
Corrosion properties of AZ31magnesium alloy and protective effects of chemical conversion layers and anodized coatings
CHENG Ying-liang(程英亮)1,WU Hai-lan(吴海兰)1, CHEN Zhen-hua(陈振华)1,
WANG Hui-min(王慧敏)1, ZHANG Zhao(张 昭)2, WU You-wu(吴有伍)1
1. College of Materials Science and Engineering, Hunan University, Changsha 410082, China;
2. Department of Chemistry, Zhejiang University, Hangzhou 310027, China
Received 28 July 2006; accepted 26 February 2007
Abstract: The corrosion properties of AZ31 magnesium alloys were studied by potentiodynamic polarization curves and electrochemical impedance spectroscopy(EIS) techniques, meanwhile, the protective properties of two environmentally protective types of chemical conversion layers and anodized coatings of AZ31 magnesium alloys were also discussed. The component of chemical conversion bath is NaH2PO4・12H2O 20 g/L, H3PO4 7.4 mL/L, NaNO2 3 g/L, Zn(NO3)2・6H2O 5 g/L and NaF 1g/L, and components of the anodization bath is Na2SiO3 60 g/L, C6H5Na3O7・2H2O 50 g/L, KOH 100 g/L and Na2B4O7・2H2O 20 g/L. The results show that the corrosion resistance of AZ31magnesium increases with the increase of pH value of the corrosive medium. For the chemical conversion layer acquired at 80 ℃, 10 min is the best processing time and the charge transfer resistance of the chemical conversion layer is enhanced nearly by 10 times. The optimum processing time for the anodization of AZ31 is 60 min, the charge transfer resistance value of the anodized sample at the early immersion stage is nearly 26 times of that of the blank sample and the corrosion type of the anodized samples is pitting.
Key words: AZ31 magnesium alloy; corrosion; chemical conversion layer; anodization; electrochemical impedance spectroscopy
1 Introduction
Magnesium alloys are the lightest metal structural materials with excellent physical and mechanical properties, are widely used in automotive industry, electronic products, aerospace industry. However, the corrosion resistance of magnesium alloys is inferior, which strongly restricts its wide applications. Thus it is important to improve the corrosion resistance of magnesium alloys, and the study on the corrosion and protection of magnesium has attracted great attention [1-4].
As the corrosion and protection of magnesium alloys are concerned, chemical conversion and anodization are two effective processes that have been widely used. The chemical conversion method owns merits such as low cost and simplicity in operation[5]. However, the traditional chemical conversion baths usually contain the poisonous Cr6+ that can cause serious environmental problems, and it is strictly limited in application. Thus the development of non-chromate chemical conversion baths has been widely explored recently. For example, DABALA et al[6], RUDD et al[7] and BRUNELLIK et al[8] studied the effect of rare earth conversion coatings applied to magnesium. UMEHARA et al[9] studied the permanganate chemical conversion coatings. CHONG and SHIH[10] studied the permaganatephosphate solution and KOUISNIA et al [11-12] studied the effects of phosphate solution. Similarly, traditional anodizing crafts, such as Dow17, HAE and Cr-22, can well offer protection, make use of the electrolyte bath that often contains deleterious substance such as Cr6+. Therefore the development of the low cost electrolyte without heavy metal ion pollutant and other substance that can cause ecological problems and health problems will be an important issue to the magnesium anodization craft and various of new magnesium anodizing processes have been explored in recent studies[13-19].
In this study, the corrosion properties of AZ31 magnesium alloy in sodium chloride solution were studied by potentiodynamic polarization and EIS methods. Then, two surface protection methods, chemical conversion process (phosphatation) and anodization were applied to AZ31 magnesium alloy, and the protection effects of the chemical conversion layers and anodization were studied by electrochemical tests.
