A universal thermodynamic model of calculating mass action concentrations for structural units or ion couples in aqueous solutions and its applications in binary and ternary aqueous solutions
来源期刊:中国有色金属学报(英文版)2011年第3期
论文作者:杨学民 赵伟洁 柴国明 郭汉杰 张强
文章页码:626 - 641
关键词:通用热力学模型;质量作用浓度;活度;二元水溶液;三元水溶液;离子?分子共存理论;结构单元;离子对;组元
Key words:universal thermodynamic model; mass action concentration; activity; ternary aqueous solution; binary aqueous solution; ion and molecule coexistence theory; structural units; ion couples; components
摘 要:基于离子与分子共存理论,建立了计算二元和三元强电解质水溶液中结构单元或离子对的质量作用浓度的通用热力学模型;同时,采用4种二元水溶液和2种三元水溶液验证该通用热力学模型。通过转换标准态和浓度单位,用所建立的通用热力学模型计算出的298.15 K时4种二元水溶液和2种三元水溶液中结构单元或离子对的质量作用浓度和文献中报道的活度值吻合得很好。因此,可采用本研究提出的通用热力学模型计算出的二元和三元水溶液结构单元或离子对的质量作用浓度预报二元和三元强电解质水溶液中组元的反应能力;同样,也可证实本研究提出的通用热力学模型的假设条件是正确和合理的,即强电解质水溶液是由阳离子和阴离子、H2O分子和其他水合盐复杂分子组成的。基于该通用热力学模型计算出的二元和三元强电解质水溶液结构单元或离子对的质量作用浓度严格服从质量作用定律。
Abstract: A universal thermodynamic model of calculating mass action concentrations for structural units or ion couples in ternary and binary strong electrolyte aqueous solution was developed based on the ion and molecule coexistence theory and verified in four kinds of binary aqueous solutions and two kinds of ternary aqueous solutions. The calculated mass action concentrations of structural units or ion couples in four binary aqueous solutions and two ternary solutions at 298.15 K have good agreement with the reported activity data from literatures after shifting the standard state and concentration unit. Therefore, the calculated mass action concentrations of structural units or ion couples from the developed universal thermodynamic model for ternary and binary aqueous solutions can be applied to predict reaction ability of components in ternary and binary strong electrolyte aqueous solutions. It is also proved that the assumptions applied in the developed thermodynamic model are correct and reasonable, i.e., strong electrolyte aqueous solution is composed of cations and anions as simple ions, H2O as simple molecule and other hydrous salt compounds as complex molecules. The calculated mass action concentrations of structural units or ion couples in ternary and binary strong electrolyte aqueous solutions strictly follow the mass action law.

YANG Xue-min1, ZHAO Wei-jie2, 3, CHAI Guo-ming2, GUO Han-jie2, ZHANG Qiang3
1. State Key Laboratory of Multiphase Complex Systems, Institute of Process Engineering,
Chinese Academy of Sciences, Beijing 100190, China;
2. School of Metallurgical and Ecological Engineering,
University of Science and Technology Beijing, Beijing 100083, China;
3. Beijing Metallurgical Equipment Research & Design Corporation Ltd.,
China Metallurgical Group Corporation, Beijing 100029, China
Received 16 May 2010; accepted 28 November 2010
Abstract: A universal thermodynamic model of calculating mass action concentrations for structural units or ion couples in ternary and binary strong electrolyte aqueous solution was developed based on the ion and molecule coexistence theory and verified in four kinds of binary aqueous solutions and two kinds of ternary aqueous solutions. The calculated mass action concentrations of structural units or ion couples in four binary aqueous solutions and two ternary solutions at 298.15 K have good agreement with the reported activity data from literatures after shifting the standard state and concentration unit. Therefore, the calculated mass action concentrations of structural units or ion couples from the developed universal thermodynamic model for ternary and binary aqueous solutions can be applied to predict reaction ability of components in ternary and binary strong electrolyte aqueous solutions. It is also proved that the assumptions applied in the developed thermodynamic model are correct and reasonable, i.e., strong electrolyte aqueous solution is composed of cations and anions as simple ions, H2O as simple molecule and other hydrous salt compounds as complex molecules. The calculated mass action concentrations of structural units or ion couples in ternary and binary strong electrolyte aqueous solutions strictly follow the mass action law.
Key words: universal thermodynamic model; mass action concentration; activity; ternary aqueous solution; binary aqueous solution; ion and molecule coexistence theory; structural units; ion couples; components
1 Introduction
The thermodynamic properties of strong electrolyte aqueous solutions, such as activity of component, have been widely applied to biological and industrial processes[1], hydrometallurgy, environmental chemistry[2-5], geochemistry and so on[6]. However, with respect to abundant kinds of electrolyte aqueous solutions, the activity data are not enough to fulfill practical application, or the correlations between activity coefficient and concentration for various solutions have several formulas. In addition, not only ions, but also molecules, such as hydrous salt molecules, can exist in strong electrolyte aqueous solutions. Therefore, it is not very easy to determine activities of components in strong electrolyte aqueous solutions by using traditional activity calculation methods. To accurately predicate activities of components with limited data, it is of importance to develop a universal method to calculate activity of components in strong electrolyte aqueous solutions.
According to the fact that not only ions but also molecules can be found in metallurgical slags or melts at high temperature, the ion and molecule coexistence theory (IMCT) proposed by ZHANG[7-9] has been successfully applied to calculate mass action concentrations of structural units or ion couples to describe reaction ability, like classical concept of activity, for various metallurgical slags and metal melts. The essential cores of IMCT[7-9] are summarized as follows: 1) ions, simple and complex molecules exist in a solution simultaneously as structural units; 2) there are dynamic equilibriums of chemical reactions between ion couples and simple molecules to form complex molecules in a solution; 3) simple ions take part in chemical reactions of forming complex molecules with simple molecules as ion couples, which are composed of cations and anions based on electrovalence balance principle; 4) the calculated equilibrium mole fractions of structural units or ion couples are defined as mass action concentration, which are similar to activities of corresponding components.
The mass action concentrations of structural units or ion couples in binary aqueous solutions of KCl-H2O, CsCl-H2O, NaCl-H2O and BaCl2-H2O[10]; NaBr-H2O, LiNO3-H2O, HNO3-H2O and KF-H2O[11]; KBr-H2O, NH4Br-H2O and ZnBr2-H2O[11]; and ternary aqueous solutions of NaCl-KCl-H2O[13]; RbCl-RbNO3- H2O[14]; NaClO4-NaF-H2O[15] at 298.15 K have been successfully calculated by using the developed thermodynamic model of calculating mass action concentrations for structural units or ion couples from IMCT[7-9]. The results[10-15] show that the calculated mass action concentrations of structural units or ion couples in above-mentioned binary and ternary aqueous solutions have good agreement with the reported activities of corresponding components in literatures. Therefore, it is verified that the application scope of IMCT[7-9] can be expanded from metallurgical slag and metal melts at high temperature to electrolyte aqueous solutions at low or ambient temperature. However, a universal thermodynamic model for binary and ternary aqueous solutions has not been reported and summarized in previous publications[10-15].
In this work, a universal thermodynamic model of calculating mass action concentrations for structural units or ion couples in ternary electrolyte aqueous solutions with hydrous salt compounds as complex molecules was developed based on IMCT[7-9]. Meanwhile, a universal thermodynamic model for binary aqueous solutions with hydrous salt compounds as complex molecules was also developed by simplifying the universal thermodynamic model for ternary solutions, and four kinds of binary aqueous solutions were chosen to verify the developed thermodynamic model as: 1) binary aqueous solutions without any hydrous salt compound formed; 2) binary solutions with ECl?2H2O as formed hydrous salt compound with unknown standard molar Gibbs free energy ![]() ; 3) binary aqueous solutions with ECl?2H2O as formed hydrous salt compound with known
; 3) binary aqueous solutions with ECl?2H2O as formed hydrous salt compound with known ![]() ; 4) binary aqueous solutions with ECl?H2O and ECl?3H2O as formed hydrous salt compounds with known
; 4) binary aqueous solutions with ECl?H2O and ECl?3H2O as formed hydrous salt compounds with known ![]() . All universal thermodynamic models were verified from the previous published data of calculating mass action concentrations for structural units or ion couples in binary and ternary solutions[10-15] as examples. The ultimate aims of this work are to develop a universal thermodynamic model of calculating mass action concentrations for structural units or ion couples in binary and ternary electrolyte aqueous solutions and verify it in various special cases.
. All universal thermodynamic models were verified from the previous published data of calculating mass action concentrations for structural units or ion couples in binary and ternary solutions[10-15] as examples. The ultimate aims of this work are to develop a universal thermodynamic model of calculating mass action concentrations for structural units or ion couples in binary and ternary electrolyte aqueous solutions and verify it in various special cases.
2 Establishment of universal thermodynamic model
2.1 Hypotheses
According to the facts that there are ions and molecules simultaneously in strong electrolyte aqueous solutions, the assumptions applied in the developed universal thermodynamic model of calculating mass action concentrations for structural units or ion couples in ternary strong electrolyte aqueous solutions can be proposed based on IMCT[7-9] by taking ECl-FCl-H2O as an example aqueous solution as follows.
1) The structural units in ECl-FCl-H2O ternary solutions are composed of E+, F+ and Cl- as simple ions, H2O as simple molecule, ECl?H2O, ECl?2H2O, ![]() , ECl?nH2O or FCl?H2O, FCl?2H2O,
, ECl?nH2O or FCl?H2O, FCl?2H2O, ![]() , FCl?mH2O as complex molecules. Each cation or anion of the simple ions in ECl-FCl-H2O ternary solutions occupies only one position of structural units, but will take part in reaction of forming hydrous salt molecules in the form of ion couple as (E++Cl-), and (F++Cl-). For example, ECl in ECl-FCl-H2O ternary solutions can be electrolyzed or separated into two simple ions as E+ and Cl- as two structural units, respectively; but ions of E+ and Cl- will take part in reaction of forming hydrous salt molecules as an ion couple (E++Cl-) in ECl-FCl-H2O ternary solutions.
