J. Cent. South Univ. Technol. (2009) 16: 0936-0941
DOI: 10.1007/s11771-009-0156-6
Technological conditions and kinetics of leaching copper from
complex copper oxide ore
SUN Xi-liang(孙锡良)1, 2, CHEN Bai-zhen(陈白珍)2, YANG Xi-yun(杨喜云)2 , LIU You-yuan(刘有源)2
(1. School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China;
2. School of Physics Science and Technology, Central South University, Changsha 410083, China)
Abstract: The kinetic behavior of leaching copper from low grade copper oxide ore was investigated. The effects of leaching temperature, H2SO4 concentration, particle size of crude ore and agitation rate on the leaching efficiency of copper were also evaluated. And the kinetic equations of the leaching process were obtained. The results show that the leaching process can be described with a reaction model of shrinking core. The reaction can be divided into three stages. The first stage is the dissolution of free copper oxide and copper oxide wrapped by hematite-limonite ore. At this stage, the leaching efficiency is very fast (leaching efficiency is larger than 60%). The second stage is the leaching of diffluent copper oxides, whose apparent activation energy is 43.26 kJ/mol. During this process, the chemical reaction is the control step, and the reaction order of H2SO4 is 0.433 84. The third stage is the leaching of copper oxide wrapped by hematite-limonite and silicate ore with apparent activation energy of 16.08 kJ/mol, which belongs to the mixed control.
Key words: copper oxide ore; H2SO4; leaching; kinetics; activation energy
1 Introduction
Due to the reduction of high grade copper ore and severe SO2 pollution resulting from processing low grade copper ore, the wet process for extracting copper from low grade copper oxide is applied widely[1]. Direct leaching technology of oxide ore and unmanageable ore attracts much attention, which plays an important role in the sustainable development of copper industry. Sulfuric acid (H2SO4) and ammonia (NH3?H2O) are usually used as the leaching reagents in the process of leaching copper ore, such as chalcopyrite (CuFeS2), black copper mine (CuO). Copper oxide ore is dissoluble in H2SO4 solution at room temperature. As a result, the dosage of leaching reagent, which is often H2SO4, is the main factor of economy in the process of leaching. But energy consumption can also become an important factor of economy when the leaching process runs under the condition of agitation and heating. It is often necessary to study the kinetics and mechanism of copper leaching for determining the factors that affect the leaching efficiency. The kinetics of leaching copper oxide mineral was studied. BING?L and CANBAZO?LU[2-3] investigated kinetics of leaching copper from malachite and the results showed that the process of copper leaching was under the control of pervasion kinetics. Quast[4] made kinetics study on the course of leaching atacamite. However, so far there have been few researches on hydrometallurgy technology for processing copper oxide wrapped by silicate and limonite. This kind of ore is usually processed by mill running and then smelting, but the total recovery of copper is about 35% during the mill running process. As a result, further research is still needed for effective exploitation of this kind of ore[5-7]. To develop a H2SO4 leaching technology for extracting copper from low grade multi-metal oxide ore, the influence of leaching conditions on the leaching efficiency was investigated, and the leaching kinetics was also discussed in this work.
2 Experimental
2.1 Composition of ore
The ore used in the experiments was offered by a company in the west of China. Table 1 shows the composition of the crude copper ore.
Table 1 Phase analysis results of copper ore (mass fraction, %)

2.2 Methods of experiment
The leaching experiments were carried out in a Three-mouth vitreous reactor of 2 L. Temperature was controlled by a constant temperature water-bath heater and the temperature error was ±0.5 ℃. Agitation speed was controlled by a motor controller. The effects of agitation speed, temperature, acidity and particle size on leaching copper from the ore were investigated, respectively. The leaching solutions were sampled by a time-basis sampling method. The copper contents in the samples were analyzed by an atomic spectral analyzer.
2.3 Principle of reaction
Because oxide ores are all compact, the reaction interface is in the cascading shape, and shifts from surface to centre with the reaction proceeding. This can be described by a reaction model of shrinking core[8]. For a liquid-solid reaction, the reaction rate can expressed as v=kAcn, where A is the surface area of the ore particle. When the geometrical shape of reaction interface is globular, the equation of reaction rate can be expressed as follows[9]:
 (1)
(1)
where η is the reaction fraction; M is the relative atomic mass; k is the constant of reaction rate; c is the concentration of reaction solution; n is the chemical reaction order; r0 is the original radius of granule; t is the reaction time; ρ is the density of reaction solid granule; and K is the constant of apparent reaction rate.
