ОДХВ±аєЕЈє1004-0609(2010)02-0301-07
ДЙГЧTiO2¶Фов(VI)µДОьёЅРФДЬ
ХЕ АЩЈ¬БхС©СТЈ¬ЅЄПюЗмЈ¬Ао зІЈ¬їµЖЅАы
(БЙДюґуС§ »ЇС§С§ФєЈ¬ЙтСф 110036)
ХЄ ТЄЈєСРѕїДЙГЧTiO2¶ФMo(VI)µДОьёЅ№эіМЈ¬їјІмИЬТєµДpHЦµЎўК±јдЎўОВ¶ИµИТтЛШ¶ФОьёЅµДУ°ПмЎЈЅб№ы±нГчЈєФЪpH 1~8·¶О§ДЪЈ¬ДЙГЧTiO2¶ФMo(VI)µДОьёЅВКѕщі¬№э99%Ј¬ОьёЅУЪДЙГЧTiO2ЙПµДMo(VI)їЙУГ2.0 mLµД0.1 mol/L NaOHИЬТє¶ЁБїПґНСЈ»ёГОьёЅ№эіМ·ыєПЧј¶юј¶·ґУ¦¶ЇБ¦С§ДЈРНЈ¬Жд·ґУ¦µД±н№Ы»о»ЇДЬОЄ22.7 kJ/molЈ¬БЈЧУДЪІїА©Йў№эіМКЗЖдОьёЅїШЦЖІЅЦиЈ¬µ«ТєПа±ЯЅзІгПтБЈЧУ±нГжµДА©Йў№эіМІ»ДЬєцВФЈ»ОьёЅРРОЄ·юґУLangmuirєНD-RµИОВДЈРНЎЈФЪКТОВПВЈ¬ДЙГЧTiO2¶ФMo(VI)µД±ҐєНОьёЅИЭБїОЄ12.74 mg/gЈ¬ЖЅѕщОьёЅДЬОЄ17.36 kJ/molЈ»ОьёЅ·ґУ¦мК±дєНмШ±дѕщОЄХэЦµЈ¬ЧФУЙДЬ±дОЄёєЦµЈ¬ЛµГчёГОьёЅ№эіМОЄЧФ·ўµДОьИИ№эіМЎЈ
№ШјьґКЈєMo(VI)Ј»ДЙГЧTiO2Ј»ОьёЅЈ»¶ЇБ¦С§Ј»ИИБ¦С§
ЦРНј·ЦАаєЕЈєO 647.3ЎЎЎЎ ОДПЧ±кК¶ВлЈєA
Adsorption properties of nano-TiO2 for Mo(VI)
ZHANG Lei, LIU Xue-yan, JIANG Xiao-qing, LI Qi, KANG Ping-li
(College of Chemistry, Liaoning University, Shenyang 110036, China)
Abstract: Titanium dioxide nanoparticles were employed for the sorption of Mo(VI) ions from aqueous solution. The process was studied in detail by varying the pH value, sorption time and temperature. The adsorption rate of Mo(VI) by the sorbent is over 99% at pH 1?8. Adsorbed Mo(VI) were desorbed by 2.0 mL of 0.1 mol/L NaOH. The kinetic experimental data properly correlate with the second-order kinetic model. The apparent activation energy is 22.7 kJ/mol. The overall rate process is mainly controlled by the intraparticle diffusion, while the boundary layer resistance cannot be ignored. The sorption data could be well interpreted by the Langmuir and D-R type sorption isotherms. At room temperature, the static saturated adsorption capacity is 12.74 mg/g, and the average sorption energy is 17.36 kJ/mol. The thermodynamic parameters: ¦¤H¦ЁЈѕ0, ¦¤S¦ЁЈѕ0 and ¦¤G¦ЁЈј0, which indicates a spontaneous and decalescence adsorption process.
Key words: Mo(VI); nano-TiO2; adsorption; kinetics; thermodynamics