2 Experimental
The material used in the AZ31 corrosion tests was extruded AZ31magnesium alloy plates, whose composition (mass fraction) was 3% Al, 1% Zn, 0.2% Mn, and Mg balance. The samples were cut into rectangular shape; the working surface was the cross-sectional surface that was perpendicular to the extruded direction. The samples was first connected with wire and sealed by epoxy resin with only one surface exposed as the working surface. The area of the working surface was 0.46 cm2. The samples were polished successively to 600 and 1 200 grit finish. They were cleaned by acetone and distilled water, and dried in a stream of warm air. The corrosion medium is 3.5% sodium chloride solution, prepared by analytical grade reagent and distilled water. The pH value of the sodium chloride solution was adjusted by HCl and NaOH solution. The corrosion behaviour was studied by potentiodynamic polarization and EIS methods, the testing equipment was a CHI660C electrochemical workstation.
The samples for the chemical conversion treatment were the same as the samples used in corrosion tests. The chemical conversion bath was a kind of phosphate mixing solution containing a component of NaH2PO4・12H2O 20 g/L, H3PO4 7.4 mL/L, NaNO2 3 g/L, Zn(NO3)2・6H2O 5 g/L, NaF 1 g/L, the pH value of the bath was approximately 3±0.2, the temperature of the phosphating electrolyte was kept at 80 ℃. The composition of the bath contains no chromate, it is an environmentally protective chemical conversion bath. After the chemical conversion process, the conversion layers were examined by electrochemical methods.
The samples for the anodizing process were cut from a rolled AZ31 magnesium alloy plate, whose chemical composition was the same as that of the extruded magnesium alloy. The samples were cut into a dimension of 10 mm×10 mm×2 mm, connected with copper wire and sealed with epoxy resin, with only a working surface exposed. The dimension of the working surface was 10 mm×10 mm. The samples were ultimately polished by 1 200 grit metallographic papers and cleaned before anodization. Based on our former research, the anodization bath used in this study was also an environmentally protective type electrolyte, whose composition was as follows: Na2SiO3 60 g/L, C6H5Na3O7・2H2O 50 g/L and KOH 100 g/L, Na2B4O7・2H2O 20 g/L. The temperature of the anodization process was kept at 20 ℃ and the current density used in the anodization process was fixed at 20 mA/cm2. The effects of the time of anodization and the corrosion resistance of the anodized coatings were examined by electrochemical methods.
3 Results and discussion
3.1 Corrosion tests of AZ31magnesium alloy
The corrosion properties of the AZ31 magnesium alloy were evaluated by potentiodynamic polarization and EIS measurements. The measuring instrument is composed of a three-electrode system. A saturated calomel electrode(SCE) was used as a reference electrode, a large platinum sheet was used as an auxiliary electrode, and the sample was the working electrode. The measurements of potentiodynamic polarization curves and EIS of AZ31 magnesium alloy were carried out in 3.5% sodium chloride solution with pH values of 2, 7 and 11, and the results are shown in Figs.1 and 2, respectively. The scan rate of the poetntiodynamic polarization tests was 0.2 mV/s. The EIS measurements were carried out at open circuit potential after 25 min immersion in the sodium chloride solution, the amplitude of the perturbative signal is 5 mV and the measuring frequency range is 105-0.01 Hz.

Fig.1 Effect of pH values on polarization curves of AZ31magnesium alloy

Fig.2 EIS results of AZ31magnesium alloy recorded after 25 min immersion at different pH values: (a) Nyquist plots; (b) Bode phase angle plots
Fig.1 shows that the stable potential of magnesium alloy in acid solution is more negative than that in neutral and basic solutions, this can be explained by the fact that the passive films on magnesium alloy can not exist in acid solutions but there is partial protective oxide film in neutral and basic solutions[20]. The partial protective oxide film can provide moderate protection to the matrix alloy, resulting in the increase of corrosion potential. According to the fitted results of the potentiodynamic polarization curves, the free corrosion current densities of AZ31 magnesium alloy in the solution of pH 2, 7 and 11 are 1.912×10-3, 1.021×10-3 and 8.853×10-4 A/cm2, respectively. It can be seen that the corrosion current density, i.e. the corrosion resistance of AZ31 magnesium alloy, increases with the increases of pH values of corrosive medium.
