
Preservation of moderately thermophilic culture by freeze drying and
frozen preservation way and effect on subsequent bioleaching of chalcopyrite
ZENG Wei-min(曾伟民)1, 2, ZHOU Hong-bo(周洪波)1, 2, LIU Xue-duan(刘学端)1, 2, QIU Guan-zhou(邱冠周)1, 2
1. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China;
2. Key Laboratory of Biometallurgy, Ministry of Education, Central South University, Changsha 410083, China
Received 21 April 2009; accepted 26 July 2009
Abstract: Freeze drying and frozen preservation way was used to preserve a moderately thermophilic culture for bioleaching of chalcopyrite concentrate. After preservation of 15 months, the cell viability rate decreases to 22% with a cell density of 7×107 mL-1. When the growth time was extended from 8 days to 14 days, cell density would increase in a large scale to about 3×108 mL-1. In the bioleaching experiments, unpreserved and preserved cultures were compared for dissolving chalcopyrite concentrate. Before 44 days, the unpreserved culture can reach a high copper extraction of about 17.4 g/L. While the preserved culture shows a rather low copper extraction, which is only 9.7 g/L. When the bioleaching time was extended to 80 days, copper extraction by preserved culture increases remarkably, and the concentration of copper finally achieves up to 18.3 g/L. On the other hand, copper extraction by the unpreserved culture does not show remarkable increase from 44th to the 80th day, and finally the total copper extraction is 19.8 g/L. As a result, total copper extraction in 80 days by preserved culture approaches that by unpreserved culture and freeze drying and frozen preservation even after 15 months does not bring much decrease of bioleaching ability.
Key words: freeze drying and frozen preservation; moderately thermophilic culture; bioleaching; chalcopyrite
1 Introduction
Bioleaching of chalcopyrite, a primary sulphide mineral, is considered to be a major challenge in the bio-hydrometallurgy due to slow kinetics and poor extraction. Many researchers have investigated the feasibility of using moderately thermophilic microorganisms to improve the bioleaching rate of chalcopyrite[1-2]. Using moderate thermophiles to leach sulphide minerals not only greatly improves the reaction kinetics, but also avoids excessive chalcopyrite passivation which hinders the durative bioleaching[3-5]. Furthermore, moderately thermophilic microorganisms can tolerate rather high pulp density[6]. Thus, they show advantage in application in industry for bioleaching of chalcopyrite. In our previous study, a moderately thermophlic culture was used to bioleach chalcopyrite, and copper extraction percentage achieved up to 75% in 44 days at 8% pulp density[7]. However, the cell loss of this culture and the degeneration of its bioleach ability during passage culture often occur in the laboratory research and industrial applications. As a result, it is very important to find a most effective preservation method which would preserve this useful culture in a long time.
The methods for storing microorganisms generally include passage culture, sterile sand tube preservation, freezing preservation, freeze drying and frozen preservation, and so on. However, freeze drying and frozen preservation is considered to be the most feasible and effective storage way for pure strains and mixed cultures in a long time[8-9]. Freeze drying (also known as lyophilization or cryodesiccation) is a dehydration process typically used to preserve a bacterium. After freeze drying, frozen preservation at -20 ℃ or -70 ℃ could obviously increase the storing time of the microorganism and improve the cell viability for long periods[10]. On the other hand, freeze drying and frozen preservation would cause major of cells to die during freeze drying and the death rate generally is very different according to the equipment and operation[11]. However, the remained cells get the surrounds of lacking air, nutrient and water, and so they can stay in a dormant phase. If only the original cell population is sufficient, 0.1% survival rate is considered to be efficient to continue the growth of cells and retain their metabolically ability after cell reviving[12].
There are few reports about preservation of bioleaching microorganisms especially for moderately thermophilic microorganisms. In this study, freeze drying and frozen preservation way was used to preserve a moderately thermophilic culture and its characterization for bioleaching of chalcopyrite before and after preservation was studied to investigate the effect of bacterial preservation on the ability of this culture for chalcopyrite bioleaching.