·єК№УГЈ¬№КДїЗ°¶ФовµДСРѕї·ЗіЈ»оФѕЎЈовµДјЫёсЅП°є№уЈ¬ЖдУГБїФЪІ»¶ПЙПЙэЈ¬¶шФЪµШїЗЦРµДє¬БїЅц2 g/tЈ¬ЗТовїуЧКФґЦРовµДЖ·О»ТІовУл»ъРµ№¤ТµЎўКЇУН»Ї№¤µИµДЙъІъУл·ўХ№ГЬЗРПа№ШЈ¬Н¬К±Ј¬ов»ЇС§Ж·ФЪДЙГЧІДБПµДСРЦЖј°ФЪЙ±ѕъјБЎўСХБПЎўИХУГЖ·µИ·ЅГж±»№гФЪІ»¶ППВЅµЈ¬Тт¶шовµДМбИЎєН»ШКХИХПФЦШТЄЎЈ·ЦАлё»јЇовµД·Ѕ·ЁєЬ¶аЈ¬ЦчТЄУРТєЭНИЎ[1]Ўў№ІЅбѕ§[2]ЎўЕЭДёЎ СЎ[3]ЎўТєД¤·Ё[4]єНАлЧУЅ»»»·Ё[5]µИЎЈ
ДЙГЧІДБПКЗЅьДкАґ·ўХ№ЖрАґµДТ»ЦЦРВРН№¦ДЬІД БП[6]Ј¬ЛьѕЯУР±И±нГж»эґуЎў±нГжФЧУКэДї¶аµИУЕµгЈ¬ТСКЬµЅСРѕї№¤ЧчХЯµД№г·є№ШЧўЎЈЛь¶ФРн¶аЅрКфАлЧУѕЯУРєЬЗїµДОьёЅДЬБ¦Ј¬КЗєЫБїФЄЛШ·ЦОцЦРЅПОЄАнПлµД·ЦАлё»јЇІДБП[7]ЎЈ
LIµИ[8]±ЁµАБЛДЙГЧTiO2·ЦАлё»јЇОЩЈ¬АоґєПгµИ[9]±ЁµАБЛДЙГЧTiO2·ЦАлё»јЇовєНОЩµДМхјюЎЈУР№ШДЙГЧTiO2ОьёЅРФДЬµДСРѕї±ЁµАІ»¶аЈ¬HADJIIVANOVµИ[10] СРѕїБЛTiO2УГУЪОьёЅCoЎўNiЎўCuЅрКфАлЧУµДїЙРРРФЈ¬МЅМЦБЛЅрКфАлЧУµДОьёЅДЈРНІўСРѕїБЛТ»Р©ЅрКфАлЧУµДОьёЅРРОЄЎЈLIANGµИ[11]±ЁµАБЛДЙГЧTiO2¶ФПЎНБАлЧУєН№э¶ЙЅрКфАлЧУОьёЅРФДЬµДСРѕїЎЈ
УЙУЪУР№ШДЙГЧTiO2ОьёЅовµДСРѕїПКјы±ЁµАЈ¬ТтґЛ±ѕОДЧчХЯТФДЙГЧTiO2ЧчОЄОьёЅјБЈ¬МЅМЦpH¶ФОьёЅµДУ°Пмј°УР№ШОьёЅ»ъАнЈ¬¶ФДЙГЧTiO2ОьёЅовµДИИБ¦С§Ул¶ЇБ¦С§РРОЄЅшРРЅППµНіµДСРѕїЎЈёГПоСРѕїіЙ№ыОЄовµДУРР§МбИЎМṩБЛІОїјНѕѕ¶ЎЈ
1 КµСй
1.1 КµСйТЗЖчУлКФјБ
1.1.1 КµСйТЗЖч
ЛщУГКµСйТЗЖчИзПВЈєUV-Vis-NIR Cary 5000·Ц№в№в¶ИјЖЈ¬PHS?3CРНѕ«ГЬpHјЖЈ¬ KQ?100BРНі¬ЙщІЁЗеПґЖчЈ¬TDL80?2BМЁКЅАлРД»ъЈ¬TDA?8002РНµзИИєгОВЛ®ФЎ№шЎЈ
1.1.2 ЦчТЄКФјБ
ЛщУГЦчТЄКФјБИзПВЈєЛ®Со»щУ«№вНЄ(SAF)Ј»де»ЇК®БщНй»щИэјЧп§(CTMAB)Ј¬HClЈ¬NaOHЈ»ДЙГЧTiO2 (ИсоСРН20~50 nm)Ј¬АґЧФЦЫЙЅГчИХДЙГЧІДБПУРПЮ№«ЛѕЎЈКµСйЛщУГКФјБЅФОЄ·ЦОцґїЈ¬Л®ОЄ¶юґОХфБуЛ®ЎЈ
ов±кЧјґў±ёИЬТє(УЕј¶ґї)µДЦКБїЕЁ¶ИОЄ1.000 mg/mLЈ¬іЖИЎ·ЦОцґїовЛбп§[(NH4)6Mo7O24Ў¤4H2O] 3.6804 gЈ¬УГ1?9(V(NH3)?V(H2O))°±Л®ИЬЅвЈ¬ТЖИл2 000 mLµДИЭБїЖїЦРЈ¬ФЩУГ1?9(V(NH3)?V(H2O))°±Л®¶ЁИЭЈ¬ТЎФИЎЈ
1.2 КµСй·Ѕ·Ё
1.2.1 овµДІвБї·Ѕ·Ё
ИЎККБїµДMo(VI)±кЧјИЬТєЈ¬ФЪHClЅйЦКЦРЈ¬јУИлЛ®Со»щУ«№вНЄєНде»ЇК®БщНй»щИэјЧп§ИЬТєЈ¬УГЛ®ПЎКНєуПФЙ«Ј¬УЪІЁі¤522 nmґ¦Ј¬Ів¶ЁОь№в¶ИЎЈ
1.2.2 овµДОьёЅ·Ѕ·Ё
БїИЎ10 mLТ»¶ЁЕЁ¶ИMo(VI)±кЧјИЬТєБїУЪ50 mLѕЯИыЧ¶РОЖїЦРЈ¬УГHClєНNaOHИЬТєµчЅЪMo(VI)±кЧјИЬТєµДpHЦµЈ¬И»єујУИлТ»¶ЁБїµДДЙГЧTiO2Ј¬і¬Йщ·ЦЙў3 minЈ¬ѕІЦГ10 minЈ¬ТФ4 000 r/minЧЄЛЩАлРД5 minЈ¬·ЦОцЛ®ПаЦРMo(VI)µДЕЁ¶ИЈ¬°ґХХ1.2.1Ів¶Ё·Ѕ·ЁЈ¬Ів¶ЁОь№в¶ИAЈ¬јЖЛгMo(VI)µДє¬БїєНОьёЅВКЎЈ
ОьёЅВК¦ЗµДјЖЛ㹫ʽΪ