, FCl?mH2O as complex molecules. Each cation or anion of the simple ions in ECl-FCl-H2O ternary solutions occupies only one position of structural units, but will take part in reaction of forming hydrous salt molecules in the form of ion couple as (E++Cl-), and (F++Cl-). For example, ECl in ECl-FCl-H2O ternary solutions can be electrolyzed or separated into two simple ions as E+ and Cl- as two structural units, respectively; but ions of E+ and Cl- will take part in reaction of forming hydrous salt molecules as an ion couple (E++Cl-) in ECl-FCl-H2O ternary solutions.
2) Formation of complex hydrous salt molecules by ion couples and simple molecule of H2O is under dynamic equilibrium as
(E++Cl-)+H2O=ECl?H2O (1.1)
(E++Cl-)+2H2O=ECl?2H2O (1.2)
![]()
(E++Cl-)+nH2O=ECl?nH2O (1.n)
(F++Cl-)+H2O=FCl?H2O (2.1)
(F++Cl-)+2H2O=FCl?2H2O (2.2)
![]()
(F++Cl-)+mH2O=FCl?mH2O (2.m)
3) The structural units in ECl-FCl-H2O strong solutions bear continuity in the investigated concentration range of solutes.
4) All chemical reactions of forming complex hydrous salt molecules in ECl-FCl-H2O strong electrolyte ternary aqueous solutions as shown in Eq.(1) and Eq.(2) follow the mass action law.
2.2 Universal thermodynamic model for ternary systems
2.2.1. Model for ternary systems with all hydrous salt molecules formed
Based on the above-mentioned hypotheses, the structural units in a chloride ternary aqueous solution with all hydrous salt molecules formed as an example, i.e., ECl-FCl-H2O ternary solution, are composed of E+, F+ and Cl- as simple ions, H2O as simple molecule, ECl?H2O, ECl?2H2O, ![]() , ECl?nH2O, and FCl?H2O, FCl?2H2O,
, ECl?nH2O, and FCl?H2O, FCl?2H2O, ![]() , FCl?mH2O as complex molecules or hydrous salt molecules.
, FCl?mH2O as complex molecules or hydrous salt molecules.
The morality ![]() (mol/kg (H2O)) is usually applied to represent concentration of components in strong electrolyte aqueous solution, i.e., mole number of solutes in 1 kg solvent of H2O. Therefore, mole number of ECl, FCl and H2O in ECl-FCl-H2O ternary solution based on 1 kg H2O before equilibrium is defined as
(mol/kg (H2O)) is usually applied to represent concentration of components in strong electrolyte aqueous solution, i.e., mole number of solutes in 1 kg solvent of H2O. Therefore, mole number of ECl, FCl and H2O in ECl-FCl-H2O ternary solution based on 1 kg H2O before equilibrium is defined as ![]()
![]() and
and ![]()
![]() respectively. The mole fractions of ECl, FCl and H2O in ECl-FCl-H2O ternary solution before equilibrium can be easily
respectively. The mole fractions of ECl, FCl and H2O in ECl-FCl-H2O ternary solution before equilibrium can be easily
determined as![]()
![]()
The mole numbers of structural units in ECl-FCl-H2O ternary solution under dynamic reaction equilibrium condition based on 1 kg H2O are defined according to IMCT[7-9] as follows:
![]()
![]()
![]()
![]()
![]()
![]() ,
,
![]()
![]()
![]()
![]() ,
,
![]() (3)
(3)
Hence, the total mole number of all structural units ![]() in ECl-FCl-H2O ternary solution under equilibrium condition based on 1 kg H2O can be calculated according to the mass balance principle as
in ECl-FCl-H2O ternary solution under equilibrium condition based on 1 kg H2O can be calculated according to the mass balance principle as
![]() (4)
(4)
The mass action concentration of structural unit is defined as a ratio of equilibrium mole number of structural unit i to the total equilibrium mole number of all structural units in a system with a fixed amount according to IMCT[7-9], and can be calculated by
![]() (-) (5)
(-) (5)
It should be emphasized that mass action concentrations of all structural units in forms of ions, simple and complex molecules can be calculated from Eq.(5) directly[7-9]; however, mass action concentration of an ion couple from cation and anion, such as (E++Cl-), should be calculated as follows[7-9]:
![]() (-) (6)
(-) (6)
Therefore, the mass action concentrations of structural units or ion couples in ECl-FCl-H2O ternary solution under equilibrium condition can be expressed as follows:
 (-) (7)
(-) (7)
The possible dynamic reactions of forming various hydrous salt molecules and their corresponding equilibrium constants and mass action concentrations can be describled as follows:
(E++Cl-)+H2O=ECl?H2O,
![]()
![]() (8.1)
(8.1)
(E++Cl-)+2H2O=ECl?2H2O,
![]() ,
, ![]() (8.2)
(8.2)
![]()
(E++Cl-)+nH2O=ECl?nH2O,
![]() (8.n)
(8.n)
(F++Cl-)+H2O=FCl?H2O,
![]() (9.1)
(9.1)
(F++Cl-)+2H2O=FCl?2H2O,
![]() ,
, ![]() (9.2)
(9.2)
![]()
(F++Cl-)+mH2O=FCl?mH2O,
![]() (9.m)
(9.m)
The mass balance of three components as ECl, FCl and H2O in ECl-FCl-H2O ternary solution based on 1 kg H2O according to IMCT[7-9] can be represented as follows:
 (10a)
(10a)
 (10b)
(10b)
 (10c)
(10c)
Combining Eq.(10a)-Eq.(10b) to eliminate ![]() , the following formulations can be obtained as
, the following formulations can be obtained as
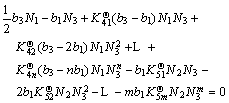 (11a)
(11a)
 (11b)
(11b)
According to the fact that the total equilibrium mole fraction of all structural units in a system with a fixed amount is 1.0, the following equation can be obtained as
 (-) (12)
(-) (12)
The equilibrium constant ![]() of all dynamic chemical reactions describled in Eq.(1) and Eq.(2) can be calculated as follows:
of all dynamic chemical reactions describled in Eq.(1) and Eq.(2) can be calculated as follows:
![]() (-) (13)
(-) (13)
Therefore, the equation groups of Eq.(10) and Eq.(12) or Eq.(11) and Eq.(12) are composed of the universal thermodynamic model of calculation mass action concentrations for structural units or ion couples ![]() in ECl-FCl-H2O ternary aqueous solution, respectively. There are four unknown parameters as
in ECl-FCl-H2O ternary aqueous solution, respectively. There are four unknown parameters as ![]() ,
, ![]() ,
, ![]() and
and ![]() with four independent equations in the universal thermodynamic model composing of Eq.(10) and Eq.(12), or three unknown parameters as N1, N2 and N3 with three independent equations in the universal thermodynamic model composing of Eq.(11) and Eq.(12). It is clear that the real solutions of above-mentioned mass action concentrations of structural units or ion couples Ni and total mole fraction of all structural units
with four independent equations in the universal thermodynamic model composing of Eq.(10) and Eq.(12), or three unknown parameters as N1, N2 and N3 with three independent equations in the universal thermodynamic model composing of Eq.(11) and Eq.(12). It is clear that the real solutions of above-mentioned mass action concentrations of structural units or ion couples Ni and total mole fraction of all structural units ![]() in ECl-FCl-H2O ternary aqueous solution can be solely solved by combining Eq.(10) and Eq.(12) or Eq.(11) and Eq.(12) under condition of knowing equilibrium constant
in ECl-FCl-H2O ternary aqueous solution can be solely solved by combining Eq.(10) and Eq.(12) or Eq.(11) and Eq.(12) under condition of knowing equilibrium constant ![]() . The equilibrium mole number of each structural unit ni can be also determined by Eq.(5) or Eq.(6) after knowing Ni and
. The equilibrium mole number of each structural unit ni can be also determined by Eq.(5) or Eq.(6) after knowing Ni and ![]() .
.
The calculated mass action concentration ![]() is based on pure mater as standard state and mole fraction xi as concentration unit; however, the reported activity data of strong electrolyte aqueous solutions in literatures usually choose infinite dilute solution as standard state and molality mi (mol/kg (H2O)) as concentration unit. To compare the calculated mass action concentrations Ni and reported activity data ai, a transformation coefficient Li for solvent or
is based on pure mater as standard state and mole fraction xi as concentration unit; however, the reported activity data of strong electrolyte aqueous solutions in literatures usually choose infinite dilute solution as standard state and molality mi (mol/kg (H2O)) as concentration unit. To compare the calculated mass action concentrations Ni and reported activity data ai, a transformation coefficient Li for solvent or ![]() for solutes should be introduced to shift standard state and concentration unit[10-15], and the transformed mass action concentration of components
for solutes should be introduced to shift standard state and concentration unit[10-15], and the transformed mass action concentration of components ![]() can be calculated as follows[10-15]:
can be calculated as follows[10-15]:
![]() (-) (14)
(-) (14)
It should be specially pointed out that mass action concentration of all ion couples, simple and complex (hydrous salts) molecules, such as ECl, FCl, H2O, ECl?H2O, ECl?2H2O, ![]() , ECl?nH2O, and FCl?H2O, FCl?2H2O,
, ECl?nH2O, and FCl?H2O, FCl?2H2O, ![]() , FCl?mH2O in ECl-FCl-H2O ternary solution under equilibrium condition can be calculated from the developed universal thermodynamic model based on IMCT[7-9]. However, only activity data of three components in ECl-FCl-H2O ternary solution as ECl, FCl and H2O can be determined from viewpoints of classical experimental tests and traditional thermodynamics. Although concept of mass action concentration of components Ni, taking ECl as an example, has been applied in previous publications[7-15] and this study, the real meaning of mass action concentration for ECl NECl is the sum of mass action concentration of two contained structural units in ECl as E+ and Cl-. From absolute viewpoint of IMCT[10-15], there is no mass action concentration of ECl as an independent component, because no independent ECl molecules can exist in ECl-FCl-H2O ternary solution. Adopting concept of mass action concentration for ECl is just for convenient comparison of the reported ECl activity data with calculated mass action concentration of the same components.
