ˮ����Һ����KBX��ADD �յ�ϸ������п������Ÿ�ѡ
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2014���5��
�������ߣ��� �� ������ ����� ��ˮ��
����ҳ�룺1578 - 1586
�ؼ��ʣ���ˮ������ϸ������п��zeta��λ���Ӵ��ǣ�������ҩ(KBX)����狀�ҩ(ADD)��ú��
Key words��hydrophobic flocculation; marmatite fines; zeta potential; contact angle; butyl xanthate (KBX); ammonium dibutyl dithiophosphate (ADD); kerosene
ժ Ҫ�����ü������ȷ�����������������Ӿ��ɢ�䡢�Ӵ��Dz����ͺ�������о���ˮ����Һ���ö�����ҩ(KBX)�Ͷ�狀�ҩ(ADD)�յ�ϸ������п�����ˮ������ͨ���ı���ֲ��������а���pHֵ�����ռ�Ũ�Ⱥ�ú���������о�������С��20 ��m�ĵ���������Ÿ�ѡ��Ϊ��������������Ÿ�ѡЧ�����������ȺͿ�����ˮ��������أ��������ڲ��ռ����������µĿ��������������û�н������Ÿ�ѡЧ�������Ż��IJ��������£���KBX��ADD��������ϸ������п������Ÿ�ѡ�����ʾ�����90%�������渡ѡ�Ļ�����ֻ��60%���ҡ����黹���֣���������п�����������Ĥ���γɣ�����ú�͵����ӿɴ���ȸĽ���ѡָ�ꡣFTIR���������������ҩ��������п������������Ϊ��ѧ������
Abstract: The hydrophobic flocculation of marmatite fines in aqueous suspensions induced by butyl xanthate (KBX) and ammonium dibutyl dithiophosphate (ADD) was investigated using laser particle size analysis, microscopy analysis, electrophoretic light scattering, contact angle measurement and infrared spectroscopy. The studies were performed on single minerals with size ��20 ��m by varying several parameters, including pH, collector concentration and kerosene addition. The results show that the floc flotation closely correlated with the size of flocs and the particle hydrophobicity, but was not lowered with increasing the particle surface charges due to collector adsorption. Under good operating conditions, the floc flotation of marmatite fines as a function of KBX and ADD can all reach floatability over 90%, in comparison with conventional flotation obtaining floatability of about 60%. It also has been found that a small addition of kerosene greatly improved the floc flotation because of the formation of oil films on marmatite particles. The results of FTIR spectra indicate that adsorption of the two collectors onto marmatite were chemical adsorption.
Trans. Nonferrous Met. Soc. China 24(2014) 1578-1586
Ting ZHANG, Wen-qing QIN, Cong-ren YANG, Shui-peng HUANG
School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
Received 20 June 2013; accepted 5 September 2013
Abstract: The hydrophobic flocculation of marmatite fines in aqueous suspensions induced by butyl xanthate (KBX) and ammonium dibutyl dithiophosphate (ADD) was investigated using laser particle size analysis, microscopy analysis, electrophoretic light scattering, contact angle measurement and infrared spectroscopy. The studies were performed on single minerals with size ��20 ��m by varying several parameters, including pH, collector concentration and kerosene addition. The results show that the floc flotation closely correlated with the size of flocs and the particle hydrophobicity, but was not lowered with increasing the particle surface charges due to collector adsorption. Under good operating conditions, the floc flotation of marmatite fines as a function of KBX and ADD can all reach floatability over 90%, in comparison with conventional flotation obtaining floatability of about 60%. It also has been found that a small addition of kerosene greatly improved the floc flotation because of the formation of oil films on marmatite particles. The results of FTIR spectra indicate that adsorption of the two collectors onto marmatite were chemical adsorption.
Key words: hydrophobic flocculation; marmatite fines; zeta potential; contact angle; butyl xanthate (KBX); ammonium dibutyl dithiophosphate (ADD); kerosene
1 Introduction
Froth flotation only well responds to mineral particles in a narrow size range, out of which flotation response falls substantially [1-3]. This size range varies with minerals and reagent conditions. For example, sulfide minerals flotation was found to have the lower size limit of 6 ��m for galena, 8 ��m for sphalerite, 9 ��m for pyrrhotite, 15 ��m for chalcopyrite and 20 ��m for pyrite through investigations in laboratory as well as in operating mills [3]. It is generally accepted that the problem of mineral fines flotation is mainly due to the low mass and high surface area, leading to a low probability of collision and adhesion of mineral particles to air bubbles. However, other factors, such as surface composition, oxidation, mineralogical alterations, and dissolved ions concentration, are also attributed to the problem [4,5].