2 Experimental
2.1 Microorganisms enrichment
Mixed moderately thermophilic cultures from several chalcopyrite mines in China were collected and enriched. The medium used for enrichment consisted of the following compounds: (NH4)2SO4 3.0 g/L, Na2SO4 2.1 g/L, MgSO4?7H2O 0.5 g/L, K2HPO4 0.05 g/L, KCl 0.1 g/L, Ca(NO3)2 0.01 g/L. 10g/L FeSO4 and 5 g/L S0 were added as the energy source. The samples were enriched at 48 ℃ and initial pH 2.0.
2.2 Mineral components
The concentrate sample was collected from Meizhou Copper Mine in Guangdong Province, China, with diameter less than 75 μm. The mineral sample mainly consisted of chalcopyrite (62.2%), galena (25.6%) and chalcocite (10.8%).
2.3 Freeze drying and frozen preservation
Cells were harvested and concentrated by centrifugation (104g, 10 min and 4 ℃). Concentrated cells were re-suspended into 20% (mass fraction) mannitol as cryoprotectant. 40 ?L aliquot containing approximately 1×109 cells/mL organisms in cryoprotectant solution with 20% mannitol was dispensed into 1.5 mL ampoule and then was frozen by direct immersion in a liquid nitrogen (-196 ℃). Then, the frozen samples were dried by a freezing-dryer for a period of 14 h at 4 ℃. After that, the freezing dry samples were stored at -72 ℃ in a refrigerator-freezer. When storing for 3, 6, 9, 12 and 15 months, one of the tubes was taken out for cell reviving. For reviving, the frozen samples were thawed for 10 min at 30 ℃ in a water bath, then inoculated in medium including 10 g/L FeSO4 and 5 g/L S0 for 8 d. The cell density was analyzed under an optical microscope.
2.4 Cell viability
Cell viability was measured by counting the cell density before preservation (N0) and after different times of storage (Nt). The viability rate (V) was expressed by cell survival rate that was calculated as follows: V=100%×Nt/N0, where t takes values of 3, 6, 9, 12 and 15, indicating 3, 6, 9, 12 and 15 months, respectively.
2.5 Bioleaching of chalcopyrite
Bioleaching experiments were carried out in a 3 L glass cylindrical reactor with a mechanic stirrer operating at 500 r/min. 1 980 mL medium and 0.4 g/L yeast extract were added into the reactor; and 20 mL seed culture was inoculated. Both the preserved and unpreserved cultures used as inocula in the bioleaching were cultured for 8 d and the inoculum concentration got a same cell density of 106 mL-1. The reactor was placed in a thermostatic bath to keep the constant temperature at (48±0.2) ℃. Air was blown into the base of the reactor at an approximate rate of 360 mL/min. The experiments were performed at initial pH 2.0 and the pulp density of chalcopyrite of 10 g/L. The acid consumption was compensated by 10 mol/L sulfuric acid to keep pH value at around 2. Distilled water was added to the reactor through a peristaltic pump in order to compensate for evaporation losses.
Redox potential, cell density and concentrations of Cu2+, Fe3+ and total iron in solution were analyzed every two days, while pH value was analyzed each day.
2.6 Analysis methods
The components of the mineral sample were analyzed by XRD. Copper ion and total iron concentrations in solution were determined by atomic absorption spectrophotometer. Ferrous ion concentration in solution was assayed by titration with potassium dichromate. The pH value was measured with pH S-3C acid meter. The redox potential was measured with a platinum electrode with an Ag/AgCl reference electrode. Free cells in solution were observed and counted by the blood counting chamber under an optical microscope.
3 Results and discussion
3.1 Freeze drying and frozen preservation of moderate thermophiles
Freeze drying has been used to preserve microorganisms for decades and it is the preferred method for culture strains worldwide. The products after freeze drying are available for storage[8]. Therefore, in this experiment, freeze drying and frozen preservation way was used to preserve a moderately thermophilic culture.