, FCl?mH2O in ECl-FCl-H2O ternary solution under equilibrium condition can be calculated from the developed universal thermodynamic model based on IMCT[7-9]. However, only activity data of three components in ECl-FCl-H2O ternary solution as ECl, FCl and H2O can be determined from viewpoints of classical experimental tests and traditional thermodynamics. Although concept of mass action concentration of components Ni, taking ECl as an example, has been applied in previous publications[7-15] and this study, the real meaning of mass action concentration for ECl NECl is the sum of mass action concentration of two contained structural units in ECl as E+ and Cl-. From absolute viewpoint of IMCT[10-15], there is no mass action concentration of ECl as an independent component, because no independent ECl molecules can exist in ECl-FCl-H2O ternary solution. Adopting concept of mass action concentration for ECl is just for convenient comparison of the reported ECl activity data with calculated mass action concentration of the same components.
2.2.2 Model for ternary systems without any hydrous salt molecule formed
As an extreme example, a universal thermodynamic model of calculating mass action concentrations for structural units or ion couples in ECl-FCl-H2O ternary solution without any complex hydrous salt molecule formed can be also obtained by considering n, m and ![]() as zero simultaneously in the equation groups of Eq.(10) and Eq.(12) or Eq.(11) and Eq.(12) choosing ECl-FCl-H2O ternary solution as a representative.
as zero simultaneously in the equation groups of Eq.(10) and Eq.(12) or Eq.(11) and Eq.(12) choosing ECl-FCl-H2O ternary solution as a representative.
The mole numbers of ECl, FCl and H2O in ECl-FCl-H2O ternary solution before reaction equilibrium based on 1 kg H2O are represented as ![]()
![]()
![]() =55.6 mol, respectively. The structural units in ECl-FCl-H2O ternary solution without any hydrous salt molecule formed are composed of E+, F+ and Cl- as simple ions, H2O as simple molecules. The mole numbers of structural units in ECl-FCl-H2O ternary solution without any hydrous salt molecule formed under dynamic reaction equilibrium condition based on 1 kg H2O can be expressed as
=55.6 mol, respectively. The structural units in ECl-FCl-H2O ternary solution without any hydrous salt molecule formed are composed of E+, F+ and Cl- as simple ions, H2O as simple molecules. The mole numbers of structural units in ECl-FCl-H2O ternary solution without any hydrous salt molecule formed under dynamic reaction equilibrium condition based on 1 kg H2O can be expressed as ![]()
![]()
![]()
![]() Hence, the total mole number of all structural units
Hence, the total mole number of all structural units![]() based on 1 kg H2O in ECl-FCl-H2O ternary solution without any hydrous salt molecule formed under equilibrium condition can be calculated according to the mass balance principle as
based on 1 kg H2O in ECl-FCl-H2O ternary solution without any hydrous salt molecule formed under equilibrium condition can be calculated according to the mass balance principle as
![]() (15)
(15)
The mass balance of ECl, FCl and H2O in ECl-FCl-H2O ternary solution can be described by considering definition of mass action concentrations of structural units or ion couples in ECl-FCl-H2O ternary solution shown in Eq.(10) as
![]() (16a)
(16a)
![]() (16b)
(16b)
![]() (16c)
(16c)
The following equations can be obtained by combining Eq.(16a)-Eq.(16c) to eliminate ![]() as
as
![]() (17a)
(17a)
![]() (17b)
(17b)
The following expression can be obtained according to the principle that the sum of all mole fraction of structural units in ECl-FCl-H2O ternary solution with a fixed amount under equilibrium condition is equal to 1.0, as
![]() (18)
(18)
Therefore, the thermodynamic model of calculating mass action concentrations for structural units or ion couples in ECl-FCl-H2O ternary solution without any hydrous salt molecule formed can be described by equation groups of Eq.(16) and Eq.(18) or Eq.(17) and Eq.(18). Substituting the known parameters as ![]()
![]() and b3=
and b3= ![]() =55.6 mol into Eq.(17)-Eq.(18), the thermodynamic model of calculating mass action concentrations for structural units or ion couples in ECl-FCl-H2O ternary solution without any hydrous salt molecule formed can be simplified as
=55.6 mol into Eq.(17)-Eq.(18), the thermodynamic model of calculating mass action concentrations for structural units or ion couples in ECl-FCl-H2O ternary solution without any hydrous salt molecule formed can be simplified as
![]()
![]()
![]() (19)
(19)
The transformation coefficient Li or ![]() and transformed mass action concentration of components
and transformed mass action concentration of components ![]() can be obtained by Eq.(14) as described in Section 2.2.1.
can be obtained by Eq.(14) as described in Section 2.2.1.
2.3 Universal thermodynamic model for binary systems
2.3.1 Model for binary systems with all hydrous salt molecules formed
The structural units in ECl-H2O binary strong electrolyte aqueous solution with all hydrous salt molecules formed are composed of E+ and Cl- as simple ions, H2O as simple molecule and ECl?H2O, ECl?2H2O, ![]() , and ECl?nH2O as complex hydrous salt molecules. Therefore, the mole numbers of ECl and H2O in ECl-H2O binary solution based on 1 kg H2O before equilibrium is defined as
, and ECl?nH2O as complex hydrous salt molecules. Therefore, the mole numbers of ECl and H2O in ECl-H2O binary solution based on 1 kg H2O before equilibrium is defined as![]() and b3=
and b3= ![]()
![]() respectively. The mole fractions of ECl and H2O in ECl-H2O binary solution before equilibrium can be easily determined by
respectively. The mole fractions of ECl and H2O in ECl-H2O binary solution before equilibrium can be easily determined by ![]()
![]()
The mole number of structural units in ECl-H2O binary solution under dynamic reaction equilibrium condition based on 1 kg H2O is defined according to IMCT[7-9] as follows:
![]()
![]()
![]()
![]() ,
, ![]() (20)
(20)
Hence, the total mole number of all structural units ![]() based on 1 kg H2O in ECl-H2O binary solution under equilibrium condition can be calculated according to the mass balance principle as
based on 1 kg H2O in ECl-H2O binary solution under equilibrium condition can be calculated according to the mass balance principle as
![]() (21)
(21)
Therefore, Eq.(10) can be rewritten to present the mass balance of ECl and H2O in ECl-H2O binary solution with all hydrous salt molecules formed under equilibrium condition based on 1 kg H2O by ignoring parameters b2, m and ![]() as
as
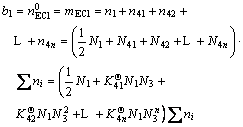 (22a)
(22a)
 (22b)
(22b)
The following equation can be obtained by combining Eq.(22a) and Eq.(22b) to eliminate ![]() as
as
 (23)
(23)
According to the fact that the total equilibrium mole fraction of all structural units in a system with a fixed amount is 1.0, Eq.(12) can be simplified for ECl-H2O binary solution as
![]() (-) (24)
(-) (24)
Therefore, Eq.(22) and Eq.(24) or Eq.(23) and Eq.(24) are the developed universal thermodynamic model of calculating mass action concentrations for structural units or ion couples Ni in ECl-H2O binary solution with all hydrous salt molecules formed, respectively. There are three unknown parameters as N1, N3 and ![]() with three independent equations in the universal thermodynamic model composing of Eq.(22) and Eq.(24), or two unknown parameters as N1 and N3 with two independent equations in the universal thermodynamic model composing of Eq.(23) and Eq.(24). The real solutions of Ni and
with three independent equations in the universal thermodynamic model composing of Eq.(22) and Eq.(24), or two unknown parameters as N1 and N3 with two independent equations in the universal thermodynamic model composing of Eq.(23) and Eq.(24). The real solutions of Ni and ![]() for ECl-H2O binary solution can be solely solved by combing Eq.(22) and Eq.(24) or Eq.(23) and Eq.(24) under condition of knowing
for ECl-H2O binary solution can be solely solved by combing Eq.(22) and Eq.(24) or Eq.(23) and Eq.(24) under condition of knowing ![]() . The equilibrium mole number of all structural units or ion couples
. The equilibrium mole number of all structural units or ion couples ![]() under equilibrium condition can be also calculated from definition formula of Ni in Eq.(5) or Eq.(6) after knowing Ni and
under equilibrium condition can be also calculated from definition formula of Ni in Eq.(5) or Eq.(6) after knowing Ni and ![]() . The expressions of
. The expressions of ![]() Li or
Li or ![]() and
and ![]() for ECl-H2O binary solution are the same as equations described in Eq.(13) and Eq.(14).
for ECl-H2O binary solution are the same as equations described in Eq.(13) and Eq.(14).
