.1% respectively. A-EMD-LMO has the highest discharge capacity but the worse cycleability, which may be due to the decrease of Na content in EMD. MITSUI and PANASONIC believed that the residual Na in the EMD is beneficial to the cycleability of as-prepared LiMn2O4 [19]. C-EMD-LMO has the best cycleability mainly because the impregnated Cr3+ shrinks the lattice and improves the stability of the structure [20,21]. Meanwhile, the decrease of Mn3+ is beneficial to the suppression of Jahn-Teller distortion and improvement of the cycleability of as-prepared LiMn2O4.

Fig. 5 Initial charge-discharge curves of as-prepared samples

Fig. 6 Cycling performance of as-prepared samples
4 Conclusions
1) Pretreatment of EMD has a certain effect on the as-prepared LiMn2O4: acid treating can decrease the contents of impurities and increase the initial discharge capacity, but the cycleability is deteriorated.
2) After presintering, the water and content of organic impurities infused into EMD decrease and its crystal structure transforms from γ-type to β-type, which decreases the crystal defect and is beneficial to the formation of spinel structure, and therefore, the structural stability and electrochemistry performance of the as-prepared LiMn2O4 are improved.
3) To C-EMD, the initial discharge capacity decreases, but the stability and cycleability of the as-prepared LiMn2O4 improve greatly.
References
[1] TANG S B, LAI M O, LU L. Li-ion diffusion in highly (003) oriented LiCoO2 thin film cathode prepared by pulsed laser deposition [J]. Journal of Alloys and Compounds, 2008, 449(1-2): 300-303.
[2] Nimisha C S, Ganapathi M, Munichandraiah N, Rao G M. Study on target conditioning for depositing LiCoO2 thin films [J]. Vacuum, 2009, 83(6): 1001-1006.
[3] KIM C J, AHN I S, CHO K K, LEE S G, CHUNG J K. Characteristics of LiNiO2 thin films synthesized by Li diffusion on the surface oxidized epitaxial layer of Ni-alloy [J]. Journal of Alloys and Compounds, 2008, 449(1-2): 335-338.
[4] FERGUS J W. Recent developments in cathode materials for lithium ion batteries [J]. Journal of Power Sources, 2010, 195(4): 939-954.
[5] GOODENOUGH J B. Cathode materials: A personal perspective [J]. Journal of Power Sources, 2007, 174(2): 996-1000.
[6] CHEN Ze-hua, HUANG Ke-long, LIU Su-qin, WANG Hai-yan. Preparation and characterization of spinel of LiMn2O4 nanorods as lithium-ion battery cathodes [J]. Transactions of Nonferrous Metals Society of China, 2010, 20(12): 2309-2313.
[7] MICHALSKA M, LIPINSKA L, MIRKOWSKA M, AKSIENIONEK M, DIDUSZKO R, WASUCIONEK M. Nanocrystalline lithium-manganese oxide spinels for Li-ion batteries ―Sol-gel synthesis and characterization of their structure and selected physical properties [J]. Solid State Ionics, 2011, 188(1): 160-164.
[8] XIONG L, XU Y, TAO T, GOODENOUGH J B. Synthesis and electrochemical characterization of multi-cations doped spinel LiMn2O4 used for lithium ion batteries [J]. Journal of Power Sources, 2012, 199(1): 214-219.
[9] ZHOU W J, BAO S J, HE B L, LIANG Y Y, LI H L. Synthesis and electrochemical properties of LiAl0.05Mn1.95O4 by the ultrasonic assisted rheological phase method [J]. Electrochimica Acta, 2006, 51(22): 4701-4708.
[10] XIAO L F, ZHAO Y Q, YANG Y Y, CAO Y L, AI X P, YANG H X. Enhanced electrochemical stability of Al-doped LiMn2O4 synthesized by a polymer-pyrolysis method [J]. Electrochimica Acta, 2008, 54(2): 545-550.
[11] LI Xiang-qun, WANG Zhi-xing, LIANG Ru-fu, GUO Hua-jun, LI Xin-hai, CHEN Qi-yuan. Electrochemical properties of high-power lithium ion batteries made from modified spinel LiMn2O4 [J]. Transactions of Nonferrous Metals Society of China, 2009, 19(6): 1494-1498.