The unpreserved culture could achieve to the maximum cell density of 3.2×108 mL-1 in 8 d with an initial inocular cell amount of 106 mL-1. When the growth continues to the 16th day, cell density would decrease in a large scale to about 4.6×107 mL-1.
Fig.1 shows the growth curve of preserved culture after reviving at the 3rd, 6th, 9th, 12th and 15th months. When the culture was preserved for 3 months, the maximum cell density decreased to 1.7×108 mL-1 after reviving for 8 d, and the cell viability rate is only 54%. The longer the storing time, the lower the maximum cell density. When the culture was preserved for 15 months, the cell viability rate decreased to 22%. This indicates that the growth of culture after preservation is inhibited in a large scale. On the other hand, it can also be seen from Fig.1 that the growth of preserved culture has a longer lag phase than the growth of the unpreserved culture. And at the 8th day, the growth of preserved cultures mostly stays in a logarithmic phase, but not in the stable growth phase. These show that the cell density of preserved culture should have an increase after 8 d. As a result, another experiment for prolonging the culture time was carried out. The result shows that when the culture time was prolonged to 14 d, the cell density of preserved culture for 3, 6, 9, 12 and 15 months would increase to the maximum cell density of 3×108, 2.9×108, 2.8×108, 2.5×108 and 2.4×108 mL-1, respectively, which is almost the same as the unpreserved culture. These indicate that freeze drying and frozen preservation could cause many cells death, but it is not obvious to decrease the growth ability of the living cells.
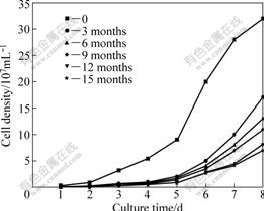
Fig.1 Growth curves of moderately thermophlic preserved after 0(unpreserved), 3, 6, 9, 12 and 15 months
3.2 Bioleaching of chalcopyrite concentrates with unpreserved and preserved cultures
Fig.2 shows the copper concentrations by the unpreserved culture and preserved culture after 15 months in the bioleaching reactor as a function of time. In the bioleaching with the unpreserved culture, copper extraction increases quickly to 9.7g/L before the 15th day. Then, the soluble copper decreases slightly, which may be due to locking of some of the extracted copper with the jarosite formed[13]. After then, the soluble copper increases continuously till the 32nd day, when the copper concentration is 16.3g/L. At the end of bioleaching (at the 44th day), the total concentration of copper ion is 17.4g/L,and the copper recovery percentage achieves up to 75%. However, the preserved culture after 15 months doesn’t show a good performance for extracting copper. At the first 20 days, dissolution of ore sample is slow and only 5.8 g/L of copper ion is extracted. After a lag phase, from the 20th to the 34th day, the dissolution of ore sample increases and gets a finally copper ion concentration of 9.7 g/L, and the copper extraction percentage is only 42%.

Fig.2 Copper extractions in chalcopyrite bioleaching experiment in 44 days with original and preserved cultures after 15 months
In the bioleaching experiment, copper ion in solution is extracted from chalcocite and chalcopyrite. Chalcocite could be dissolved more easily by sulfuric acid than chalcopyrite, which has a complex crystal lattice structure, and is mainly dissolved by ferric ion generated from ferrous ion by microorganisms[14]. It can be seen from Fig.3 that the cell density of preserved culture is much lower than that of unpreserved culture before the 21st day, and in this period, the increase of copper extraction by the preserved culture is mainly due to acid dissolution of chalcocite. After then, when most of chalcocite is leached out, copper extraction enters into a lag phase till the biomass obviously increases (Fig.3). After 30 days, the cell density of preserved culture is more than 108 mL-1, and bioleaching of chalcopyrite begins to increase.
The concentration of total iron is an important parameter in bioleaching of chalcopyrite concentrate. As can be seen from Fig.4, high concentration of total iron would be beneficial to copper extraction. In the Fig.4(a), iron in solution at the first stage is mainly as ferrous ion and the presence of ferric ion is rather notable only at the
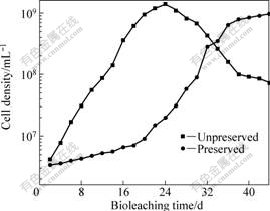
Fig.3 Variation of biomass in solution as function of time

Fig.4 Variations of total iron, ferrous and ferric ion concentrations in bioleaching experiment: (a) Original culture; (b) Preserved culture after 15 months
final stage. When almost 66% of copper is leached (from the 28th day and on), the concentration of ferrous ion begins to decrease and that of ferric ion begins to increase, which corresponds to an increase of redox potential in the solution over 460 mV. While in the Fig.4(b), the total ion concentration increases gradually and finally reached 4.9 g/L. Ferrous ion concentration increases before 20 days and then decreases slightly till the 30th day. During this period, ferrous ion is continually oxidized to ferric ion but no enough ore sample is dissolved to generate ferrous ion. After that, ferrous ion concentration increases sharply to 4 g/L, mainly due to increase of chalcopyrite dissolution. Ferric ion concentration always retains at a low value and it is only 0.9 g/L at the end.
Ion plays a key role in the bioleaching of chalcopyrite concentrate. Ferric ion dissolves in chalcopyrite concentrate to generate copper and ferrous ion[15]. The latter as the energy source of bioleaching microorganisms is oxidized to regenerate ferric ion. In the bioleaching with unpreserved culture, the concentration of ferric ion is very low at the beginning since it is continuously used to dissolve chalcopyrite concentrate. On the other hand, at the final stage of bioleaching, the dissolution of chalcopyrite concentrate is hindered, and the concentration of ferric ion begins to increase since the surface of chalcopyrite concentrate is covered with passivation substances such as jarosite[16]. Meanwhile, ferrous ion is oxidized continuously by microorganisms and reaches a low concentration at the end. However, in the bioleaching with preserved culture, the microorganisms after reviving should pass through a long lag phase to get an enough biomass, which is necessary for chalcopyrite bioleaching. When the biomass increases to a remarkable value, the bioleaching experiment had been carried out to the 44th day, and there is no enough time to allow complete chalcopyrite dissolution.
In the experiment, the sulfuric acid consumption is necessary, because the oxidation of chalcopyrite consumes lots of acid. The reaction is shown as follows:
CuFeS2+O2+4H+=Cu2++Fe2++2S0+2H2O (1)
Acid is also consumed during the oxidation of the released ferrous ion to ferric ion, which is catalysed by microorganisms during the bioleaching of chalcopyrite:
4Fe2++O2+4H+=4Fe3++2H2O (2)
Therefore, in the first stage of bioleaching experiment, sulfuric acid was added into reactor to keep pH value at around 2.0 for acid consumption of chalcopyrite concentrates[17]. After that, the pH decreased continuously to a final value of 1.32 (for unpreserved culture) and 1.42 (for preserved culture after 15 months) since acid was produced during the oxidation of sulphur and the hydrolysis of ferric ion to ferric species such as Fe(OH)2+ and Fe(OH)2+[2-3]. In the bioleaching with the unpreserved culture, the total consumption of sulfuric acid achieves to about 10.5 g. While in the bioleaching with preserved culture, the total acid consumption is more than 21 g, which is due to the lack of acid production at the beginning of bioleaching. When the amount of acid-production was more than that of acid-consumption, the pH value began to decrease[18]. As a result, the experiments of acid consumption shows that the addition of large amount of acid is necessary for efficient bioleaching of chalcopyrite concentrate especially for the preserved culture.
After freeze drying and frozen preservation, bioleaching microorganisms have to take a longer time to adapt the growth environment and reviving. Through prolonging the culture time, microorganisms maybe recover their growth and metabolic ability. As a result, the additional experiment for prolonging the bioleaching time to 80 days was carried out. Fig.5 shows the copper extraction in 80 days. For the unpreserved culture, copper concentration in solution increased slightly from 44th to the 80th day, and the copper extraction finally increased to 19.6 g/L. While bioleaching of chalcopyrite concentrate with the preserved culture after 80 days also showed a high copper extraction of 18.3 g/L at the end. Furthermore, during bioleaching with the reserved culture, the total ion concentration increased to 7.2 g/L at the 50th day, and the high concentration of total ion would benefit the copper extraction at the later stage of bioleaching. However, at the end of bioleaching, the total iron concentration decreased in a large scale, including 2.4 g/L of ferric ion and 1.0 g/L of ferrous ion. The cell density of unpreserved culture for 8 days kept a stable value after the 36th day, and then decreased. Finally, the cell density was 3.1×107 mL-1. Compared with the unpreserved culture, the preserved culture shows an approached total cooper extraction at the 80th day. The unpreserved culture dissolves the ore sample mainly before 44 days and later only 2 g/L copper ion is extracted. It is considered that chalcopyrite passivation seriously affects the total copper extraction and it is unnecessary to prolong the culture time for the unpreserved culture[7, 19]. However, in the bioleaching with preserved culture, microorganisms passed through a long growth lag phase till the 34th day. After then, the cell density increased remarkably, and it brought an
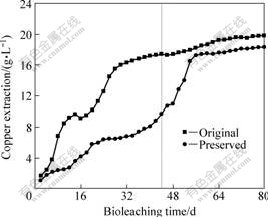
Fig.5 Copper extractions in chalcopyrite bioleaching in 80 d with original and preserved cultures
increase of chalcopyrite dissolution. Finally, preserved culture got 18.3 g/L copper ion concentration in solution. These indicate that although freeze drying and frozen preservation prolongs the lag phase of cell growth, the ability of preserved culture for bioleaching of chalcopyrite concentrate approaches that of the unpreserved culture.
4 Conclusions
1) Freeze drying and frozen preservation is a feasible and effective way for storing the special bioleaching microorganism in a long time. While storing in a short time, this method is not considered to be a good way due to causing serious cells death.
2) Freeze drying and frozen preservation for a long time prolongs the lag phase of cell growth, but the ability of preserved culture for bioleaching of chalcopyrite concentrate is not decreased in a much scale.
References
[1] HUGUES P D, JOULIAN C, SPOLAORE P, MICHEL C, GARRIDO F, MORIN D. Continuous bioleaching of a pyrite concentrate in stirred reactors: Population dynamics and exopolysaccharide production vs. bioleaching performance [J]. Hydrometallurgy, 2008, 94(1/4): 34-41.
[2] GOMEZ E, BLAZQUEZ M L, BALLESTER A, GONZALEZ F. Study of SEM and EDS of chalcopyrite bioleaching using new thermophilic bacteria [J]. Minerals Engineering, 1996, 9(9): 985-999.
[3] SANDSTORM A, PETERSSON S. Bioleaching of a complex sulphide ore with moderate thermophilic and extreme thermophilic microorganism [J]. Hydrometallurgy, 1997, 46(1/2): 181-190.
[4] GOMEZ E, BALLESTER A, GONZALEZ F, BLAZQUEZ M L. Leaching capacity of a new extremely thermophilic microorganism, Sulfolobus rivotinct [J]. Hydrometallurgy, 1999, 52(3): 349-366.
[5] GOMEZ E, BALLESTER A, GONZALEZ F, BLAZQUEZ M L. Silver-catalysed bioleaching of a chalcopyrite concentrate with mixed cultures of moderately thermophilic microorganisms [J]. Hydrometallurgy, 1999, 51(1): 37-46.
[6] EHRLICH H L. Past, present and future of biohydrometallurgy [J]. Hydrometallurgy, 2001, 59(2): 127-134.
[7] ZHOU H B, ZENG W M, YANG Z F, XIE Y J, QIU G Z. Bioleaching of chalcopyrite concentrate by a moderately thermophilic culture in a stirred tank reactor [J]. Bioresource Technology, 2009, 100(2): 515-520.
[8] MORGAN C A, HERMAN N, WHITE P A, VESEY G. Preservation of micro-organisms by drying: A review [J]. Journal of Microbiological Methods, 2006, 66: 183-193.
[9] OTERO M C, ESPECHE M C, NADER-MACIAS M E. Optimization of the freeze-drying media and survival throughout storage of freeze-dried Lactobacillus gasseri and Lactobacillus delbrueckii subsp. delbrueckii for veterinarian probiotic applications [J]. Process Biochemistry, 2007, 42(10): 1406-1411.
[10] ZENG Wei-min, ZHOU Hong-bo, WAN Min-xi, CHAO Wei-liang, XU Ai-ling, LIU Xue-duan, QIU Guan-zhou. Preservation of Acidithiobacillus caldus: A moderately thermophilic bacterium and the effect on subsequent bioleaching of chalcopyrite [J]. Hydrometallurgy, 2009, 96(4): 333-336.
[11] HAYS H C W, MILLNER P A, JONES J K. A novel and convenient self-drying system for bacterial preservation [J]. Journal of Microbiological Methods, 2005, 63: 29-35.
[12] BOZOGLU T F, OZILGEN M, BAKIR U. Survival kinetic of lactic acid starter cultures during and after freeze drying [J]. Enzyme Microbiology Technology, 1987, 9: 531-537.
[13] ACEVEDO F. The use of reactors in biomining processes [J]. Electronic Journal of Biotechnology, 2000, 3(3): 1-11.
[14] LEAHY M J, DAVIDSON M R, SCHWARZ M P. A model for heap bioleaching of chalcocite with heat balance: Mesophiles and moderate thermophiles [J]. Hydrometallurgy, 2007, 85(1): 24-41.
[15] ZENG Wei-min, WU Chang-bin, ZHANG Ru-bing, HU Pei-lei, QIU Guan-zhou, GU Guo-hua, ZHOU Hong-bo. Isolation and identification of moderately thermophilic acidophilic iron-oxidizing bacterium and its bioleaching characterization [J]. Transactions of Nonferrous Metals Society of China, 2009, 19(1): 222-227
[16] DOPSON M, LOVGREN L, BOSTROM D. Silicate mineral dissolution in the presence of acidophilic microorganisms: Implications for heap bioleaching [J]. Hydrometallurgy, 2009, 96(4): 288-293.
[17] MOUSAVI S M, JAFARI A, YAGHMAEI S, VOSSOGHI M. Bioleaching of low-grade sphalerite using a column reactor [J]. Hydrometallurgy, 2006, 82(1/2): 75-82.
[18] OKIBE N, GERICKE M, HALLBERG K B, JOHNSON D B. Enumeration and characterization of acidophilic microorganisms isolated from a pilot plant stirred-tank bioleaching operation [J]. Applied and Environmental Microbiology, 2003, 69(4): 1936-1943.
[19] HACKL R P, DREISINGER D B, PETERS E. Passivation of chalcopyrite during oxidative leaching in sulfate media [J]. Hydrometallurgy, 1995, 39(1): 25-48.
(Edited by YANG Bing)
Foundation item: Project(2004CB619201) supported by the National Basic Research Program of China; Projects(50321402, 30428014) supported by the National Natural Science Foundation of China; Project(NCET-06-0691) supported by the Program for New Century Excellent Talents in Chinese Universities
Corresponding author: QIU Guan-zhou; Tel: +86-731-88879212; E-mail: qgzhoucsu@163.com
DOI: 10.1016/S1003-6326(09)60230-2