2.3.2 Model for binary system without any hydrous salt molecule formed
The structural units in ECl-H2O binary strong electrolyte aqueous solution without any hydrous salt complex molecule generated are composed of E+ and Cl- as simple ions, and H2O as simple molecule. Hence, no equilibrium constant of forming hydrous salt complex molecules ![]() can exist in the universal thermodynamic model of calculating mass action concentrations for structural units or ion couples Ni in ECl-H2O binary solution, as shown in Eq.(22) and Eq.(24) or Eq.(23) and Eq.(24), respectively. The mole numbers of ECl and H2O in ECl-H2O binary solution without any hydrous salt complex molecule generated before reaction equilibrium based on 1 kg H2O are presented as
can exist in the universal thermodynamic model of calculating mass action concentrations for structural units or ion couples Ni in ECl-H2O binary solution, as shown in Eq.(22) and Eq.(24) or Eq.(23) and Eq.(24), respectively. The mole numbers of ECl and H2O in ECl-H2O binary solution without any hydrous salt complex molecule generated before reaction equilibrium based on 1 kg H2O are presented as ![]()
![]() 55.6 mol. The mole numbers of structural units in ECl-H2O ternary solution without any hydrous salt molecule formed under dynamic reaction equilibrium condition based on 1 kg H2O can be expressed[7-9] as
55.6 mol. The mole numbers of structural units in ECl-H2O ternary solution without any hydrous salt molecule formed under dynamic reaction equilibrium condition based on 1 kg H2O can be expressed[7-9] as ![]()
![]()
![]() 55.6. Hence, the total mole number of all structural units
55.6. Hence, the total mole number of all structural units ![]() based on 1 kg H2O in ECl-H2O ternary solution without any hydrous salt molecule formed under equilibrium condition can be calculated according to the mass balance principle as
based on 1 kg H2O in ECl-H2O ternary solution without any hydrous salt molecule formed under equilibrium condition can be calculated according to the mass balance principle as
![]() (25)
(25)
The mass balance of ECl and H2O in ECl-H2O binary solution without any hydrous salt molecule formed under equilibrium condition based on 1 kg H2O can be presented by simplifying Eq.(22) as
![]() (26a)
(26a)
![]() (26b)
(26b)
The following equation can be obtained by combining Eq.(26a) and Eq.(26b) to eliminate ![]() , as
, as
![]() (27)
(27)
According to the fact that the total equilibrium mole fraction of all structural units in a system with a fixed amount is 1.0, Eq.(24) can be simplified for ECl-H2O binary solution without any complex hydrous salt molecule formed as
![]() (-) (28)
(-) (28)
The equation groups of Eq.(26) and Eq.(28) or Eq.(27) and Eq.(28) are the thermodynamic model of calculating mass action concentrations for structural units or ion couples Ni in ECl-H2O binary solution without any complex hydrous salt molecule formed, respectively. Ni can be calculated by solving Eq.(26) and Eq.(28) or Eq.(27) and Eq.(28) as follows:
 (29)
(29)
The expressions of Li or ![]() and
and ![]() for ECl-H2O binary solution without any complex hydrous salt molecule formed are the same as equations described in Eq.(14).
for ECl-H2O binary solution without any complex hydrous salt molecule formed are the same as equations described in Eq.(14).
2.3.3 Model for binary system with ECl?2H2O as formed hydrous salt molecule with unknown ![]()
In the case of no standard molar Gibbs free energy ![]() of the existed complex hydrous salt molecules in ECl-H2O binary solution can be found from related literatures, the common procedure of developing the thermodynamic model of calculating mass action concentrations for structural units or ion couples
of the existed complex hydrous salt molecules in ECl-H2O binary solution can be found from related literatures, the common procedure of developing the thermodynamic model of calculating mass action concentrations for structural units or ion couples ![]() from IMCT[7-9] is summarized as follows: 1) confirming the chemical formulas of possibly formed hydrous salt molecules from phase diagram of the studied ECl-H2O binary solution; 2) establishing the thermodynamic model from the proposed universal thermodynamic model; 3) substituting the mass action concentrations Ni of ECl and H2O in ECl-H2O binary solution by the reported activities of ECl and H2O in formulas of equilibrium constant
from IMCT[7-9] is summarized as follows: 1) confirming the chemical formulas of possibly formed hydrous salt molecules from phase diagram of the studied ECl-H2O binary solution; 2) establishing the thermodynamic model from the proposed universal thermodynamic model; 3) substituting the mass action concentrations Ni of ECl and H2O in ECl-H2O binary solution by the reported activities of ECl and H2O in formulas of equilibrium constant ![]() for reactions of possibly formed hydrous salt molecules. When a relatively stable equilibrium constant
for reactions of possibly formed hydrous salt molecules. When a relatively stable equilibrium constant ![]() is obtained for the reaction of formed hydrous salt molecule, the thermodynamic model can be solved by using the calculated
is obtained for the reaction of formed hydrous salt molecule, the thermodynamic model can be solved by using the calculated ![]() to determine mass action concentrations of structural units or ion couples. Taking ECl?2H2O as formed hydrous salt molecule in ECl-H2O binary solution with unknown
to determine mass action concentrations of structural units or ion couples. Taking ECl?2H2O as formed hydrous salt molecule in ECl-H2O binary solution with unknown ![]() as an example, the procedure of developing thermodynamic model is explained in the following text.
as an example, the procedure of developing thermodynamic model is explained in the following text.
Certainly, the structural units in ECl-H2O binary solution with ECl?2H2O as hydrous salt molecule are E+ and Cl- as simple ions, H2O as simple molecule and ECl?2H2O as complex hydrous salt molecule. The mole numbers of ECl and H2O in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt molecule before reaction equilibrium based on 1 kg H2O are presented as ![]() ,
, ![]() =55.6 mol. The mole numbers of structural units in ECl-H2O ternary solution with ECl?2H2O as formed hydrous salt molecule formed under dynamic reaction equilibrium condition based on 1 kg H2O can be expressed as
=55.6 mol. The mole numbers of structural units in ECl-H2O ternary solution with ECl?2H2O as formed hydrous salt molecule formed under dynamic reaction equilibrium condition based on 1 kg H2O can be expressed as ![]()
![]() 55.6,
55.6, ![]() Hence, the total mole number of all structural units
Hence, the total mole number of all structural units ![]() in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt molecules under equilibrium condition based on 1 kg H2O can be calculated according to the mass balance principle as
in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt molecules under equilibrium condition based on 1 kg H2O can be calculated according to the mass balance principle as
![]() (30)
(30)
The mass balance of ECl and H2O in ECl-H2O binary solution based on 1 kg H2O by Eq.(22) can be rewritten as
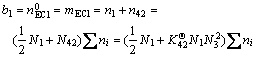 (31a)
(31a)
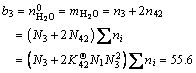 (31b)
(31b)
The developed universal thermodynamic model of ECl-H2O binary solution shown in Eq.(23) and Eq.(24) can be simplified as
![]() (32)
(32)
![]() (33)
(33)
The chemical reaction equilibrium constant ![]() of reaction for forming ECl?2H2O by Eq.(1.2) can be presented by considering Eq.(32) or Eq.(33), respectively, as
of reaction for forming ECl?2H2O by Eq.(1.2) can be presented by considering Eq.(32) or Eq.(33), respectively, as

![]()

![]()
![]() (34a)
(34a)
![]() (34b)
(34b)
It should be specially pointed out that Ni is calculated based on pure matter as standard state and mole fraction xi as concentration unit, which is the same as the reported ![]() from literatures. Usually, activity of H2O,
from literatures. Usually, activity of H2O, ![]() , but not activity coefficient of H2O, is reported in related literatures. Unfortunately, only activity coefficient
, but not activity coefficient of H2O, is reported in related literatures. Unfortunately, only activity coefficient ![]() not ai, is reported from literatures for solute i, which is based on infinite dilute solution as standard state and molality mi as concentration unit. Hence, only
not ai, is reported from literatures for solute i, which is based on infinite dilute solution as standard state and molality mi as concentration unit. Hence, only ![]() can be applied to replace
can be applied to replace ![]() , while, aECl, i.e., product of the reported activity coefficient
, while, aECl, i.e., product of the reported activity coefficient ![]() and mECl, cannot be used to substitute for NECl because NECl is less than 1.0 while aECl can be much larger than 1.0 when
and mECl, cannot be used to substitute for NECl because NECl is less than 1.0 while aECl can be much larger than 1.0 when ![]() is high enough. Under the circumstances, the calculated
is high enough. Under the circumstances, the calculated ![]() from multiplying the reported
from multiplying the reported ![]() by xECl, which is less than 1.0, is recommended to substitute NECl in Eq.(34a) or Eq.(34b)[7-9]. However, the calculated
by xECl, which is less than 1.0, is recommended to substitute NECl in Eq.(34a) or Eq.(34b)[7-9]. However, the calculated ![]() from Eq.(34a) is certainly not equal to that from Eq.(34b) because Eq.(34a) is formulated from mass balance of solute i and solvent H2O, while Eq.(34b) is based on mole fraction of all structural units in a close system as 1.0 by using Ni as concentration unit. Therefore, the reported
from Eq.(34a) is certainly not equal to that from Eq.(34b) because Eq.(34a) is formulated from mass balance of solute i and solvent H2O, while Eq.(34b) is based on mole fraction of all structural units in a close system as 1.0 by using Ni as concentration unit. Therefore, the reported ![]() from literature and product of the reported
from literature and product of the reported ![]() and xECl as
and xECl as ![]() are applied in Eq.(34a) to determine
are applied in Eq.(34a) to determine ![]() according to IMCT[7-9].
according to IMCT[7-9].
When the calculated ![]() from Eq. (34a) by using reported
from Eq. (34a) by using reported ![]() and
and ![]()
![]() from literatures is constant with a very narrow fluctuation in various xECl or mECl, the calculated
from literatures is constant with a very narrow fluctuation in various xECl or mECl, the calculated ![]() can be applied in the developed thermodynamic model as shown in equation groups of Eq.(31) and Eq.(33) or Eq.(32) and Eq.(33) to solve Ni,
can be applied in the developed thermodynamic model as shown in equation groups of Eq.(31) and Eq.(33) or Eq.(32) and Eq.(33) to solve Ni, ![]() and ni. The expressions of Li or
and ni. The expressions of Li or ![]() and
and ![]() for ECl-H2O binary solution with ECl?2H2O as complex hydrous salt molecule are the same as equations described in Eq.(14).
for ECl-H2O binary solution with ECl?2H2O as complex hydrous salt molecule are the same as equations described in Eq.(14).
