Trans. Nonferrous Met. Soc. China 23(2013) 1815-1821
Kinetics of hydrothermal sulfidation of synthetic hemimorphite with elemental sulfur
Cun-xiong LI, Chang WEI, Zhi-gan DENG, Xing-bin LI, Min-ting LI, Gang FAN, Hong-sheng XU
College of Metallurgical and Energy Engineering, Kunming University of Science and Technology, Kunming 650093, China
Received 9 August 2012; accepted 24 December 2012
Abstract: The kinetics of hydrothermal sulfidation of synthetic hemimorphite with elemental sulfur was investigated. The effects of stirring speed (100-600 r/min), temperature (120-220 ��C), sulfur dosage (mole ratio of 0.6-1.6), and particle size (48-246 ��m) on the hemimorphite conversion rate were studied. The results show that the conversion rate of hemimorphite increases with increasing temperature and decreasing particle size. A stirring speed above 400 r/min and a sulfur dosage above 1 have very little effect on the conversion rate. The kinetics analyses of the experimental data under various conditions indicate that the sulfidation process is controlled by the chemical reaction at the early stage of process with activation energy of 50.23 kJ/mol, and then controlled by diffusion through the product layer with activation energy of 11.12 kJ/mol.
Key words: hydrothermal sulfidation; kinetics; synthetic hemimorphite; elemental sulfur
1 Introduction
At present, zinc is produced mostly from zinc sulphide ores because the sulphides are easy to separate from the gangue by conventional flotation techniques. In the case of flotation of oxide zinc ores, the process will be extremely complex because of the similarities in the physicochemical and surface chemistry of the constituent minerals. There are no known direct-acting collectors which can be capable of producing single metal concentrates [1-3]. Therefore, it is common practice to sulfurize oxidized zinc minerals prior to flotation to prepare their surfaces to receive the collectors generally adopted for concentrating sulfide.
There have been extensive investigations on the sulfidation of oxidized zinc ore and zinc ores containing oxidized constituents. The ore is subjected to a sulfidizing operation, such as a treatment with hydrogen sulfide gas or its equivalents [4] or sodium sulfide [5-11] or elemental sulfur[12]. Based on the reported studies, sodium sulfide is one of the most widely used sulfides in the sulfidization-flotation process. Nevertheless, the use of sodium sulfide as a sulfidizing agent is accompanied
by the emission of secondary wastes and the release of toxic gases, and its effectiveness is not entirely satisfactory, possibly due to the fact that the sulfidized layer detaches readily. Insufficient sulfide gives poor recovery because of inadequate sulfidization, while excess sulfide causes poor flotation due to the depressant action of sulfide ions. Hence, sulfur has been suggested as a substitute for sulfide, and the use of sulfur as sulfidation agent has received much attention [13-18].
Hemimorphite (Zn4Si2O7(OH)2��H2O), a zinc hydroxyl silicate hydrate, was chosen because it is one of the most commercially important minerals for the extraction of zinc. There are few reports on hydrothermal sulfidation of pure synthetic zinc silicate as the hemimorphite phase with elemental sulfur and the kinetics of this sulfidation process. In this work, as a fundamental study which has high significance for industrial production using sulfidation of low grade oxidized zinc-lead ore, the hydrothermal sulfidation behavior of zinc from synthetic hemimorphite was studied systematically. The sulfidation kinetics of synthetic hemimorphite was investigated and the reaction-controlled process was determined.
2 Experimental
2.1 Materials
The synthetic hemimorphite used in the present study was prepared in the laboratory. The XRD pattern of the prepared materials is shown in Fig. 1. The XRD analyses show that hemimorphite is the only chemical component in the synthetic hemimorphite.
