Anisotropic surface broken bond properties and wettability of calcite and fluorite crystals
来源期刊:中国有色金属学报(英文版)2012年第5期
论文作者:高志勇 孙伟 胡岳华 刘晓文
文章页码:1203 - 1208
关键词:方解石;萤石;表面断裂键;解理;表面能;接触角;润湿性
Key words:calcite; fluorite; surface broken bonds; cleavage; surface energy; contact angle; wettability
摘 要:计算方解石6个晶面和萤石3个晶面的表面断裂键密度,借助各向异性的表面断裂键密度预测两种含钙矿物的常见暴露面,分析表面断裂键密度与表面能之间的关系,利用接触角实验研究两种矿物常见暴露面的润湿性。表面断裂键密度计算分析表明,(1014)、(2134)和(0118)面及 (111) 面分别是方解石和萤石最常见的暴露面;两种矿物表面断裂键密度与表面能之间存在线性关系,其相关系数R2分别为0.9613和0.9969。接触角测量实验表明,两种矿物常见暴露面和水作用后的润湿性与表面断裂键密度有关,表面断裂键密度越大,亲水性越强,接触角越小;和油酸钠作用后的润湿性与表面Ca活性质点密度及质点的空间分布有关。
Abstract:
Anisotropic surface broken bond densities of six different surfaces of calcite and three surfaces of fluorite were calculated. In terms of the calculated results, the commonly exposed surfaces of the two minerals were predicted and the relations between surface broken bonds densities and surface energies were analyzed. Then the anisotropic wettability of the commonly exposed surfaces was studied by means of contact angle measurement. The calculation results show that the (1014),(2134)and (0118) surfaces for calcite and (111) for fluorite are the most commonly exposed surfaces and there is a good rectilinear relation between surface broken bond density and surface energy with correlation of determination (R2) of 0.9613 and 0.9969, respectively. The anisotropic wettability of different surfaces after immersing in distilled water and sodium oleate solutions at different concentrations can be explained by anisotropic surface broken bond densities and active Ca sites densities, respectively.

![]()

Trans. Nonferrous Met. Soc. China 22(2012) 1203-1208
GAO Zhi-yong, SUN Wei, HU Yue-hua, LIU Xiao-wen
School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
Received 5 May 2011; accepted 5 September 2011
Abstract: Anisotropic surface broken bond densities of six different surfaces of calcite and three surfaces of fluorite were calculated. In terms of the calculated results, the commonly exposed surfaces of the two minerals were predicted and the relations between surface broken bonds densities and surface energies were analyzed. Then the anisotropic wettability of the commonly exposed surfaces was studied by means of contact angle measurement. The calculation results show that the![]() ,
, ![]() and
and ![]() surfaces for calcite and (111) for fluorite are the most commonly exposed surfaces and there is a good rectilinear relation between surface broken bond density and surface energy with correlation of determination (R2) of 0.9613 and 0.9969, respectively. The anisotropic wettability of different surfaces after immersing in distilled water and sodium oleate solutions at different concentrations can be explained by anisotropic surface broken bond densities and active Ca sites densities, respectively.
surfaces for calcite and (111) for fluorite are the most commonly exposed surfaces and there is a good rectilinear relation between surface broken bond density and surface energy with correlation of determination (R2) of 0.9613 and 0.9969, respectively. The anisotropic wettability of different surfaces after immersing in distilled water and sodium oleate solutions at different concentrations can be explained by anisotropic surface broken bond densities and active Ca sites densities, respectively.
Key words: calcite; fluorite; surface broken bonds; cleavage; surface energy; contact angle; wettability
1 Introduction
Anisotropic surface chemistry of solids has been actively studied in many research fields including bionic engineering [1], micro- or nano-scale surface materials [2-3] and pharmaceuticals in recent years [4]. The research results of the anisotropic wettability or wetting phenomenon of surfaces with different micro- and nano-structures provided theoretical evidence for controlling the anisotropic wettability and the design of materials and devices with anisotropic wetting surface in microfluidics, micro- or nano-optics, antifouling, and so forth.
Flotation is a surface-chemistry based process for the separation of fine solids that takes advantage of the differences in wettability at the solid particle surfaces. The flotation separation of calcium minerals from each other is an important area of separation science in the field of mineral processing. Because most of calcium minerals, such as fluorite (CaF2), calcite (CaCO3), and scheelite (CaWO4), occur in nature in association with each other and possess very similar surface properties and semisoluble nature [5], the separation of them from each other is still a worldwide challenging problem to date. Calcite and fluorite are two of the most common calcium minerals and they show a remarkable variety of forms [6], particularly calcite crystals, and hence several anisotropic exposed surfaces with different physicochemical characteristics. Earlier studies on the adsorption of molecular waters on different calcite surfaces employing atomistic simulation technique by LEEUW and PARKER [7] indicated that these hydrated surface structures and hydration energies exhibited anisotropy. It was also reported by PRADIP et al [8] that the interactions of alkyl hydroxamates with three different fluorite surfaces are anisotropic. However, few experimental results have been reported on the anisotropy of surface properties of calcium minerals to date. Since the challenge for flotation separation is that the separation has to be based on subtle differences on the anisotropic exposed surfaces amongst various minerals, the study of anisotropic surface chemistry through both molecular modeling computations and experimental tests may be of great importance in the flotation separation of Ca-bearing minerals.
The anisotropic densities of surface broken bonds were calculated to predict the most commonly exposed surfaces of the morphologies of calcite and fluorite crystals in the current work. These commonly exposed surfaces were carefully selected or cleaved from pure crystal samples to study the anisotropic wetting behavior of water or sodium oleate at them through contact angle experiments. This work has two aims: 1) To establish the validity and suitability of the calculation of surface broken bonds densities for predicting the most commonly exposed surfaces of the calcite and fluorite crystals; 2) To study the anisotropic wetting behavior of water and oleate on different calcite and fluorite surfaces and establish the link between anisotropic wetting behavior and surface chemistry.