2.3.4 Model for binary system with ECl?2H2O as formed hydrous salt molecule with known ![]()
The structural units in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt molecule with known ![]() consist of E+ and Cl- as simple ions, H2O as simple molecule and ECl?2H2O as complex molecule. The mole numbers of ECl and H2O in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt molecule before reaction equilibrium based on 1 kg H2O are presented as
consist of E+ and Cl- as simple ions, H2O as simple molecule and ECl?2H2O as complex molecule. The mole numbers of ECl and H2O in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt molecule before reaction equilibrium based on 1 kg H2O are presented as ![]() ,
, ![]() =55.6 mol. The mole number of structural units in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt molecule under dynamic reaction equilibrium condition based on 1 kg H2O can be expressed as
=55.6 mol. The mole number of structural units in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt molecule under dynamic reaction equilibrium condition based on 1 kg H2O can be expressed as ![]()
![]()
![]() =55.6, n42=
=55.6, n42=![]() Hence, the total mole number of all structural units
Hence, the total mole number of all structural units![]() in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt molecule under equilibrium condition based on 1 kg H2O can be calculated according to the mass balance principle as
in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt molecule under equilibrium condition based on 1 kg H2O can be calculated according to the mass balance principle as
![]() (35)
(35)
The mass balance of ECl and H2O in ECl-H2O binary solution based on 1 kg H2O can be presented by Eq.(31) or Eq.(32), and equilibrium constant ![]() can be calculated from
can be calculated from ![]() by Eq.(13).
by Eq.(13).
Therefore, the thermodynamic model of ECl-H2O binary solution with ECl?2H2O as formed hydrous salt with known ![]() is presented by combining of Eq.(31) and Eq.(33) or Eq.(32) and Eq.(33). Therefore, parameters as Ni,
is presented by combining of Eq.(31) and Eq.(33) or Eq.(32) and Eq.(33). Therefore, parameters as Ni, ![]() and ni in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt with known
and ni in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt with known ![]() can be solved. The expressions of Li or
can be solved. The expressions of Li or ![]() and
and ![]() for ECl-H2O binary solution with ECl?2H2O as complex hydrous salt molecule with known
for ECl-H2O binary solution with ECl?2H2O as complex hydrous salt molecule with known ![]() are the same as equations described in Eq.(14).
are the same as equations described in Eq.(14).
2.3.5 Model for binary system with ECl?H2O and ECl?3H2O as formed hydrous salt molecule with known ![]()
In the case of forming ECl?H2O and ECl?3H2O as hydrous salt molecules with known ![]() , the structural units in ECl-H2O binary solution are composed of E+ and Cl- as simple ions, H2O as simple molecule, ECl?H2O and ECl?3H2O as complex molecules. The mole numbers of ECl and H2O in ECl-H2O binary with ECl?H2O and ECl?3H2O as formed hydrous salt molecules before reaction equilibrium based on 1 kg H2O is presented as
, the structural units in ECl-H2O binary solution are composed of E+ and Cl- as simple ions, H2O as simple molecule, ECl?H2O and ECl?3H2O as complex molecules. The mole numbers of ECl and H2O in ECl-H2O binary with ECl?H2O and ECl?3H2O as formed hydrous salt molecules before reaction equilibrium based on 1 kg H2O is presented as ![]()
![]() =55.6 mol. The mole number of structural units in ECl-H2O binary solution with ECl?H2O and ECl?3H2O as formed hydrous salt molecules under dynamic reaction equilibrium condition based on 1 kg H2O can be expressed as
=55.6 mol. The mole number of structural units in ECl-H2O binary solution with ECl?H2O and ECl?3H2O as formed hydrous salt molecules under dynamic reaction equilibrium condition based on 1 kg H2O can be expressed as ![]()
![]() =55.6,
=55.6, ![]()
![]() Hence, the total mole number of all structural units
Hence, the total mole number of all structural units ![]() based on 1 kg H2O in ECl-H2O binary solution with ECl?H2O and ECl?3H2O as formed hydrous salt molecules under equilibrium condition can be calculated according to the mass balance principle as
based on 1 kg H2O in ECl-H2O binary solution with ECl?H2O and ECl?3H2O as formed hydrous salt molecules under equilibrium condition can be calculated according to the mass balance principle as
![]() (36)
(36)
The mass balance of ECl and H2O in ECl-H2O binary solution with ECl?H2O and ECl?3H2O as formed hydrous salt molecules with known ![]() can be presented by simplifying Eq.(22) as
can be presented by simplifying Eq.(22) as
 (37a)
(37a)
 (37b)
(37b)
Eq.(23) and Eq.(24) can be rewritten for ECl-H2O binary solution with ECl?H2O and ECl?3H2O as formed hydrous salt molecules with known ![]() as follows:
as follows:
![]()
![]() (38)
(38)
![]() (39)
(39)
The thermodynamic model of ECl-H2O binary solution with ECl?H2O and ECl?3H2O as formed hydrous salt molecules with known ![]() is represented by combining Eq.(37) and Eq.(39) or Eq.(38) and Eq.(39). The parameters as Ni,
is represented by combining Eq.(37) and Eq.(39) or Eq.(38) and Eq.(39). The parameters as Ni, ![]() and ni can be solved by combining Eq.(37) and Eq.(39) or Eq.(38) and Eq.(39). The equilibrium constants of
and ni can be solved by combining Eq.(37) and Eq.(39) or Eq.(38) and Eq.(39). The equilibrium constants of ![]() and
and ![]() , Li or
, Li or ![]() and
and ![]() for ECl-H2O binary solution with ECl?H2O and ECl?3H2O as formed hydrous salt molecules with known
for ECl-H2O binary solution with ECl?H2O and ECl?3H2O as formed hydrous salt molecules with known ![]() are also the same as equations described in Eq.(13) and Eq.(14), respectively.
are also the same as equations described in Eq.(13) and Eq.(14), respectively.
3 Application of universal thermodynamic model
3.1 Application of developed models in binary aqueous solutions
3.1.1 Model application in binary aqueous solutions without any hydrous salt molecule formed
The representatives of ECl-H2O binary solutions without any hydrous salt complex molecule generated are KCl-H2O, CsCl-H2O, KBr-H2O and NH4Br-H2O according to their phase diagrams[16]. Hence, the mass action concentrations of structural units or ion couples in KCl-H2O[10], CsCl-H2O[10], KBr-H2O[12] and NH4Br-H2O[12] binary aqueous solutions at 298.15 K have been calculated by the developed thermodynamic model of calculating mass action concentrations for structural units or ion couples in ECl-H2O binary solution without any hydrous salt complex molecule generated. Taking KCl-H2O[10] binary solution as an example, the application of the developed universal thermodynamic model is briefly demonstrated hereinbelow.
The mass action concentrations of KCl and H2O in KCl-H2O binary solution without any hydrous salt molecule formed at 298.15 K can be calculated by Eq.(29) directly. The transformation coefficients ![]() and
and ![]() in various molalities of KCl are listed in Table 1, the relationships between
in various molalities of KCl are listed in Table 1, the relationships between ![]() and reported activity[17] aKCl
and reported activity[17] aKCl ![]() against xKCl,
against xKCl, ![]()
![]() and reported
and reported ![]() [17] against
[17] against ![]() , are illustrated in Fig.1, respectively.
, are illustrated in Fig.1, respectively.

Fig.1 Comparison of calculated mass action concentrations ![]() and
and ![]() with measured activities of KCl and H2O as aKCl
with measured activities of KCl and H2O as aKCl ![]() and
and ![]() in Ref.[17] for KCl-H2O binary solution without forming any complex hydrous salt molecule at 298.15 K
in Ref.[17] for KCl-H2O binary solution without forming any complex hydrous salt molecule at 298.15 K
It can be seen from Table 1 that the transformation coefficients of KCl (![]() ) keep constant with a small fluctuation range with an average datum of 18.099; meanwhile,
) keep constant with a small fluctuation range with an average datum of 18.099; meanwhile, ![]() is also stable with 1.000 as average datum. It can be also observed from Fig.1 that
is also stable with 1.000 as average datum. It can be also observed from Fig.1 that ![]() and
and ![]() are in good agreement with the report in Ref.[17] aKCl
are in good agreement with the report in Ref.[17] aKCl ![]() and
and ![]() . Therefore, it can be suggested that the calculated mass action concentrations can be applied to present reaction ability of KCl and H2O, and have a close corresponding relation with reported activity data; all hypotheses used
. Therefore, it can be suggested that the calculated mass action concentrations can be applied to present reaction ability of KCl and H2O, and have a close corresponding relation with reported activity data; all hypotheses used
Table 1 Transformation coefficients between calculated mass action concentrations and reported activities of structural units or ion couples for investigated six strong electrolyte aqueous solutions with various molalities of solutes at 298.15 K and equilibrium constant of forming NaCl?2H2O in NaCl-H2O binary solution at 298.15 K

in the developed thermodynamic model are reasonable and can depict the intrinsic structure of KCl-H2O binary aqueous solution.
3.1.2 Model application in binary system with ECl?2H2O as formed hydrous salt molecule with unknown ![]()
NaCl-H2O is a representative of ECl-H2O binary aqueous solution with ECl?2H2O as hydrous salt molecule with unknown ![]() based on its phase diagram[18]. The developed thermodynamic model of calculating mass action concentrations for structural units or ion couples in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt molecule without
based on its phase diagram[18]. The developed thermodynamic model of calculating mass action concentrations for structural units or ion couples in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt molecule without ![]() has been successfully applied in NaCl-H2O binary solution[10] at 298.15 K by using the determined stable equilibrium constant
has been successfully applied in NaCl-H2O binary solution[10] at 298.15 K by using the determined stable equilibrium constant ![]() through substituting
through substituting ![]() in formula of
in formula of ![]() by product of reported
by product of reported ![]() [17] and xNaCl as aNaCl and replacing
[17] and xNaCl as aNaCl and replacing ![]() in formula of
in formula of ![]() by the reported
by the reported ![]() respectively. The determined
respectively. The determined ![]() has an average datum as 1.043 with a small fluctuation in various molalities of NaCl in NaCl-H2O binary solution as shown in Table 1 at 298.15 K. The transformation coefficient
has an average datum as 1.043 with a small fluctuation in various molalities of NaCl in NaCl-H2O binary solution as shown in Table 1 at 298.15 K. The transformation coefficient ![]() and
and ![]() are stable with 58.897 and 0.960 as average datum, respectively, as shown in Table 1.
are stable with 58.897 and 0.960 as average datum, respectively, as shown in Table 1.