It can be seen in Fig.2 that the EIS plots of magnesium consist of a capacitive arc in high frequency range and an inductive arc in low frequency range. According to relevant studies[20], the high frequency capacitive arc can be related to the parallel combination of the interface capacitance (it can be substituted by a CPE element, ZCPE=1/[Y0(jw)-n], 0<n<1) and the charge transfer resistance(Rct), where the charge transfer resistance(Rct) can be used to evaluate the corrosion resistance of samples. The high frequency region of Fig.2 was fitted as listed in Table 1, where Rs is the solution resistance. The inductive loop in low frequency region can be ascribed to the partially protective film on the surface of magnesium alloys and it can be explained with the theory proposed by CAO and ZHANG[21]. According to Ref.[21], if the electrode system is controlled by only a surface state variable X besides electrode potential E, the non-faradaic and faradaic admittance of the electrode- solution interface can be expressed as follows:
Y=jωCdl+1/Rct+(?IF/?X)(dX/dE)/[1-jω/(?X′/?X)] (1)
where X′=dX/dt and Rct is the charge transfer resistance.
The high frequency region in Fig.2 was contributed by the first two parts in the right side of Eqn.(1) while the low frequency part of Fig.2 was contributed by the last part of Eqn.(1). When (?IF/?X)/(dX/dE)>0, there will be an inductive loop in the low frequency region of EIS.
It was confirmed that there are partial films on the magnesium alloy in neutral and basic solutions[20], then the surface state variable can be assigned to the ratio of the surface area covered by film to the film free area of magnesium alloy, which is designated by θ. The enhancement of electrode potential(E) can lead to the break of the film, then (dθ/dE)<0, which shows that the area that is covered by film decrease with the increase of electrode potential. Moreover, the anodic current passing through the electrode will lead to the dissolution of the oxide film, thus the covering ratio of the oxide film will decrease with the increase of anodic current density, that is, (?IF/?θ)<0. As a result, (?IF/?θ)(dθ/dE)>0. According to Ref.[21], there will be an inductive loop at the low frequency region of the EIS of AZ31 magnesium alloy.
It can be seen in Table 1 that the charge transfer resistance(Rct) increases with the increase of pH values, the charge transfer resistance at pH=11 is nearly two times of the value at pH=2. Compared with the result of the potentiodynamic polarization experiment, the trend shown by the results of EIS is similar.
Table 1 Fitted result of high frequency part of EIS result in Fig.2

3.2 Experimental results of phosphatation
The phosphating of the AZ31magnesium alloy was carried out at a thermostat. The samples were pretreated by 60% NaOH and 75% H3PO4 solutions, NaOH solution was used to degrease the samples, H3PO4 solution was used mainly to dissolve the surface MgO film. The phosphating layers were acquired at 80 ℃ for different processing time of 5, 10, 20 and 30 min. The properties of different phospahting layers were examined by EIS and the results are shown in Fig.3.
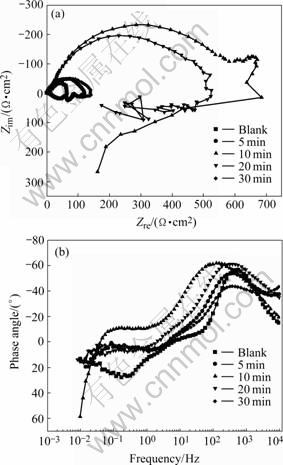
Fig.3 EIS of phosphated films for different processing time acquired at 80 ℃: (a) Nyquist plots; (b) Bode phase angle plots
It can be seen that the EIS plots still consist of a high frequency capacitive arc and an inductive loop in low frequency region. Table 2 lists the fitted data of the high frequency part in Fig.3.
Table 2 Fitted results of EIS of phosphated samples for different processing time at 80 ℃
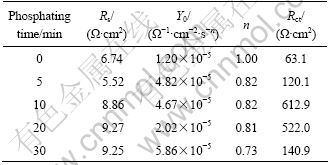
It can be seen in Table 2 that the sample processed for 10 min has the largest Rct value. Thus the phosphated film processed for 10 min has the best corrosion resistance and the charge transfer resistance is nearly enlarged by 10 times.
In this experiment, the processing craft at 80 ℃ was considered only, because phosphatation reaction is an endothermic reaction, the phosphating films acquired at medium and low temperature will be inferior to the films acquired at high temperature.
3.3 Experimental results of anodization
In order to clarify the effect of anodizing time, the samples were first prepared with different anodizing time then the corresponding samples were examined by EIS in 3.5% NaCl solution, the results are shown in Fig.4.

Fig.4 EIS of AZ31magnesium alloy with different anodizing time: (a) Nyquist plots; (b) Bode phase angle plots
It can be seen in the relationship between frequency and modulus in Fig.4 that the value of impedance increases with the increase of processing time, the sample anodized for 60 min has the highest value and the sample anodized for 40 min is the second. Although the sample with 60 min anodization has the highest impedance value, the amount of the increment is not apparent compared with that of the sample with 40 min anodization. It can be concluded that the anodization time of 60 min is sufficient for the forming of high quality anodizing films.
In order to study the corrosion mechanism of the anodized AZ31 alloy, the sample prepared by 60 min anodization was immersed in 3.5% NaCl solution and the evolution of the EIS with time were measured. The results are shown in Fig.5. For the purpose of comparison, the EIS of the blank AZ31magnesium alloy during the early immersion stage is also plotted in Fig.5. The experimental process was also been examined by naked eyes, it was found that macroscopic pits appear after 24 h immersion. The surface and cross-section SEM morphologies of the anodized film formed by 60 min anodization are shown in Fig.6. According to Fig.6, the microscopic features are porous, this may be caused by the contraction of the anodic film when the molten oxides formed by anodization are cooled by electrolyte [22].
It can be seen in Fig.5 that the EIS of the anodized samples have two time constants in the high and medium frequency region besides an inductive loop in the low frequency region. Especially at the later immersion stage, there are two apparent peaks at the frequency-phase angle plots. In addition, it can be seen in Fig.6 that the microstructure of the anodized film is porous, as a result, the electrolyte will continuously permeate into the pores. Thus, according to the EIS spectra and the physical structure of the electrode and solution interface, the equivalent circuit of Fig.7 can be put forward to fit the high and medium part of the EIS of Fig.5. In Fig.7, Rs is the solution resistance, CPE1 is the capacitance of the film, Rpore is the resistance of the pores in anodized film, CPE2 is the double layer capacitance between the magnesium alloy matrix and the electrolyte in pores. The fitted results are listed in Table 3. The EIS of the high frequency region of the blank sample cannot be fitted to the equivalent circuit of Fig.7, there is only one time constant in the high and medium frequency region, thus it should be fitted according to the way in 3.1 and 3.2 sections the fitted results of the high frequency part of the blank sample are: Rs=8.64 Ω/cm2, Y0=1.42×10-5 Ω-1?cm-2?s-n, n=0.92, Rct=695.8 Ω/cm2.

Fig.5 EIS features of sample with 60 min anodization in 3.5% NaCl solution: (a) Nyquist plots; (b) Bode phase angle plots

Fig.6 SEM morphologies of anodized film of AZ31 magnesium with 60 min anodization: (a) Surface; (b) Cross-section

Fig.7 Equivalent circuit of anodized samples immersed in sodium chloride solution
Table 3 Fitted results of EIS of anodized sample with different immersion time

It can be seen in Table 3 that the resistance of the pores and the charge transfer resistance decrease with immersion time. This can be explained that the electrolyte will reach the matrix through the pores of the anodized film for the elongation of immersion time, causing the pitting of the sample. The pits on the sample will be enlarged and new pits will appear at the new sites on the sample surface, as a result, the resistance of the pores and the charge transfer resistance will decrease with the increase of immersion time. In the early immersion time, the charge transfer resistance of the anodized sample at 1 h is 18 519 Ω/cm2, while Rct of the blank sample is 695.8 Ω/cm2, it can be seen that Rct of the anodized sample is enhanced nearly 26 times. It can also be seen in Table 3 that the impedance of the anodized sample after 120 h immersion in corrosive medium is still higher than that of the blank sample, indicating the high corrosion protection of the anodized film.
It should be noticed that charge transfer resistance of the blank sample used in anodization is 695.8 Ω/cm2, while the samples used in corrosion experiment and chemical conversion experiment is only 63.1 Ω/cm2, this is due to the samples used in anodization is rolled AZ31magnesium alloy while the materials used in corrosion experiment and chemical conversion experiments is extruded AZ31magnesium alloy. The higher corrosion resistance of the rolled AZ31 is ascribed to the fact that the grain of the rolled magnesium is finer and the microstructure of rolled AZ31 is more homogenous. Compared with the chemical conversion layers on AZ31 magnesium alloy, the anodized films on AZ31 magnesium alloy have an excellent protective function, showing greater enhancement of the impedance of the anodized films.
4 Conclusions
1) The free corrosion potential of AZ31 magnesium alloy increases with the increase of pH values of corrosive medium and the free corrosion current density decreases with the increase of pH values, indicating that the corrosion resistance of AZ31 magnesium alloy increases with the increase of pH value.
2) The samples prepared by phosphating for 10 min at 80 ℃ have the largest Rct value, showing the best corrosion resistance property. Compared with blank sample, the charge transfer resistance increases nearly 10 times.
3) The corrosion resistance of the anodized films increases with the increase of processing time. According to the EIS tests, Rct of the sample with 60 min anodization at the early immersion stage is nearly 26 times of that of the blank sample and the impedance of the anodized samples decreases with the increase of immersion time in corrosive medium. The corrosion type of the anodized samples is pitting.
4) Compared with the chemical conversion layers, the anodization process for AZ31 magnesium alloys can afford better protective effect.
References
[1] GRAY J E, LUAN B. Protective coatings on magnesium and its alloys―A critical reviews [J]. J Alloys Compd, 2002, 336: 88-113.
[2] XIAO Yang, ZHANG Xin-ming, CHEN Jian-mei, JIANG Hao, DENG Zhen-zhen. Performance of Mg-9Gd-4Y-0.6Zr alloy with high strength and heat resistance [J]. Journal of Central South University: Science and Technology, 2006, 37(5): 850-855. (in Chinese).
[3] CHEN Jian-mei, ZHANG Xin-ming, DENG Yun-lai, XIAO Yang XIONG Chuang-xian. Thermodynamics of melding and refining of magnesium alloys [J]. Journal of Central South University: Science and Technology, 2006, 37(3): 427-432. (in Chinese).
[4] ROSALBINO F, ANGELINI E, de NEGRI S. Effect of erbium addition on the corrosion behaviour of Mg-Al alloys [J]. Intermetallics, 2005, 13: 55-60.
[5] CHIU L H, CHEN C C, YANG C F. Improvement of corrosion properties in an aluminum-sprayed AZ31 magnesium alloy by a post-hot pressing and anodizing treatment [J]. Surface and Coatings Technology, 2005, 191: 181-187.
[6] DABALA M, BRUNELLI K, NAPOLITANI E, MAGRINI M. Cerium-based chemical conversion coating on AZ63 magnesium alloy [J]. Surface and Coatings Technology, 2003, 172: 227-232.
[7] RUDD A L, BRESLIN C B, MANSFELD F. The corrosion protection afforded by rare earth conversion coatings applied to magnesium [J]. Corrosion Science, 2000, 42(2): 275-288.
[8] BRUNELLI K, DABALA M, CALLIARI I, MAGRINI M. Effect of HCl pre-treatment on conversion resistance of cerium-based conversion coatings on magnesium and magnesium alloys [J]. Corrosion Science, 2005, 47(4): 989-1000.
[9] UMEHARA H, TAKAYA M, TERAUCHI S. Chrome-free surface treatments for magnesium alloy [J]. Surface and Coatings Technology, 2003, 169/170: 666-669.
[10] CHONG K Z, SHIH T S. Conversion-coating treatment for magnesium alloys by a permanganate-phosphate solution [J]. Materials Chemistry and Physics, 2003, 80: 191-200.
[11] KOUISNIA L, AZZI M, ZERTOUBI M, DAIARD F, MAXIMOVITCH S. Phosphate coatings on magnesium alloy AM60 (part 1): Study of the formation and the growth of zinc phosphate films [J]. Surface and Coatings Technology, 2004, 185: 58-67.
[12] KOUISNIA L, AZZI M, MAXIMOVITCH S. Phosphate coatings on magnesium alloy AM60 (part 2): Electrochemical behaviour in borate buffer solution [J]. Surface and Coatings Technology, 2005, 192: 239-246.
[13] ZHANG Y J, YAN C W, WANG F H, LOU H Y, CAO C N. Study on the environmentally friendly anodizing of AZ91D magnesium alloy [J]. Surface and Coating Technology, 2002, 161: 36-43.
[14] ZHANG Y J, YAN C W, WANG F H, LI W F. Electrochemical behavior of anodized Mg alloy AZ91D in chloride containing aqueous solution [J]. Corrosion Science, 2005, 47(11): 2816-2831.
[15] FUKUDA H, MATSUMOTO Y. Formation of Ti-Si composite oxide films on Mg-Al-Zn alloy by electrophoretic deposition and anodization [J]. Electrochimica Acta, 2005, 50: 5329-5333.
[16] HSIAO H Y, TSUNG H C, TSAI W T. Anodization of AZ91D magnesium alloy in silicate-containing electrolytes [J]. Surface and Coatings Technology, 2005, 199: 127-134.
[17] VERDIER S, BOINET M, MAXIMOVITCH S, DALARD F. Formation, structure and composition of anodic films on AM60 magnesium alloy obtained by DC plasma anodizing [J]. Corrosion Science, 2005, 47(6): 1429-1444.
[18] HSIAO H Y, TSAI W T. Characterization of anodic films formed on AZ91D magnesium alloy [J]. Surface and Coatings Technology, 2005, 190: 299-308.
[19] FUKUDA H, MATSUMOTO Y. Effects of Na2SiO3 on anodization of Mg-Al-Zn alloy in 3 M KOH solution [J]. Corrosion Science, 2004, 46(9): 2135-2142.
[20] SONG G L, ATRENS A, STJOHN D, XU X, NAIRN J. The anodic dissolution of magnesium in chloride and sulphate solutions [J]. Corrosion Science, 1997, 39(10/11): 1981-2004.
[21] CAO C N, ZHANG J Q. An Introduction to Electrochemical Impedance Spectroscopy [M]. Beijing: Science Press, 2002. (in Chinese)
[22] ZOZULIN A J, BARTAK D E. Anodized coatings for magnesium alloys [J]. Metal Finishing, 1994, 92: 39-44.
Foundation item: Project(50471043) supported by the National Natural Science Foundation of China
Corresponding author: CHENG Ying-liang; Tel: +86-13036798588; E-mail: deepblacksea@163.com
(Edited by YANG Hua)