�Ӹ������ս��ͷ����ѡ������λ��ȡ��
��Դ�ڿ���ϡ�н���2021���4��
�������ߣ����ɹ� ���˱� κ�� ������ ��־�� ��F͢
����ҳ�룺465 - 474
�ؼ��ʣ��ս��ͷ��;��λ����;��Դ�ۺ�����;
ժ Ҫ�����ݸ������ս��ͷ�Ҹ��������ȡ�����̼�ȶ����м�Ԫ�ص��ص�,�����ս��ͷ���������е���Ԫ��Ϊ��λ��,ͨ����λ�����ķ�ʽ��ȡ����������ʵ���о���ҺpH����Ӧ�¶ȡ���λ��Ũ�ȡ���Ӧʱ�䡢Һ�̱ȵȷ�Ӧ�������������ʵ�Ӱ����ɡ�����X���������ǣ�XRD����ɨ��ʽ������-������SEM-EDS�������Ⱥͻ�ѧ�ɷ֣�ICP-MS�������ֶηֱ��ԭ�Ϻͽ��������б�����֪:�ս��ͷ���е�����Ҫ��AgCl��ʽ����,ͨ�������������̼���������ʰ���,����Ҫ��Fe2O3��Fe3O4�������;����������Ҫ�ɷ������������PԪ��̼�ȡ�ʵ��������:�������Ժ�������Һ��,��λ�����������Ȼ�����Ҫ��AgCl43-���������̬������,������̼�ȶ��ս�������Ԫ�ز���������������,�������̾���ѡ���ԡ�����ҺpHֵΪ3.0����Ӧ�¶�80�桢������Ũ��6 mol��L-1������ʱ��2 h��Һ�̱�10:1 ml��g-1�������ٶ�400 r��min-1�����������,���Ľ����ʿɴ�93.90%,���Ľ����ʽ�Ϊ1.28%,����ѡ���Է�����ȡЧ���á�
������ʱ��: 2019-11-01 12:14
ϡ�н��� 2021,45(04),465-474 DOI:10.13373/j.cnki.cjrm.xy19070002
���ɹ� ���˱� κ�� ������ ��־�� ��F͢
����������ѧұ������Դ����ѧԺ
���ݸ������ս��ͷ�Ҹ��������ȡ�����̼�ȶ����м�Ԫ�ص��ص�,�����ս��ͷ���������е���Ԫ��Ϊ��λ��,ͨ����λ�����ķ�ʽ��ȡ����������ʵ���о���ҺpH����Ӧ�¶ȡ���λ��Ũ�ȡ���Ӧʱ�䡢Һ�̱ȵȷ�Ӧ�������������ʵ�Ӱ����ɡ�����X����������(XRD)��ɨ��ʽ������-����(SEM-EDS)�����Ⱥͻ�ѧ�ɷ�(ICP-MS)�����ֶηֱ��ԭ�Ϻͽ��������б�����֪:�ս��ͷ���е�����Ҫ��AgCl��ʽ����,ͨ�������������̼���������ʰ���,����Ҫ��Fe2O3��Fe3O4�������;����������Ҫ�ɷ������������PԪ��̼�ȡ�ʵ��������:�������Ժ�������Һ��,��λ�����������Ȼ�����Ҫ��AgCl43-���������̬������,������̼�ȶ��ս�������Ԫ�ز���������������,�������̾���ѡ���ԡ�����ҺpHֵΪ3.0����Ӧ�¶�80�桢������Ũ��6 mol��L-1������ʱ��2 h��Һ�̱�10:1 ml��g-1�������ٶ�400 r��min-1�����������,���Ľ����ʿɴ�93.90%,���Ľ����ʽ�Ϊ1.28%,����ѡ���Է�����ȡЧ���á�
��ͼ����ţ� X757
����飺���ɹ���1995-�����У����������ˣ�˶ʿ�о������о�����ѹʪ��ұ�����Ʊ���E-mail:1844234601@qq.com;;*���˱����ڣ��绰��0871-65188819,E-mail:lixingbin2011@126.com;
�ո����ڣ�2019-07-01
���������ص��з��ƻ��̷���Դ��ר�2018YFC1900402��;������Ȼ��ѧ������Ŀ��51664029,51564030,51664030,51964029������;
Shi Xianguo Li Xingbin Wei Chang Wang Chenyu Deng Zhigan Li Minting
Faculty of Metallurgy and Energy Engineering,Kunming University of Science and Technology
Abstract��
As one of the solid wastes produced in steel plant,sintering dust had a huge annual output and its chemical composition was complex,so it was not suitable to directly return to sintering ingredients to avoid damaging equipment,nor was it suitable to direct--y stockpile,so as to avoid the waste of resources caused by heavy metals seeping into the land and polluting the environment.Therefore,it was urgent to develop new recovery and treatment technologies.According to the fact that the insoluble compound silver chloride could be dissolved in hydrochloric acid,and could also form the coordination ion dissolved in a high concentration of halogen ion solution,combined with the high content of chlorine element in the sintering dust.Therefore,chlorine was used as coordination agent in the experiment to change the silver in the sintering dust into a soluble complex and increased its solubility into the solution,so as to realize the chlorination coordination leaching of silver.Firstly,quantitative sintering dust was weighed,and then quantitative leaching agent(ammonium chloride) and deionized water with predetermined concentration were added.Finally,the temperature in the flask was heated to the preset experimental temperature,and stirred continuously at a constant temperature for 3 h.During stirring,pulp pH was measured every half hour until the end.After leaching,liquid-solid separation was completed by pumping filtration,and the leaching solution and leaching residue were obtained.In this experiment,the effect of reaction conditions on silver leaching ratio was investigated,such as the pH of solution,reaction temperature,coordination agent concentration,reaction time,liquid-solid ratio and so on.With the increase of pH value,the leaching ratio of silver first increased and then decreased.When pH was 3.0,the maximum silver leaching ratio was 93.9%.As pH continued to increase,the concentration of hydrogen ions in the solution decreased,the dissolved silver chloride decreased,and coordination complexation weakened.With the increase of reaction temperature,the leaching ratio of silver showed an increasing trend.As the temperature rose,the reactivity of reactants was enhanced,and the diffusion ratio of reactants and products was accelerated,the dissolution ratio of silver was accelerated,and the reaction ratio was faster.When the temperature was 80��,the silver leaching ratio reached the maximum of 93.9%.The leaching ratio of silver increased with the concentration of coordination agent increasing.When the concentration of Cl-was 2 to 6 mol��L-1,the leaching ratio of silver was from 32.6% to 93.9%.The higher the concentration of Cl-,the greater the probability of the formation of a complex