
Microstructure and properties of biodegradable
��-TCP reinforced Mg-Zn-Zr composites
HE Sheng-ying1, SUN Yue1, CHEN Min-fang1, 2, LIU De-bao1, 2, YE Xin-yu1
1. School of Materials Science and Engineering, Tianjin University of Technology, Tianjin 300384, China;
2. Tianjin Key Laboratory for Photoelectric Materials and Devices, Tianjin 300384, China
Received 23 September 2010; accepted 25 December 2010
Abstract: Magnesium alloys have good biocompatibility, but their mechanical properties and corrosion resistance may not be satisfied for using as degradable materials within bone due to its high corrosion rate in the physiological environment. Nano ��-TCP particles were added into Mg-Zn-Zr alloy to improve its microstructure and the properties. As-extruded Mg-3Zn-0.8Zr alloy and Mg-3Zn-0.8Zr/x��-TCP (x=0.5%, 1.0% and 1.5%) composites were respectively fabricated. The grains of Mg-Zn-Zr/��-TCP composites were significantly refined. The results of the tensile tests indicate that the ultimate tensile strength and the elongation of composites were improved with the addition of ��-TCP. The electrochemical test result in simulation body fluid shows that the corrosion resistance of the composites was strongly enhanced comparing with that of the alloy. The corrosion potential of Mg-3Zn0.8-Zr/1.0��-TCP composite is -1.547 V and its corrosion current density is 1.20��10-6 A/cm2.
Key words: ��-tricalcium phosphate; magnesium composite; microstructure; mechanical properties; biodegradation
1 Introduction
Biodegradable materials are very salutary in medical application[1]. They play a temporary supportive role in the restoration of tissues and can degrade gradually after healing. Therefore, they can be used as permanent implants, which may not cause mismatches between the implant and the body nor physiological inflammatory, and a secondary surgery is also unnecessary. At present, most biodegradable implants are polymer, and some metallic implants are appearing[2]. Taking the mechanical strength into account, the latter ones have more advantages. Magnesium alloys are dissolvable in aqueous solution, especially in that containing chloride ions due to their low corrosion potential[3-4]. Thus, they are generally considered as promising materials for biodegradable orthopaedic implants[5] and vascular stents[6]. Recently, a series of Mg-based biodegradable implants were developed[7-13]. Furthermore, a biodegradable Mg stent was used in clinical experiments, which demonstrated the feasibility of Mg based vascular stents[14]. However, the high corrosion rate of Mg alloys results in rapid consumption of hydrogen[13] and increasing pH value, which badly affects the pH-dependent physiological process around the implant. Moreover, the implants with high corrosion rate lose their mechanical integrity before the tissues get entirely healed. Previous studies showed that purification could reduce the corrosion rate of Mg. However, the application of pure Mg in orthopedics and the repairing of load-bearing bone is confined because of its low yield strength[13]. Therefore, it is necessary to develop Mg alloys with good corrosion resistance and high strength for the application in biomedical engineering.
Alloying elements can be added to improve the mechanical properties and the corrosion properties of pure Mg. With the aim of maintaining the biocompatibility and biodegradability of Mg alloys, SONG[13] studied the in vitro corrosion rates of several binary Mg alloys, and indicated that Ca, Mn and Zn could be suitable selections. Recently, some investigations demonstrated that Mg-Zn[9] alloy could gradually degrade within tissues and had good biocompatibility in vivo. Zr is confirmed to have a series of suitable properties to be used as metallic biomaterials such as low density, high corrosion resistance and biocompatibility[15]. Furthermore, adding Zr element into Mg-Zn alloys can effectively refine their grains. Mg alloys containing Zn and trace amount of Zr were chosen in this study to fabricate Mg-Zn-Zr alloys.
Recent research indicated that the addition of ceramic reinforcement can enhance the mechanical and corrosion resistance properties of Mg alloys[11, 16]. ��-tricalcium phosphate (��-TCP) is a bioactive material which has good biocompatibility and biodegradability. Moreover, the degrading of ��-TCP can offer abundant calcium and phosphorus elements to the osteoblasts, and then promotes the formation of new bone. Nowadays, the application of ��-TCP in composites focused on the fracture fixation material using polylactice acid as matrix and ��-TCP particles as reinforcement[17-20]. Based on the aforementioned considerations, ��-TCP particles are selected to improve the corrosion resistance and the mechanical properties of Mg-Zn-Zr alloys.
In this study, Mg-Zn-Zr based composites reinforced with different contents of ��-TCP were successfully synthesized, and the influence of ��-TCP on the microstructure and the properties of Mg-Zn-Zr alloy was investigated.
2 Experimental
2.1 Material preparation
High purity Mg (99.99%), Zn (99.99%), Mg-30Zr(mass fraction, %) alloy and nano ��-TCP particles with a diameter of about 100 nm were used to prepare Mg-3Zn-0.8Zr alloy and Mg-3Zn-0.8Zr/x��-TCP (x=0.5, 1 and 1.5, mass fraction, %) composites. The nano ��-TCP particles were synthesized by chemical reactions in the aqueous solutions of Ca(NO3)2 and (NH4)2HPO3, respectively. The chemical deposition was carried out at 25 ��C, and the pH value of the aqueous solutions was controlled to be about 8 by adding NaOH solution during the course of the reaction. The chemical compositions of the melted Mg-Zn-Zr alloy and Mg-Zn-Zr/x��-TCP composites are listed in Table 1.
The melting was carried out in a vacuum inductive furnace (ZG-10) under the protection of argon gas. When the temperature rose to 780 ��C, it was kept in a magnetic stirring for about 10 min, then the melt was cast into ingots with a diameter of 60 mm at about 680 ��C. After a homogenizing annealing at 420 ��C for 13 h, the ingots were extruded at 400 ��C into bars with an extrusion ratio of 56. Then the extruded bars were aged at 150 ��C for 24 h.
Table 1 Compositions of samples (mass fraction, %)

2.2 Microstructure and mechanical properties
The samples with a size of d8 mm �� 3 mm were cut from the extruded bars of Mg-Zn-Zr alloy and Mg-Zn-Zr/��-TCP composites respectively for microstructure observation. All samples were polished by SiC papers up to 1000 grit, and then ultrasonically cleaned in the absolute ethanol and distilled water for 5 min each, and etched in a solution of 5 g of picric acid, 0.5 mL of acetic acid, 5 mL of distilled water and 25 mL of ethanol, then ultrasonically cleaned in the alcohol for 5 min. Microstructure observation was conducted on a optical microscope (OLYMPUS U-TV0.5XC-3) and a scanning electron microscope (FE-SEM, JSM-6700F) equipped with energy dispersive spectrum (EDS). X-ray diffraction analysis (XRD��D/MAX-2000PC) was used to examine the phases of the alloy and composites.
Mechanical testing samples were cut from the extruded bars along the extrusion direction, with a size of d20 mm�� 4 mm. Tension tests were carried out on a WDW-100 electron universal testing machine with a strain rate of 1 mm/min. For each type of the samples, three tests were conducted. A sclerometer (Japan HMV-2T) was used to mensurate the Vickers hardness of the samples with an utmost load of 9.8 N and a loading time of 20 s.
2.3 Electrochemical measurements
The electrochemical measurements of the samples were performed in simulated body fluid (SBF) at 37 ��C in a three electrode measurement system (CHI660C), in which a saturated calomel electrode (SCE) was used as the reference, a graphite electrode as counter electrode and the sample as the working electrode. Wafer specimens for the electrochemical test were encapsulated into epoxy resin with a surface of 10 mm��10 mm exposed to the SBF. The testing surface was polished by SiC papers up to 1000 grit, and then ultrasonically cleaned in the absolute ethanol and distilled water for 5 min each. Potentiodynamic polarization curves were measured at a scan rate of 10 mV/min.
3 Results and discussion
3.1 Microstructure and mechanical properties
Normally, as for the composites, the grain refinement can be achieved by adding ceramic particles to the Mg alloys. The results show that the average grain size of the as-cast Mg-3Zn-0.8Zr/1.0��-TCP composite is much smaller than that of the as-cast Mg-3Zn-0.8Zr alloy as shown in Fig.1. Furthermore, the grain size systematically decreases with increasing the amount of ��-TCP from about 80 ��m (Fig.1(a)) to about 30 ��m (Fig.1(b)). The result demonstrates that the presence of ��-TCP particles has a positive influence on the refinement of grain size. Referring to the classical theory of heterogeneous nucleation, the refinement of grain size can be explained by the addition of ��-TCP particles. With the addition of ��-TCP particles, the solid/liquid interfacial energy is reduced and the embryo turns into the shape of a spherical cap with a wetting angle ��. Therefore, for a given volume of solid, the energy barrier to nucleation is decreased and thereby the nuclei can be formed at higher undercooling. In other words, heterogeneous nuclei can form at a higher temperature than homogeneous nuclei. It is obvious that the grains of samples are refined after hot-extruding with a large extrusion ratio (see Fig.2).