КЅЦРЈє¦С0ОЄФИЬТєЦРОьёЅЦКµДЦКБїЕЁ¶ИЈ¬mg/LЈ»¦СОЄОьёЅЖЅєвК±ИЬТєЦРКЈУаОьёЅЦКµДЦКБїЕЁ¶ИЈ¬mg/LЎЈ
1.2.3 ¶ЇБ¦С§КµСй
БїИЎ10 mLЦКБїЕЁ¶ИОЄ50 mg/LµДMo(VI)ИЬТєЦГУЪТ»ПµБР50 mLѕЯИыЧ¶РОЖїЦРЈ¬µчИЬТєpHОЄ6.0Ј¬јУИл50 mgДЙГЧTiO2Ј¬·Ц±рФЪ273Ўў296єН323 KМхјюПВЈ¬Ів¶ЁІ»Н¬К±јдјдёфПВДЙГЧTiO2µДОьёЅБїЎЈ
1.2.4 ИИБ¦С§КµСй
ФЪТ»ПµБР50 mLѕЯИыЧ¶РОЖїЦРЈ¬јУИлІ»Н¬ЦКБїЕЁ¶ИµДMo(VI)ИЬТє10 mLЈ¬УГHClєНNaOHµчИЬТєµДpHОЄ6.0Ј¬јУИл50 mgДЙГЧTiO2Ј¬·Ц±рФЪ275Ўў290єН323 KМхјюПВЅшРРОьёЅКµСйЎЈ
2 Ѕб№ыУлМЦВЫ
2.1 pHµДУ°Пм
pHЦµ¶ФДЙГЧTiO2ОьёЅMo(VI)ЖрЧЕЦШТЄµДЧчУГЎЈФЪІ»Н¬pHЦµМхјюПВЈ¬ДЙГЧTiO2¶ФMo(VI)µДОьёЅВКµДУ°ПмИзНј1ЛщКѕЎЈУЙНј1їЙЦЄЈ¬pHЦµФЪ1~8·¶О§ДЪЈ¬ДЙГЧTiO2¶ФMo(VI)ѕЯУРБјєГµДОьёЅРФДЬЈ¬ОьёЅВКґп99%ТФЙПЈ¬јґїЙТФ±»¶ЁБїОьёЅЈ»µ±pHЈѕ8К±Ј¬ДЙГЧTiO2¶ФMo(VI)µДОьёЅДЬБ¦ПВЅµЎЈ
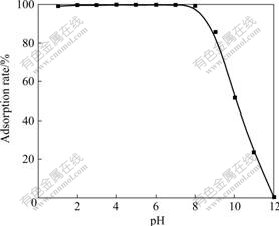
Нј1 pHЦµ¶ФДЙГЧTiO2ОьёЅMo(VI)µДУ°Пм
Fig.1 Effect of pH on adsorption rate of Mo(VI) for nano-TiO2
Mo(VI) АлЧУФЪИЬТєЦРµДґжФЪРОКЅУлИЬТєµДpHЦµГЬЗРПа№ШЎЈФЪЛбРФМхјюПВЈ¬Mo(VI)ТФMo7O246?ЎўMo8O264?µИРОКЅґжФЪЈ¬ґшЅП¶аµДёєµзєЙЈ¬¶шДЙГЧTiO2±нГжґшХэµзєЙ[12]Ј¬ИЭТЧОьёЅMo(VI)АлЧУЎЈФЪјоРФМхјюПВЈ¬Mo(VI)ФтТФЛДГжМеТхАлЧУMoO42?РОКЅґжФЪЈ¬ДЙГЧTiO2±нГжґшёєµзєЙ[12]Ј¬¶ФMo(VI)ОьёЅВКЅПµНЎЈКµСйСЎФсpH 6.0МхјюПВЅшРРОьёЅЎЈ
ґУНј1їЙТФЦЄµАЈ¬јоРФМхјюПВMo(VI)µДОьёЅВКЅПµНЈ¬±ѕКµСйСЎФсNaOHИЬТєЅвОцMo(VI)ЎЈКµСй·ўПЦУГ2.0 mL 0.1 mol/LNaOHИЬТєїЙ¶ЁБїПґНСMo(VI)Ј¬ПґНСВКОЄ97%ЎЈ
2.2 ОьёЅ¶ЇБ¦С§СРѕї
2.2.1 ОьёЅ¶ЇБ¦С§ДЈРН
іЈУГУЪГиКцОьёЅ¶ЇБ¦С§·ЅіМµДКэС§ДЈРН[13]ИзПВЈє
1) Lagergren ЧјТ»ј¶¶ЇБ¦С§·ЅіМ

2) Ho Чј¶юј¶¶ЇБ¦С§·ЅіМ

КЅЦРЈєk1(min?1)Ўўk2(g?mg?1?min?1)·Ц±рОЄТ»Ўў¶юј¶ОьёЅЛЩВКіЈКэЈ¬qt(mg?g?1)ОЄtК±їМµДОьёЅБїЈ¬q1(mg?g?1)Ўўq2(mg?g?1)ОЄЖЅєвОьёЅБїЎЈ
·Ц±рТФlg( q1?qt )¶ФtЎўt/qt¶ФtЧчНјЈ¬¶ФЛщУРКэѕЭЅшРР»Ш№й·ЦОцЈ¬ґУР±ВКєНЅШѕШїЙµГµЅІ»Н¬ОВ¶ИПВДЙГЧTiO2µДk1Ўўk2Ўўq1Ўўq2ТФј°Па№ШПµКэr1Ўўr2ЦµЈ¬КµСйКэѕЭИз±н1ЛщБРЎЈ
±н1 І»Н¬ОВ¶ИПВДЙГЧTiO2ОьёЅMo(VI)µД¶ЇБ¦С§ІОКэ
Table 1 Kinetic parameters for Mo(VI) adsorption on nano-TiO2 at different temperatures
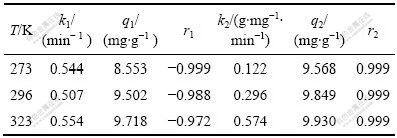
ґУ±н1їЙїґіцЈ¬ФЪ3ЦЦОВ¶ИПВЈ¬¶ЇБ¦С§КµСйКэѕЭУл¶юј¶·ґУ¦¶ЇБ¦С§ДЈРНУРЅПєГµДДвєП(r2Јѕ0.999)ЎЈґЛНвЈ¬ЛжЧЕ·ґУ¦ОВ¶ИµДЙэёЯЈ¬·ґУ¦ЛЩВКФцґуЈ¬УЙґЛїЙЦЄЈ¬ДЙГЧTiO2¶ФMo(VI)µДОьёЅ№эіМКЗОьИИ№эіМЎЈ
2.2.2 ОьёЅ·ґУ¦µД»о»ЇДЬ
Arrhenius№«КЅіЈУГУЪ¶Ф·ґУ¦»о»ЇДЬµДЗуЅв[14]Ј¬Жд±нґпКЅОЄ

КЅЦРЈєkОЄ·ґУ¦ЛЩВКіЈКэЈ¬g?mg?1?min?1Ј»AОЄЦёЗ°ТтЧУЈ»EaОЄ·ґУ¦»о»ЇДЬЈ¬kJ?mol?1Ј»RОЄАнПлЖшМеіЈКэЈ¬J?mol?1?K?1Ј»TОЄѕш¶ФОВ¶ИЈ¬KЎЈ
ёщѕЭІ»Н¬ОВ¶ИПВMo(VI)ОьёЅ¶ЇБ¦С§КµСйКэѕЭЈ¬ТФln k¶Ф1000/TЧчНјЈ¬їЙµГµЅТ»МхЦ±ПЯ(Нј2)Ј¬ёщѕЭЦ±ПЯµДР±ВКїЙЗуµГДЙГЧTiO2ОьёЅMo(VI)µД±н№Ы»о»ЇДЬEaОЄ22.7 kJ/molЎЈ

Нј2 »о»ЇДЬµД°ўВЧДбОЪЛ№ЗъПЯ
Fig.2 Arrhenius curve of activation energy
2.2.3 А©ЙўДЈРН
ОЄБЛИ·¶ЁОьёЅµДЛЩВКїШЦЖІЅЦиЈ¬Ѕ«¶ЇБ¦С§КэѕЭУГД¤А©ЙўєНБЈЧУА©Йў·ЅіМЅшРРДвєПЈ¬·ЅіМИзПВЈє
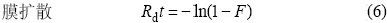
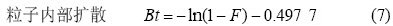
КЅЦРЈєF=qt /qeЈ»qtЈ¬qe·Ц±рОЄtК±їМµДОьёЅИЭБїєНЖЅєвОьёЅИЭБїЈ¬mg?g?1Ј»RdОЄД¤А©ЙўµДЛЩВКіЈКэЈ»B=¦Р2Di /d2 (DiОЄДЪІїА©ЙўПµКэЈ¬dОЄБЈЧУ°лѕ¶)ЎЈёщѕЭКЅ(6)єНКЅ(7)Ј¬УГ?ln(1?F)єН?ln(1?F)?0.497 7¶ФtЧцНјЈ¬·Ц±рИзНј3єН4ЛщКѕЎЈНј3ЦРЦ±ПЯГ»УРНЁ№эФµгЈ¬±нГчД¤А©ЙўІ»КЗДЙГЧTiO2ОьёЅMo(VI)ОьёЅ№эіМµДїШЦЖІЅЦиЎЈНј4ЦРЦ±ПЯЅьЛЖНЁ№эФµгЈ¬±нГчБЈЧУДЪІїА©ЙўКЗОьёЅ№эіМµДЦчТЄїШЦЖІЅЦи[15]ЎЈ

Нј3 ТєД¤А©ЙўДвєПЗъПЯ
Fig.3 Fitted curve of liquid film diffusion

Нј4 БЈЧУДЪІїА©ЙўДвєПЗъПЯ
Fig.4 Fitted curve of intraparticle diffusion
2.2.4 ДЪІїБЈЧУА©ЙўДЈРН
Mo(VI)ФЪОьёЅјБДЪІїµДА©Йў№эіМїЙУГWeber-MorrisДЈРНАґГиКц[16]Ј¬·ЅіМКЅИзПВЈє

КЅЦРЈєI±нКѕОьёЅјБЦЬО§±ЯЅзІг¶ФОьёЅ№эіМµДУ°ПмЈ¬IЦµФЅґуЈ¬±ЯЅзІг¶ФОьёЅµДУ°ПмФЅґу[17]ЎЈ·ыєПКЅ(9)µДОьёЅ»ъАн±ИЅПёґФУЈ¬±ЯЅзІгПтБЈЧУ±нГжµДА©Йў№эіМ(»тБЈЧУНвІїґ«ЦКЧи°¶ФА©Йў№эіМµДУ°Пм)І»їЙТФєцВФ[18]ЎЈ
Ѕ«Weber-MorrisДЈРНКэѕЭЅшРР·Ц¶ОПЯРФДвєПЈ¬ДвєПЗъПЯИзНј5ЛщКѕЈ¬УЙДЈРНµГµЅµДДЪІїА©ЙўЛЩ¶ИіЈКэєНПЯРФПа№ШПµКэrИз±н2ЛщБРЎЈґУНј5їЙТФїґіцЈ¬Weber-MorrisДЈРНЗъПЯµД2¶ОПЯРФДвєПЗйїцЅПєГЈ¬ЛµГчБЈЧУДЪІїА©Йў№эіМКЗОьёЅ№эіМµДїШЦЖІЅЦиЈ»µ«А©ЙўДЈРНДвєПЦ±ПЯІўІ»ѕ№эФµгЈ¬ЛµГчДЙГЧTiO2ОьёЅMo(VI)»ъАн±ИЅПёґФУЈ¬ОьёЅјБЦЬО§ТєПа±ЯЅзІгПтБЈЧУ±нГжµДА©Йў№эіМІ»їЙТФєцВФЎЈ
2.3 ОьёЅИИБ¦С§СРѕї
2.3.1 ОьёЅµИОВДЈРНєНОьёЅИЭБї
¶ФУЪ№М?ТєМеПµµДОьёЅРРОЄЈ¬іЈУГLangmuirєНFreundlichОьёЅµИОВДЈРНАґГиКц[19]ЎЈ
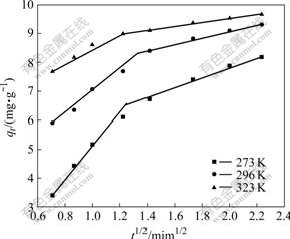
Нј5 ФЪІ»Н¬ОВ¶ИПВWeber-MorrisДЈРНДвєПЗъПЯ
Fig.5 Fitted curves of Weber-Morris at different temperatures
LangmuirµИОВКЅјЩЙиОьёЅКЗµҐ·ЦЧУІгОьёЅДЈРНЈ¬МеПаИЬТєєНОьёЅІгѕщїЙКУОЄАнПлИЬТєЈ¬ИЬЦКУлИЬјБ·ЦЧУМе»эПаµИ»тУРПаН¬µДОьёЅО»ЎЈ

КЅЦРЈєqmОЄ±ҐєНОьёЅБїЈ¬mg?g?1Ј»¦СeОЄMo(VI)ОьёЅЖЅєвК±µДЦКБїЕЁ¶ИЈ¬mg?L?1Ј»bОЄОьёЅЖЅєвіЈКэЈ¬L?mg?1ЎЈТФ¦Сe/qe¶Ф¦СeЧцЦ±ПЯїЙµГqmєНbЎЈ
FreundlichДЈРНКЗ±нХч¶аІгОьёЅµДѕСйКЅЈє

КЅЦРЈєKFОЄОьёЅИЭБїЈ¬mg?g?1Ј»nОЄОьёЅіЈКэЎЈ
2¶ФMo(VI)µДОьёЅКЗОьИИ№эіМЎЈ
2.3.2 ОьёЅ№эіМЦРµДИИБ¦С§ІОКэ
ДЙГЧTiO2¶ФMo(VI)µДОьёЅБїЛжОВ¶ИµДЙэёЯ¶шФцјУЈ¬ХвїЙНЁ№эОьёЅИИБ¦С§єЇКэ?G¦ЁЎў?H¦ЁЎў?S¦ЁµДјЖЛгЦµјУТФЅвКНЎЈ
±кЧјјЄІјЛ№ЧФУЙДЬ±дЎў±кЧј·ґУ¦мК±дЎў±кЧј·ґУ¦мШ±дУл№М?Тє·ЦЕдПµКэKDЦ®јдµД№ШПµОЄ

±н2 І»Н¬ОВ¶ИПВWeber-MorrisДЈРН¶а¶ОПЯРФДвєПЗъПЯПµКэј°Па№ШПµКэ
Table 2 Calculated parameters for Mo(VI) adsorption on nano-TiO2 at different temperatures

±н3 І»Н¬ОВ¶ИПВLangmuirєНFreundlichОьёЅµИОВПЯОьёЅіЈКэєНПЯРФПа№ШПµКэ
Table 3 Langmuir and Freundlich isotherm constants and correlation coefficients at different temperatures