coordinated with free Ag+ and the greater the solubility of silver chloride,the higher the concentration of silver in the leaching solution and the higher the leaching ratio of silver.With the increase of leaching reaction time,the leaching of silver first increased and then decreased.When the leaching time was from2 to 4 h,the leaching ratio was from 94.8% to 84.5%.With the extension of leaching reaction time,the volatilization loss of leaching agent would increase,the coordination effect of Ag-Cl would be weakened gradually,and the silver leaching ratio would decrease.The silver leaching ratio increased with the increase of the liquid-solid ratio.When the liquid-solid ratio was from 10 to 12 ml��g-1,the leaching ratio was from 93.9% to 95%.From the use of experimental raw materials and reagents,the larger the liquid-solid ratio was,the greater the consumption of raw materials and reagents would be,resulting in higher cost and complicated subsequent treatment of the solution.The compositions and phase results of raw material and leaching residue were characterized by X-ray diffraction(XRD),scanning electron microscope-energy dispersive spectrometer(SEM-EDS),particle size analysis and chemical composition analysis(ICP-MS).According to the characterization results,the silver content in the sintering dust reached 610.4 g��t-1 and mainly existed in the form of AgCl,which was usually wrapped by ferric oxides,carbon particles and other substances.The chlorine content was 1.5%;the iron content in the sintering dust was 28.5% and mainly existed in the form of Fe2 O3 and Fe304.The average diameters of raw materials and leaching residue were 9.63 and 3.59 ��m respectively,and the average diameters of specific surface areas were 3.74 and 0.78 ��m respectively.Compared with the size distribution of the raw material,the size distribution of the leaching residue was smaller,which proved that the soluble material in the raw material could get a more adequate reaction.The leaching residue was iron oxide in phase,and its chemical composition was as follows:Ag content decreased to 78.2 g��t-1,chlorine content was only 0.096%,iron content increased to 52.6%,and element carbon content was 4.5%.The metal elements which were harmful to sintering process were almost all leached out,while the iron leach ratio was low and remained in the leaching residue.The leaching ratio of chlorine element was the