There are two ways to improve the poor flotation response of mineral fines, namely increasing the size of floated particles and reducing the size of air bubbles [6]. The former is realized through floc flotation, which includes carrier flotation, emulsion flotation and shear flocculation flotation, etc; the latter is represented by vacuum flotation and electrolytic flotation [7-9]. Floc flotation is based on the size enlargement of desired mineral fines through selective aggregation, followed by conventional flotation of the fines in the form of flocs, instead of individual fine particles that interact with air bubbles, avoiding the problem of low probability of collision and adhesion of mineral fines to air bubbles. Hydrophobic flocculation is mostly suited for this purpose because the hydrophobic surfaces have more compact structure and higher floc-strength in comparison with electrolytic coagulation and polymer flocculation [10]. Except for naturally hydrophobic minerals, selective hydrophobization is induced by surfactants upon their adsorption on the particles and sufficient input of kinetic energy to the system to overcome the energy barrier, and is greatly enhanced by adding non-polar oil [11]. For example, SONG et al [12,13] reported studies on the hydrophobic flocculation of galena and sphalerite fines. It has been found that amyl xanthate (a short hydrocarbon chain collector) can induce a powerful hydrophobic flocculation to the mineral fines by means of kinetic energy input, which can be greatly enhanced by adding a small amount of kerosene.
Except for sphalerite, marmatite is the most common zinc mineral, and its flotation with alkyl xanthates and aerofloats as collectors has been practiced worldwide since early 2000. Numerous reviews are available in this regard, including the recent ones by LIU [14], ZHANG et al [15] and QIN et al [16], whose objectives are to research the flotation behavior of marmatite >38 ��m. While reports about marmatite <38 ��m are very little.
In this work, the hydrophobic flocculation of marmatite fines and the adsorption of butyl xanthate and ammonium dibutyl dithiophosphate on marmatite were studied using laser particle size analysis, microscopy analysis, electrophoretic light scattering measurements and infrared spectroscopy, versatile techniques in studying colloidal particles. The particle hydrophobicity was determined through contact angle measurements. The objectives are to investigate the possibility of floc flotation applied to the beneficiation of marmatite fines, to correlate the floc flotation with various parameters such as pH, collector��s concentration and kerosene addition, and to study the mechanisms of the hydrophobic flocculation.
2 Experimental
2.1 Materials
The sample of marmatite used in this study was received from the Dachang Mine in Guangxi Autonomous Region, China. The chunk mineral was first crushed by a hand hammer, and then purified by hand sorting. Several pieces of marmatite were cut to obtain rectangles with about 2 cm in length and 1 cm in width for contact angle measurements. Marmatite of less than 38 ��m in size was first produced by grinding in a vibrating cup mill, followed by size classification and then regrinding to obtain marmatite fines <20 ��m. The size distribution of the sample is illustrated in Fig. 1. Marmatite less than 20 ��m takes up about 90% and has a volumetric diameter (d50) of 4.24 ��m, which was used for floc flotation tests. Chemical composition analysis of marmatite gave the following chemical compositions: 50.53% Zn, 14.76% Fe, 30.02% S and 4.69% others.
Butyl xanthate and ammonium dibutyl dithiophosphate were used as the collectors for single mineral tests. 0.1 mol/L solutions of hydrochloric acid and sodium hydroxide analytical grade were used to adjust pH. Without further purification, kerosene was used as an oil emulsion made by an ultrasonic treatment. 2# oil was used as a frother. All experiments were carried out with distilled water only.
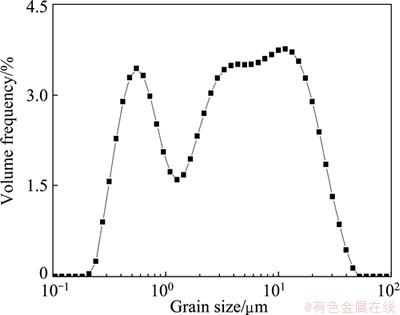
Fig. 1 Size distribution of fine marmatite particles
2.2 Experimental methods
2.2.1 Hydrophobic floc flotation
Hydrophobic floc flotation of marmatite fines in aqueous suspensions was carried out in a microflotation cell with a 40 mL effective volume. The contents of the cell were stirred by a four-bladed impeller. The amount of sample used for each experiment was 2.0 g. After adjusting the pH to the appropriate value with hydrochloric acid or sodium hydroxide solution, mineral suspension of 2.0 g minerals and 32 mL water was conditioned with collector for 2 min, sometimes with kerosene emulsion for 2 min. Lastly, 2# oil was added as frother with a consumption of 10 mg/L and its agitating time was 1 min. The flotation time was 4 min. At last, concentrate and bottom products were filtered, dried, weighed and analyzed.
2.2.2 Floc size determination
A Master sizer 2000 (Malvern/ England) instrument was used to determine the size distributions of dispersed and flocculated mineral fines in this study. In order to prevent flocs from breaking in the measurements, ultrasonic treatment was not applied to the suspensions.
