J. Cent. South Univ. Technol. (2008) 15(s1): 492-495
DOI: 10.1007/s11771-008-407-y
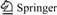
Factors influencing HPAM solution viscosity prepared by produced water using orthogonal method
KANG Wan-li(康万利)1, WANG Zhi-wei(王志伟)1, ZHOU Yang(周 阳)1,
MENG Ling-wei(孟令伟)1, LIU Shu-ren(刘述忍)1, BAI Bao-jun(白宝君)2
(1. Enhanced Oil Recovery Research Center, China University of Petroleum, Qingdao 266555, China;
2. University of Missouri of Science and Technology, Rolla MO 65401, USA)
Abstract: The effect of temperature and metal ion components on the viscosity of HPAM solution was studied by means of orthogonal method. Five factors and 4 levels were considered for the orthogonal design. The five factors included temperature, contents of Na+, Mg2+, Ca2+ and S2-, and the four levels mainly considered the level of produced water components in main oilfields in China. The experiment results show that temperature is the most important factor to control HPAM solution viscosity prepared by produced water. The effect of ions was in order of Na+>S2->Mg2+>Ca2+. The mechanism of each factor influencing viscosity was also discussed. Based on the actual condition of Nanyang oilfield, the desulfurization was used to improve the solution viscosity, and satisfactory result was obtained.
Key words: polymer; viscosity; oil produced water; orthogonal method; O3
1 Introduction
The consumption of fresh water in oil field has increased dramatically as the extension of polymer flooding industrialization. It will be feasible and economical if we can use produced water to prepare polymer fluid. Produced water from oil wells is very complex because it has more suspended substances, greater fraction oil mass, more dissolved oxygen, greater bacteria mass fraction and greater salinity than standardized injection water, especially if it contains produced HPAM. Therefore, it’s more difficult to treat oilfield produced water[1]. Although the current treatment techniques for produced water, such as oil separation, coalescence, coagulation, filtration, and sterilization could improve HPAM solution viscosity, there are still great salinity and some existing reducing substances which have great effect on HPAM solution viscosity. These factors determined the ground viscosity level of HPAM solution with produced water. A lot of studies have been conducted on some of these factors[2-5], but none of them offered a comprehensive method that could show the degree of each factor for a specified oilfield. In this paper, these contents were studied by means of orthogonal test, and the result could be a theoretical support to improve HPAM solution viscosity with oil produced water. Satisfactory result was obtained by the research on the method of desulfurization.
2 Experimental
2.1 Experiment instruments
Brookfield DV-II+ viscometer.
O3 producer.
2.2 Chemicals
NaCl, NaHCO3, Na2CO3, Na2S・9H2O, FeSO4・7H2O, MgSO4・7H2O, FeCl3・6H2O, CaCl2 (above were C.P. or A.P.), deionized water, HPAM (M=2.2×107) were used.
2.3 Experimental methods
2.3.1 Effect of various ions on HPAM solution viscosity
Solutions with various concentrations of Na+, Ca2+, Mg2+, Fe2+, Fe3+, S2- were prepared, and then HPAM solutions with the same concentration were prepared using solutions above. The effect of six types of ions on HPAM solution viscosity was studied.
2.3.2 Orthogonal experiment
The factors for orthogonal experiment were temperature, concentrations of Na+, Mg2+, Ca2+, S2-, and the levels of factors were determined according to component analysis of main oilfield produced water in China[6-9]. L16(45) orthogonal table was used for the experiment, that is to say, 16 tests were done totally. Table 1 shows the factors and levels used.
Table 1 Conditions selected for orthogonal experiment
The HPAM concentrations in both groups of experiments were 1 400 mg/L. After 4 h of stirring and 1 h of aging, the viscosities were measured by Brookfield DV-II+ viscometer. The rotating speed was 6 r/min while the measuring time was 5 min. The results were the average value of 3 measurements for each sample.