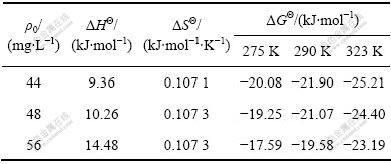
КЅЦРЈє¦С0ОЄMo(VI)ИЬТєіхКјЕЁ¶И(mg/L)Ј¬¦СtОЄtК±їМИЬТєЦРMo(VI)µДЕЁ¶И(mg/L)Ј¬VОЄИЬТєМе»э(mL)Ј¬mОЄДЙГЧ¶юСх»ЇоСµДјУИлБї(g)ЎЈУЙУЪКЗКµјКИЬТєЈ¬ТтґЛЈ¬ёщѕЭКЅ(13)єН(14)јЖЛгµГµЅµДКЗ±н№ЫјЄІјЛ№ЧФУЙДЬ±д(¦¤G¦Ё)Ј¬±н№ЫмК±д(¦¤H¦Ё)єН±н№ЫмШ±д(¦¤S¦Ё)ЎЈ
±н4ЛщБРОЄІ»Н¬іхКјЕЁ¶ИµДовОьёЅУЪДЙГЧTiO2µДИИБ¦С§±н№ЫІОКэЎЈУЙ±н4їЙЦЄЈ¬ДЙГЧTiO2ОьёЅMo(VI)µДОьёЅмК±дОЄХэЦµЈ¬±нГчёГОьёЅОЄмКНЖ¶ЇµДОьИИ№эіМЈ¬ХвУлОьёЅµИОВПЯµДЅб№ыТІКЗТ»ЦВµДЎЈОьёЅЧФУЙДЬОЄёєЦµЈ¬±нГчОьёЅ№эіМКЗЧФ·ўРРОЄЈ¬ЛжЧЕОВ¶ИЙэёЯЈ¬¦¤G¦ЁјхРЎЈ¬ЛµГчёЯОВУРАыУЪёГ·ґУ¦ЅшРРЎЈАлЧУФЪ№ММе±нГжµДОьёЅµјЦВЧФУЙ¶И±дРЎЈ¬µ«КЗКµСйЅб№ы¦¤S¦ЁЈѕ0Ј¬їЙДЬКЗТтОЄФЪИЬТєЦРЈ¬ОьёЅЦК(Изов)ІўІ»ТФјтµҐµДАлЧУРОКЅґжФЪЈ¬ЛьГЗѕіЈ»бУлЛ®ЅбєП(Л®»ЇЧчУГ»тЛ®ЅвЧчУГ) ЙъіЙЛ®єПАлЧУ»т·ЦЧУЈ¬µ±ОьёЅЦКµДЛ®єПАлЧУ»т·ЦЧУ±»ОьёЅК±Ј¬ЖдЅбєПЛ®їЙДЬ»бАлЅвіцАґЈ¬ЦШРВ»ШµЅИЬТєЦРЈ¬Хвёц№эіМ»бФцјУПµНіµДмШЦµЈ¬µјЦВ¦¤S¦ЁЈѕ0[20]ЎЈ
±н4 І»Н¬іхКјЕЁ¶ИµДовОьёЅУЪДЙГЧTiO2µДИИБ¦С§±н№ЫІОКэ
Table 4 Thermodynamic parameters for adsorption of Mo(VI) on nano-TiO2
2.3.3 ЖЅѕщОьёЅДЬ
ЖЅѕщОьёЅДЬEКЗЕР¶ПОьёЅАаРНµДЦШТЄЦё±кЈ¬ЖдјЖЛгКЅОЄ[21]

ёщѕЭD-RДЈРНµДПЯРФРОКЅ(КЅ16)Ј¬ТФlnqe¶Ф¦Е2ЧчЦ±ПЯ(јыНј6)Ј¬УЙЦ±ПЯР±ВКЗуµГKЈє

КЅЦРЈєEОЄЖЅѕщОьёЅДЬ(kJ/mol)Ј¬KОЄУлОьёЅДЬУР№ШµДіЈКэЈ¬¦ЕОЄPolanyiКЖДЬ(kJ)ЎЈ
ёщѕЭКЅ(15)Ј¬ФЪ275Ўў290єН323 KМхјюПВЈ¬ДЙГЧTiO2¶ФMo(VI)µДЖЅѕщОьёЅДЬ·Ц±рОЄ16.0Ўў17.4єН18.7 kJ/molЎЈEЈѕ16 kJ/molЈ¬№КёГОьёЅ·ґУ¦КфУЪ»ЇС§ОьёЅ[22]ЎЈ

Нј6 ФЪІ»Н¬ОВ¶ИПВD-RµДОьёЅµИОВПЯ
Fig.6 D-R adsorption isotherms at different temperatures
4 ЅбВЫ
1) ДЙГЧTiO2¶ФMo(VI)µДОьёЅФЪpHОЄ1~8·¶О§ДЪ,ОьёЅВКїЙґпµЅ99%Ј¬јоРФМхјюПВІ»АыУЪMo(VI)µДОьёЅЈ¬КµСй·ўПЦ2.0 mL 0.1 mol/L NaOHИЬТєїЙ¶ЁБїПґНСMo(VI)Ј¬ПґНСВКОЄ97%ЎЈ
2) ёГОьёЅ№эіМ·ыєПЧј¶юј¶·ґУ¦¶ЇБ¦С§ДЈРНЈ¬іЈОВПВЖд·ґУ¦ЛЩВКіЈКэОЄ0.296 g?mg?1?min?1Ј¬·ґУ¦µД»о»ЇДЬОЄ22.7 kJ/molЈ¬ОьёЅ»ъАн±ИЅПёґФУЈ¬БЈЧУДЪІїА©Йў№эіМКЗЖдОьёЅїШЦЖІЅЦиЈ¬µ«ТєПа±ЯЅзІгПтБЈЧУ±нГжµДА©Йў№эіМІ»ДЬєцВФЎЈ
3) ДЙГЧTiO2¶ФMo(VI)µДОьёЅ·ыєПLangmiurЎўD-RµИОВКЅЈ¬ФЪ275Ўў290єН323 KМхјюПВЈ¬Ж䱥єНОьёЅИЭБї·Ц±рОЄ11.51Ўў12.74єН14.19 mg/gЈ¬ЖЅѕщОьёЅДЬ·Ц±рОЄ16.0 Ўў17.4 єН18.7 kJ/molЈ¬±ҐєНОьёЅИЭБїЛжМеПµОВ¶ИЙэёЯ¶шФцґуЈ¬ЛµГчґЛОьёЅ·ґУ¦ОЄОьИИ№эіМЈ¬ЖЅѕщОьёЅДЬEЈѕ16 kJ/molЈ¬№КёГОьёЅ·ґУ¦КфУЪ»ЇС§ОьёЅ№эіМЎЈ
4) ДЙГЧTiO2¶ФMo(VI)µДОьёЅИИБ¦С§ІОКэ·Ц±рОЄЈє¦¤H¦ЁЈѕ0Ј¬¦¤G¦ЁЈј0Ј¬¦¤S¦ЁЈѕ0Ј¬±нГчОьёЅ·ґУ¦ОЄЧФ·ўµДОьИИ№эіМЎЈ
REFERENCES
[1] ХЕЧУСТ, јтґ»БЦ. ИЬјБЭНИЎ·ЁФЪОЩКЄ·ЁТ±ЅрЦРµДУ¦УГ[J]. КЄ·ЁТ±Ѕр, 2006, 25(1): 1?9.
ZHANG Zi-yan, JIAN Chun-lin. Application of solvent extraction in tungsten hydrometallurgy[J]. Hydrometallurgy of China, 2006, 25(1): 1?9.
[2] Нх»Эѕэ, РЬґє»Є, Т¦ІКЖј, №ЛХсУо. D201ЎБ4 КчЦ¬ОьёЅов(VI)µДРФДЬј°»ъАн[J]. УРЙ«ЅрКф, 2006, 58(4): 29?32.
WANG Hui-jun, XIONG Chun-hua, YAO Cai-ping, GU Zhen-yu. Adsorption performance and mechanism of Mo(VI) on D201 ЎБ4 resin[J]. Nonferrous Metals, 2006, 58(4): 29?32.
[3] ј§ МО, ЅрЖжНҐ, №щРВі¬, Лпі¤Лі. овЛбп§ЙъІъЛбПґ·ПЛ®µДЦОАн[J]. №¤ТµУГЛ®Ул·ПЛ®, 2007, 38(4): 12?14.
JI Tao, JIN Qi-ting, GUO Xin-chao, SUN Chang-shun. Treatment of acid-washing wastewater from ammonium molybdate production[J]. Industrial Water & Wastewater, 2007, 38(4): 12?14.
[4] НхПЧїЖ, АоУсЖј, АоАт·Т. ТєД¤·ЦАлё»јЇов[J]. ЦР№ъовТµ, 2002, 26(5): 34?36.
WANG Xian-ke, LI Yu-ping, LI Li-fen. Separation enrichment of molybdenum with liquid memberanes[J]. China Molybdenum Industry, 2002, 26(5): 34?36.
[5] Бє єк, В¬»щѕф. АлЧУЅ»»»·ЁґУє¬овЛбРФ·ПТєЦР»ШКХов[J]. ЦР№ъовТµ, 1999, 23(3): 43?45.
LIANG Hong, LU Ji-jue. Recovery of molybdenum from contains molybdenum acid waste liquors with ion exchanging method[J]. China Molybdenum Industry, 1999, 23(3): 43?44.
[6] іВЛЙМО, гЖУАК¤, РмНсХд, Бх »Є, ѕЈїЎЅЬ, Р»јЄГс. ДЙГЧTiO2Ф¤·ЦАл/ё»јЇFAAS·ЁН¬К±Ів¶ЁCr(III)єНCr(VI)µДСРѕї[J]. №вЖЧС§Ул№вЖЧ·ЦОц, 2007, 27(5): 1018?1020.
CHEN Song-tao, YAN Yong-sheng, XU Wan-zhen, LIU Hua, JING Jun-jie, XIE Ji-min. Combination of separation/ preconcentration based on nanoscale TiO2 and FAAS for the simultaneous determination of Cr(III)/Cr(VI) in water[J]. 2007, 27(5): 1018?1020.
[7] ХЕЦРМ«, БЦФЄ»Є, МЖЧУБъ, ХЕїЎУў. ДЙГЧІДБПј°ЖдјјКхµДУ¦УГЗ°ѕ°[J]. ІДБП№¤іМ, 2000, 3: 42?48.
ZHANG Zhong-tai, LIN Yuan-hua, TANG Zi-long, ZHANG Jun-ying. Nanometer materials & nanotechnology and their application prospect[J]. Materials Engineering, 2000, 3: 42?48.
[8] LI Shun-xing, DENG Nan-sheng, ZHENG Feng-ying, HUANG Yu-zhen. Spectrophotometric determination of tungsten(VI) enriched by nanometer-size titanium dioxide in water and sediment[J]. Talanta, 2003, 60(6): 1097?1104.
[9] АоґєПг, ЗШУАі¬, Бє Еж, єъ ±у, ЕнМмУТ, ЅЧжіЙ. ДЙГЧTiO2·ЦАлё»јЇєНICP-A ESІв¶ЁµШЦКСщЖ·ЦРµДовєНОЩ[J]. ·ЦОцїЖѧѧ±Ё, 2002, 18(3): 186?189.