Fig.1 OM images of as-cast samples: (a) Sample 1; (b) Sample 3
Figure 3 shows the XRD patterns of the ��-TCP powder, Mg-Zn-Zr alloy and sample 3. The XRD pattern of ��-TCP shows that all the strong peaks of ��-TCP appear in the range from 10�� to 42��. By analyzing the XRD pattern of Mg-Zn-Zr alloy, it is found that all the peaks are caused by the Mg matrix phase and the MgZn2 second phase. In the diffraction pattern of Mg-3Zn-0.8Zr/1.0��-TCP composite, the peak of ��-TCP is not apparent, because the amount of the ��-TCP is little. The result of the XRD analysis demonstrates that no other phases are detected from the reaction between the matrix and the reinforcement.

Fig.2 OM images of extruded samples: (a) Sample 1; (b) Sample 3
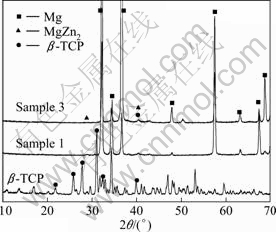
Fig.3 XRD patterns of ��-TCP powders, Mg-Zn-Zr alloy (sample 1) and Mg-Zn-Zr/1.0��-TCP composite (sample 3) in as-extruded state
Figure 4 shows the SEM images of Mg-Zn- Zr/��-TCP composites with different ��-TCP contents. It can be observed that ��-TCP particles distribute along the grain boundaries and within the grains of the alloy matrix phase, and a small amount of clustering ��-TCP particles can be observed in the composite. Their corresponding EDX results are also shown in Fig.4, which demonstrates the presence of ��-TCP particles in composites.

Fig.4 SEM images and corresponding EDX analysis for marked points of samples: (a, b) Sample 2; (c, d) Sample 3; (e, f) Sample 4
Previous researches demonstrated that the reinforcement with a micron dimension can improve the tensile strength of the Mg-based composite, but its plasticity would decline[21]. When the size of reinforcement particle is reduced to nanometer scale, the tensile strength and plasticity of the composites would be both enhanced[22], which agrees with that of this experiment. As shown in Fig.5, the tensile strength and plasticity of sample 3 are enhanced by 35 MPa and 1.87%, respectively, compared with those of sample 1. It is generally believed that nanoparticles can be used as heterogeneity nucleus to promote the formation nuclei of ��-��g and limit the growth of the grains. In the process of deformation, the existence of grain boundaries hinders the dislocations from slipping, resulting in dislocation accumulating. So it is needed to increase the stress for the macroscopical plastic deformation. From above, it can be concluded that the grain refinement can impede dislocation movement, and then increase the strength of the material. Furthermore, the MgZn2 phase formed during aging and ��-TCP particles distributing along the grain boundaries can be considered strengthening phases which are used as barrels to prevent dislocations from moving and enhance the strength of matrix. With the addition of ��-TCP ceramic particles, the grains of Mg-Zn-Zr matrix are refined, in which the formation and expansion of crack are not apt to occur, that is the reason why the material with a finer grain size has a higher plasticity. However, when the content of ��-TCP is more than 1.0%, the elongation of composite is decreased by 27%. It can be explained by the enhanced agglomeration of ��-TCP particles and the formation of pores and defects. The mechanical properties demonstrate that the composite with a suitable quantity of ��-TCP particles is adapted to biomedical application.
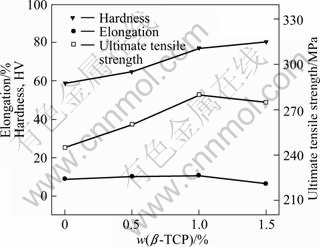
Fig.5 Average tensile properties of samples with different ��-TCP content
3.2 Electrochemical measurements
The electrochemical polarization curves of the alloy and composites in the SBF are shown in Fig.6. Normally, anodic polarization curves reveal the corrosion behavior of Mg alloys. A higher corrosion potential and a lower corrosion current density in the polarization curve indicate a lower average corrosion rate of the material. Table 2 lists the corrosion potential ��corr and the corrosion current density Jcorr which are derived from Fig.6. As for sample 3, the corrosion potential is heightened by about 100 mV compared with sample 1 due to the incorporation of ��-TCP, and it significantly reduces the corrosion current density of sample 1. It is demonstrated that sample 3 has a better corrosion resistance. Previous study showed that the corrosion mechanism of ZK60 alloys is a typical micro-galvanic corrosion using ��-Mg phase as anode and MgZn phase as cathode[23]. The grain size of composites was refined by adding ��-TCP particles, and the organizational structure became more uniform as well, therefore, the activity of micro-galvanic battery consisting of matrix and second phase decreased. It indicates that the ��-TCP particles can effectively increase the corrosion resistance of the matrix and control the formation of pitting corrosion. Moreover, the addition of ��-TCP particles increases the density of Mg(OH)2 layer, which can slow down the corrosion of samples. It is feasible that the corrosion rate of Mg-Zn-Zr/��-TCP can be adjusted by adding suitable content of ��-TCP particles, further investigation is needed.

