
Synthesis and electrochemical properties of LiMn2O4/Li4Ti5O12 composite
HE Ze-qiang(ļő‘Ú«Ņ)1, 2, XIONG Li-zhi(–‹ņŻ÷•)1, 2,
WU Xian-ming(ő‚Ō‘√ų)1, CHEN Shang(≥¬ …Ō)1, HUANG Ke-long(Ľ∆Ņ…Ńķ)2
1. College of Biology and Environmental Sciences, Jishou University, Jishou 416000, China;
2. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China
Received 6 July 2009; accepted 30 December 2009
_____________________________________________________________________________________________________
Abstract: LiMn2O4/Li4Ti5O12 composite was synthesized by in-situ composite technique using LiMn2O4, lithium acetate, tetrabutyl titanate as starting materials and characterized by various electrochemical methods in combination with X-ray diffractometry (XRD), infrared (IR) spectroscopy and scanning electron microscopy (SEM). The results show that Li4Ti5O12 is coated on the surface of crystalline LiMn2O4 to form LiMn2O4/Li4Ti5O12 composite. The structure of LiMn2O4 does not change due to the introduction of Li4Ti5O12. By being coated with Li4Ti5O12, the rate capability and high temperature cyclability of LiMn2O4 is improved greatly. At room temperature, the discharge capacity of LiMn2O4/Li4Ti5O12 composite is more than 108.4 mA?h/g and the capacity loss per cycle is only 0.053% after 20 cycles at 2.0C.While at 55 °ś, the discharge capacity of LiMn2O4/Li4Ti5O12 composite is more than 109.9 mA?h/g and the capacity loss per cycle is only 0.036% after 60 cycles at 1.0C.
Key words: Li4Ti5O12; LiMn2O4; in-situ composite technique; lithium ion batteries
_____________________________________________________________________________________________________
1 Introduction
LiMn2O4 is of great interest for the replacement of LiCoO2 in Li ion batteries due to its low cost, nontoxicity and environmentally-friendly properties[1-2]. However, there are still some problems in terms of the wider application of LiMn2O4. The main problem is the capacity loss upon cycling at elevated temperature above 50 °ś[3-6]. Recently, this problem has partially been resolved by metal cation doping[7-8] and surface modification treatment[9-10].
Besides the long cycling life and safety, the high rate performance is also important for commercial batteries. In spite of the success in improving cycle stability, little has been reported on the enhanced rate capability of LiMn2O4. In particular, it is necessary to improve the rate capability of LiMn2O4 in order to be used as a cathode material of Li ion batteries for electric vehicles. The reason for poor rate capability of LiMn2O4 is not clear but it may be attributed to its low chemical diffusion coefficient (10-9-10-12 cm2/s)[11-12]. The low diffusion coefficient of LiMn2O4 can limit the current flow between particles, which may decrease its rate capability. Therefore, the increase in diffusion coefficient of LiMn2O4 probably improves its rate property.
LiMn2O4 was surface modified by nano-Li4Ti5O12 particles to improve the high rate capability, in consideration of the following factors: 1) the chemical diffusion coefficient of Li4Ti5O12 (10-6 cm2/s)[13] being larger than that of LiMn2O4; 2) both Li4Ti5O12 and LiMn2O4 sharing the same spinel structure and space group of Fd3m[14-15]; 3) Li4Ti5O12 showing exceptional rate capacity retention even as high as 10C[16]; 4) Li4Ti5O12 being a zero-strain material, which means that there is no structural change during the insertion/extraction of lithium ions[15].
In the present work, LiMn2O4/Li4Ti5O12 composite was synthesized by in-situ composite technique and its electrochemical properties were investigated in detail.
2 Experimental
2.1 Preparation of LiMn2O4/Li4Ti5O12 composite
A certain quantity of tetrabutyl titanate was solved in ethanol absolute with the volume ratio of tetrabutyl titanate) to ethanol absolute of 1?5. The solution of tetrabutyl titanate was added to an aqueous ethanol solution of lithium acetate by stirring strongly according to the stoichiometric ratio of Li4Ti5O12 to get a clear yellow sol. A certain quantity of LiMn2O4 (the mass ratio of Li4Ti5O12 to LiMn2O4 is 1?19) used in lithium ion batteries was dispersed uniformly in this sol with ultrasonic wave and stirrers. After stirring for 2-3 h, a black gel was obtained. The gel was dried at 105 °ś for 4 h in vacuum to get a black precursor. The precursor was calcinated at 800 °ś in Ar atmosphere for 6 h to obtain LiMn2O4/Li4Ti5O12 composite.
2.2 Characterization of LiMn2O4/Li4Ti5O12 composite
Phase identification studies of the samples were carried out by an X-ray diffractometer (Rigaku D/MAX-gA) with Cu K¶Ń radiation. The surface morphology was done by scanning electron microscope (JSM 5600LV). The IR spectra of samples were conducted on a Fourier transform infrared spectrometer (Nicolet).
2.3 Electrochemical tests of LiMn2O4/Li4Ti5O12 composite
A slurry containing 80% LiMn2O4/Li4Ti5O12 composite, 10% acetylene black and 10% PVDF (polyvinylidene fluoride) was made using N-methylprrolidinone (NMP) as the solvent. Electrodes with an area of 1 cm2 for the measurements of electrochemical characterization were prepared by coating slurries (about 100 ¶Őm in thickness) on aluminum foils followed by drying in vacuum at 60 °ś for 12 h. Electrochemical tests were conducted using a conventional coin-type cell, employing lithium foil as a counter electrode and utilizing 1.0 mol/L LiPF6 in ethylene carbonate/dimethyl carbonate (EC/DMC) (with an volume ratio of 1?1 of EC to DMC) as the electrolyte. The assembly was carried out in an Ar-filled glove box. The electrochemical analyses were carried out with an electrochemical analysis system.
3 Results and discussion
3.1 XRD analysis
Fig.1 shows the X-ray diffraction patterns of LiMn2O4 and LiMn2O4/Li4Ti5O12. As can be seen, all those materials display nearly the same XRD patterns without any peak shift, which indicates that there is no structural change after the Li4Ti5O12 surface modification treatment. In the modified samples, no impurity phase is detected, suggesting that Li4Ti5O12 just exists on the surface of LiMn2O4 and is not incorporated into the host structure since no changes are detected in the XRD patterns.
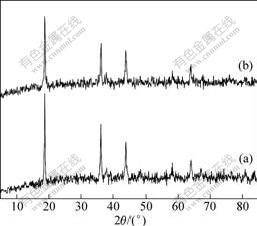
Fig.1 XRD patterns of LiMn2O4 (a) and LiMn2O4/Li4Ti5O12 (b)
3.2 IR analysis
Fig.2 shows the IR spectra of LiMn2O4 and LiMn2O4/Li4Ti5O12. It is obvious that the spectra of LiMn2O4 and LiMn2O4/Li4Ti5O12 agrees well, suggesting that Li4Ti5O12 just exists on the surface of LiMn2O4.The peaks at 3 200-3 600 cm-1 and 1 600 cm-1 are due to the absorbed water on the surface of samples. The peaks at 2 300-2 500 cm-1 is caused by CO2 in air. The peaks between 400 and 1 200 cm-1 are attributed to the metal-oxygen bond in samples.

Fig.2 IR spectra of LiMn2O4 (a) and LiMn2O4/Li4Ti5O12 (b)
3.3 SEM analysis
The SEM images of LiMn2O4 and LiMn2O4/ Li4Ti5O12 are shown in Fig.3. It can be seen clearly that the surface of the bare LiMn2O4 is smooth, but after surface modification treatment, a lot of nanoparticles are observed on the surface of the core materials. Therefore, it is speculated that Li4Ti5O12 particles coated on LiMn2O4 and Li4Ti5O12 coating would minimize the direct contact area between the cathode and the electrolyte.

Fig.3 SEM images of LiMn2O4 (a) and LiMn2O4/Li4Ti5O12 (b)
3.4 Electrochemical properties
Fig.4 shows the initial discharge curves of the uncoated LiMn2O4 and LiMn2O4/LiMn2O4/Li4Ti5O12 electrode at 1C between 3.4 and 4.3 V. It is evident that all these electrodes display the characteristic two 4 V plateaus, and similar results are also observed under 2C and 5C, which means that Li4Ti5O12 coating does not change the intrinsic properties of LiMn2O4, such as charge/discharge behavior and crystal structure. In addition, the first-cycle discharge capacity of the LiMn2O4 is 122.2 mA?h/g, while that of the LiMn2O4/Li4Ti5O12 is 119.3 mA?h/g. The capacity decreases with the increase of Li4Ti5O12 coating amount, which can be attributed to the less active spinel in the electrode.
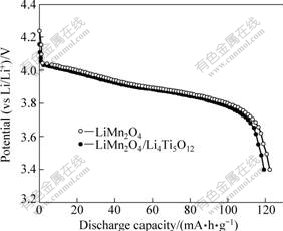
Fig.4 Initial discharge capacities of LiMn2O4 and LiMn2O4/Li4Ti5O12 at 0.1C
Fig.5 compares the cycling performance of LiMn2O4 and LiMn2O4/Li4Ti5O12 at 0.1C. After 100 cycles at 0.1C, the discharge capacity of LiMn2O4 decreases from 122.2 mA?h/g to 118.3 mA?h/g and the capacity loss per cycle is 0.032%. Under the same conditions, the discharge capacity of LiMn2O4/Li4Ti5O12 decreases from 119.3 mA?h/g to 117.1 mA?h/g and the capacity loss per cycle is 0.018%, suggesting that the loading of Li4Ti5O12 on LiMn2O4 helps to improve the cycling stability of LiMn2O4.

Fig.5 Cycling performance of LiMn2O4 and LiMn2O4/Li4Ti5O12 at 0.1C
Table 1 presents the comparison of cycling performance of LiMn2O4 and LiMn2O4/Li4Ti5O12 at various current rates. With the increase of discharge current rate, the discharge capacities of both samples decrease. This may be due to the increase of the electrode polarization at higher discharge current rate. In the point of capacity loss after 20 cycles, it can be seen obviously that the discharge capacity loss of LiMn2O4/Li4Ti5O12 is lower than that of LiMn2O4. After 20 cycles at 2.0C, the discharge capacity of LiMn2O4 is 108.3 mA?h/g and the capacity loss per cycle is 0.108%. Under the same conditions, the discharge capacity of LiMn2O4/Li4Ti5O12 keeps at 108.4 mA?h/g and the capacity loss per cycle is more than 0.053%, suggesting that the loading of Li4Ti5O12 on LiMn2O4 improves the rate stability of LiMn2O4. This may be explained that Li4Ti5O12 coating minimizes the direct contact area between the cathode and the electrolyte[17] and increases the chemical diffusion coefficient of lithium ion, accordingly improves the high rate performance of LiMn2O4.
Table 1 Comparison of cycling performance for LiMn2O4 and LiMn2O4/Li4Ti5O12 composite at various current rates

Fig.6 shows the high temperature cycling performance of LiMn2O4 and LiMn2O4/Li4Ti5O12 at 55 °ś and 1.0C. The discharge capacities of LiMn2O4 and LiMn2O4/Li4Ti5O12 are 113.1 mA?h/g and 112.3 mA?h/g at 55 °ś, respectively, lower than their discharge capacities at room temperature (as shown in Table 1).
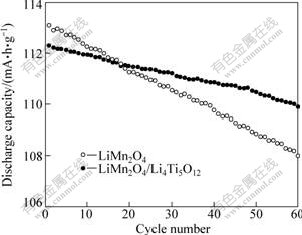
Fig.6 High temperature cycling performance of LiMn2O4 and LiMn2O4/Li4Ti5O12 at 1.0C
After 20 cycles at 55 °ś, the discharge capacities of LiMn2O4 and LiMn2O4/Li4Ti5O12 are 111.3 mA?h/g and 111.5 mA?h/g, and the capacity losses are 0.080% and 0.036% per cycle, respectively. After 60 cycles at 55 °ś, the discharge capacities of LiMn2O4 and LiMn2O4/Li4Ti5O12 are 108.0 mA?h/g and 109.9 mA?h/g, and the capacity losses are 0.075% and 0.036% per cycle, respectively. The high temperature cycling performance of LiMn2O4 is improved significantly by coating Li4Ti5O12. Surface modification of Li4Ti5O12 can obstruct the direct contact and suppress the side reactions between the electrode and electrolyte to prevent the spinel LiMn2O4 from dissolving into the electrolyte, resulting in improved high temperature cycling performance of LiMn2O4[8, 18-20].
4 Conclusions
1) LiMn2O4/Li4Ti5O12 composite was prepared by in-situ composite technique using LiMn2O4, lithium acetate, tetrabutyl titanate as starting materials. Li4Ti5O12 just exists on the surface of LiMn2O4 and is not incorporated into the host structure of LiMn2O4.
2) By coating Li4Ti5O12 on the surface of LiMn2O4, the rate capability and high temperature cyclability of LiMn2O4 are improved greatly. This is because that the surface modification of Li4Ti5O12 can minimize the direct contact area between the cathode and the electrolyte, increase the chemical diffusion coefficient of lithium ion and suppress the side reactions between the electrode and electrolyte to prevent the spinel LiMn2O4 from dissolving into the electrolyte.
References
[1] THACKERAY M M, DAVID W I F, BRUCE P G, GOODENOUGH J B. Lithium insertion into manganese spinels [J]. Mater Res Bull, 1983, 18(4): 461-472.
[2] GUYOMARD D, TARASCON J M. Li metal-free rechargeable LiMn2O4/carbon cells: Their understanding and optimization [J]. J Electrochem Soc, 1992, 139(4): 937-948.
[3] XIA Y, YOSHIO M. Studies on Li-Mn-O spinel system (obtained from melt-impregnation method) as a cathode for 4 V lithium batteries (Part IV): High and low temperature performance of LiMn2O4 [J]. J Power Sources, 1997, 66(1/2): 129-133.
[4] WU S H, SU H J. Electrochemical characteristics of partially cobalt-substituted LiMn2-yCoyO4 spinels synthesized by Pechini process [J]. Mater Chem Phys, 2002, 78(1): 189-195.
[5] AMATUCCI G G, BLYR A, SIGALA C, ALFONSE P, TARASCON J M. Surface treatments of Li1+xMn2-xO4 spinels for improved elevated temperature performance [J]. Solid State Ionics, 1997, 104(1/2): 13-25.
[6] ANTONINI A, BELLITTO C, PASQUALI M, PISTOIA G. Factors affecting the stabilization of Mn spinel capacity upon staring and cycling at high temperatures [J] J Electrochem Soc, 1998, 145(8): 2726-2732.
[7] EIN-ELI Y, HOWARD W F Jr, LU S H, MUKERJEE S, MCBREEN J, VAUGHEY J T, THACKERAY M M. LiMn2-xCuxO4 spinels (0.1£ľx£ľ0.5): A new class of 5 V cathode materials for Li batteries [J]. J Electrochem Soc, 1998, 145(4): 1238-1244.
[8] DOKKO K, HORIKOSHI S, ITOH T, NISHIZAWA M, MOHAMEDI M, UCHIDA I. Microvoltammetry for cathode materials at elevated temperatures: electrochemical stability of single particles [J]. J Power Sources, 2000, 90(1): 109-115.