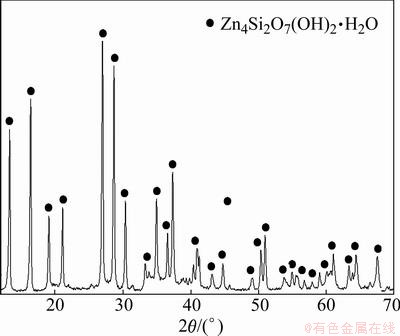
Fig. 1 XRD pattern of synthetic hemimorphite
For the hydrothermal sulfidation experiments, the synthetic hemimorphite was ground and sieved to different sizes, 147-246, 106-147, 74-106, 57-74 and 48-57 ��m. Elemental sulfur of analytical grade (99% S) was used as the sulfidizing agent. Synthetic hemimorphite and sulfur powder were mixed thoroughly at various mole ratios to decide the effect of sulfur dosage on conversion rate of hemimorphite.
2.2 Apparatus and procedure
The experimental apparatus used for hydrothermal sulfidation experiments consisted of a 2 L-autoclave which was equipped with a heating coil, a PID temperature controller, a variable speed stirrer and an internally mounted cooling coil.
150 g of mixture sample of synthetic hemimorphite and sulfur was dissolved in water, and then the pH of the solution was adjusted to 9.0��0.10 with dilute NaOH aqueous solution, finally the total volume was made up to 1500 mL. The pH of 9.0��0.10 was chosen with respect to formation of as much S2- as possible by disproportionation reaction of sulfur. Then, the slurry was poured into the autoclave and the sealed autoclave was heated to the set temperature under slow N2 flow. Once it reached the set temperature, the nitrogen flow was shut off and the stirrer was turned on. The experiment was run for a preset time. At the end of the predetermined time of reaction, the autoclave was rapidly water-cooled to about 30 ��C under nitrogen atmosphere, and then the slurry was filtered. The filtrate and filter cake obtained were used for further analysis.
The conversion rate (x) of synthetic hemimorphite was calculated by
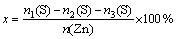 (1)
(1)
where n1(S) is the initial amount of elemental sulfur, n2(S) is the amount of sulfide ions consumed during blank experiment, n3(S) is the amount of residual sulfide ions and n(Zn) is the total amount of zinc involved in simulation.
The amount of the sulfide ions, n2(S), consumed by the conversion of S2- ions to S2O32- or SO42- in water (blank experiment) was determined as the difference between the initial amount of sulfide ions and the residual amount of sulfide ions after performing the blank experiment. In the present study, the blank experiments were exactly the same as in the case of experiments performed with synthetic hemimorphite.
3 Results and discussion
3.1 Effect of agitation
The influence of the stirring speed on synthetic hemimorphite conversion was studied at 100-600 r/min, and the results are shown in Fig. 2.

Fig. 2 Effect of agitation speed on conversion rate of hemimorphite
The results show that the distribution of S2- and suspension of particles at 400 r/min or above are adequate. To eliminate the stirring speed as a variable in the kinetics study and the negative effect of boundary layers, a stirring speed of 500 r/min was selected for the subsequent experiments.
3.2 Effect of temperature
Figure 3 shows effect of temperature on hydrothermal sulfidation of synthetic hemimorphite at 120�C220 ��C. The results indicate that the temperature has a pronounced effect on the conversion of hemimorphite. The hemimorphite conversion rate increases greatly with the temperature increasing in the range and about 71% hemimorphite was sulfidized at 220 ��C in 180 min.

Fig. 3 Effect of temperature on conversion rate of hemimorphite
3.3 Effect of sulfur dosage
A series of experiments were carried out with various mole ratios of sulfur to hemimorphite. The mole ratio ranged from 0.6 to 1.6. Figure 4 indicates that the sulfur dosage also has a great influence on hemimorphite conversion. By increasing the sulfur dosage from 0.6 to 1.2, the hemimorphite conversion rate increases from 54% to 73% in 180 min of hydrothermal sulfidation.