Fig.6 Polarization curves of samples in SBF at 37 ��C
Table 2 Electrochemical parameters of samples

4 Conclusions
1) Mg-3Zn-0.8Zr/x��-TCP composites with favorable mechanical and corrosive properties were successfully prepared as a biomedical degradable material.
2) The ��-TCP particles distribution in the composites is homogeneous. Moreover, the composites present a higher ultimate tensile strength compared with the Mg-Zn-Zr alloy due to the fine grain size and the accumulating of the dislocation.
3) With the increase of nano ��-TCP, the corrosion potential of the composites increases, which means a superior corrosion resistance of the composites is obtained. It is expected that the Mg-Zn-Zr/��-TCP composites can be used as a candidate material at biodegradable implant application due to their appropriate microstructure and properties.
References
[1] LEVESQUE J, DUBE D, FISET M, MANTOVANI D. Materials and properties for coronary stents [J]. Adv Mater Process, 2004, 162(9): 45-48.
[2] MANI G, FELDMAN M D, PATEL D, AGRAWAL M C. Coronary stents: A materials perspective [J]. Biomaterials, 2007, 28(9): 1689-1710.
[3] SONG G L, ATRENS A, St JOHN D, NAIRN J, LI Y. The electrochemical corrosion of pure magnesium in 1 N NaCl [J]. Corros Sci, 1997, 39(5): 855-875.
[4] SONG G L, ATRENS A, St JOHN D, WU X, NAIRN J. The anodic dissolution of magnesium in chloride and sulphate solutions [J]. Corros Sci, 1997, 39(10-11): 1981-2004.
[5] STAIGER M P, PIETAK A M, HUADMAI J, DIAS G. Magnesium and its alloys as orthopedic biomaterials: A review [J]. Biomaterial, 2006, 27(9): 1728-1734.
[6] HEUBLEIN B, ROHDE R, KAESE V, NIEMEYER M, HARTUNG W, HAVERICH A. Biocorrosion of magnesium alloys: a new principle in cardiovascular implant technology [J]. Heart, 2003, 89(6): 651-656.
[7] WITTE F, KAESE V, HAFERKAMP H, SWITZER E, MEYER- LINDENBERG A, WIRTH C J, WINDHAGEN H. In vivo corrosion of four magnesium alloys and the associated bone response [J]. Biomaterials, 2005, 26(17): 3557-3563.
[8] WITTE F, FISCHER J, NELLESEN J, CROSTACK H A, KAESE V, PISCH A, BECKMANN F, WINDHAGEN H. In vitro and in vivo corrosion measurements of magnesium alloys [J]. Biomaterials, 2006, 27(7): 1013-1018.
[9] ZHANG Shao-xiang, ZHANG Xiao-nong, ZHAO Chang-li, LI Jia-nan, SONG Yang, XIE Chao-ying, TAO Hai-rong, ZHANG Yan, HE Yao-hua, JIANG Yao, BIAN Yu-jun. Research on an Mg-Zn alloy as a degradable biomaterial [J]. Acta Biomaterialia, 2010, 6(2): 626-640.
[10] KIM W C, KIM J G, LEE J Y, SEOK H K. Influence of Ca on the corrosion properties of magnesium for biomaterials [J]. Materials Letters, 2008, 62(25): 4146-4148.
[11] WITTE F, FEYERABEND F, FISCHER J, MAIER P, STORMER M, BLAWERT C, DIETZEL W, HORT N. Biodegradable magnesium�Chydroxyapatite metal matrix composites [J]. Biomaterials, 2007, 28(13): 2163-2174.
[12] ZHANG Er-lin, HE Wei-wei, DU Hui, YANG Ke. Microstructure, mechanical properties and corrosion properties of Mg-Zn-Y alloys with low Zn content [J]. Materials Science and Engineering A, 2008, 488(1-2): 102-111.
[13] SONG Guang-ling. Control of biodegradation of biocompatible magnesium alloys [J]. Corros Sci, 2007, 49(4): 1696-1701.