2.2.3 Microscopy imaging
A microscope, OLYMPUS CX31, was used to observe flocculation state of particles in suspension. The preparation of suspension was the same as flotation operation, just without adding frother. A small amount of the suspension drops on the microslide and makes it spread. After waiting for air drying, the microslide was transferred to microscope to observe. The magnification times of microscope objective lens and ocular lens were 40 and 10, respectively.
2.2.4 �� potential measurement
�� potentials of marmatite particles were determined using a JS94H micro-electrophoresis apparatus, which was produced by the digital technology equipment of Zhongchen Co., Ltd. in Shanghai, China. In this study, marmatite suspensions at 0.1% solids <5 ��m were first conditioned for 5 min with pH regulating reagents in the absence or presence of KBX and ADD. The suspensions were then transferred to the rectangular capillary cell of the instrument to measure the zeta potential four times. The average value of �� potentials of four measurements was reported as final results.
2.2.5 Contact angle measurement
A France GBX company contact angle tester was used for contact angles measurements. The surface of the marmatite piece used for the measurements was polished using first a fine sandpaper and then a diamond paste. The piece was conditioned in a collector solution at a given pH for 5 min.
2.2.6 Infrared spectroscopy
Infrared spectra were used to study the mechanism of mineral-reagent interaction. The interaction product was obtained by contacting fine marmatite minerals (<2 ��m) with or without collector solution using an agate mortar and pestle. After an additional 30 min for settling, the sample was filtered and washed 3 times with distilled water. The solid was then vacuum dried. Fourier transform infra-red (FTIR) spectra of the samples were recorded with Nicolet Model Nexus 670 (USA) instrument.
3 Results and discussion
3.1 Hydrophobic flocculation
The size distributions of the marmatite before and after the hydrophobic flocculation induced by KBX and ADD, sometimes with kerosene, are shown in Figs. 2 and 3, respectively. The stirring speed was 1800 r/min. As can be seen, hydrophobic flocculation increased the size of marmatite. Figure 2 shows that the 50% volumetric diameters of marmatite induced by 2��10-4 mol/L and 1.2��10-3 mol/L KBX were increased to 6.95 ��m and 8.92 ��m from 4.24 ��m, respectively. And according to the particle size analysis, hydrophobic flocculation in the function of the two concentrations increased the volumetric mean diameters (D(4, 3)) to 9.98 ��m and 12.27 ��m from 7.58 ��m, respectively. From Fig. 3, it can be found that the marmatite has the volumetric diameters (d50) of 7.36 ��m and 9.45 ��m after conditioning with 1��10-4 mol/L and 4��10-4 mol/L ADD, respectively. The volumetric mean diameters D(4, 3) were increased to 10.81 ��m and 46.70 ��m, respectively. This indicates that hydrophobic flocculation makes the size of marmatite larger and increase with increasing the dosage of collectors.
The sizes of marmatite induced by KBX and ADD were increased by the addition of kerosene. Under the operating conditions in the two figures, the 50% volumetric diameters of marmatite in the absence of kerosene were 6.95 ��m for KBX and 7.36 ��m for ADD, respectively. While in the presence of 250 mg/L kerosene, the 50% volumetric diameters of marmatite induced by KBX and ADD were increased to 12.73 ��m and 11.80 ��m, respectively. According to the particle size analysis, hydrophobic flocculation in the function of 250 mg/L kerosene increased the volumetric mean diameter D(4, 3) to 33.89 ��m from 10.81 ��m for KBX and to 25.79 ��m from 13.80 ��m for ADD, respectively.
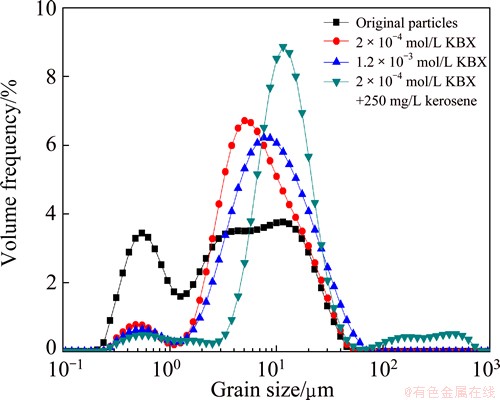
Fig. 2 Size distributions of marmatite with KBX before and after addition of kerosene at pH 7.4
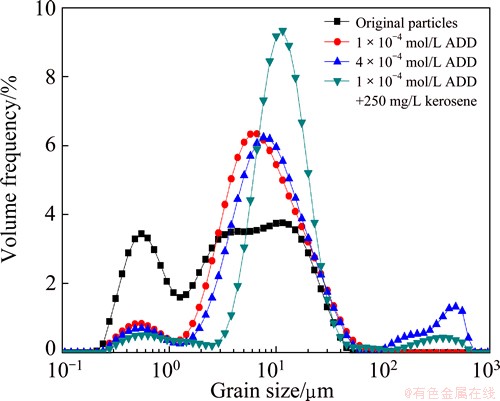
Fig. 3 Size distributions of marmatite with ADD before and after addition of kerosene at pH 7.4
The flocculation images of the marmatite before and after the hydrophobic flocculation induced by KBX and ADD, sometimes with kerosene, are shown in Fig. 4 and Fig. 5, respectively. From these two figures, it can be seen obviously that the flocculation phenomenon under the action of collectors, sometimes with kerosene, happens between particles. The level of the suspended particulate flocculation is increased with increasing the dosage of collectors by comparing the two figures. The level of the suspended particulate flocculation is more intense by adding the kerosene in comparison with just by collectors.