highest,indicating that in the process of leaching of sintering dust,chlorine ion and silver element were selectively coordinated to form soluble compounds into the solution.High content of iron and carbon in the leaching residue,heavy metals such as zinc and alkali metals such as potassium were dissolved into the solution,the efficient leaching of zinc and alkali metals could avoid the cycle enrich?ment of zinc and alkali metals in the recycling process of leaching residue,and reduced the harm of zinc and alkali metals to sintering,blast furnace and other ironmaking processes.The experiment results showed that in weak acidic and neutral solution,silver chloride was leached mainly in the form of AgCl43- complex ions during the process of coordinate leaching,while iron and carbon were not leached into the residue,which meant that the leaching process was selective.Under the optimal conditions of the solution pH of 3.0,the reaction temperature of 80��,chloride ion concentration of 6 mol��L-1,the reaction time of 2 h,liquid-solid ratio of 10-1 ml��g-1and stirring velocity of 400 r��min-1,the leaching ratio of silver was up to 93.90%,and the leaching ratio of iron was only 1.28%,the selective separation and extraction of silver was effective.Compared with other methods,chlorine self-coordination method was effective in extracting silver from sintered ash,and it was a new method to treat sintered dust.The extraction of silver from silver-containing solution was also a worthy research direction.Meanwhile,other ionic applications,such as potassium ion could be used to produce potash fertilizer,could also be studied.
Keyword��
sintering filtrated dust; coordinate leaching; comprehensive utilization of resources;
Received�� 2019-07-01
�ս��ͷ����������ʯ�ս�����У�ͨ����������ռ����̳������̳�����һ��ռ�ս�������2%��4%
��Ȼ�������Ĵ�����ʽ���ֶ�������Ҫ����������ʽ�����ں�ɫ������ʯ��
����������ij���������ս��ͷ��Ϊԭ�ϣ��ڷ������ﻯ���ԵĻ����ϣ������ս��ͷ���������е���Ԫ��Ϊ��λ����ͨ����λ�����ķ�ʽ��ȡ��������Ϊ�ս��ͷ���������ۺϻ����ṩһ�ָ�Ч��������ȡ������
1 ʵ��
1.1 �ս��ͷ�һ�ѧ�ɷֺ�X�������䣨XRD������
ʵ�������ս��ͷ��Դ������ij����������۳ʻҺ�ɫϸС������ԭ����Ҫ�Ļ�ѧ�ɷּ���1,X�������䣨XRD��������ͼ1��ʾ��
�ɱ�1��֪���ս��ͷ�����������ﵽ610.4g��t-1�����ռ�ֵ�ߡ���ͼ1��֪���ս��ͷ��������Ǧ���ȵ���Ҫ����ֱ�ΪKPb2Cl5,Fe2O3��Fe3O4���ս��ͷ���е�����Ҫ�������Ȼ���������������������ʽ���ڣ��ұ��ս��ͷ���е����Ļ����������ͳ�����Ǧ������Ȱ����������ս��ͷ����
��1 �ս��ͷ�ҵĻ�ѧ�ɷַ��� ����ԭͼ