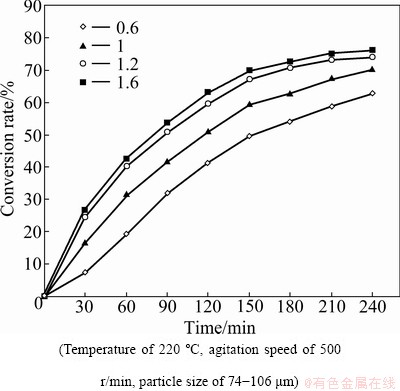
Fig. 4 Effect of sulfur dosage on conversion rate of hemimorphite
3.4 Particle size
The influence of particle size on synthetic hemimorphite conversion was studied using different particle sizes, 147-246, 106-147, 74-106, 57-74 and 48-57 ��m, and the results are shown in Fig. 5.

Fig. 5 Effect of particle size on conversion rate of hemimorphite
These results present that particle size has a significant effect on the conversion of hemimorphite and show that a decrease in particle size enhances the conversion rate. However, when the particle size is smaller than 74 ��m, it plays only a minor role in the sulfidation process. To investigate the other sulfidation parameters, a size range of 74-106 ��m was chosen.
3.5 Morphology of sulfidized product
The morphology of the hemimorphite particles before and after hydrothermal sulfidation was examined by SEM. The solid particles present a rough and porous surface, as observed in Fig. 6(a). The micrograph of the sulfidized product indicates a progressive increase in the roughness and porosity of the solid. Figure 6 also shows that the particle surface presents a reaction product layer with the progress of hydrothermal sulfidation. And the mineral surface is covered completely by porous product. Therefore, the hemimorphite sulfidation process is controlled both by interfacial transfer and diffusion of the reagent in the porous structure of the solid particles.
3.6 Kinetics analysis
The hydrothermal sulfidation reaction of hemimorphite with elemental sulfur is given as follows:
3Zn4Si2O7(OH)2��H2O(s)+16S(s)+10H2O(l)=12ZnS(s)+4SO42-(aq)+8H+(aq)+6H4SiO4(aq) (2)
Hydrothermal sulfidation of hemimorphite can proceed in a heterogeneous reaction manner, with a progressively thickening outer shell of insoluble product, while the inner core of unreacted particle decreases.

Fig. 6 SEM images of synthetic hemimorphite (a) and hydrothermal sulfidized product (b)
The shrinking core model (SCM) considers that the rate controlling step of reaction process may be controlled by chemical reaction on the particle surface, diffusion through the product layer and a combination of both.
3.6.1 Rate control by chemical reaction
Assuming that the hydrothermal sulfidation of hemimorphite is controlled by the chemical reaction, the following expression of shrinking core model can be used to describe the conversion kinetics of the process:
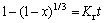 (3)
(3)
When the chemical reaction is the rate controlling step, a plot of 1-(1-x)1/3 vs time is a straight line with a slope Kr:
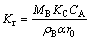 (4)
(4)
where KC is the chemical rate constant; MB is the relative molecular mass of the solid hemimorphite; CA is the concentration of liquid reactant; ��B the is density of hemimorphite;  is the stoichiometric coefficient (dimensionless); r0 is the radius of unreacted particle.
is the stoichiometric coefficient (dimensionless); r0 is the radius of unreacted particle.
A plot of 1-(1-x)1/3 against time, based on the experimental data in Fig. 3, is given in Fig. 7. The results indicate that the correlation coefficient (R2) for reaction control is above 0.97 and below about x=0.4 the data are linear with respect to the chemical model. From the slope of the curves in Fig. 7, the apparent kinetic constants for the various temperatures can be determined, which are used to draw an Arrhenius plot as shown in Fig. 8. The activation energy of the early stages of the hydrothermal sulfidation process is 50.23 kJ/mol.