3 Results and discussion
3.1 Effect of various ions on HPAM solution viscosity
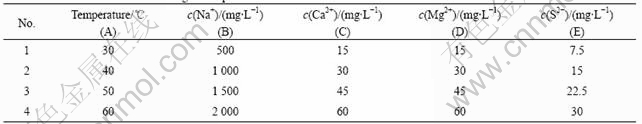
Fig.1 shows the effect of various ions on HPAM solution viscosity. The capability of each ion to reduce the viscosity can be determined by the concentration (that is, more reducing the viscosity in lower- concentration), and it is ranked as Fe2+>Fe3+>Mg2+>Ca2+>S2->Na+. The mechanisms of Fe2+ and S2- were due to good reducibility. The redox reaction could accelerate the formation of free radicals in the solution. The free radical reactions were very active and could lead to the fracture of polymer chain. As a result of degradation of polymer, HPAM solution viscosity reduced seriously. The mechanism of Fe3+>Mg2+>Ca2+>Na+ was due to the sensibility to salinity, and the higher ion valence was, the stronger sensibility to salinity the polymer solution was. Moreover, polymer in solution could be crosslinked by high concentration of high valent cationic, which could result in precipitation of polymer. A serious lose of viscosity would be manifested because of the precipitation.
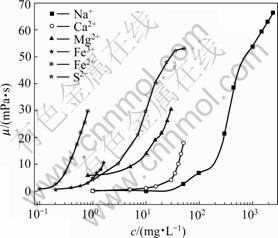
Fig.1 Relationships between polymer solution viscosity and ion concentrations
3.2 Orthogonal experiment and differential analysis
Table 2 lists the differential analysis on the result of orthogonal experiment.
The differential represents the importance of factors. The higher differential is, the greater effect on polymer solution viscosity the factor is. In addition, Kij represents the importance of levels of the same factor. The higher the Kij is, the greater effect the factor of this level is than other levels.
The orthogonal experiments were conducted because it can be used not only to compare the viscosity reduction ability of different ions ranging from different concentrations but also can to rank the relative level of each parameter that influences the polymer viscosity.
Fig.2 shows the effect curve of each factor. The intercept of each curve represents the differential of each factor and reflects the viscosity reduction ability of each factor. The highest the curve intercept is, the stronger the viscosity reduction ability of this factor is. Each curve slope represents the effect degree ranging from experimental concentrations of ion used to prepare HPAM solution. For the factor with largest curve slope, it will have a great effect on HPAM solution viscosity if a little change happens to this factor within the experimental concentration.
Two aspects were indicated from Fig.2. On one hand, for this oilfield produced water, the sequence of ion reduction effect on polymer solution viscosity was Na+>S2->Mg2+>Ca2+. Among them, S2- has little effect on solution viscosity when its concentration ranges from 15 to 30 mg/L. On the other hand, preparing HPAM solution with high viscosity, the process should be conducted at a low temperature, and the content of ions should be decreased as far as possible. It is easy to discover that S2- and divalent ions can cause obvious loss in viscosity at a low concentration level. A large amount of money will be spent on the desalination of oil produced water. Therefore, it is economical and reasonable to increase HPAM solution viscosity by eliminating sulfides.
Table 2 Calculation and experimental results of L16(45)
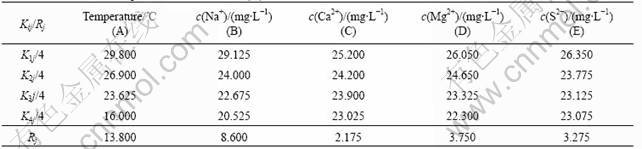
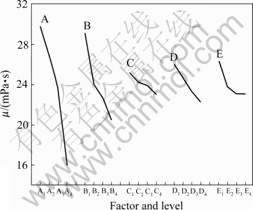
Fig.2 Differential tendency of each factor
4 Applications
The results of orthogonal experiment were applied to analyze the effect of produced water on HPAM solution viscosity in Jianghe block of Nanyang Oilfield. Table 3 lists the components in produced water of Jianghe block, and the corresponding value according to Fig.2.
Table 3 Components and corresponding value in produced water of Jianghe block

It is shown that temperature has the greatest effect on HPAM solution viscosity. However, the temperature is constant in the reservoir, so it is not taken into consideration. The ions effect sequence was Na+>S2->Mg2+. Therefore, the technique to improve HPAM solution viscosity is to eliminate S2-.