Table 1 Chemical composition of analysis of sintering dust(%,mass fraction)

ͼ1 �ս��ͷ�ҵ�XRDͼ��
Fig.1 XRD pattern of sintering dust
1.2 ʵ��ԭ��
�������ܻ������Ȼ��������ܽ��������У������γ��������ܽ��ڸ�Ũ�ȵ�±������Һ��
�������ȶ���һ����������������ͽṹ���ܲ������������
����������ڴ�������ѧƽ���Ag-Cl--H2Oϵ��ˮ��Һ��Ag2Cl+���٣����Ժ��Բ���
Ag+��Cl-��ˮ��Һ�з�Ӧ�γ�Ag Cli1-i(i=1,2,3,4�����ܷ�Ӧʽ����
ˮ��Һ��Ag+��Ũ��[Ag+]TΪ��
����ˮ��Һ�е�Ag+,Ag Cl,Ag Cl2-,Ag Cl32-�Լ�Ag Cl43-��[Ag+]T�����æ�i(i=0,1,2,3,4�������棬����
������Һ�еĽ�������Mj(j=0,1,2����k���ܺ������ӽ�������������Mj Cli(i=0,1,2��n������ˮ��Һ�е�Cl-����Ũ��[Cl-]TΪ
��ôʽ��11������ת��Ϊʽ��14):
ͼ2��Ag-Cl-H2Oϵ�����ȵ������������Һ��[Cl-]�仯������ͼ
��ͼ2�ɼ�����Һ��Ag+��Cl-�γɵ����������[Cl-]�ı仯���仯����[Cl-]��Ũ����������3.0 mol��L-1ʱ����Һ��80%���϶���Ag Cl43-��
1.3 ʵ���豸�뷽��
�������Ժ���ˮԡ������̳�д���Զ���������HH-S24S�����ƽ����¶ȣ�ʹ��������ƿ�����Ͽ��ǻ�������˾��Ϊ������Ӧ���������õ�����ƽ��JE1001���Ϻ��ִ�������������˾������һ���������ս��ͷ�ң�ͬʱ��ȡ�����Ȼ����Һ�����Ⱥ����������ƿ�У����ڽ���ת��Ϊ400 r��min-1������������ڿ�p Hֵ�����Ʒ�Ӧ�¶ȵı仯��Χ��0.5�档
����p H�ƣ�PHSJ-5���Ϻ���������ѧ��������˾��������Һ��p Hֵ����Ӧ������ֹͣ���裬������Ƭ��ձã�2XZ-2�ͣ��㽭̨������ձ�����˾�����ˣ����������������ˮϴ�ӳ��˺��ڵ��ȹķ�������ڣ�DZF-6090���Ϻ�һ���ѧ��������˾�����¸���¶�75�棩12 h�������ͻ��������
ͼ2 ���ȵ������ֲ�
Fig.2 Distribution of silver chloride complex
���Ľ����ʣ��ǣ����㹫ʽΪ��
ʽ�У���Ϊ���Ľ����ʣ���1Ϊ���ϵ���������2Ϊ����������������1Ϊԭ��������������2Ϊ����������������
1.4 �ս��ͷ��ɨ��羵��SEM��ͼ�����ȷ���
���ս��ͷ�ҽ���ɨ��羵��SEM�������ȷ����������ͼ3��4��ʾ��
ͼ3 ��ͬ�Ŵ������ս��ͷ�ҵ�SEMͼ
Fig.3 SEM images of sintering dust with different magnifica-tion(a-d)
ͼ4 �ս��ͷ�����ȷֲ�ͼ
Fig.4 Particle size distribution of sintering dust
��ͼ3���Կ������ս��ͷ�ҵ�����С���ֿ�����������ܣ��������ԣ�������ճ����ٲ��ֿ�����������״��ճ���������ɶ�ס���ͼ4���ȷ��������֪���ս��ͷ�ҵ�������Χ��0.38��66.2��m�������ֲ��ϼ��У����ƽ��ֱ��Ϊ9.63��m���ȱ����ƽ��ֱ��Ϊ3.74��m,90%�������������Ϊ21.00��m������Ŀ�������С������ʽ���ڡ�
2 ���������
2.1 ��Һp H���������ʵ�Ӱ��
ʵ����������Ӧ�¶�80�棬������Ũ��6 mol��L-1������3 h��Һ�̱�10��1 ml��g-1���ı���Һp Hֵ�����첻ͬp H���������ʵ�Ӱ�졣ʵ������ͼ5��ʾ��
��ͼ5�ɼ���������Һp Hֵ���������Ľ����ʳ������������½������ơ���p H=1.5ʱ����������Ϊ82.7%����p H=2.0ʱ����������Ϊ93.2%����p H=3.0ʱ���������ʴﵽ���Ϊ93.9%�����ŷ�Ӧ�Ľ��У���Һp H����������Һ�е�H+Ũ����С��Cl-��Ag+����λ�����������Ȼ������ܽ�ȼ�С
2.2 ��Ӧ�¶ȶ��������ʵ�Ӱ��
ʵ����������Һp H=3.0��������Ũ��6 mol��L-1������3 h��Һ�̱�10��1 ml��g-1���ı������Ӧ�¶ȣ������¶ȶ��������ʵ�Ӱ�졣ʵ������ͼ6��ʾ��
��ͼ6�ɼ������߷�Ӧ�¶ȣ����Ľ����ʳ����������ơ��¶����ߣ���Ӧ��ķ�Ӧ������ǿ����Ӧ��Ͳ�����ɢ�ٶȼӿ�
ͼ5 ��Һp H���������ʵ�Ӱ��
Fig.5 Effect of solution p H on leaching ratio of silver
ͼ6 ��Ӧ�¶ȶ��������ʵ�Ӱ��
Fig.6 Effect of reaction temperature on leaching ratio of silver
2.3 ��λ��Ũ�ȶ��������ʵ�Ӱ��
ʵ���������£���Һp H=3.0����Ӧ�¶�80�棬����3 h��Һ�̱�10��1 ml��g-1���ı���������ӵ�Ũ�ȣ������ڲ�ͬŨ�����������ʵ�Ӱ�졣ʵ������ͼ7��ʾ��
��ͼ7�ɼ�����һ����Χ�ڣ�������λ��Ũ�ȵ����ӣ����Ľ����ʳ��������ơ���Cl-Ũ��Ϊ2mol��L-1ʱ����������Ϊ32.6%������Ũ�ȵ����ߣ���Cl-��Ũ��Ϊ6 mol��L-1ʱ�����Ľ��������Ϊ93.9%��Cl-Ũ��Խ�ߣ�������Ag+��λ���������ļ��ʾ�Խ���Ȼ������ܽ��ҲԽ��
ͼ7 ������Ũ�ȶ��������ʵ�Ӱ��
Fig.7 Effect of chlorideion concentration on leaching ratio of silver
2.4 ��Ӧʱ����������ʵ�Ӱ��
ʵ����������Һp H=3.0����Ӧ�¶�80�棬������Ũ��6 mol��L-1��Һ�̱�10��1 ml��g-1���ı䷴Ӧʱ�䣬����ʱ����������ʵ�Ӱ�죬ʵ������ͼ8��ʾ��
��ͼ8�ɼ������Ž�����Ӧʱ������ӣ����Ľ������ȱ��͡�������ʱ��Ϊ0.5 hʱ����������Ϊ79.48%��������ʱ��Ϊ1 hʱ����������Ϊ89.4%��������ʱ��Ϊ2 hʱ�������ʴﵽ���Ϊ94.8%�����Ž�����Ӧʱ����ӳ���������ʱ��ﵽ4 hʱ��������ֻ��84.5%�����ڽ���ʱ��Խ�����������Ļӷ���ʧ�����࣬���ȵ���λ�����������������ʽ��ͣ�ͬʱҲ�����ں������ա���˽���ʱ��ѡ��2 hΪ�ˡ�
2.5 ��ӦҺ�̱ȣ�L/S�����������ʵ�Ӱ��
ʵ����������Һp H=3.0����Ӧ�¶�80�棬������Ũ��6 mol��L-1������2 h���ı䷴ӦҺ�̱ȣ�����Һ�̱ȣ�L/S�����������ʵ�Ӱ�죬ʵ������ͼ9��ʾ��
��ͼ9�ɼ���Һ�̱ȶ���������Ӱ��ϴ����ŷ�ӦҺ�̱ȵ�����������������������L/S=4 ml��g-1ʱ����������Ϊ53.1%����L/S=8 ml��g-1ʱ������������Ϊ75.3%�����ŷ�ӦҺ�̱��������������������ߡ���L/S=10 ml��g-1ʱ�����Ľ����ʴﵽ93.9%���˺�Һ�̱ȼ����������ʴﵽ���ֵ95%��������1.1%�������ķ��Ƚ�С�����Ҵ�ʵ��ԭ�Ϻ�ʵ���Լ���ʹ�����������Һ�̱�Խ��ԭ�Ϻ��Լ�����Խ����ɳɱ����ߣ���Һ�����������ӣ���ѡ��Һ�̱�10:1 ml��g-1����ʡ�
ͼ8 ��Ӧʱ����������ʵ�Ӱ��
Fig.8 Effect of reaction time on leaching ratio of silver
ͼ9 ��ӦҺ�̱ȶ��������ʵ�Ӱ��
Fig.9 Effect of reaction L/S on leaching ratio of silver
3 ����������
Ϊ�˸�������˽��ս��ͷ���ڽ���ǰ����������̬�仯���������о�������ѡ����ѹ�����������Һp H=3.0����Ӧ�¶�80�棬������Ũ��6 mol��L-1��Һ�̱�10:1 ml��g-1������ʱ��2 h��չ�ۺ���֤ʵ�顣���ۺ���֤ʵ��õ��Ľ������ֱ���л�ѧ�ɷַ�����XRD������SEM������
3.1 �������ɷַ���
�Խ������Ļ�ѧ�ɷֽ��м�⣬������2��ʾ������ֵĽ�������ͼ10��ʾ��
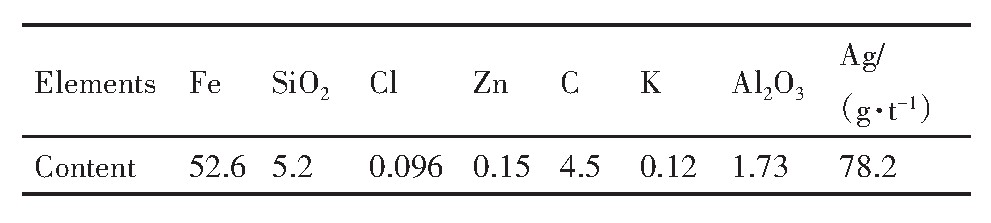
�ɱ�2�ɼ���������λ������Ӧ�������е������ȵ�Ԫ�غ�����ԭ�ϳɷ����Խ��ͣ����ͼ10��֪������п���ء��ȵĽ����ʷֱ�Ϊ88.7%,93.5%,97.7%��99.1%�����������衢̼�Ľ����ʷֱ���1.3%,2.6%,5.01%��10.8%�����ս�����к��Ľ���Ԫ�ؼ���ȫ�������������Ľ����ʵͣ������ڽ������С���Ԫ�صĽ���������ߵģ��������ս��ͷ�ҽ���������������������Ԫ�ؽ���ѡ������λ�������γɿ��ܽ�Ļ����������Һ�С�������������̼Ԫ�غ����ߣ��ֱ�Ϊ52.6%��4.5%��п���ؽ����ͼصȼ�������ܽ������Һ��п��������ĸ�Ч�������Ա����������ѭ�����ù��������п�ͼ������ѭ������������п�ͼ�����ȶ��սᡢ��¯���������̵�Σ����
��2 ����������Ҫ�ɷ� ����ԭͼ
Table 2 Main components of leaching residue(%,mass fraction)
ͼ1 0 �������м�����Ҫ��ֽ�����
Fig.10 Leaching ratio of several main components in leaching residue
3.2 ������XRD���������ȷ���
Ϊ�˽�һ��������������������ɼ����ȷֲ�������Խ���������XRD�����ȷ�����⣬�����ͼ11��12��ʾ��
��ͼ11��XRD�����������֪�����������е�������ɼ��������м�Ԫ����Ҫ�������衢Ǧ�ȡ����У�����Ҫ��Fe2O3��Fe3O4������̬���ڣ��ҿ��Կ������д�ΪFe2O3����ͼ12�����ȼ������֪����������������Χ��0.38��21.53��m�������ֲ��Ƚϼ��У������������ƽ��ֱ��Ϊ3.59��m���ȱ����ƽ��ֱ����0.78��m,90%���ӵ����������8.20��m������С�����ԭ�ϵ������ֲ����ԣ��������������ֲ���С��֤��ԭ���еĿ��ܽ����ʵõ��ϳ�ֵķ�Ӧ��ͬʱ��������������нϴ���ս��ͷ�ҿ����ڽ��������������С�������ʣ��ҷ�Ӧ��ɺ�ϸС�Ŀ����ž��ڽϴ������Χ���γ��ž��帽���ڲ����ս��������档
ͼ1 1 ��������XRDͼ��
Fig.11 XRD pattern of leaching residue
ͼ1 2 ���������ȷֲ�ͼ
Fig.12 Particle size distribution of leaching residue
3.3 ���������ף�SEM-EDS������
Ϊ�˽�һ������������������ò�ͻ�ѧ�ɷ֣��Խ������������ף�SEM-EDS����⣬�����ͼ13��ʾ��
��ͼ13�Ľ�����SEMͼ��EDS��ͼ�ɼ����ս��ͷ�ҽ�����������Ϊ��������Χ������״��ճ�����Ҹ����������֪���������������ڿ���������Ƿ�Ӧ��֮������������һ����ž��壬�еĿ�������Ϲ⻬���ܣ��еĿ�������ֲ����ɡ�������Ҫ��Fe,Ca,C,O 4��Ԫ�أ���������Ԫ�غ����ﵽ52.6%����Ҫ��̬Ϊ���Ļ�����Fe2O3��Fe3O4�������ս��ͷ�������������ڽ���ʱ���������뷴Ӧ�������������ڽ������еõ�������
4 ����
���IJ����Ȼ���λ�����ķ������о����ս��ͷ����������λ������Ϊ����õ���Ҫ�������£�
1.��XRD,FESEM-EDS�ȷ���֪���ս��ͷ����Ag�����ﵽ610.4 g?t-1����Ҫ��Ag Cl����̬���ڣ�ԭ���е��Ⱥ����ﵽ7.52%����Ҫ���Ȼ�����̬���ڣ��ս��ͷ���е�����Ҫ��Fe3O4��Fe2O3������̬���ڡ�
ͼ1 3 ��������SEMͼƬ��EDS��
Fig.13 SEM images(a,b)and EDS spectra of selected positions(c,d)
2.�����ս��ͷ���е��Ⱥ��������ߵ��ص㣬������λ������ȡ�ս��ͷ���е�����ʹ��ת��ΪAg Cl43-���������̬ѡ�����ܽ������Һ��ͨ��������ʵ��õ������������Һp H=3.0����Ӧ�¶�80�棬������Ũ��Ϊ6 mol��L-1����Ӧʱ��2 h��Һ�̱�10:1 ml��g-1�������ٶ�400 r��min-1���ڴ������£����Ľ����ʿɴ�93.90%�����Ľ����ʽ�Ϊ1.28%��ʵ��������ѡ������λ�����������������������٣�����������ҪΪ�Ը���ұ�����õ�����̼��Ԫ�أ��������к���п�ͼ����������Ч�ܳ���ʵ��������̼�ķ��롣������ѳ��к�����Ԫ�صĽ��������Է���������������ѭ�����ã�ʵ�ֶ�Ԫ�ص��ۺϻ��ա�
3.�����ս��ͷ���б�������������λ����Ϊ��λ����ֱ����λ�������ļ���·�ߣ����в����������ʸߡ�������ȾС���ɱ��͵��ŵ㣬Ϊ�����ս��ͷ�������Ľ����ṩ��һ���µļ���;����
�����

