�������ʶ�Al-Zn-Mg�Ͻ�����֯�����ʷֲ���Ӱ��
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2015���5��
�������ߣ�A. L. RAMIREZ-LEDESMA R. A. RODR��GUEZ-D��AZ J. CHAVEZ-CARVAYAR O. ALVAREZ-FREGOSO J. A. JUAREZ-ISLAS
����ҳ�룺1391 - 1398
�ؼ��ʣ����Ͻ𣻶������̣��������ʣ����ʷֲ���֦������
Key words��aluminum alloy; unidirectional solidification; growth rate; solute distribution; dendrite growth model
ժ Ҫ�����������Ʊ�Al-5.3% Zn-5.3%Mg�Ͻ�����������Ϊ4~500 ��m/s���¶��ݶ�Ϊ25 K/cm�������£����øĽ��ľ�������Bridgman�����о��úϽ����òת������ʷֲ����������������������Ϊ6.5~9.5 ��m/s�������£������˾���������������ʱ������ǰ������֯ת�䣬������������ʱ������֯ת��Ϊ��״֦������30~ 500 ��m/s���������̵ĺϽ�������֯��Ҫ����״֦����ɣ�Ȼ���ڴ���500 ��m/s���������̵ĺϽ�������֯Ϊ���ᾧ����Ԫ�Ͻ�֦�����̵����ʷֲ�ʵ������Rappaz �� Boettingerģ��Ԥ������Ǻϡ��������ʷֲ���������Եõ�t ��ij�������������t ����Ϊ��������������ߺϽ�ĵ绯ѧ���ܡ�
Abstract: An Al-5.3%Zn-5.3%Mg alloy was unidirectionally solidified to determine morphological transition and solute distribution by a modification of the Bridgman technique for crystal growth with growth rates ranging from 4-500 ��m/s and a temperature gradient of 25 K/cm. It was determined that growth rates from 6.5-9.5 ��m/s generated a cell morphology, where the lower limit corresponds to the plane front to cellular transition and the upper limit indicates the cellular to columnar dendrite transition. The microstructures of the alloys solidified from 30 ��m/s to growth rates less than 500 ��m/s were mainly composed of columnar dendrites, while the microstructures solidified at growth rates greater than 500 ��m/s were equiaxed. Regarding experimental results on solute distribution, a prediction of the model developed by Rappaz and Boettinger for dendrite solidification of multicomponent alloys was applied with excellent agreement. Results of solute distribution were employed to derive the precipitation fraction of t-phase needed to increase the electrochemical properties of the alloy to be used as an Al-sacrificial anode.
Trans. Nonferrous Met. Soc. China 25(2015) 1391-1398
A. L. RAMIREZ-LEDESMA1,2, R. A.  1, J. CHAVEZ-CARVAYAR1,
1, J. CHAVEZ-CARVAYAR1,
O. ALVAREZ-FREGOSO1, J. A. JUAREZ-ISLAS1
1. Materials Research Institute, National Autonomous Universtiy of Mexico, Circuito Exterior S/N, Cd. Universitaria, C.P. 04510, M��xico, D.F.;
2. Polytechnic University of the State of Morelos, Boulevard Cuauhnahuac 556, Col. Lomas del Texcal, 62574 Jiutepec, Morelos M��xico
Received 19 June 2014; accepted 11 November 2014
Abstract: An Al-5.3%Zn-5.3%Mg alloy was unidirectionally solidified to determine morphological transition and solute distribution by a modification of the Bridgman technique for crystal growth with growth rates ranging from 4-500 ��m/s and a temperature gradient of 25 K/cm. It was determined that growth rates from 6.5-9.5 ��m/s generated a cell morphology, where the lower limit corresponds to the plane front to cellular transition and the upper limit indicates the cellular to columnar dendrite transition. The microstructures of the alloys solidified from 30 ��m/s to growth rates less than 500 ��m/s were mainly composed of columnar dendrites, while the microstructures solidified at growth rates greater than 500 ��m/s were equiaxed. Regarding experimental results on solute distribution, a prediction of the model developed by Rappaz and Boettinger for dendrite solidification of multicomponent alloys was applied with excellent agreement. Results of solute distribution were employed to derive the precipitation fraction of ��-phase needed to increase the electrochemical properties of the alloy to be used as an Al-sacrificial anode.