Fig. 4 Microscopic images by OLYMPUS CX31 for marmatite at pH 7.4

Fig. 5 Microscopic images OLYMPUS CX31 for marmatite at pH 7.4
3.2 Effect of pH
The floatability of marmatite flocs induced by KBX and ADD as a function of pH is shown in Fig. 6, in which the results for the conventional flotation of the same marmatite sample under the same reagent conditions are also given. For the conventional flotation, the suspension was only mildly stirred at 1600 r/min. But for floc flotation, the stirring speed was 1800 r/min. As is noted, the flotation response of the mineral fines is markedly improved due to the formation of hydrophobic flocs. At the given reagent and operating conditions, the floc flotation induced by KBX and ADD increases the floatability of the marmatite fines over 25% and 30% in the range of pH 3-8, respectively, in comparison with the corresponding conventional flotation. Clearly, floc flotation is a very effective means of recovering marmatite fines. Also, the graphs show that the floc flotation changed with pH in a similar manner to conventional flotation. As is noted in Fig. 6(a), the floatability induced by KBX sharply increases with increasing pH from 2 to 4, and then there is a plateau in the range of pH 4-7.5, immediately markedly decreases with increasing pH. For floc flotation, the floatability can be up to a maximum of 97%. From Fig. 6(b), it can be seen that there is a plateau in the range of pH 2-7.5 for the floc flotation, out of which the floatability fails sharply.
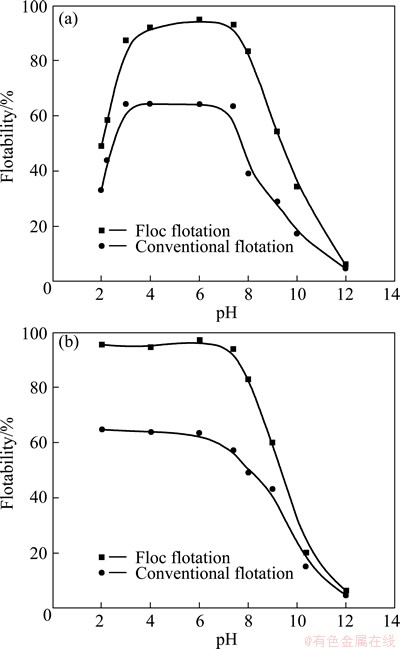
Fig. 6 Flotability of marmatite fines induced by 1.2��10-3 mol/L KBX (a) and 4��10-4 mol/L ADD (b) as function of pH
The adsorptions of both butyl xanthate ions and dibutyl dithiophosphate ions on marmatite were studied through electrokinetics. The zeta potentials of the marmatite were measured as a function of pH in the absence or presence of KBX and ADD. The results are illustrated in Fig. 7. It shows that the isoelectric point (IEP) of marmatite in aqueous solutions was at pH 4.7. In the presence of 2��10-4 mol/L KBX or 1��10-4 mol/L ADD, the IEP shifts to a lower pH and the �� potential becomes more negative. As is known, the adsorptions of KBX and ADD are due to the chemical forces between the polar group and the zinc on the surfaces [16-18]. This adsorption (chemical adsorption) occurs in the Stern plane, resulting in a more negative zeta potential. The increment in the negative �� potential (��|��|) has been related directly to adsorption density of the ions at the solid/water interface [12,13]. The greater the difference, the higher the adsorption density. Hence, from Fig. 7, as is noted, the adsorption density of ADD is higher than that of KBX.
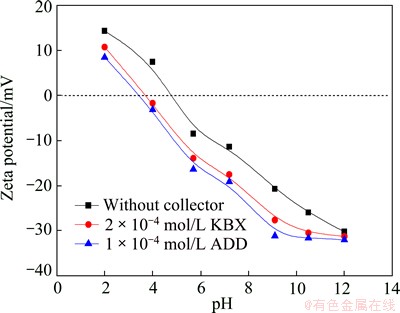
Fig. 7 �� potential of marmatite as function of pH in the absence and presence of KBX and ADD
Figure 8 presents the contact angles (which is usually used to represent hydrophobicity) of the marmatite as a function of pH in the absence and presence of 1��10-3 mol/L KBX and 4��10-4 mol/L ADD. Indeed, the addition of KBX and ADD all greatly increased the marmatite contact angles. This increase varied markedly with pH. The maximum contact angle increase all occurred at pH 7.4, being about 26��. Clearly, marmatite contact angles closely correlated with the adsorption density of butyl xanthate ions and dibutyl dithiophosphate ions. The higher the adsorption density, the greater the contact angle, which means that the hydrophobic flocculation is more powerful and the floc floatability is higher [12]. These observations indicate that the marmatite hydrophobicity (which is usually represented by contact angle) is a dominant factor for the hydrophobic flocculation. From the point of view of surface forces, the particle hydrophobicity effect is due to the fact that elevating the contact angles of the surfaces increases the hydrophobic attractive force, and thereby the hydrophobic flocculation [12,13].