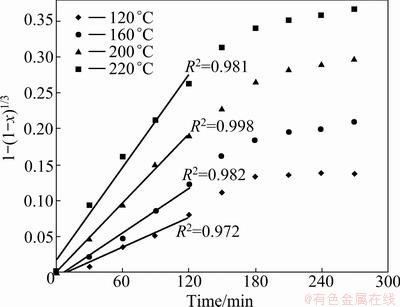
Fig. 7 Plot of 1-(1-x)1/3 against time at different temperatures
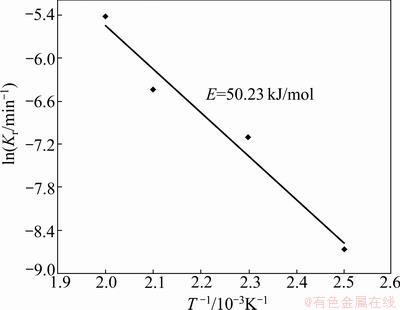
Fig. 8 Arrhenius plot at early stage of reaction
3.6.2 Rate control by diffusion through product layer
Similarly, when the diffusion of the reagent (sulphuric acid) through a product layer is the slowest step, the following expression of the shrinking core model can be used:
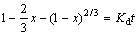 (5)
(5)
where Kd is the reaction rate constant.
If the hydrothermal sulfidation of hemimorphite is controlled by diffusion through a product layer, experimental data from Fig. 3 should yield a straight line when 1-(2/3)x-(1-x)2/3 is plotted against time. As indicated in Fig. 9, above x=0.4 the data give a straight line, assuming a diffusion controlled model. The slope of the straight lines is Kd,
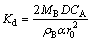 (6)
(6)
where D is the diffusion coefficient of zinc ions in porous medium.
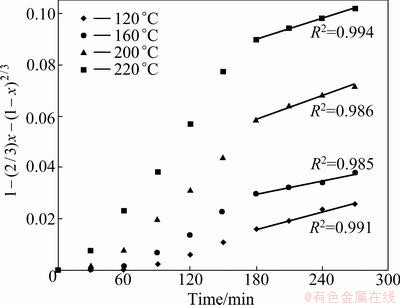
Fig. 9 Plot of 1-(2/3)x-(1-x)2/3 versus time at different temperatures
The slopes of the curves in Fig. 9 can be used to determine the apparent reaction rate constants at various temperatures at later stages. The Arrhenius plots are shown in Fig. 10. The calculated activation energy is 11.12 kJ/mol for the later stages.

Fig. 10 Arrhenius plot at later stage of reaction
3.6.3 Rate control by both diffusion and chemical reactions
No model gives a straight line relationship throughout the hydrothermal sulfidation process. Thus, the rate of reaction is controlled by the chemical reaction rate at the early stage and by diffusion at the later stage of the reaction. A mixed kinetics equation was used to describe the conversion kinetics of the process:

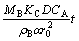 (7)
(7)
where
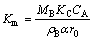 (8)
(8)
 (9)
(9)
where Km is the rate constant, B is a constant.
Thus, the following kinetic expression can be concluded:
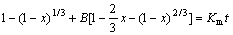 (10)
(10)
The two terms on the left side of Eq.(10) represent the chemical and diffusion resistances. To plot 1-(1-x)1/3+B[1-(2/3)x-(1-x)2/3] against time, the constant B must be determined, which contains the chemical rate constant KC, and diffusion coefficient D.
3.7 Determination of chemical rate constant KC and diffusion coefficient D
KC can be determined from the slope of the linear portion of the curves given in Fig. 7, from which KC can be obtained by calculation from known values of MB, CA, ��B,  and r0. The chemical rate constant KC is estimated to be about 4.95��10-7 m/s at 220 ��C.
and r0. The chemical rate constant KC is estimated to be about 4.95��10-7 m/s at 220 ��C.
Similarly, D can be determined from the slope of the linear portion of the curves given in Fig. 9. The slope is Kd, from which D can be calculated from known values of the other parameters. And the diffusion coefficient D is estimated to be about 2.33��10-12 m2/s at 220 ��C.
According to KC and D, the calculated value of constant B by Eq.(9) is 8.498. As a result, the following kinetic expression can be used to describe the hydrothermal sulfidation process of hemimorphite with elemental sulfur:
 (11)
(11)
A plot of the left side of Eq.(11) against time is given in Fig.11. Figure 11 shows the good fit (R2>0.98) obtained by plotting 1-(1-x)1/3+8.498[1-(2/3)x-(1-x)2/3] vs time at different temperatures.
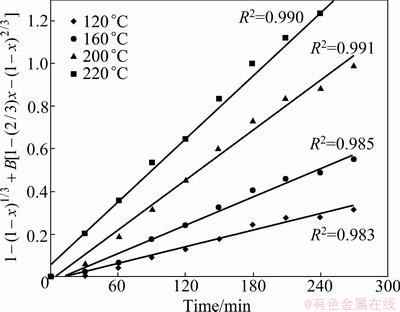
Fig. 11 Plots of 1-(1-x)1/3+B[1-(2/3)x -(1-x)2/3] vs time at different temperatures
The Arrhenius plot constructed with the rate constant value Km calculated from the data presented in Fig. 11 is shown in Fig. 12. The activation energy of the overall reaction given from Fig. 12g is 28.96 kJ/mol.

Fig. 12 Arrhenius plot of hemimorphite sulphidation reaction
4 Conclusions
1) A stirring speed greater than 400 r/min is sufficient to eliminate the effects of this variable on the reaction rate. The hemimorphite conversion rate increases significantly with increase in temperature or decrease in particle size.
2) The sulfidation process is controlled by two steps. At the initial stage of sulfidation, the process is controlled by chemical reaction but diffusion governs the later stages. The calculated activation energies are 50.23 kJ/mol and 11.12 kJ/mol, which are typical values for reaction-controlled and diffusion-controlled processes, respectively.
3) A mixed kinetics model equation which has features of both chemical reaction and diffusion through the product layer is used to well describe the conversion kinetics of hemimorphite under hydrothermal conditions. The activation energy of the overall reaction is 28.96 kJ/mol.
References
[1] REY M. The flotation of oxidized ores of lead copper and zinc [C]// IMM Recent Developments in Mineral Dressing Symposium. London: Technology Press, 1953: 541-548.
[2] BARBARO M. Lead and zinc ores-flotation [M]. Rome: Academic Press, 2000: 15-34.
[3] IRANNAJAD M, EJTEMAEI M, GHARABAGHI M. The effect of reagents on selective flotation of smithsonite-calcite-quartz [J]. Minerals Engineering, 2009, 22(9-10): 766-771.
[4] THOMPSON J W. Sulfidation and flotation of ores [M]. New York: Wiley, 1960: 133-156.
[5] LAPTEV Y V, SHEVCHENKO V S, URAKAEV F K. Sulphidation of valleriite in SO2 solutions [J]. Hydrometallurgy, 2009, 98(3-4): 201-205.
[6] CASTRO S, GOLDFARB J, LASKOWSKI J. Sulphidizing reactions in the flotation of oxidized copper minerals [J]. International Journal of Mineral Processing, 1974, 1(2): 141�C161.
[7] YAMADA M, SHOJI T, ONADA T, SHIMOIIZAKA J. Flotation of zinc carbonate [J]. Chemical Abstracts, 1974, 84(2): 182-194.
[8] CAPRONI G, CICCU R, GHIANI M, TRUDU I. The processing of oxidized lead and zinc ores in the Campo Pisano and San Giovanni Plants [J]. Minerals Engineering, 1979, 3(13): 71-91.
[9]  A, PEREK K T, ARSLAN F. Flotation of Aladag oxide lead�Czinc ores [J]. Minerals Engineering, 2005, 18(2): 279-282.
A, PEREK K T, ARSLAN F. Flotation of Aladag oxide lead�Czinc ores [J]. Minerals Engineering, 2005, 18(2): 279-282.