Key words: aluminum alloy; unidirectional solidification; growth rate; solute distribution; dendrite growth model
1 Introduction
In unidirectional solidification of metallic alloys, planar growth can be obtained when growth rates are smaller than those dictated by constitutional supercooling. As the growth rate is increased, a transition from planar to cellular or dendrite structure is observed. With further increase in growth rate, the dendrite structure transforms again to cellular and then to a microstructure free of segregation. Alloy composition (C0), temperature gradient (GL) and growth rate (v) are the main variables which determine the morphology of the solid during solidification. Several theoretical and experimental studies have determined the conditions of microstructural transitions [1,2]. The theory of constitutional supercooling (Vcs) expressed with the equation of GL/Vcs=��To/DL is an important tool that analyzes the stability of a growing solid-liquid interface, where ��To is the solidus-liquidus range to the initial composition C0, DL is the diffusion coefficient of solute in the liquid, (TL-TS)=mLC0(k-1)/k, mL is the slope of the liquidus in the equilibrium phase diagram and k(CS/CL) is the distribution coefficient in equilibrium. When an alloy is unidirectionally solidified at low Vcs and with a positive GL, a planar-cellular transition can be formed. On the other hand, a cellular-microstructure free of segregation transition can be formed at high growth rates, which is predicted according to the criterion of absolute stability limit and is given for Vabs=mL(1-k)C0DL/k��, where �� is the Gibbs-Thomson parameter.
Cellular structures become stable at low and high growth rates (v). The GL effect at low growth rates and the capillarity effect at high growth rates are responsible for the formation of the cellular structure. The cellular- dendrite transition at low growth rates has been expressed with the equation Vtran=DLGL/[mL(1-k)C0], while the dendrite-to-cell transition at high growth rates has not been yet specified by a simple expression; however, the general form is DL/v=����/��T0, where �� is a constant and corresponds to the currently available theoretical results [3]. The growth rates employed to solidify conventional metallic materials are higher than those established by the constitutional undercooling criterion and lower than those of dendrite-cellular transition. Therefore, the dendrite microstructure with eutectic at the grain boundaries is the most frequently observed structure when an alloy is solidified. The growth rate during the solidification as well as the solute distribution plays an important role in the formation and morphology of the microstructures [4]. A control of these two parameters is essential for designing engineering alloys for different applications such as sacrificial anodes for cathodic protection of undersea vessels and pipelines.
Over the past years, dendrite growth models for multicomponent alloys have been developed in order to determine the profile of composition at the solid-liquid interface [5-10]. The model for dendrite solidification of multicomponent alloys with unequal liquid diffusion coefficients developed by RAPPAZ and BOETTINGER [11] can be used to predict the distribution of solute as function of the growth rate. This model states that during the solidification of any given alloy, the growth of the dendrite tip is governed by the supersaturation:
Wj=C*L, j�CC0,j/C*L, j�CC*S, j=C*L, j�CC0, j/C*L, j(1-kj)=Iv(Pj),
j=1, n (1)
where ��j is the supersaturation associated with solute element j; C0, jis the nominal concentration of the alloy; C*L, j and C*S, j are the concentrations at the tip in the liquid and solid phases, respectively; kj is the partition coefficient; Iv(Pj) is the Ivantsov function Iv(Pj)= Pjexp(Pj)E1(Pj), where E1(Pj) is the first exponential integral. Pj is the Peclet number given by Pj=Rv/2DL,j, where R is the dendrite tip radius. Under the assumption of growth at the marginal stability, the following has been proposed: 1) no thermal gradient at the scale of the grain; 2) negligible thermal under-cooling; 3) low grow velocity; 4) independent solute fields; 5) neglecting off-diagonal diffusion terms. Thus, the dendrite tip radius is expressed by
 (2)
(2)
where Gc,j is the solute gradient at the tip, with a dependence given by
Gc, j=-v/DL, j(C*L, j-C*S, j)=(v/DL, j){C0, j(1-kj)/[1-(1-kj)Iv(Pj)]} (3)
Combining Eqs. (2) and (3), the radius of the dendrite tip is obtained as a function of the Peclet number, and from that, the solute concentration at the tip is given as follows:
C*L, j=C0, j/[1-(1-kj)Iv(Pj)] (4)
The Al-Zn-Mg alloy has a complex equilibrium diagram. The first complete investigation of this alloy was made by Eger in 1913, since then, a number of studies have been carried out due to its excellent mechanical properties, low density and high strength [12]. However, studies on the morphological transitions and solute distribution varying the growth rates are scare in the literature. The type of microstructure and the distribution of solute should be considered in order to improve the physical, mechanical and cathodic properties of this alloy. For instance, the knowledge of the solute distribution in solid solution can be used to find the relationship between the content of solute in the matrix and the maximum fraction of precipitates that are formed during aging [13].