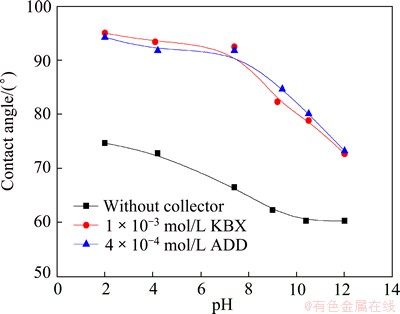
Fig. 8 Contact angle of marmatite as function of pH in the absence and presence of KBX and ADD
The collapses of the floc flotability of the marmatite fines under alkaline conditions (see Fig. 6) should not only be due to the decline of the contact angle (see Fig. 8), but also to the sharp increase of the surface charges (see Fig. 7). According to the DLVO theory [19], the electrical double-layer repulsion is proportional to the square of the surface charge, so that the repulsion between KBX-adsorbed or ADD-absorbed marmatite fines substantially increases with increasing pH, resulting in a much higher energy barrier. Hence, although the contact angle did not strongly decrease, the hydrophobic flocculation under alkaline conditions fell substantially.
3.3 Effect of collector concentrations
Figure 9 illustrates the flotability of flocculated marmatite fines as a function of collector concentrations at pH 7.4. It can be seen that the flotability of flocculated marmatite fines increased with increasing the collector concentrations until reaching 93.13% at 1.2��10-3 mol/L KBX and 94.02% at 4��10-4 mol/L ADD, respectively, and then flattened out. Relatively speaking, the growth trend of the curve for ADD is more apparent than that for KBX. As shown, the flotability is higher for ADD, indicating that hydrophobic flocculation induced by ADD is more effective than that by KBX. It suggests that the floc flotation closely correlates with the adsorption of collectors on the minerals, through which the mineral surfaces are rendered hydrophobic. Because particle hydrophobicity plays a dominant role in the formation of hydrophobic flocs [6], the increase of floc floatability with increasing collector concentrations should not only be due to the increase of the degree of hydrophobicity of the flocs, but also to the increase of floc size.
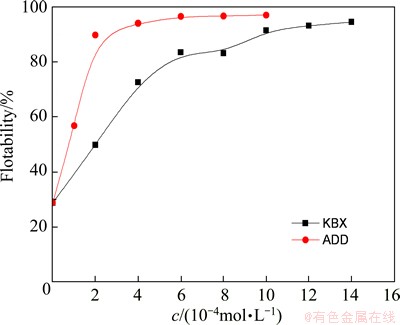
Fig. 9 Flotability of flocculated marmatite fines as function of collector concentrations at pH 7.4
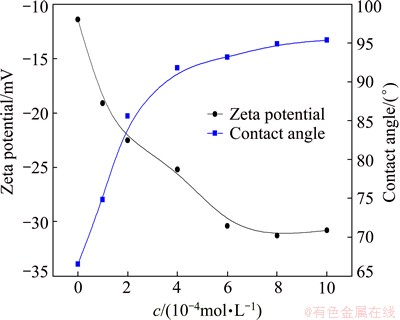
Fig. 10 Zeta potential and contact angle of marmatite particles as function of ADD concentration at pH 7.4
The zeta potentials and contact angles of marmatite particles were also measured as functions of ADD concentration at pH about 7.4. The results are shown in Fig. 10. As can be seen, the marmatite contact angles substantially increased and the zeta potentials dramatically went down to a large negative value with increasing ADD concentration. As already stated, these observations are attributed to the adsorption of ADD on marmatite surfaces. The adsorption density increased with increasing ADD concentration, raising the increases of the contact angle and the zeta potential in absolute value.
From the results presented in the two graphs, it is clear that the floc flotation of the marmatite fines had responses to the change of ADD concentration quite similar to that of the marmatite contact angle, suggesting a close correlation between the hydrophobic flocculation and the particle hydrophobicity. However, the correlation between the flocculation and the zeta potential was not found. According to the DLVO theory [19], the electric double layer interaction between particles arisen from particle surface charges (which is usually determined by the �� potential) plays an important role in the aggregative stability of fine particle suspensions. The greater the surface charge, the higher the energy barrier and the harder the aggregation. In the case of ADD adsorption on marmatite, the hydrophobic flocculation of the marmatite fines was not depressed, but was enhanced upon increasing the surface charges, which is contrary to the DLVO theory. It may be due to the fact that the ADD adsorption on marmatite increased the hydrophobic attraction much more strongly than the electric double-layer repulsion.