Three techniques were used to eliminate S2-: pumping into air, H2O2 and O3. Fig.3 shows S2- concentrations change with time. It can be seen that O3 has the greatest effect on eliminating S2-. Therefore, viscosity recovery experiments were conducted via ozone generator and oilfield produced water. Preliminary gas-liquid ratio was also studied. The results were shown in Fig.4. It is shown that HPAM solution has the highest viscosity when the gas-liquid ratio is 12 mg/L. The highest viscosity is 44.9 mPa・s, which is improved by 22% compared with the initial viscosity of 36.8 mPa・s. The result shows that desulfurization can effectively improve HPAM solution viscosity.
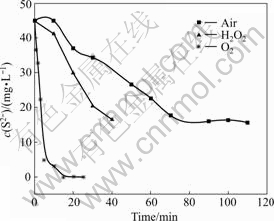
Fig.3 Effects of different desulfurizing methods on polymer viscosity recovery
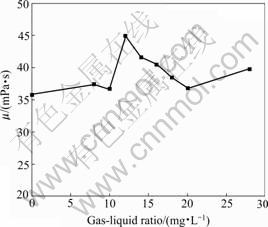
Fig.4 Relationship between viscosity and gas-liquid ratio
5 Conclusions
1) The temperature drastically affects polymer solution viscosity. The ions order to reduce polymer solution viscosity is Fe2+>Fe3+>Mg2+>Ca2+>S2->Na+.
2) Orthogonal test can be used to analyze the effect of components on HPAM solution viscosity for a specified oilfield produced water. If the factors and levels in experiments are corresponded with the real oilfield situation, then the analysis will show the degree of each influencing factor.
3) Orthogonal experiment was applied to analyzing the effect of produced water on HPAM solution viscosity in Jianghe block of Nanyang oilfield. The result shows that desulfurization can improve HPAM solution viscosity by 22% and it is feasible.
References
[1] YANG Huai-jun, LUO Ping-ya. Factors for and control of polymer degradation in recycled produced water solutions [J]. Oilfield Chemistry, 2005, 22(2): 158-162. (in Chinese)
[2] LIU Yu-wen. Influence of salinity on the viscosity of hydrophobic associated polymer solution [J]. Petroleum Geology and Recovery Efficiency, 2003, 10(3): 62-64. (in Chinese)
[3] ZHAN Ya-li, GUO Shao-hui, YAN Guang-xu. Study on degradation of partially hydrolyzed polyacrylamide [J]. Polymer Bulletin, 2004, 4: 70-74. (in Chinese)
[4] YAO Hui, ZHANG Zhong-jian. The oily sewage matches the note polymer applied research [J]. The Oil-Gas Field Ground Project, 2007, 26(5): 3-4. (in Chinese)
[5] KANG Wan-li, MENG Ling-wei, NIU Jing-gang, XU Dian-ping. Mechanism of the effect of salinity on HPAM solution viscosity [J] Polymer Materials Science and Engineering, 2006, 22(5): 175-177. (in Chinese)
[6] XU Fu, YIN Xian-qing. The compatibleness and scaling of recycled injection water at dagang oil field [J]. Journal of Pingdingshan University, 2007, 22(5): 77-79. (in Chinese)
[7] FAN Ping, WANG Xue-jun, WANG Xiao-yu, HU Jun-qing. Research on demineralization method of wastewater from oilfield [J]. Environmental Science and Management, 2007, 32(1): 90-93. (in Chinese)
[8] PU Peng-cheng. Present state and development trend of oily waste water disposal in Jidong oilfield [J]. Journal of Chengde Petroleum College, 2007, 9(3): 1-3. (in Chinese)
[9] ZHANG Ben-yan, ZHANG Ji-chao, TU Wen-li. Current situation of water injection and its influence on reservoirs in Shengli oilfield [J]. Petroleum Exploration and Development, 2007, 24(3): 364-368. (in Chinese)
(Edited by CHEN Ai-hua)
Foundation item: Project(2007AA06Z214) supported by the National High-Tech Research and Development Program of China; Project(ts20070704) supported by Taishan Scholars Construction Engineering; Project(200873181) supported by National Natural Science Foundation of China
Received date: 2008-06-25; Accepted date: 2008-08-05
Corresponding author: KANG Wang-li, Professor, PhD, Taishan Scholar; Tel: +86-13589332193; E-mail: Kangwanli@126.com