The purpose of the present work is to determine the microstructural transitions and solute distribution of an Al-5.3Zn-5.3Mg (mole fraction, %) alloy at low growth rates. Then, the model proposed by RAPPAZ and BOETTINGER [11] for dendrite solidification of multicomponent alloys with unequal liquid diffusion coefficients was used to predict solid solute composition to derive the precipitation fraction of ��-phase needed to increase the electrochemical properties of the alloy to be used as an Al-sacrificial anode according to
Dfp=k1[Cm- ]3 (5)
]3 (5)
where k1=0.1399; Cm is the solute concentration in a-phase and  is the solute concentration in the base material [14].
is the solute concentration in the base material [14].
2 Experimental
The master Al-5.3Zn-5.3Mg (mole fraction, %) alloy composition indicated in Fig. 1 [15] was prepared from high purity (99.99%) aluminum, zinc (99.9%) and magnesium (99.9%). The alloy was melted in an alumina crucible in an induction furnace under an inert Ar atmosphere and poured into a copper mold with a cavity dimensions of 25 mm in thickness, 50 mm in width and 0.12 m in height. The alloy composition of the as-cast ingots was determined by optical emission spectrometry.
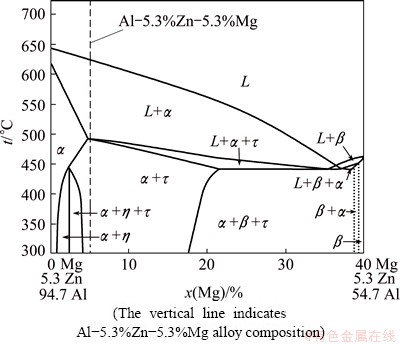
Fig. 1 Vertical section with constant 5.3% Zn of ternary Al-Zn-Mg phase diagram [15]
In order to produce the material suitable for the unidirectional solidification experiments, rods of 3 mm in diameter and 0.12 m in length were produced from the cast ingots, which were introduced into cylindrical graphite crucibles (6 mm in outer diameter, 3 mm in inner diameter and 0.12 m in length). The samples were unidirectional solidified using the Bridgman technique for crystal growth. During the unidirectional solidification experiments, a period of 45 min at 373 K above the liquidus temperature of the alloy was kept so that the furnace could reach thermal equilibrium. The liquid alloy was unidirectionally solidified at a selected withdrawal rate in the range of 4-500 ��m/s with a temperature gradient of 25 K/cm, which was experimentally measured using two thermocouples type-K inserted directly into the sample, with a separation distance of 1 cm from tip to tip.
The longitudinal sections of the specimens were ground from 80 to 1500 mesh grit, polished with Al2O3 of 1 ��m and etched in Keller��s reagent (1 mL HF, 1.5 mL HCl, 2.5 mL HNO3 and 95 mL H2O) for 30 s. Wavelength dispersion X-ray (WDX) microanalysis and line scan were performed along the longitudinal axis of dendrites to determine the solute distribution of Mg and Zn elements, using a scanning electron microscope (FEI-7600 Jeol) equipped with microanalysis facilities. The experimental results of the solute content were compared with predictions according to the model for dendrite solidification of multicomponent alloys with unequal liquid diffusion coefficients and used to derive the precipitation fraction of ��-phase needed to increase the electrochemical properties of the alloy to be used as an Al-sacrificial anode.