The transformed mass action concentrations ![]() and
and ![]() have been compared with the reported activities[17], aNaCl
have been compared with the reported activities[17], aNaCl ![]() and
and ![]() in Fig.2 in various mole fractions of NaCl and H2O before equilibrium, respectively. There is a good corresponding relation between
in Fig.2 in various mole fractions of NaCl and H2O before equilibrium, respectively. There is a good corresponding relation between ![]() and aNaCl,
and aNaCl, ![]() and
and ![]() with small deviation, respectively. Therefore, NaCl?2H2O surely exists in NaCl-H2O binary solution as described in phase diagram of NaCl-H2O binary solution[18], the developed thermodynamic model of NaCl-H2O binary solution can represent the intrinsic structure of NaCl-H2O binary solution.
with small deviation, respectively. Therefore, NaCl?2H2O surely exists in NaCl-H2O binary solution as described in phase diagram of NaCl-H2O binary solution[18], the developed thermodynamic model of NaCl-H2O binary solution can represent the intrinsic structure of NaCl-H2O binary solution.
3.1.3 Model application in binary system with ECl?2H2O as formed hydrous salt molecule with known ![]()
NaBr-H2O, KF-H2O and ZnBr2-H2O are representatives of ECl-H2O binary aqueous solution with ECl?2H2O as hydrous salt molecule with known ![]() from their phase diagrams[16]. The established thermodynamic model of calculating mass action concentrations for structural units or ion couples in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt molecule with known
from their phase diagrams[16]. The established thermodynamic model of calculating mass action concentrations for structural units or ion couples in ECl-H2O binary solution with ECl?2H2O as formed hydrous salt molecule with known ![]() has been successfully applied in NaBr-H2O and KF-H2O[11], and ZnBr2-H2O[12] binary aqueous solutions at 298.15 K. Taking KF-H2O[11] binary solution as an example to demonstrate the thermodynamic model of binary solution with KF?2H2O as formed hydrous salt molecule with known
has been successfully applied in NaBr-H2O and KF-H2O[11], and ZnBr2-H2O[12] binary aqueous solutions at 298.15 K. Taking KF-H2O[11] binary solution as an example to demonstrate the thermodynamic model of binary solution with KF?2H2O as formed hydrous salt molecule with known ![]() at 298.15 K, the calculated
at 298.15 K, the calculated ![]() in various molalities of KF has an average datum of 1611.0 as summarized in Table 1.
in various molalities of KF has an average datum of 1611.0 as summarized in Table 1.
The transformed mass action concentration ![]() and reported activity[19] aKF
and reported activity[19] aKF![]() are compared in Fig.3. There is a good corresponding relation between
are compared in Fig.3. There is a good corresponding relation between ![]() and aKF
and aKF ![]() with a small deviation. It can be seen from Table 1 that the transformation coefficient of KF (
with a small deviation. It can be seen from Table 1 that the transformation coefficient of KF (![]() ) are relatively constant with 1611.0 as an average value. Hence, the hypotheses in the developed thermodynamic model of KF-H2O binary system are reasonable; the calculated mass action concentration of KF can represent reaction ability of KF in KF-H2O binary system; KCl?2H2O surely exists in KF-H2O binary solution as described in phase diagram of KF-H2O binary solution[16]; the established thermodynamic model of KCl-H2O binary solution can
) are relatively constant with 1611.0 as an average value. Hence, the hypotheses in the developed thermodynamic model of KF-H2O binary system are reasonable; the calculated mass action concentration of KF can represent reaction ability of KF in KF-H2O binary system; KCl?2H2O surely exists in KF-H2O binary solution as described in phase diagram of KF-H2O binary solution[16]; the established thermodynamic model of KCl-H2O binary solution can

Fig.2 Comparison of calculated mass action concentrations ![]() and
and ![]() with measured activities of NaCl and H2O as aNaCl
with measured activities of NaCl and H2O as aNaCl ![]() and
and ![]() in Ref.[17] for NaCl-H2O binary solution with NaF?2H2O as formed hydrous salt molecule at 298.15 K
in Ref.[17] for NaCl-H2O binary solution with NaF?2H2O as formed hydrous salt molecule at 298.15 K

Fig.3 Comparison of calculated mass action concentration ![]() with measured activity of KF as aKF
with measured activity of KF as aKF ![]() in Ref.[19] for KF-H2O binary solution with KF?2H2O as formed hydrous salt molecule at 298.15 K
in Ref.[19] for KF-H2O binary solution with KF?2H2O as formed hydrous salt molecule at 298.15 K
represent the intrinsic structural properties of KCl-H2O binary solution.
3.1.4 Model application in binary system with ECl?H2O and ECl?3H2O as formed hydrous salt molecules with known ![]()
HNO3-H2O is a representative of ECl-H2O binary aqueous solution with ECl?H2O and ECl?3H2O as formed hydrous salt molecules with known![]() from its phase diagram[16]. The developed thermodynamic model of calculating mass action concentrations for structural units or ion couples in ECl-H2O binary with ECl?H2O and ECl?3H2O as formed hydrous salt molecules with known
from its phase diagram[16]. The developed thermodynamic model of calculating mass action concentrations for structural units or ion couples in ECl-H2O binary with ECl?H2O and ECl?3H2O as formed hydrous salt molecules with known ![]() has been successfully applied in HNO3-H2O binary solution at 298.15 K as reported in previous publication[11]. The calculated
has been successfully applied in HNO3-H2O binary solution at 298.15 K as reported in previous publication[11]. The calculated ![]() has a narrow deviation with an average of 80 753.0 in large molality range as listed in Table 1. The transformed mass action concentrations of HNO3
has a narrow deviation with an average of 80 753.0 in large molality range as listed in Table 1. The transformed mass action concentrations of HNO3 ![]() and reported activity[19]
and reported activity[19] ![]()
![]() with mole fraction
with mole fraction ![]() before equilibrium as horizontal axis are compared in Fig.4. There is a good corresponding relationship between
before equilibrium as horizontal axis are compared in Fig.4. There is a good corresponding relationship between ![]() and
and ![]()
![]() with a small deviation. It can be deduced from Fig.4 that the structural properties of considering HNO3?H2O and HNO3?3H2O as formed hydrous salt molecules in HNO3-H2O binary solution from phase diagram[16] are reasonable.
with a small deviation. It can be deduced from Fig.4 that the structural properties of considering HNO3?H2O and HNO3?3H2O as formed hydrous salt molecules in HNO3-H2O binary solution from phase diagram[16] are reasonable.
3.2 Application of developed models in ternary aqueous solutions
3.2.1 Model application in ternary systems with formed hydrous salt molecules
The developed thermodynamic model of calculating mass action concentrations for structural units or ion

Fig.4 Comparison of calculated mass action concentration ![]() with measured activity of HNO3
with measured activity of HNO3 ![]()
![]() in Ref.[19] for HNO3-H2O binary solution with HNO3?H2O and HNO3?3H2O as formed hydrous salt molecules at 298.15 K
in Ref.[19] for HNO3-H2O binary solution with HNO3?H2O and HNO3?3H2O as formed hydrous salt molecules at 298.15 K
couples in ternary aqueous solution has been successfully applied in NaCl-KCl-H2O[13] and NaClO4-NaF-H2O [15] ternary solutions with formed hydrous salt molecules at 298.15 K. Choosing NaCl-KCl-H2O ternary solution with NaCl・2H2O as hydrous salt molecule at 298.15 K as an example, the structural units in NaCl-KCl-H2O ternary solution are composed of Na+, K+, and Cl- as simple ions, H2O as simple and NaCl・2H2O as complex molecules from the phase diagram[18]. The mass balance of NaCl, KCl and H2O in NaCl-KCl-H2O ternary solution based on 1 kg H2O shown in Eq.(10) can be simplified as
 (40a)
(40a)
![]() (40b)
(40b)
 (40c)
(40c)
Eq.(11) and Eq.(12) can be rewritten for NaCl-KCl-H2O ternary solution as follows:
![]() (41a)
(41a)
![]() (41b)
(41b)
![]()
![]() (42)
(42)
Therefore, the equation groups of Eq.(40) and Eq.(42) or Eq.(41) and Eq.(42) are composed of the universal thermodynamic model of calculation mass action concentrations for structural units or ion couples Ni in NaCl-KCl-H2O ternary solution, respectively. The mass action concentrations of NaCl, KCl and H2O in NaCl-KCl-H2O ternary solution have been calculated and reported in detail in previous publication[13] in different mole number ratios of NaCl to KCl, i.e., r=mNaCl/mKCl, as 0.2, 0.5, 1.0, 2.0, 3.0, and 4.0, respectively, under condition of knowing ![]() as describled in Section 3.1.2 as 1.042[13]. The transformed mass action concentration
as describled in Section 3.1.2 as 1.042[13]. The transformed mass action concentration ![]()
![]() and
and![]() have been compared with the reported activities[20], aNaCl
have been compared with the reported activities[20], aNaCl ![]() aKCl
aKCl ![]() and
and ![]() , with different r.
, with different r.
As a representative of results, the transformation coefficients ![]() and
and ![]() with r of 4.0 are listed in Table 1 as 55.606 and 16.157, respectively; meanwhile, comparison of
with r of 4.0 are listed in Table 1 as 55.606 and 16.157, respectively; meanwhile, comparison of ![]()
![]() and
and ![]() with the reported activities[20], aNaCl
with the reported activities[20], aNaCl ![]() aKCl
aKCl ![]() and
and ![]() , is also given in Fig.5 with r of 4.0, respectively.