When the liquid-solid reaction produces compact solid, the process of leaching must be controlled by pervasion, and its kinetic equation can be expressed as follows[10]:
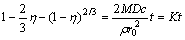 (2)
(2)
where D is the pervasion coefficient.
3 Results and discussion
3.1 Factors influencing reaction rates
3.1.1 Effects of agitation rate on leaching process of copper
At leaching time of 1 and 3 h, and the mass ratio of liquid to solid of 5?1, H2SO4 concentration of 50 g/L and core sample granularity of 0.200-0.125 mm, the influence of agitation rate on the leaching efficiency of copper was evaluated at 95 ℃. The results are shown in Fig.1.
From the principle of hydrometallurgy kinetics [11-12], it is well known that in a process of leaching reaction (solid-liquid multiphase reaction), the influence of agitation rate on leaching efficiency will be great if the control step is liquid pervasion. As a result, agitation can usually make leaching efficiency increased by 40%-
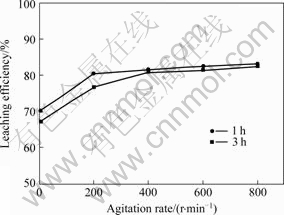
Fig.1 Influence of agitation rate on leaching efficiency of copper
70%. But it can be seen from Fig.1 that the influence of agitation rate on the leaching efficiency is not obvious. The leaching efficiency increases by 5%-10%, which may be attributed to the solid granulates dispersing in the liquid sufficiently. Therefore, it can be concluded that for this leaching reaction, the control step is not liquid film pervasion. An agitation rate of 400 r/min is suitable for the leaching experiment. In this experiment, agitation strengthens the flowing state of two-phase, and thus makes the mineral dispersed completely in the liquid. Furthermore, it may also reduce the thickness of mass transfer boundary layer, accelerating the course of mass transfer.
3.1.2 Effect of reaction temperature on leaching process of copper
For kinetic analysis, it is necessary to keep the reactant concentrations constant before and after the reaction occurs. For the sake of convenience, the amounts of leaching reagents used greatly exceeded the theoretical amounts. Thus, the concentrations of the leaching reagents could be regarded as constants during reaction. When the H2SO4 concentration was 200 g/L, core sample granularity was 0.200-0.125 mm, agitation rate was 400 r/min, and mass ratio of liquid to solid was 20?1, the influence of leaching temperature on leaching efficiency of copper was investigated. The results are presented in Fig.2. It can be seen that the influence of temperature on the leaching efficiency of copper is obvious. The leaching efficiency increases with increasing temperature, which is especially obvious between 323 and 338 K.
In addition, Fig.2 shows that each leaching efficiency―time relationship curve consists of two beelines with different slopes. This indicates that the constant of reaction rate (k) is related to time at different temperatures. Thus, the process of leaching copper can be divided into three stages. The first stage is in the leaching time range of 0-2 min. At this stage, the reaction rate is very fast, and leaching efficiency is larger than 60%. Obviously, the kinetic mechanism at this stage is not the same as that at the other two stages. Because the reaction rate is too fast to be sampled, the kinetic model for this stage is not established, and thus the second and third stage’s kinetic curves do not go through the origin. The second stage is in the leaching time range of 2-15 min. k value is very large and reaction rate is fast in this stage. This indicates that the leaching reaction is mainly due to the dissolution of free copper oxide and copper oxide wrapped by hematite-limonite ore at this time. The third stage is in the leaching time range of 15-240 min. The reaction rate is comparatively slow at this time. This may be attributed to the leaching of the copper oxide wrapped by hematite-limonite ore and silicate.
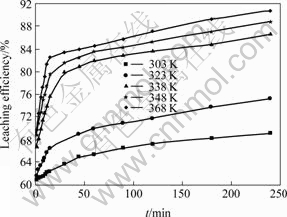
Fig.2 Influence of temperature on leaching efficiency of copper
3.1.3 Effect of particle size on leaching efficiency
Keeping the leaching temperature of 348 K, H2SO4 concentration of 200 g/L, agitation rate of 400 r/min, and mass ratio of liquid to solid of 20?1, the effect of original particle size of crude ore on leaching efficiency was evaluated. The results are shown in Fig.3.