3 Results and discussion
Figure 2(a) shows the microstructure of the master alloy, which is constituted by ��(Al) dendrites and ��-Al2Zn3Mg3 intermetallic compounds in the form of fine precipitates distributed on the ��(Al) matrix and the eutectic (��+��-Al2Zn3Mg3) in interdendritic regions (Figs. 2(a) and (b)). From the micrographs, it can be established that the ��(Al) phase was the first to grow, since it developed a dendritic pattern. As the solidification continued, the liquid surrounding the advancing solid-liquid interface was enriched with solute (Zn and Mg), and phases such as ��-Al2Zn3Mg3 intermetallic compound and (��+��-Al2Zn3Mg3) eutectic phase were formed as reported in Refs. [4,12,13].

Fig. 2 SEM images of master alloy (a) and ��+��-Al2Zn3Mg3 eutectic phase (b)
Figure 3 shows the optical micrographs of Al-5.3%Zn-5.3%Mg alloy unidirectional solidified at different growth rates by the Bridgman technique. At the bottom of Fig. 3(a), a morphological transition from plane front to columnar cell can be observed. The alloy was initially solidified at a growth rate of 4 ��m/s for 5 h and then the growth rate was increased to 8 ��m/s. The change in growth rate caused the breakup of the plane front and the creation of a cellular structure. Figure 3(b) shows that the alloy was solidified at 30 ��m/s, the microstructure is entirely constituted by columnar dendrites of ��(Al) and ��+�� eutectic phase in the interdendritic region. From this, it can be established that there is a transition from cellular to dendrite columnar at growth rates between 8 and 30 ��m/s. Figures 3(c)-(e) show that the alloy was solidified at 70, 130 and 170 ��m/s, respectively. At these three growth rates, the microstructures are constituted by columnar dendrites, similar to those obtained at 30 ��m/s, but showing a refinement in the structure with increasing the growth rate. In the alloy solidified at growth rates of 500 ��m/s (Fig. 3(f)), a type of equiaxed microstructure as the temperature gradient was not large enough to maintain the directionality of the structure. It is worth mentioning that unidirectional solidification of Al-Zn-Mg alloys with solute content more than 10% (mole fraction) has not been reported.

Fig. 3 Optical micrographs of Al-5.3%Zn-5.3%Mg alloy solidified at 4 ��m/s (a), 30 ��m/s (b), 70 ��m/s (c), 130 ��m/s (d), 170 ��m/s (e) and 500 ��m/s (f)
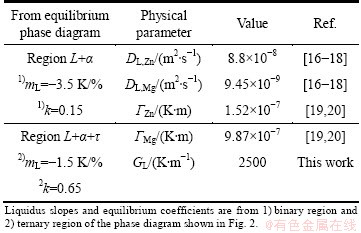
Regarding the equations for microstructural transitions which were solved to estimate the growth rates of morphological transition using the physical parameters of Mg and Zn, as well as the mL and k parameters calculated from the L+a region of the phase diagram (Fig. 1). The input data used in the calculations of the microstructural transitions and solute distribution are given in Table 1. From the experimental results, an apparent morphological transition from plane front to cellular microstructure, at a low velocity close to 4 ��m/s was observed. According to the constitutional supercooling criterion, growth rates of 6.5 and 62 ��m/s are required to achieve the plane front-to-cell transition, considering the physical parameters of Mg and Zn, respectively. As mentioned above, the alloy solidified at a growth rate of 30 ��m/s was mainly constituted by columnar dendrites of ��(Al). In addition, the possibility of a cellular�Cdendrite transition at growth rates between 8 and 30 ��m/s was established. According to the criterion for cellular-dendrite transition, this transition could be achieved at growth rate of 9.5 or 91 ��m/s. From the results, it can be mentioned that there is a reasonable correlation between experimental and predicted transition growth rates determined in the region L+��. The estimated growth rate range for the formation of cellular microstructures might be from 6.5 to 9.5 ��m/s. The plane front transition at high growth rates (absolute stability) might be achieved at growth rates higher than 0.3 m/s. These high growth rates are common even for diluted metallic alloys, which have been achieved by rapid solidification as laser melting [21,22].
Table 1 Input data used for calculation of morphologic transition and solute distribution
Directional microstructures are mainly characterized by microstructural parameters such as cellular spacing, primary and secondary dendrite spacing, and dendrite tip radius. Primary dendrite arm spacing was obtained from the transverse section by the measurement of the average separation of neighboring primary arms, while the tip radius was measured by fitting a suitable circle on the dendrite tip side. For each velocity, at least 25-50 values were measured. As the growth rate was increased, the microstructural parameters were found to decrease gradually. The microstructure of the alloy is mainly constituted by columnar dendrites, the primary dendrite spacing decreased from (250��3) to (140��2) ��m and the dendrite tip radius decreased from (50��5) to (20��3) ��m at growth rates of 4 and 17 ��m/s, respectively. This behavior has been reported in Refs. [23,24].
Figure 4 shows the solute distribution of Mg and Zn elements in the ��(Al) solid solution, which was obtained by line scan and punctual microanalyses. The solute profiles obtained by line scan reveal that the concentrations of the solutes Mg and Zn are kept almost constant along the longitudinal axis of dendrites, where the concentration of Mg is slightly higher than that of Zn. This is due to the fact that the concentrations are strongly affected by diffusion in the liquid and the difference in concentration of solutes is because the magnitudes of the diffusion coefficients are not the same. The results of punctual microanalysis performed along the longitudinal axis from the bottom of dendrites toward the dendrite tips are listed in Table 2. The compositional analysis showed that the amount of solutes Mg+Zn measured at the dendrite bottom and at the center of dendrites kept invariant, while in the dendrite tips the amount of solutes Mg+Zn increased considerably. When the growth rate is increased, a slight increase of the average amount of solutes Mg+Zn retained in all dendrites was observed, reaching the maximum value of 8.06% (mole fraction) at 170 ��m/s.
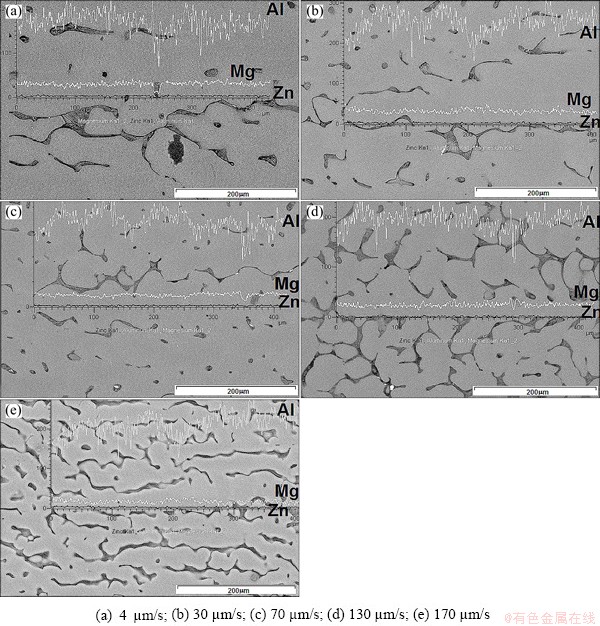
Fig. 4 Distribution of Mg and Zn solutes along longitudinal axis of dendrites solidified at different growth rates
Table 2 Concentrations of solutes Zn and Mg along longitudinal axis of dendrites solidified at different growth rates (The experimental error is ��0.2%)

The solute distribution of Zn+Mg in ��(Al) solid solution during dendritic solidification was predicted using the model for dendritic solidification of multi- component alloys with unequal liquid diffusion coefficients (Eqs. (1)-(4)). The input data used in the calculations for the model are given in Table 1. Just as in previous works [4,20,24], the vertical section at constant 5.3% Zn (mole fraction) of the ternary Al-Zn-Mg phase diagram (Fig. 1) was used to derivate the slopes of the liquidus mL and the partition coefficients k. The results are shown in Fig. 5, where predictions for CS* are given for L+�� region shown in the phase diagram. The predicted values of CS* showed that the maximum concentration of Zn+Mg in solution solid is close to 7.5% (mole fraction) in a growth rate range of 4-170 ��m/s. In the same figure, the full circles and the full squares represent the amount of solute Zn+Mg retained in the dendrite bottom and dendrite tip, respectively. The open circles indicate the experimental average of Zn+Mg solute concentration detected by WDX microanalysis in the whole dendrite. As can be observed, predictions of CS* are closer to the experimental results of Zn+Mg retained in solid solution, as determined by the microanalysis.
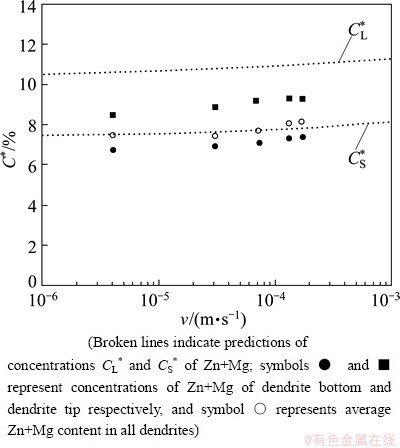
Fig. 5 Predictions of concentrations C* of Zn+Mg as function of growth rate in L+�� region of vertical section at 5.3% Zn of Al-Zn-Mg phase diagram
From Eq. (5), it was derived the maximum precipitation fraction of ��-phase which will be formed during aging, giving a value of 5.94% (volume fraction) close to the experimental value of 5.50% (volume fraction). Figure 6 shows the results of electrochemical efficiency of sand cast (170 ��m/s) and aged experimental anodes. As can be observed, the maximum value of electrochemical efficiency (~87%) was obtained when the precipitation fraction of ��-phase in ��(Al) solid solution reached 5.4% (volume fraction), close to the predicted value of 5.94% (volume fraction). The role of t precipitates in solid solution is to avoid the formation of the continuous and adherent oxide film of the anode surface. The value of electrochemical efficiency is relatively high as compared with Al-Zn+(In+Hg) or with additions of oxides [25].
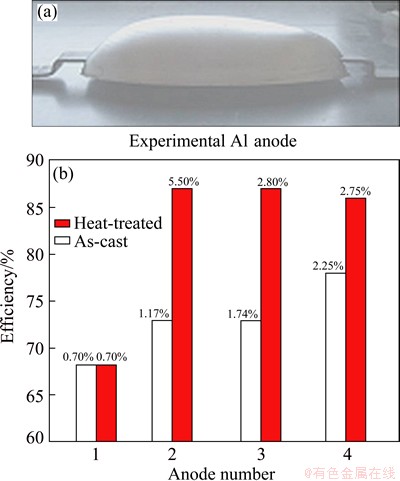
Fig. 6 Electrochemical efficiency of Al-5.3%Zn-5.3%Mg alloy for several experimental anodes with different volume fractions of ��-phase
4 Conclusions
1) The Al-5.3%Zn-5.3%Mg alloy was solidified unidirectionally with a temperature gradient of 25 K/cm at different growth rates. From the experimental and theoretical results, a plane front-to-cell transition at 6.5 ��m/s was estimated that the cellular microstructure could be formed in the range of 6.5-9.5 ��m/s.
2) The microstructures solidified at 30, 70, 130 and 170 ��m/s were mainly constituted by columnar dendrites and were constituted by equiaxial structures at 500 ��m/s, where the temperature gradient in liquid was not high enough to form directional structures.
3) The increase of growth rate causes not only a decrease in the dendrite arm spacing and dendrite tip radius, but also a small increase of solute Zn+Mg retained in solution, registering values of 7.5% at 4 ��m/s and 8.06% at 170 ��m/s.
4) The experimental results of the Zn+Mg solute distribution showed good agreement with the predictions carried out with the model for dendrite solidification of multicomponent alloys in the studied growth range and used to predict the maximum amount of t precipitates (6.84%, volume fraction) in solid solution to increase electrochemical efficiency of Al-sacrificial anodes close to 90%.
Acknowledgments
The authors acknowledge the financial support from Consejo Nacional de Ciencia y Tecnolog��a (CONACYT) Project N�� 129780 and PAPIIT-UNAM project N�� IN213912-3. We also thank to C. ZORRILLA, for the technical support.
References
[1] XIE F Y, KRAFT T, ZUO Y, MOON C H, CHANG Y A. Microstructure and microsegregation in Al-rich Al-Cu-Mg alloys [J]. Acta Materialia, 1999, 47: 489-500.
[2] TRIVEDI R, KURZ W. Dendritic growth [J]. International Materials Reviews, 1994, 39: 49-74.
[3] TRIVEDI R, KURZ W. Solidification microstructures: A conceptual approach [J]. Acta Metallurgica et Materialia, 1994, 42: 15-23.
[4] SOTO J, ARAMBURO G, GONZALEZ C, GENESCA J, HERRERA R, JUAREZ-ISLAS J A. Distribution and prediction of solute in Al-Zn-Mg alloys [J]. Materials Science and Engineering A, 2005, 408: 303-308.
[5] KURZ W, GIOVANOLA B, TRIVEDI R. Theory of microstructural development during rapid solidification [J]. Acta Metallurgica, 1981, 34: 823-830.
[6] ESAKA H, KURZ W. Columnar dendrite growth: A comparison of theory [J]. Journal of Crystal Growth, 1984, 69: 362-366.
[7] RAPPAZ M, THEVOZ P H. Solute diffusion model for equiaxed dendritic growth: Analytical solution [J]. Acta Metallurgica, 1987, 35: 2929-2933.
[8]  M, TRIVEDI R, KURZ W. Nucleation ahead of the advancing interface in directional solidification [J]. Materials Science and Engineering A, 1997, 226: 763-769.
M, TRIVEDI R, KURZ W. Nucleation ahead of the advancing interface in directional solidification [J]. Materials Science and Engineering A, 1997, 226: 763-769.
[9] BOETTINGER W J, CORIELL S R, GREER A L, KARMA A, KURZ W, RAPPAZ M, TRIVEDI R. Solidification microstructures: Recent developments, future directions [J]. Acta Materialia, 2000, 48: 43-70.
[10] CONTIERI R J, RIOS C T, ZANOTELLO M, CARAM R. Growth and three-dimensional analysis of a Nb-Al-Ni ternary eutectic [J]. Materials Characterization, 2008, 59: 693-699.
[11] RAPPAZ M, BOETTINGER W J. On dendritic solidification of multicomponent alloys with unequal liquid diffusion coefficients [J]. Acta Materialia, 1999, 47: 3205-3219.
[12] OGURA T, HIROSAWA S, HIROSE A, SATO T. Effects of microalloying tin and combined addition of silver and tin on the formation of precipitate free zones and mechanical properties in Al-Zn-Mg alloys [J]. Materials Transactions, 2011, 52: 900-905.
[13] BJORNEKLETT B J, GRONG O, MYHR O R, KLUKEN A O. A process model for the heat-affected zone microstructure evolution in Al-Zn-Mg weldments [J]. Metallurgical and Materials Transactions A, 1999, 30(10): 2667-2677.
[14] OROZCO R, GENESCA J, JUAREZ-ISLAS J A. Effect of Mg content on the performance of Al-Zn-Mg sacrificial anodes [J]. Materials Engineering and Performance, 2007, 16(2): 229-235.
[15] PETROV D A, PETZOW G. Ternary alloys [M]. 2nd ed. Germany: Weinheim, 1986.
[16] PETERSON N L, ROTHMAN S J. Impurity diffusion in aluminium [J]. Physical Review B: Solid State, 1970, 1: 3264-3273.
[17] DERYAGIN B V, FRIDLYAND R O. The thickness of the oxide film formed on the aluminum during its forming in the electrolytes Group-1 [J]. Technical Physics, 1948, 18(11): 1443-1448.
[18] DU Y, CHANG Y A, HUANG B, GONG W, JIN Z, XU H, YUAN Z, LIU Y, HE Y, XIE F Y. Diffusion coefficients of some solutes in fcc and liquid Al: Critical evaluation and correlation [J]. Materials Science and Engineering A, 2003, 363: 140-151.
[19] LIANG H, CHEN S L, CHANG Y A. A thermodynamic description of the Al-Mg-Zn system [J]. Metallurgical and Materials Transactions A, 1997, 28: 1725-1734.
[20] GONZALEZ C, ALVAREZ O, GENESCA J, JUAREZ-ISLAS J A. Solidification of chill cast Al-Zn-Mg alloys to be used as sacrificial anodes [J]. Metallurgical and Materials Transactions A, 2003, 34: 2991-2997.
[21] GENG X, FU H. The transition mode of dilute binary alloys during directional solidification near to the absolute stability limit [J]. Science and Technology of Advanced Materials, 2001, 2: 209-212.
[22] SU Yun-peng, LIN Xin, WUANG Meng, XUE Lei, HUANG Wei-dong. Absolute stability of the solidification interface in a laser resolidified Zn-2wt.%Cu hypoperitectic alloy [J]. Chinese Physical Society, 2006, 15(7): 1631-1637.
[23]  , CARDILI E. Directional solidification of aluminium copper alloys [J]. Materials Science and Engineering A, 2002, 327: 167-185.
, CARDILI E. Directional solidification of aluminium copper alloys [J]. Materials Science and Engineering A, 2002, 327: 167-185.
[24] SUAREZ M A, CAMPILLO B, RODRIGUEZ-DIAZ R A, ALVAREZ-FREGOSO O, JUAREZ-ISLAS J A. The effect of Mg content on microstructure in Al-12wt.%Zn-xMg alloy [J]. Journal of Applied Research and Technology, 2009, 7(1): 153-162.
[25] SHIBLI S M, BINOJ K K. Developments of MnO2-incorporated high performance aluminium alloy matrix sacrificial anodes [J]. Journal of Applied Electrochemistry, 2009, 39: 159-166.
A. L. RAMIREZ-LEDESMA1,2, R. A.  1, J. CHAVEZ-CARVAYAR1,
1, J. CHAVEZ-CARVAYAR1,
O. ALVAREZ-FREGOSO1, J. A. JUAREZ-ISLAS1
1. Materials Research Institute, National Autonomous Universtiy of Mexico, Circuito Exterior S/N, Cd. Universitaria, C.P. 04510, M��xico, D.F.;
2. Polytechnic University of the State of Morelos, Boulevard Cuauhnahuac 556, Col. Lomas del Texcal, 62574 Jiutepec, Morelos M��xico
ժ Ҫ�����������Ʊ�Al-5.3% Zn-5.3%Mg�Ͻ�����������Ϊ4~500 ��m/s���¶��ݶ�Ϊ25 K/cm�������£����øĽ��ľ�������Bridgman�����о��úϽ����òת������ʷֲ����������������������Ϊ6.5~9.5 ��m/s�������£������˾���������������ʱ������ǰ������֯ת�䣬������������ʱ������֯ת��Ϊ��״֦������30~ 500 ��m/s���������̵ĺϽ�������֯��Ҫ����״֦����ɣ�Ȼ���ڴ���500 ��m/s���������̵ĺϽ�������֯Ϊ���ᾧ����Ԫ�Ͻ�֦�����̵����ʷֲ�ʵ������Rappaz �� Boettingerģ��Ԥ������Ǻϡ��������ʷֲ���������Եõ�t ��ij�������������t ����Ϊ��������������ߺϽ�ĵ绯ѧ���ܡ�
�ؼ��ʣ����Ͻ𣻶������̣��������ʣ����ʷֲ���֦������
(Edited by Xiang-qun LI)
Corresponding author: R. A.  ; Tel/Fax: +52-5556224645; E-mail: rdiaz.unam@gmail.com
; Tel/Fax: +52-5556224645; E-mail: rdiaz.unam@gmail.com
DOI: 10.1016/S1003-6326(15)63738-4