LI Chun-xiang, QIN Yong-chao, LIANG Pei, HU Bin, PENG Tian-you, JIANG Zu-cheng. Nanometer-size TiO2 separation/ preconcentration and ICP-AES determination of trace Mo and W in geological samples[J]. Journal of Analytical Science, 2002, 18(3): 186?189.
[10] HADJIIVANOV K, KLISSURSKI D, KANTCHEVA M. State and focalization of cobalt, nickel and copper ions adsorbed on titania(anatase)[J]. Journal of the Chemical Society, Faraday Transactions, 1991, 87(6): 907?912.
[11] LIANG P, JIANG Z C, HU B, QIN Y C. Study on adsorption behaviour of rare earth complexes on namometer-size titanium dioxide with ICP-AES[J]. Journal of Rare Earths, 2003, 21(4): 474?477.
[12] ZHANG L, LIU N, YANG L J, LIN Q. Sorption behavior of nano-TiO2 for the removal of selenium ions from aqueous solution [J]. Journal of Hazardous Materials, 2009, 17(2/3): 1197?1203.
[13] єјТеЖј, ЗШУАі¬, ЅЧжіЙ, єъ ±у. ICP-AESСРѕїДЙГЧTiO2ІДБП¶ФGa, In, TlµДОьёЅРФДЬ[J]. №вЖЧС§Ул№вЖЧ·ЦОц, 2005, 25(7): 1131?1134.
HANG Yi-ping, QIN Yong-chao, JIANG Zu-cheng, HU Bin. Study on the adsorption behavior of Ga, In and Tl on nanometer-size titanium dioxide by ICP-AES[J]. Spectroscopy and Spectral Analysis, 2005, 25(7): 1131?1134.
[14] Рн№вГј, К© ЦЬ, µЛ ѕь. КЇУўЙ°ёєФШСх»ЇМъОьёЅіэБЧµДИИ¶ЇС§СРѕї[J]. »·ѕі№¤іМС§±Ё, 2007, 1(6): 15?18.
XU Guang-mei, SHI Zhou, DENG Jun. Thermodynamic and kinetic investigation of phosphate adsorption on iron-oxide coated sand [J]. Chinese Journal of Environmental Engineering, 2007, 1(6): 15?18.
[15] AKSU Z. Application of biosorption for the removal of organic pollutants: A review[J]. Process Biochemistry, 2005, 40(3/4): 997?1026.
[16] SHAUHA M S, SARWAR M I, QADEER R. Adsorption of strontium ions from aqueous solution on Pakistani coal[J]. Journal of Radioanalytical and Nuclear Chemistry, 2005, 265(1): 73?79.
[17] ?ZER A, AKKAYA G, TURABIK M. The biosorption of acid red 337 and acid blue 324 on enteromorpha prolifera: The application of nonlinear regression analysis to dye biosorption[J]. Chemical Engineering Journal, 2005, 112(1/3): 181?190.
[18] MALL I D, SRIVASTAVA V C, AGARWAL N K, MISHRA I M. Removal of Congo red from aqueous solution by bagasse fly ash and activated carbon: Kinetic study and equilibrium isotherm analyses[J]. Chemosphere, 2005, 61(4): 492?501.
[19] ЧЮОА»Є, іВЧЪи°, є«ИуЖЅ, Р» ЛЄ, КЇ ЅЬ. ГМСх»ЇОп/КЇУўЙ°(MOCS)¶ФНєНЗ¦АлЧУµДОьёЅСРѕї[J]. »·ѕіїЖѧѧ±Ё, 2005, 25(6): 779?784.
ZOU Wei-hua, CHEN Zong-zhang, HAN Run-ping, XIE Shuang, SHI Jie. Removal of copper cation and lead cation from aqueous solution by manganese-oxide-coated-sand [J]. Acta Scientiae Circumstantiae, 2005, 25(6): 779?784.
[20] ?ZER A, AKKAYA G, TURABIK M. Biosorption of acid blue (AB290) acid blue 324 (AB324) dyes on spirogyra rhizopus[J]. Journal of Hazardous Material, 2006, 135(1/3): 355?364.
[21] KILISLIOGLB A, BILGIN B. Thermodynamic and kinetic investigations of uranium adsorption on amberlite IR-118H resin[J]. Applied Radiation and Isotopes, 2003, 58(2): 155?160.
[22] EL-SHAHAWI M S, NASSIF H A. Retention and thermodynamic characteristics of mercury(II) complexes onto polyurethane foams[J]. Analytica Chimica Acta, 2003, 481(1): 29?39.
»щЅрПоДїЈєБЙДюКЎЅМУэМьїЖјјґґРВНЕ¶УЧКЦъПоДї(2007T053)Ј»БЙДюКЎЎ°°ЩЗ§НтИЛІЕ№¤іМЎ±ЧКЦъПоДї
КХёеИХЖЪЈє2008-12-03Ј»РЮ¶©ИХЖЪЈє2009-04-21
НЁРЕЧчХЯЈєХЕ АЩЈ¬ЅМКЪЈ¬І©КїЈ»µз»°Јє024-62207809Ј»E-mail: zhanglei63@126.com
(±ај АоПтИє)