, is also given in Fig.5 with r of 4.0, respectively.
It can be observed from Fig.5 that the transformed mass action concentrations ![]()
![]() and
and ![]() , are in good agreement with the reported activities[20], aNaCl
, are in good agreement with the reported activities[20], aNaCl ![]() aKCl
aKCl ![]() and
and ![]() with r of 4.0. It is shown from Table 1 that
with r of 4.0. It is shown from Table 1 that ![]() and
and ![]() remain constant with small deviations with r of 4.0. Hence, the developed thermodynamic model of calculating mass action concentrations for structural units or ion couples in NaCl-KCl-H2O ternary solution can be successfully applied to predict reaction ability and reflect structural characteristics.
remain constant with small deviations with r of 4.0. Hence, the developed thermodynamic model of calculating mass action concentrations for structural units or ion couples in NaCl-KCl-H2O ternary solution can be successfully applied to predict reaction ability and reflect structural characteristics.
3.2.2 Model application in ternary systems without any hydrous salt molecule formed
The developed thermodynamic model for ternary solution without any hydrous salt molecule formed has been successfully applied in RbCl-RbNO3-H2O ternary solution[14] at 298.15 K. Certainly, the structural units in RbCl-RbNO3-H2O ternary solution at 298.15 K are composed of Rb+, Cl- and ![]() as simple ions, H2O as simple molecule from the phase diagram[18]. The mass balance of RbCl, RbNO3 and H2O in RbCl-RbNO3-H2O ternary solution based on 1 kg H2O shown in Eq.(10) can be simplified as
as simple ions, H2O as simple molecule from the phase diagram[18]. The mass balance of RbCl, RbNO3 and H2O in RbCl-RbNO3-H2O ternary solution based on 1 kg H2O shown in Eq.(10) can be simplified as
![]() (43a)
(43a)
![]() (43b)
(43b)

Fig.5 Comparison of calculated mass action concentrations ![]()
![]() and
and ![]() with reported activities of NaCl, KCl and H2O as aNaCl
with reported activities of NaCl, KCl and H2O as aNaCl ![]() aKCl
aKCl ![]() and
and ![]() in Ref.[20] for NaCl-KCl-H2O ternary system with r of 4.0 at 298.15 K
in Ref.[20] for NaCl-KCl-H2O ternary system with r of 4.0 at 298.15 K
![]() (43c)
(43c)
Eq.(11) and Eq.(12) can be expressed for RbCl-RbNO3-H2O ternary solution, respectively, as
![]() (44a)
(44a)
![]() (44b)
(44b)
![]() (45)
(45)
Therefore, the equation groups of Eq.(43) and Eq.(45) or Eq.(44) and Eq.(45) are composed of the universal thermodynamic model of calculation mass action concentrations for structural units or ion couples Ni in RbCl-RbNO3-H2O ternary solution, respectively. The mass action concentrations of RbCl, RbNO3 and H2O in RbCl-RbNO3-H2O ternary solution have been calculated and reported in detail in previous publication[14] in a range of the total ionic strength, i.e., ![]() , from 0.1 to 0.9 mol/kg (H2O) in 0.1 mol/kg (H2O) step with different ionic strength fractions of NaClO4,
, from 0.1 to 0.9 mol/kg (H2O) in 0.1 mol/kg (H2O) step with different ionic strength fractions of NaClO4, ![]()
![]() as 0, 0.2, 0.4, 0.5, 0.6 and 0.8, respectively[14]. The transformed mass action concentrations
as 0, 0.2, 0.4, 0.5, 0.6 and 0.8, respectively[14]. The transformed mass action concentrations ![]() and
and ![]() have been compared with the reported activities[21], aRbCl
have been compared with the reported activities[21], aRbCl ![]() and
and ![]()
![]() with different I values reported elsewhere[14].
with different I values reported elsewhere[14].
As a representative of results, the transformation coefficients ![]() and
and ![]() with I of 0.5 mol/kg (H2O) are listed in Table 1, meanwhile, comparison of
with I of 0.5 mol/kg (H2O) are listed in Table 1, meanwhile, comparison of ![]() and
and ![]() with the reported activities[21], aRbCl
with the reported activities[21], aRbCl ![]() and
and ![]()
![]() , are also given in Fig.6 under condition of I of 0.5 mol/kg (H2O), respectively.
, are also given in Fig.6 under condition of I of 0.5 mol/kg (H2O), respectively.
It can be obtained from Table 1, Fig.6 and other results described in previous publication elsewhere[14] that ![]() and
and ![]() are in good agreement of reported activities[21], aRbCl
are in good agreement of reported activities[21], aRbCl ![]() and
and ![]()
![]() in a large change range of I ; the transformation coefficients,
in a large change range of I ; the transformation coefficients, ![]() and
and ![]() , listed in Table 1, keep constant with 17.29 and 15.61 as average datum with small deviations under condition of I as 0.50 mol/kg (H2O) for various
, listed in Table 1, keep constant with 17.29 and 15.61 as average datum with small deviations under condition of I as 0.50 mol/kg (H2O) for various ![]() , respectively. Therefore, the developed thermodynamic model of calculation mass action concentrations for structural units or ion couples in RbCl-RbNO3-H2O ternary solution can be successfully applied to predict reaction ability and reflect structural characteristics.
, respectively. Therefore, the developed thermodynamic model of calculation mass action concentrations for structural units or ion couples in RbCl-RbNO3-H2O ternary solution can be successfully applied to predict reaction ability and reflect structural characteristics.
4 Discussion and error analysis
Although the transformed mass action concentrations of structural units or ion couples ![]() in various binary and ternary aqueous solutions have good consistency with the reported activity ai
in various binary and ternary aqueous solutions have good consistency with the reported activity ai ![]() , small difference between
, small difference between ![]() and ai can be observed for some solutions in high concentration range of solute. The
and ai can be observed for some solutions in high concentration range of solute. The

Fig.6 Comparison of mass action concentrations ![]() and
and ![]() with reported activities of RbCl and NbNO3 as aRbCl and
with reported activities of RbCl and NbNO3 as aRbCl and ![]() in Ref.[21] for RbCl-RbNO3-H2O ternary solution with total ionic strength I of 0.5 mol/kg (H2O) at 298.15 K
in Ref.[21] for RbCl-RbNO3-H2O ternary solution with total ionic strength I of 0.5 mol/kg (H2O) at 298.15 K
reasons of small errors of the transformed mass action concentrations of structural units or ion couples with the reported activities can be summarized as follows:
1) Solute in high concentration range almost reaches saturation.
2) A transformation coefficient ![]() must be used when comparing the calculated mass action concentration Ni and the reported activity ai
must be used when comparing the calculated mass action concentration Ni and the reported activity ai ![]() of solute i. However, the calculated
of solute i. However, the calculated ![]() is based on pure matter as standard state and mole fraction
is based on pure matter as standard state and mole fraction ![]() as concentration unit, while the reported ai
as concentration unit, while the reported ai ![]() is relative to infinite dilution as standard state and molality mi as concentration unit. The average transformation coefficient
is relative to infinite dilution as standard state and molality mi as concentration unit. The average transformation coefficient ![]() is the average of ratios for ai to Ni with various solute concentrations in the investigated concentration range, some errors can be certainly generated from the average calculated transformation coefficient
is the average of ratios for ai to Ni with various solute concentrations in the investigated concentration range, some errors can be certainly generated from the average calculated transformation coefficient ![]() . The idea method is to compare the calculated mass action concentrations of structural units or ion couples and reported activities relative to pure matter as standard state and mole fraction xi as concentration unit for solutes.
. The idea method is to compare the calculated mass action concentrations of structural units or ion couples and reported activities relative to pure matter as standard state and mole fraction xi as concentration unit for solutes.
3) To some strong electrolyte aqueous solutions, equilibrium constant ![]() of reactions for forming hydrous salt molecules cannot be accurately calculated because its free formation energy
of reactions for forming hydrous salt molecules cannot be accurately calculated because its free formation energy ![]() cannot be found from related literatures. The suggested method to get
cannot be found from related literatures. The suggested method to get ![]() by ZHANG[7-9] is calculating
by ZHANG[7-9] is calculating ![]() by substituting
by substituting ![]() in formula of
in formula of ![]() by product of reported activity coefficient f′i multiplying by xi as a′i
by product of reported activity coefficient f′i multiplying by xi as a′i ![]() for solute, but not traditional activity ai
for solute, but not traditional activity ai ![]() for solute, with a relatively stable in an acceptable fluctuation range. It is not an absolutely correct, but an acceptable method. The average datum
for solute, with a relatively stable in an acceptable fluctuation range. It is not an absolutely correct, but an acceptable method. The average datum ![]() can certainly induce some deviations.
can certainly induce some deviations.
5 Conclusions
1) A universal thermodynamic model of calculating mass action concentrations for structural units or ion couples in ternary and binary strong electrolyte aqueous solutions based on the ion and molecular coexistence theory has been developed and verified in four kinds of binary and two kinds of ternary aqueous solutions. The calculated mass action concentrations of structural units or ion couples in four binary and two ternary solutions have good agreement with the reported activity data from literatures after shifting the standard state and concentration unit.
2) The mass action concentrations of structural units or ion couples calculated from the developed universal thermodynamic model for calculating mass action concentrations of structural units or ion couples in ternary and binary aqueous solutions can be applied to predict reaction ability of components in ternary and binary strong electrolyte aqueous solutions.
3) The developed universal thermodynamic model for ternary and binary aqueous solutions provides a useful method to judge structural characteristics of ternary and binary strong electrolyte aqueous solutions.