[14] ZARTNER P, CESNJEVAR R, SINGER H, WEYAND M. First successful implantation of a biodegradable metal stent into the left pulmonary artery of a preterm baby [J]. Catheterization and Cardiovascular Interventions, 2005, 66(4): 590-594.
[15] BRANZOI L V, IORDOC M, CODESCU M. Electrochemical studies on the stability and corrosion resistance of new zirconium-based alloys for biomedical applications [J]. Surface and Interface Analysis, 2008, 40(3-4): 167-173.
[16] LIU De-bao, CHEN Min-fang, WANG Xiao-wei. Fabrication and corrosion biodegradable properties of the HA/Mg biocomposite [J]. Rare Metal Materials and Engineering, 2008, 37(12): 2201-2205. (in Chinese)
[17] LI Xin-song, CHU Jun, ZHU Guo-hua, QI Xin-sheng, PU Yue-pu. In vivo degradation and tissue compatibility of poly-L-lactide/ ��-tricalcium phosphate composite rods for internal fixation of bone fractures [J]. Journal of Biomedical Engineering, 2007, 24(1): 81-86. (in Chinese)
[18] DI Li-zhi, ZHAO Hong. Preparation and orthopaedic application of ��-tricalcium phosphate [J]. Journal of Tianjin Institute of Technology, 2003, 19(2): 102-105. (in Chinese)
[19] LUO Lin, KANG Yun-qing, YIN Guang-fu, DONG Gang. Research on in vitro degradation of ��-TCP/PLLA internal fracture fixation composite material [J]. New Chemical Materials, 2006, 34(5): 57-60. (in Chinese)
[20] SHI Xiao-lin, XIE Huai-qin, ZHANG Zhen-yu. Preparation of in vivo degradable material used in bone fixation [J]. Journal of Beijing University of Aeronautics and Astronautics, 2003, 29(2): 144-146. (in Chinese)
[21] GUPTA M, LAI M O, SARAAVANARANGANATHAN D. Synthesis, microstructure and properties characterization of disintegrated melt deposited Mg/SiC composites [J]. Journal of Materials Science, 2000, 35(9): 2155-2165.
[22] ZHANG Z, CHEN D L. Consideration of Orowan strengthening effect in particulate-reinforced metal matrix nanocomposites: A model for predicting their yield strength [J]. Scripta Materialia, 2006, 54(7): 1321-1326.
[23] WANG Ping, LI Jian-ping, MA Qun. Effects of gadolinium on the microstructure and corrosion resistance properties of ZK60 magnesium alloy [J]. Rare Metal Materials and Engineering, 2008, 37(6): 1056-1059. (in Chinese)
����ɽ���Mg-Zn-Zr/��-TCP���ϲ�����֯�ṹ������
��ʤӢ1, �� ��1, ����1, 2, ���±�1, 2, Ҷ����1
1. ���������ѧ ���Ͽ�ѧ�빤��ѧԺ����� 300384��
2. ����й����ʾ�����������ص�ʵ���ң���� 300384
ժ Ҫ��þ�Ͻ������������������ԣ��������������й���ĸ�ʴ����������Լ���Ϊ�ɽ���ֲ����ϡ����⣬þ�Ͻ����ѧ����Ҳ�ϵ͡�ͨ���������צ�-TCP����������Mg-Zn-Zr�Ͻ������֯�����ܣ��Ʊ���ѹ̬��Mg-3Zn-0.8Zr�Ͻ��Mg-3Zn-0.8Zr/x��-TCP (x=0.5, 1.0, 1.5)���ϲ��ϡ��������צ�-TCP��ǿ��ĸ��ϲ����侧������ϸ��������ʵ�������������Ӧ�-TCP���ϲ��ϵļ�������ǿ�Ⱥ��쳤�ʾ�������ߡ��绯ѧ���Խ�����������ϲ�����ģ����Һ�еĿ�ʴ�ԽϺϽ����������ߣ�����Mg-3Zn0.8-Zr/1.0��-TCP���ϲ��ϵĸ�ʴ�缫��λΪ-1.547 V���丯ʴ�����ܶ�Ϊ1.20��10-6 A/cm2.
�ؼ��ʣ���-����ƣ�þ�����ϲ��ϣ�����֯����ѧ���ܣ�����ɽ���
(Edited by FANG Jing-hua)
Foundation item: Project (51071108) supported by the National Natural Science Foundation of China; Project (09JCZDJC18500) supported by the Key Project of Natural Science Foundation of Tianjin, China
Corresponding author: CHEN Min-fang; Tel: +86-22-60215845; E-mail: mfchentj@126.com
DOI: 10.1016/S1003-6326(11)60786-3