[10] PEREIRA C A, PERES A E C. Reagents in calamine zinc ores flotation [J]. Minerals Engineering, 2005, 18(2): 275-277.
[11] HOSSEINI S H, FORSSBERG E. Adsorption studies of smithsonite flotation using dodecylamine and oleic acid [J]. Minerals and Metallurgical Processing, 2006, 23(2): 87-95.
[12] KE-QING F A, MILLER J D, JIANG Tao, LI Guang-hui. Sulphidization flotation for recovery of lead and zinc from oxide�Csulfide ores [J]. Transactions of Nonferrous Metals Society of China, 2005, 15(5): 1138-1147.
[13] LI Yong, WANG Ji-kun, WEI Chang, LIU Chun-xia, JIANG Ji-bo, WANG Fan. Sulfidation roasting of low grade lead�Czinc oxide ore with elemental sulfur [J]. Minerals Engineering, 2010, 23(7): 563- 566.
[14] WANG J, LU J F, ZHANG Q W, SAITO F. Mechanochemical sulfidization of nonferrous metal oxides by grinding with sulfur and iron [J]. Industrial & Engineering Chemistry Research, 2003, 23(42): 1094-1095.
[15] PADILLA R,  M, RUIZ C. Sulfidation of chalcopyrite with elemental sulfur [J]. Metallurgy and Materials Transaction B, 2003, 34(1-2): 15-23.
M, RUIZ C. Sulfidation of chalcopyrite with elemental sulfur [J]. Metallurgy and Materials Transaction B, 2003, 34(1-2): 15-23.
[16] PADILLA R, OLIVARES E, RUIZ M C. Kinetic of the sulfidation of chalcopyrite with gaseous sulfur [J]. Metallurgy and Materials Transaction B, 2003, 34(1-2): 61�C68.
[17] PADILLA R, VEGA D, RUIZ M C. Pressure leaching of sulfidized chalcopyrite in sulfuric acid�Coxygen media [J]. Hydrometallurgy, 2007, 86(1-2): 80-88.
[18] LIANG Yan-jie, CHAI Li-yuan, LIU Hui, MIN Xiao-bo, Qaisar Mahmood, ZHANG Hai-jing, KE Yong. Hydrothermal sulfidation of zinc-containing neutralization sludge for zinc recovery and stabilization [J]. Minerals Engineering, 2012, 25(1): 14-19.
�ϳ��켫��Ԫ����ˮ������ѧ
����֣�κ �ƣ���־�ң����˱���F͢���� �գ����ʤ
����������ѧ ұ������Դ����ѧԺ������ 650093
ժ Ҫ���о���Ԫ����ˮ���ϳ��켫��Ķ���ѧ�о��������˽���ת��(100~600 r/min)���¶�(120~220 ��C)���������(Ħ����Ϊ0.6~1.6)�Ϳ�������(48~246 ��m)���켫��ת���ʵ�Ӱ�졣�������������ת���¶ȵ����ߺͿ������ȵļ�С���켫���ת���ʲ�����������ת�ٴ���400 r/min���������Ħ���ȴ���1ʱ������켫���ת����Ӱ�첻���ԡ��켫��Ԫ����ˮ����ת����Ӧ�����ܻ�ѧ��Ӧ���ƣ��ù��̵ı��ۻ��Ϊ50.23 kJ/mol��������ת��Ϊ�ܹ����������ɢ���ƣ�����ۻ��Ϊ11.12 kJ/mol��
�ؼ��ʣ�ˮ��������ѧ���ϳ��켫����
(Edited by Xiang-qun LI)
Foundation item: Project (50904030) supported by the National Natural Science Foundation of China
Corresponding author: Chang WEI; Tel/Fax: +86-871-65188819; E-mail: weichang502@sina.cn
DOI: 10.1016/S1003-6326(13)62665-5