4) It is confirmed from the developed universal thermodynamic model for ternary and binary aqueous solutions that ternary and binary strong electrolyte aqueous solutions are composed of cations and anions as simple ions, H2O as simple molecule, and other hydrous salt compounds as complex molecules.
5) The calculated mass action concentrations of structural units or ion couples in the investigated ternary and binary strong electrolyte aqueous solutions strictly follow the mass action law.
Symbol list
ai―Reported activity of component i in aqueous solutions as ![]() , (-);
, (-);
a′i―Activity of component i in aqueous solutions defined as ![]() , (-);
, (-);
bi―Total mole number of components i before chemical reaction equilibrium in aqueous solutions, mol;
f′i―Activity coefficient of component i relative to infinite dilute solution as standard state and molality mi as concentration unit, (-);
I―Total ionic strength of solutes i and j in aqueous solutions as mi+mj, mol/kg (H2O);
![]() ―Chemical equilibrium constant, (-);
―Chemical equilibrium constant, (-);
![]() ―Transformation coefficient of component i between the calculated mass action concentration chosen pure matter as standard state and mole fraction xi as concentration unit and the reported activity relative to infinite dilute solution as standard state and molality mi as concentration unit, (-);
―Transformation coefficient of component i between the calculated mass action concentration chosen pure matter as standard state and mole fraction xi as concentration unit and the reported activity relative to infinite dilute solution as standard state and molality mi as concentration unit, (-);
![]() ―Average of
―Average of ![]() , (-);
, (-);
mi―Molality of component i in aqueous solutions, mol/kg (H2O);
![]() ―Mole mass of H2O, 18 g/mol;
―Mole mass of H2O, 18 g/mol;
![]() ―Total mole number of components i before chemical reaction in aqueous solutions, same as bi, mol;
―Total mole number of components i before chemical reaction in aqueous solutions, same as bi, mol;
ni―Equilibrium mole number of structural unite i or ion couple i in aqueous solutions, mol;
Ni―Mass action concentrations of structural unite i or ion couple i in aqueous solutions, (-);
![]() ―Transformed mass action concentration of structural unit i or ion couple i in aqueous solutions, (-);
―Transformed mass action concentration of structural unit i or ion couple i in aqueous solutions, (-);
R―Gas constant, 8.314?10-3 kJ/(mol×K);
r―Ratio of molality for solute i to solute j, (-);
T―Absoulte temperature, K;
vi―Ionic strength fraction of component i as mi/I, (-);
xi―Mole fraction of components i before equilibrium, (-);
![]() ―Standard molar Gibbs free energy of forming hydrous salt compound i as complex molecule, kJ/mol;
―Standard molar Gibbs free energy of forming hydrous salt compound i as complex molecule, kJ/mol;
![]() ―Total mole number of all structural units in aqueous solutions under equilibrium condition, mol;
―Total mole number of all structural units in aqueous solutions under equilibrium condition, mol;
![]() ―Total mole number of structural units before dynamic equilibrium, mol.
―Total mole number of structural units before dynamic equilibrium, mol.
References
[1] Gokcen N A. Determination and estimation of ionic activities of metal salts in water [R]. Report of Investigations No.8372, U.S. Department of the Interior, Bureau of Mines, Washington, D C, 1979.
[2] CHAN C K, HA Z Y, CHOI MY. Study of water activities of aerosols of mixtures of sodium and magnesium salts [J]. Atmospheric Environment, 2000, 34(28): 4795-4803.
[3] HA Z Y, CHAN C K. The water activities of MgCl2, Mg(NO3)2, MgSO4, and their mixtures [J]. Aerosol Science Technology, 1999, 31(2-3): 154-169.
[4] Pruppacher H R, Klett J D. Microphysics of clouds and precipitation [M]. Dordrecht, Holland: Reidel Publishing Company, 1978.
[5] Molina M J, Zhang R, Wooldridge P J, McMahon J R, KIM J E, CHANG H Y, BEYER K D. Physical chemistry of the H2SO4/HNO3/H2O system: Implications for polar stratospheric clouds [J]. Science, 1993, 261: 1418-1423.
[6] Guendouzi M E, Dinane A. Determination of water activities, osmotic and activity coefficients in aqueous solutions using the hygrometric method [J]. J Chem Thermodynamics, 2000, 32(3): 297-310.
[7] Zhang Jian. The coexistence theory of slag structure [J]. Journal of University of Science and Technology Beijing, 1984, 6(1): 21-29. (in Chinese)
[8] ZHANG Jian. Application of the annexation principle to the thermodynamic property study of ternary metallic melts In-Bi-Cu and In-Sb-Cu [J]. Journal of University of Science and Technology Beijing, 2002, 9(3): 170-176. (in Chinese)
[9] Zhang Jian. Computational thermodynamics of metallurgical melts and solutions [M]. Beijing: Metallurgical Industry Press, 2007. (in Chinese)
[10] GUO Han-jie, Zhao Wei-jie, LI Lin, Yang Xue-min. A universal thermodynamic model of calculating mass action concentration of components in strong electrolyte binary aqueous solutions [J]. The Chinese Journal of Process Engineering, 2007, 7(2): 347-353. (in Chinese)
[11] GUO H J, Zhao W J, Yang X. M. Calculating models of mass action concentrations for NaBr(aq), LiNO3(aq), HNO3(aq), and KF(aq) binary solutions [J]. Journal of University of Science and Technology Beijing, 2007, 14(3): 204-211.
[12] Zhao Wei-jie, GuO Han-jie, DAN Zhi-gang. Calculating models of mass action concentrations for KBr(aq),NH4Br(aq) and ZnBr2(aq) binary solutions [J]. Journal of University of Science and Technology Beijing, 2008, 30(8): 926-930. (in Chinese)
[13] Zhao W J, GUO H J, Yang X M. A universal thermodynamic model of calculating mass action concentrations for components in ternary strong electrolyte aqueous solution and its application in NaCl-KCl-H2O system [J]. Journal of University of Science and Technology Beijing, 2008, 15(5): 543-551.
[14] GUO H J, YANG X M, ZHAO W J. Calculating models of mass action concentrations for RbCl-H2O binary system and RbCl-RbNO3-H2O ternary system based on ion-molecule coexistence theory [J]. Transactions of Nonferrous Metals Society of China, 2010, 20(6): 1112-1120.
[15] YANG X M, ZHAO W J, GUO H J, ZHANG Q, ZHANG J. A Thermodynamic model of calculating mass action concentrations for structural units or ion couples in NaClO4-H2O and NaF-H2O binary solutions and NaClO4-NaF-H2O ternary solution [J]. International Journal of Minerals, Metallurgy and Materials, 2010, 17(5): 546-557.
[16] WAGMAN D D, EVANS W H, PARKER V B, SCHUMM R H, HALOW I, BAILEY S M, CHURNEY K L, NUTTALL R L. The NBS tables of chemical thermodynamic properties-selected values for inorganic and C1 and C2 organic substances in SI units [M]. LIU Tian, ZHAO Meng-yue. Beijing: Chinese Standards Press, 1998. (in Chinese)
[17] Guendouzi M EI, Dinane A, Mounir A. Water activities, osmotic and activity coefficients in aqueous chloride solutions at T=298.15 K by the hygrometric method [J]. The Journal of Chemical Thermodynamics, 2001, 33(9): 1059-1072.
[18] Niu Zi-de, Cheng Fang-qin. Phase diagrams of hydrous slats and their applications [M]. Tianjin: Tianjin University Publishing House, 2002. (in Chinese)
[19] Рабинович В А. The concise handbook of chemistry [M]. YIN Cheng-lie. Beijing: Chemical Industry Press, 1983: 651-659. (in Chinese)
[20] Dinane A, Guendouzi M E, Mounir A. Hygrometric determination of water activities, osmotic and activity coefficients of (NaCl+KCl)(aq) at T=298.15 K [J]. The Journal of Chemical Thermodynamics, 2002, 34(4): 423-441.
[21] Zhang J, Huang Y, Xia S P. Experimental determination and prediction of activity coefficients of RbCl in aqueous (RbCl+RbNO3) mixture at T=298.15 K [J]. The Journal of Chemical Thermodynamics, 2005, 37(11): 1162-1167.
杨学民1, 赵伟洁2, 3, 柴国明2, 郭汉杰2, 张 强3
1. 中国科学研究院 过程工程研究所 多相复杂系统国家重点实验室,北京 100190;
2. 北京科技大学 冶金与生态工程学院,北京 100083;
3. 北京中冶设备研究设计总院有限公司, 北京 100029
摘 要:基于离子与分子共存理论,建立了计算二元和三元强电解质水溶液中结构单元或离子对的质量作用浓度的通用热力学模型;同时,采用4种二元水溶液和2种三元水溶液验证该通用热力学模型。通过转换标准态和浓度单位,用所建立的通用热力学模型计算出的298.15 K时4种二元水溶液和2种三元水溶液中结构单元或离子对的质量作用浓度和文献中报道的活度值吻合得很好。因此,可采用本研究提出的通用热力学模型计算出的二元和三元水溶液结构单元或离子对的质量作用浓度预报二元和三元强电解质水溶液中组元的反应能力;同样,也可证实本研究提出的通用热力学模型的假设条件是正确和合理的,即强电解质水溶液是由阳离子和阴离子、H2O分子和其他水合盐复杂分子组成的。基于该通用热力学模型计算出的二元和三元强电解质水溶液结构单元或离子对的质量作用浓度严格服从质量作用定律。
关键词:通用热力学模型;质量作用浓度;活度;二元水溶液;三元水溶液;离子-分子共存理论;结构单元;离子对;组元
(Edited by YANG Hua)
Foundation item: Project supported by Publication Foundation of National Science and Technology Academic Books of China
Corresponding author: YANG Xue-min; Tel: +86-10-82622893; Fax: +86-10-82622893; E-mail: yangxm71@home.ipe.ac.cn
DOI: 10.1016/S1003-6326(11)60760-7