[9] GNANARAJ J S, POL V G, GEDANKEN A, AURBACH D. Improving the high-temperature performance of LiMn2O4 spinel electrodes by coating the active mass with MgO via a sonochemical method [J]. Electrochem Comm, 2003, 5(11): 940-945.
[10] LEE S W, KIM K S, MOON H S, KIM H J, CHO B W, CHO W I, JU J B, PARK J W. Electrochemical characteristics of Al2O3-coated lithium manganese spinel as a cathode material for a lithium secondary battery [J]. J Power Sources, 2004, 126(1/2): 150-155.
[11] DEISS E. Spurious chemical diffusion coefficients of Li+ in electrode materials evaluated with GITT [J]. Electrochimica Acta, 2005, 50(14): 2927-2932.
[12] SANTIAGO E I, AMANCIO-FILHO S T, BUENO P R, BULH?ES L O S. Electrochemical performance of cathodes based on LiMn2O4 spinel obtained by combustion synthesis [J]. J Power Sources, 2001, 97/98: 447-449.
[13] TAKAI S, KAMATA M, FUJINE S, YONEDA K, KANDA K, ESAKA T. Diffusion coefficient measurement of lithium ion in sintered Li1.33Ti1.67O4 by means of neutron radiography [J]. Solid State Ionics, 1999, 123(1/4): 165-172.
[14] MIURA K, YAMADA A, TANAKA M. Electric states of spinel LixMn2O4 as a cathode of the rechargeable battery [J]. Electrochimica Acta, 1996, 41(2): 249-256.
[15] OHZUKU T, UEDA A, YAMAMOTO N. Zero-strain insertion material of Li[Li1/3Ti5/3]O4 for rechargeable lithium cells [J]. J Electorchem Soc, 1995, 142(5): 1431-1435.
[16] SINGHAL A, SKANDAN G, AMATUCCI G, BADWAY F, YE N, MANTHIRAM A, YE H, XU J J. Nanostructured electrodes for next generation rechargeable electrochemical devices [J]. J Power Sources, 2004, 129(1): 38-44.
[17] LIU Dong-qiang, YU Ji, SUN Yu-heng, HE Ze-zhen, LIU Xing-quan. Preparation and rate property of Li4Ti5O12-coated LiMn2O4 for lithium ion battery [J]. Chinese Journal of Inorganic Chemistry, 2007, 23(1): 41-45.
[18] AMATUCCI G G, SCHMUTZ C N, BLYR A, SIGALA C, GOZDZ A S, LARCHER D, TARASCON J M. Materials°Į effects on the elevated and room temperature performance of C/LiMn2O4 Li-ion batteries [J]. J Power Sources, 1997, 69(1/2): 11-25.
[19] PASQUIER A D, BLYR A, COURJAL P, LARCHER D, AMATUCCI G, GERAND B AND TARASCON J M. Mechanism for limited 55 °ś storage performance of Li1.05Mn1.95O4 electrodes [J]. J Electrochem Soc, 1999, 146(2): 428-436.
[20] HUANG H, VINCENT C A, BRUCE P G. Capacity loss of lithium manganese oxide spinel in LiPF6/ethylene carbonate-dimethyl carbonate electrolytes [J]. J Electrochem Soc, 1999, 146(2): 481-485.
________________________
Foundation item: Project(20376086) supported by the National Natural Science Foundation of China; Project(2005037700) supported by Postdoctoral Science Foundation of China; Project(07JJ3014) supported by Hunan Provincial Natural Science Foundation of China; Project(07A058) supported by Scientific Research Fund of Hunan Provincial Education Department; Project(2004107) supported by Postdoctoral Science Foundation of Central South University
Corresponding author: HE Ze-qiang; Tel: +86-743-8564416; Fax: +86-743-8564416; E-mail: csuhzq@163.com
(Edited by CHEN Ai-hua)