[12] JIANG Qing-lai, HU Guo-rong, PENG Zhong-dong, DU Ke, LIU Ye-xiang. Influence of MnO2 materials on performance of spinel LiMn2O4 prepared by solid state method [J]. Journal of Functional Materials, 2010, 41(9): 1485-1489. (in Chinese)
[13] XIA Xi. Crystal structure, preparation and discharge performance for manganese dioxides and related manganese oxides (I) [J]. Battery Bimonthly, 2004, 34(6): 411-414. (in Chinese)
[14] CHABRE Y, PANNETIER J. Structural and electrochemical properties of the proton γ-MnO2 system [J]. Progress in Solid State Chemistry, 1995, 23(1): 1-130.
[15] PISTOIA G, ANTONINI A, ZANE D, PASQUALI M. Synthesis of Mn spinels from different polymorphs of MnO2 [J]. Journal of Power Sources, 1995, 56(1): 37-43.
[16] HE Fang-yong. Study on synthesis and properties of doped lithium manganese oxide [D]. Changsha: Central South University, 2007. (in Chinese)
[17] PENG Z D, JIANG Q L, DU K, WANG W G, HU G R, LIU Y X. Effect of Cr-sources on performance of Li1.05Cr0.04Mn1.96O4 cathode materials prepared by slurry spray drying method [J]. Journal of Alloys and Compounds, 2010, 493(1-2): 640-644.
[18] GUO Hua-jun, LI Xiang-qun, HE Fang-yong, LI Xin-hai, WANG Zhi-xing, PENG Wen-jie. Effects of sodium substitution on properties of LiMn2O4 cathode for lithium ion batteries [J]. Transactions of Nonferrous Metals Society of China, 2010, 20(6): 1043-1048.
[19] NAGAYAMA M, ARIMOTO S, NUMATA K. Production method of spinel lithium manganese oxides, the cathode materials and nonaqueous secondary battery: CN Patent, 99801152.5 [P]. 1999-06-08. (in Chinese)
[20] YU Ze-min, ZHAO Lian-cheng. Preparation and electrochemical properties of LiMn1.95M0.05O4 (M=Cr, Ni) [J]. Rare Metals, 2007, 26(1): 62-67.
[21] JIANG Qing-lai, HU Guo-rong, PENG Zhong-dong, DU Ke, CAO Yan-bing, TANG Dai-chun. Preparation of spherical spinel LiCr0.04Mn1.96O4 cathode materials based on slurry spray drying method [J]. Rare Metals, 2009, 28(6): 618-623.
电解二氧化锰预处理对制备锰酸锂性能的影响
赵于前1,蒋庆来2,王伟刚3,杜 柯3,胡国荣3
1. 中南大学 地球科学与信息物理学院,长沙 410083;
2. 中南大学 粉末冶金研究院,长沙 410083;
3. 中南大学 冶金科学与工程学院,长沙 410083
摘 要:分别采用酸洗、预烧、浸渍掺铬的方式对电解二氧化锰(EMD)进行预处理,研究EMD预处理对制备锰酸锂性能的影响。采用XRD、ICP等手段对预处理的EMD及制备的锰酸锂进行表征,并通过Li/LiMn2O4电池的充放电测试对其电化学性能进行评估。结果表明,酸洗后EMD中的钠、硫等无机杂质含量显著降低;预烧能够有效去除EMD吸附的水分和有机杂质,扩大孔径,增多反应活性位点;对EMD进行浸渍掺铬的预处理,能够得到更加均质的掺铬锰酸锂材料LiCr0.05Mn1.95O4,并表现出较好的结构稳定性及容量保持率。
关键词:锂离子电池;正极材料;锰酸锂;二氧化锰
(Edited by YANG Hua)
Foundation item: Project (61172184) supported by the National Natural Science Foundation of China; Project (2007BAE12B01) supported by the National Key Technology R&D Program of China
Corresponding author: ZHAO Yu-qian; Tel: +86-731-88796789; E-mail: zhaocsu@hotmail.com
DOI: 10.1016/S1003-6326(11)61297-1