3.4 Effect of kerosene addition
The flotability of the marmatite flocs as a function of kerosene concentration at pH 7.4 is given in Fig. 11. As can be seen, the floatability increases with increasing kerosene addition until reaching over 92% at 250 mg/L for KBX and at 300 mg/L for ADD, respectively. Under the operating conditions in Fig. 11, the floc floatability in the absence of kerosene is 49.77% for KBX and 56.52% for ADD, respectively. It is clear that the floc flotation was greatly improved by the addition of a small amount of kerosene. From Figs. 9 and 11, also, it shows that kerosene replaced a large amount of collector to obtain over 90% floatability of the marmatite fines with floc flotation. 1��10-3 mol/L KBX was needed when it was used alone; while only 2��10-4 mol/L KBX was needed when 250 mg/L kerosene was added. 4��10-4 mol/L ADD was needed if it used alone; while only 1��10-4 mol/L ADD was required with 300 mg/L kerosene. From the point of view of flotation principle, the kerosene improvement of the floc flotation is not only attributed to the increase of floc size (see Figs. 2 and 3), but also to the increase of floc hydrophobicity.
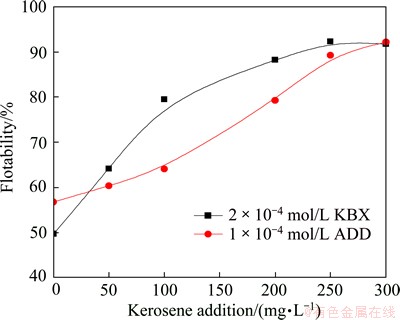
Fig. 11 Flotability of flocculated marmatite fines induced by collectors as function of kerosene addition at pH 7.4
Nonpolar oil enhancement of hydrophobic flocculation is due to the formation of oil films on the hydrophobic particles to increase the particle hydrophobicity and the bridging of the particles [20]. The oil bridges considerably increase adhesion forces between the particles involved in hydrophobic flocs (or floc strength), leading the flocs to withstanding strong floc-rupture forces from turbulent flows [21]. Owing to the two factors, nonpolar oil addition can greatly enhance hydrophobic flocculation, producing much bigger and more compact flocs, in comparison with the absence of nonpolar oil.
3.5 Fourier transform infrared (FTIR) spectra
Spectra were recorded for marmatite samples (<2 ��m) and for minerals treated with KBX and ADD at pH about 7.4 (Figs. 12 and 13). The infrared spectra show only the 500-2000 cm-1 region since this area includes most of the adsorption bands attributable to the KBX and ADD. Figure 12 shows the infrared spectra for KBX and ADD. As can be seen, the infrared spectrum of KBX shows ��CH2�� deformation vibration at 1465 cm-1. The absorption peak at 1367 cm�C1 is ��C��C�� skeletal vibration. The peak at 1109 cm�C1 is C��O��C stretching vibration. The 1061 cm�C1 peak is C=S stretching vibration [22-25]. The infrared spectrum of ADD in Fig. 12 shows that the absorption peak at 1469 cm-1 is ��CH3 or ��CH2 deformation vibration. The absorption peak at 1392 cm�C1 is the bending vibration of ��NH. The peaks at 1037 cm-1 and 993 cm-1 are P��O��C stretching vibrations. The peaks at 837 cm-1, 663 cm-1 and 563 cm-1 are P��S2 stretching vibration [22-24,26].
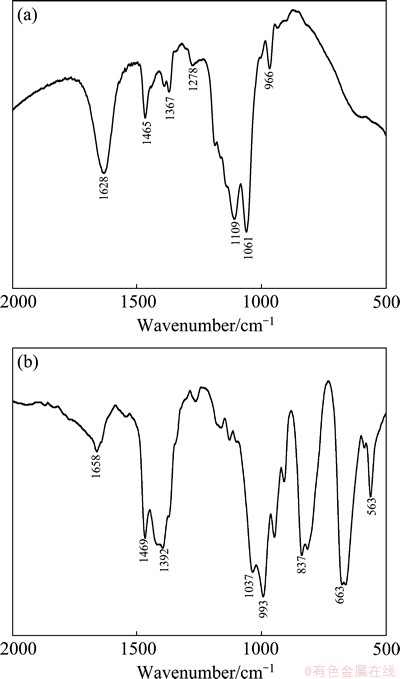
Fig. 12 Infrared spectra for KBX (a) and ADD (b)
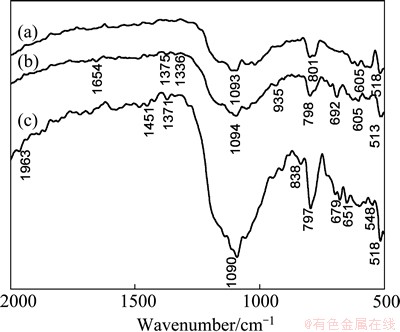
Fig. 13 Infrared spectra for marmatite (a), KBX adsorbed on marmatite (b), and ADD adsorbed on marmatite (c) at pH about 7.4
Figure 13 shows the IR spectra of marmatite and marmatite after interaction with the collector regent. The characteristic peaks of marmatite at 1093, 801, 518 cm-1 all showed a certain deviation after conditioning with KBX and ADD in Fig. 13. Except that, some faint peaks such as 1654, 1375, 1336, 935, 692 cm-1 appeared as the function of KBX in Fig. 13(b). Then we can obtain that the absorption of KBX on marmatite is chemical adsorption. In Fig. 13(c), after conditioning with ADD, there are many characteristic adsorption peaks appearing at the wavenumbers: 1451 cm-1 (��CH3 or ��CH2); 1371 cm-1 (N��H); 838, 679, 651, 548 cm-1 (P��S2). It can be seen that the interaction between ADD and marmatite also is a chemical reaction.
4 Conclusions
1) Hydrophobic flocculation induced by butyl xanthate (KBX) and ammonium dibutyl dithiophosphate (ADD) is effective on marmatite fines in aqueous suspensions. This hydrophobic flocculation arises as a result of the chemisorption of butyl xanthate ions or dibutyl dithiophosphate ions on marmatite, which imparts hydrophobicity to the particles, and thereby the hydrophobic interaction between the particles. It strongly correlates with the degree of particle hydrophobicity (which is usually represented by contact angle), namely the more hydrophobic the particles, the more powerful the hydrophobic flocculation and the higher the floc flotability. However, it is not lowered by increasing the surface charges due to collector adsorption.
2) The flotation response of marmatite fines is markedly improved due to the formation of flocs. The floc flotation strongly correlates with the floc size, and can be considerably enhanced by the addition of a small amount of kerosene. It is suggested that floc flotation is a promising means to recovery marmatite in the fine size range.
3) The IR spectra confirm that the interactions of the mineral surface with the reagent depend upon adsorption of the reagent onto the mineral surface. It can be seen that the adsorption of KBX and ADD onto marmatite is chemical adsorption.
References
[1] REN Liu-yi. The interaction of fine cassiterite particles-bubbles and its influence on flotation [D]. Changsha: Central South University, 2012. (in Chinese)
[2] COLLINS G L, JAMESON G L. Experiments on the flotation of fine particles: The influence of particle size and charge [J]. Chemical Engineering Science, 1976, 31(11): 985-991.
[3] TRAHAR W J, WARREN L J. The floatability of very fine particles��A review [J]. International Journal of Mineral Processing, 1976, 3(2): 103-131.
[4] SIVAMOHAN R. The problem of recovering very fine particles in mineral processing��A review [J]. International Journal of Mineral Processing, 1990, 28(3-4): 247-288.
[5] MIETTINEN T, RALSTON J, FOMASIERO D. The limits of fine particle flotation [J]. Minerals Engineering, 2010, 23 (5): 420-437.
[6] SONG S, LOPEZ-VALDIVIESO A, REYES-BAHENA J L, LARA-VALENZUELA C. Floc flotation of galena and sphalerite fines [J]. Minerals Engineering, 2001, 14(1): 87-98.
[7] MATIS K A, GALLIOS G P, KYDROS K A. Separation of fines by flotation techniques [J]. Separations Technology, 1993, 3(2): 76-90.
[8] FORBES E. Shear, selective and temperature responsive flocculation: A comparison of fine particle flotation techniques [J]. International Journal of Mineral Processing, 2011, 99(1-4): 1-10.
[9] OZKAN A, USLU Z, DUZYOL S, UCBEYIAY H. Correlation of shear flocculation of some salt-type minerals with their wettability parameter [J]. Chemical Engineering and Processing: Process Intensification, 2007, 46(12): 1341-1348.
[10] LU Shou-ci, DING Yu-qing, GUO Jin-yong. Kinetics of fine particle aggregation in turbulence [J]. Advances in Colloid and Interface Science, 1998, 78(3): 197-235.
[11] SONG Shao-xian. Hydrophobic flocculation theory and sorting process [M]. Beijing: Coal Industry Publishing Press, 1993. (in Chinese)
[12] SONG S, LOPEZ-VALDIVIESO A, REYES-BAHENA J L, BERMEJO-PRREZ H I, TRASS O. Hydrophobic flocculation of galena fines in aqueous suspensions [J]. Journal of Colloid Interface Science, 2000, 227(2): 272-281.
[13] SONG S, LOPEZ-VALDIVIESO A, REYES-BAHENA J L, BERMEJO-PRREZ H I. Hydrophobic flocculation of sphalerite fines in aqueous suspensions induced by ethyl and amyl xanthates [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2001, 181(1-3): 159-169.
[14] LIU Zhi-neng. Study on electrochemistry of flotation of lead-antimony-zinc sulfide ore in dithiophosphate system [D]. Changsha: Central South University, 2009. (in Chinese)
[15] ZHANG Qin, XU Jing, WANG Chang-an, CHEN Tie-jun. Study of electrochemistry flotation behavior of lead-antimony-zinc-iron sulfide ores-ADDP [J]. Express Information of Mining Industry, 2005(10): 15-17. (in Chinese)
[16] QIN Wen-qing, JIAO Fen, SUN Wei, WANG Xing-jie. Effects of sodium salt of N,N-dimethyldi-thiocarbamate on floatability of chalcopyrite, sphalerite, marmatite and its adsorption properties [J]. Colloids and Surfaces A, 2013, 421: 181-192.
[17] YANG Wei. Flotation and adsorption mechanism of marmatite on ammonium dibutyl dithiophosphate [J]. Nonferrous Metals, 2010(4): 39-42. (in Chinese)
[18] WU Bo-zeng, QIU Guan-zhou, QIN Wen-qing, CHEN Jian-ming. Floatation behavior and electrochemistry of marmatite in butyl xanthate solution [J]. Mining and Metallurgical Engineering, 2004, 24(6): 34-36. (in Chinese)
[19] WANG Dian-zuo, QIU Guan-zhou, HU Yue-hua. The interaction between particles and fine particle flotation [M]. Changsha: Central South University of Technology Press, 1993. (in Chinese)
[20] LU Shou-ci, DING Yu-qing, GUO Jin-yong. Kinetics of fine particle aggregation in turbulence [J]. Advances in Colloid and Interface Science, 1998, 78(3): 197-235.
[21] SONG S, A LOPEZ-VALDIVIESO A, DING Y. Effects of nonpolar oil on hydrophobic flocculation of hematite and rhodochrosite fines [J]. Powder Technology, 1999, 101(1): 73-80.
[22] LU Yong-quan, DENG Zhen-hua. Practical infrared spectrum analysis [M]. Beijing: Electronic Industry Press, 1989. (in Chinese)
[23] WANG Zong-ming, HE Xin-xiang, SUN Dian-qing. Practical infrared spectroscopy [M]. Beijing: Petroleum Industry Press, 1982. (in Chinese)
[24] PENG Wen-shi, LIU Gao-kui. Atlas of mineral infrared spectra [M]. Beijing: Science Press, 1982. (in Chinese)
[25] CHEN Yu-ping. The flotation study of super fine lead-antimony-zinc sulfide [D]. Changsha: Central South University, 2008. (in Chinese)
[26] SONG Quan-yuan, CHEN Xiu-zhi. The flotation mechanism of ammonium dibutyl dithiophosphate on pyrite were studied with FTIR [J]. Nonferrous Metals, 1997, 49(1): 33-38. (in Chinese).
�� �ã������죬����ʣ���ˮ��
���ϴ�ѧ ��Դ�ӹ������﹤��ѧԺ����ɳ 410083
ժ Ҫ�����ü������ȷ�����������������Ӿ��ɢ�䡢�Ӵ��Dz����ͺ�������о���ˮ����Һ���ö�����ҩ(KBX)�Ͷ�狀�ҩ(ADD)�յ�ϸ������п�����ˮ������ͨ���ı���ֲ��������а���pHֵ�����ռ�Ũ�Ⱥ�ú���������о�������С��20 ��m�ĵ���������Ÿ�ѡ��Ϊ��������������Ÿ�ѡЧ�����������ȺͿ�����ˮ��������أ��������ڲ��ռ����������µĿ��������������û�н������Ÿ�ѡЧ�������Ż��IJ��������£���KBX��ADD��������ϸ������п������Ÿ�ѡ�����ʾ�����90%�������渡ѡ�Ļ�����ֻ��60%���ҡ����黹���֣���������п�����������Ĥ���γɣ�����ú�͵����ӿɴ���ȸĽ���ѡָ�ꡣFTIR���������������ҩ��������п������������Ϊ��ѧ������
�ؼ��ʣ���ˮ������ϸ������п��zeta��λ���Ӵ��ǣ�������ҩ(KBX)����狀�ҩ(ADD)��ú��
(Edited by Hua YANG)
Foundation item: Project (51274255) supported by the National Natural Science Foundation of China; Project supported by the Foundation of State Key Laboratory of Comprehensive Utilization of Low-Grade Ores (Zijin Mining Group Co., Ltd), China
Corresponding author: Wen-qing QIN; Tel: +86-731-88830884; E-mail: qinwenqing1@126.com
DOI: 10.1016/S1003-6326(14)63228-3