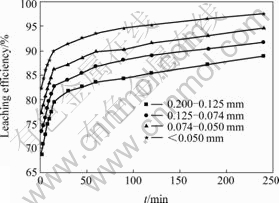
Fig.3 Influence of particle size of crude ore on leaching efficiency of copper
It can be seen from Fig.3 that the leaching efficiency of copper increases with decreasing ore particle size. In the particle size range of 0.074-0.050 mm, leaching efficiency of copper reaches a high level, so the desirable size distribution range is in the range of 0.074-0.05 mm. According to the data shown in Fig.3, 1-(1-η)1/ 3 vs t curves can be easily obtained, as shown in Fig.4. Fig.4 shows that the kinetics of leaching copper from ores with different particle sizes can be explained by the shrinking core model. Each curve consists of two beelines. The relationship between the constant of apparent reaction rate K and the reciprocal of average particle size of ore 1/d0 is shown in Fig.5. Thus parameter K can be represented as ln K=-2.124 9- 0.002 35/d0 and ln K=-3.074 0-0.015 87/d0 for the two leaching stages mentioned above, respectively. It can be clearly seen that the reaction rate constant is inversely proportional to the initial size.
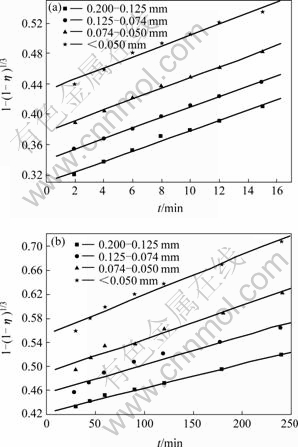
Fig.4 Relationship between 1-(1-η)1/3 and t for ores with different sizes after leaching for different time: (a) 2-15 min; (b) 15-240 min
3.2 Measurement of apparent activation energy
When the leaching efficiency shown in Fig.2 is used in the reaction model of shrinking core[13], it can be found that the leaching kinetics fits the model well, i.e., 1-(1-η)1/3=Kt. According to the data in Fig.2 and Eq.(1), we can obtain the relationship between 1-(1-η)1/3 and time at different leaching temperatures, as shown in Fig.6.
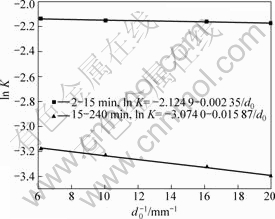
Fig.5 Relationship between constant apparent reaction rate (ln K) and reciprocal of average particle size of ore 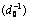
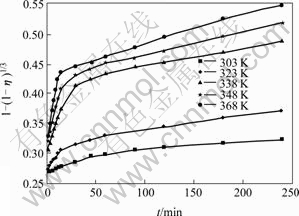
Fig.6 Relationship between 1-(1-η)1/3 and t at different leaching temperatures
It can be seen from Fig.6 that each curve consists of two beelines. This indicates that the constant of reaction rate k is related to time under different circumstances. The leaching process can also be divided into three stages. Fig.7 shows the results of linear regression analysis to the data in Fig.6, in which the slope of the straight line is the constant of apparent reaction rate K. Fig.8 shows the lnK-103/T curves, which are the Arrhenius curves for leaching copper from low grade multi-metal oxide copper.
The apparent activation energy of the leaching reaction can be obtained according to Fig.8. At the stage of 2-15 min, Ea=36.95 kJ/mol<42 kJ/mol. The result may be significantly influenced by the diffusion process at leaching time of 15 min. After canceling the data corresponding to at leaching time of 15 min, the apparent activation energy of the leaching reaction is 43.26 kJ/mol. This indicates that the chemical reaction is the control step during the leaching process[12]. While at the stage of 15-240 min, Ea=16.08 kJ/mol, which is between 12.0-41.8 kJ/mol. This means that the leaching process is under mixed control[14-15].
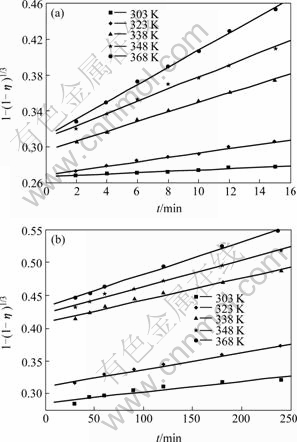
Fig.7 Relationship between 1-(1-η)1/3 and t at different leaching temperatures after leaching for different time: (a) 2-15 min; (b) 15-240 min
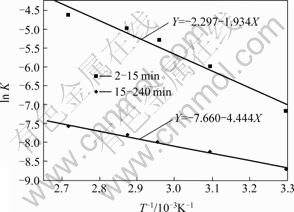
Fig.8 Relationship between ln K and 103/T
3.3 Determination of kinetics equation
Keeping temperature at 348 K, neutral leaching residue size in the range of 0.200-0.125 mm, agitation rate of 400 r/min, and mass ratio of liquid to solid of 20?1, the influence of H2SO4 concentration on leaching efficiency of copper was evaluated. The results are shown in Fig.9.
As shown in Fig.9, the leaching efficiency of copper tends to increase with increasing of H2SO4 concentration. According to the data in Fig.6 and Eq.(1), the curves of 1-(1-η)1/ 3 vs t at different H2SO4 concentrations can be obtained, as shown in Fig.10. Fig.10 shows that the leaching kinetics can also be explained by the reaction model of shrinking core model at these H2SO4 concentrations. However, each relation curve consists of two beelines which intersect at leaching time of 15 min. Fig.11 shows the results of linear regression analysis for the data in Fig.10. The copper leaching efficiency increases linearly with increasing H2SO4 concentration at the stage of 2-15 min, and increases slowly at stage of 15-240 min. Fig.12 shows the relationship between ln K and ln c(H2SO4). According to the slope of each line, the chemical reaction order of H2SO4 can be calculated, which is n(H2SO4)=0.433 84 at 2-15 min stage and n(H2SO4)=0. 241 48 at the stage of 15-240 min.
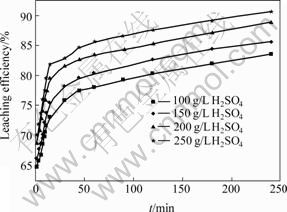
Fig.9 Influence of H2SO4 concentration on leaching efficiency of copper
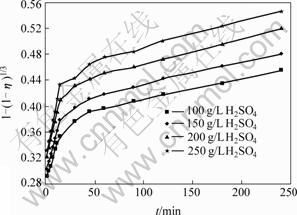
Fig.10 Relationship between 1-(1-η)1/3 and t at different H2SO4 concentrations
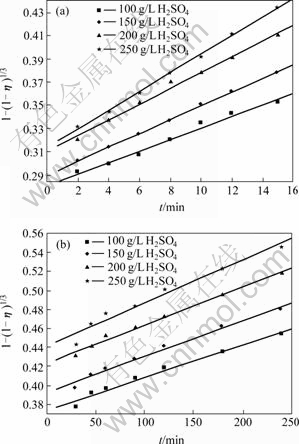
Fig.11 Relationship between 1-(1-η)1/3 and t at different H2SO4 concentrations after leaching for different time: (a) 2-15 min; (b) 15-240 min

Fig.12 Relationship between ln K and ln c(H2SO4)
The empirical kinetic equation of leaching copper from the complex multi-metal copper oxide ore can be expressed as[16-17]:
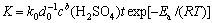 (3)
(3)
where k0 is the constant.
At the average particle size of 0.162 5 mm, values of K and activation energy obtained at different temperatures, k0 can be calculated according to Eq.(3). k0 is 3.46×103 at the stage of 2-15 min, and k0=1.63×10-2 at the stage of 15-240 min. Considering Eq.(1), macro-kinetic equations of the process of copper leaching are as follows:
At stage 2 (2-15 min):

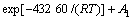 (4)
(4)
At stage 3 (15-240 min):
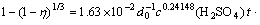
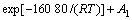 (5)
(5)
where A1 is a constant. Eqs.(4) and (5) are the leaching models of stages 2 and 3. The curves corresponding to the equations will not go through the origin of coordinate.
4 Conclusions
(1) The process of leaching copper from low grade copper oxide ore by H2SO4 can be described with a reaction model of shrinking core. The leaching process can be divided into three stages. The first stage (t<2 min) is the dissolution of free copper oxide and copper oxide wrapped by hematite-limonite ore. The leaching rate is very fast (leaching efficiency is higher than 60%) at this stage. The second stage (t=2-15 min) is mainly the leaching of dissolvable copper oxide wrapped by hematite-limonite ore, which is under chemical reaction control. The reaction rate increases with increasing of H2SO4 concentration. At this stage, the apparent activation energy is 43.26 kJ/ mol, and the reaction order of H2SO4 is 0.433 84. The third stage (t=15-240 min) is the leaching of copper oxide wrapped by hematite-limonite ore and silicate, which is under mixed control. The reaction rate increases with increasing reaction temperature and agitation rate. The apparent activation energy and chemical reaction order of H2SO4 are 16.08 kJ/mol and 0.241 48, respectively.
(2) The macro-kinetic equations for the process of leaching copper from low grade copper oxide ore are as follows:
At stage 2 (2-15min):
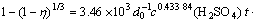
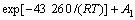
At stage 3 (15-240 min):
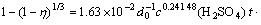

(3) The reaction rate of copper oxide ore wrapped by hematite-limonite ore increases by increasing temperature, reaction time and decreasing particle size of ore. The leaching efficiency of copper can reach up to higher than 90%. Compared with leaching efficiency of copper of 35% for mill-running technology, leaching technology exhibits better economic value. This makes it possible for copper industries to leach copper from low grad copper oxide ore and increase utilization efficiency of copper resource.
References
[1] LIU Da-xing. The development and forecast of copper hydrometallurgical process[J]. Non-Ferrous Metals Recycling and Utilization, 2005, 54(7): 37-39. (in Chinese)
[2] BING?L M, CANBAZO?LU M. Dissolution kinetics of malachite in sulphuric acid[J]. Hydrometallurgy, 2004, 72(2): 159-165.
[3] BING?L M, CANBAZO?LU M. Dissolution kinetics of malachite in ammonia/ammoniumcarbonate leaching[J]. Hydrometallurgy, 2005, 76(1): 55-62.
[4] Quast K B. Leaching of atacamite (Cu2(OH)3Cl) using dilute sulphuric acid[J]. Minerals Engineering, 2000, 13(14/15): 1647- 1652.
[5] LEE K, ARCHIBALD D, MCLEAN M A. Flotation of mixed copper oxide and sulphide minerals with xanthate and hydroxamate collectors[J]. Minerals Engineering, 2009, 22(4): 395-401.
[6] MASAHARU M, NORIHARU A. Potential for phytoextraction of copper, lead, and zinc by rice[J]. Journal of Hazardous Materials, 2009, 162(6): 1185-1192.
[7] FU Cong-sui. Nonferrous metallurgical principle[M]. Beijing: Metallurgical Industry Press, 1993. (in Chinese)
[8] JIANG Han-ying. Physical chemistry of hydrometallurgy process[M]. Beijing: Metallurgical Industry Press, 1984. (in Chinese)
[9] Rajko Z V, NATAS? V, KAMBEROVIC Z. Leaching of copper(I) sulphide by sulphuric acid solution with addition of sodium nitrate[J]. Hydrometallurgy, 2003, 70(3): 143-151.
[10] Herreros O, Quiroz R, Vin?als J. Dissolution kinetics of copper, white metal and natural chalcocite in Cl2/Cl- media[J]. Hydrometallurgy, 1999, 51(3): 345-357.
[11] Padilla R, Vega D, Ruiz M C. Pressure leaching of sulfidized chalcopyrite in sulfuricacid-oxygen media[J]. Hydrometallurgy, 2007, 86(4): 80-88.
[12] Souza A D, Pina P S, Lima E V O, DA Silvab C A, LE?O V A. Kinetics of sulphuric acid leaching of a zinc silicate calcine[J]. Hydrometallurgy, 2007, 89(12): 337-345.
[13] HU Tian-jue, ZENG Guang-ming, YUAN Xing-zhong. Leaching kinetics of silver extracted by thiourea from residue in hydrometallurgy of zinc[J]. The Chinese Journal of Nonferrous Metals, 2001, 11(5): 933-937. (in Chinese)
[14] XIA Zhi-hua, TANG Mo-tang, LI Shi-qing, LUO Yan, TANG Chao-bo. A study on the kinetics of leaching the residues from the neutral leaching process with high concentration of sulfuric acid at high temperature[J]. Mining and Metallurgical Engineering, 2005, 25(2): 53-57. (in Chinese)
[15] XIE Ke-qiang, YANG Xian-wan, WANG Ji-kun, YAN Jiang-feng, SHEN Qing-feng. Kinetic study of iron on pressure leaching marmatite[J]. China Nonferrous Metallurgy, 2007, 36(2): 37-40. (in Chinese)
[16] MO Ding-cheng. Metallurgy dynamics[M]. Changsha: Central South University of Technology Press, 1988: 293-301. (in Chinese)
[17] LI Hong-gui. Hydrometallurgy[M]. Changsha: Central South University Press, 2002. (in Chinese)
(Edited by CHEN Wei-ping)
Foundation item: Project(2005BA639C) supported by the National Science and Technology Development of China
Received date: 2009-02-25; Accepted date: 2009-05-31
Corresponding author: SUN Xi-liang, Professor, PhD; Tel: +86-13786199946; E-mail: sunxl1971@163.com