2 Experimental
2.1 Calculation method
The unit cells of calcite and fluorite were built employing the software of Material Studio, and the lattice parameters required for this work can be found in Ref. [9]. Then, a range of surfaces were created and the density of interplanar bonds broken for the creation of a certain surface was calculated according to the following formula:
Db=Nb/S (1)
where Db and Nb represent the surface broken bond density (number of broken bonds per surface unit area) and broken bonds number per surface unit cell on a certain surface, respectively; S is the unit area of the surface. It should be pointed out that the carbonate groups act as poly-anions, and these groups are kept intact when the calcite surfaces are created.
2.2 Pure crystal samples
Pure calcite and fluorite crystals were purchased from Changsha Mineral and Gem Market and used for contact angle measurements. The commonly exposed cleavage and crystal surfaces were carefully selected or cleaved to avoid cracks and inclusions. And then the selected crystals were embedded in resin and exposed the desired surfaces for further grinding and polishing. The crystallographic orientations of calcite and fluorite crystals were confirmed by single crystal X-ray diffraction analysis, polarizing microscope or/and crystal habit.
2.3 Reagents
Analytical grade sodium oleate was purchased from Tianjin Guangfu Fine Chemical Research Institute and used for contact angle measurements. For pH adjustment, dilute solutions of HCl and NaOH were employed. High purity water produced by atomic adsorption-type ultrapure water systems (manufactured by Lead R&D) was used for all experiments. The resistivity of water was more than 18 MΩ・cm.
2.4 Grinding and polishing materials
The metallographic specimen polishing machine PG-1A, from Shanghai Metallurgical Equipment Company Ltd., was used for grinding and polishing of the calcite and fluorite crystals. The grinding materials (diamond grinding disks of roughness 90, 38, 18, 6.5 and 2.5 μm), polishing materials (alumina powders of 1.0, 0.5, and 0.05 μm and Selected Silk cloth) were obtained from Xinhui (Changsha) Technology Co., Ltd.
2.5 Contact angle measurements
Wettability characteristics of minerals have a substantial influence on the efficiency of processing methods such as agglomeration, aggregation and froth flotation [10]. The contact angle is a very common measure of the wettability of a solid surface. Contact angle measurement was done with Digidrop goniometer from GBX (France) using sessile-drop technique at room temperature of 20℃, with an accuracy of ±1.5°. The desired exposed surface was ground by diamond grinding disks of roughness 90, 38, 18, 6.5 and 2.5 μm in series to obtain a flat surface. After grinding, the surface was successively polished with 1.0, 0.5, and 0.05 μm alumina powder solution on a “selected Silk” polishing cloth.
For the study of wetting by water, a prepared sample surface was immersed in water or surfactant solution under agitation for 20 min. Next, the sample was gently washed with distilled water and vacuum dried at 50 ℃ for 10 min. A water drop of about 3.5 μL was placed on the sample surface, and the readings of contact angles were taken automatically on the left and right sides of the water droplet profile by computer software.
The sample surface was large enough for 4-6 contact angle measurements. After the measurement, the sample surface was cleaned with water, acetone, and water again, and then was polished again using 0.05 μm alumina powder to restore a fresh surface before each experiment.
3 Results and discussion
3.1 Crystal structures of calcite and fluorite
Calcite has a trigonal crystal structure with space group ![]() with ah=0.4988 nm, ch=1.7061 nm, α=β=90° and γ=120°. The calcium ions are sixfold coordinated to one oxygen each of six surrounding carbonate groups at Ca―O distance of 0.236 nm. The carbon atoms are threefold coordinated to three oxygen atoms at C―O distance of 0.128 nm. It is noteworthy that
with ah=0.4988 nm, ch=1.7061 nm, α=β=90° and γ=120°. The calcium ions are sixfold coordinated to one oxygen each of six surrounding carbonate groups at Ca―O distance of 0.236 nm. The carbon atoms are threefold coordinated to three oxygen atoms at C―O distance of 0.128 nm. It is noteworthy that ![]() ,
,![]() and
and ![]() surfaces of hexagonal unit cell of calcite are the same as
surfaces of hexagonal unit cell of calcite are the same as![]() ,
, ![]() and
and ![]() surfaces of obtuse rhombohedron unit cell which was used traditionally and habitually in mineralogy.
surfaces of obtuse rhombohedron unit cell which was used traditionally and habitually in mineralogy.
Fluorite has a cubic crystal structure with space group Fm3m with a=b=c=0.5463 nm and α=β=γ=90°. The calcium ions are eightfold coordinated to eight surrounding fluorine ions at Ca―F distance of 0.237 nm. The fluorine ions are tetrahedrally coordinated to calcium ions.
3.2 Relations between surface broken bond density and cleavage property of calcite and fluorite crystals
It is well established that mineral crystal has the tendency to split along definite crystallographic structural surfaces in the presence of external stress. The property of split of mineral crystal to smooth surfaces is called cleavage and the smooth surface is known as cleavage plane which is generally in parallel to surfaces with a large interlayer spacing or/and stoichiometry and charge neutrality and is generated along the direction of the weakest strength of chemical bonds between two layers [6]. COOPER and LEEUW [11] held that when the mineral particles are crushed in the first stages of flotation process, the minerals will mainly cleave along surfaces that have large interplanar spacings and few interplanar bonds. That is, as for a certain crystal surface, the higher the density of surface broken bonds is, the greater the bonding strength with adjacent surface is, and accordingly the more difficult it is for cleavage to generate between these surface layers, and vice versa.
From Table 1, for calcite, the density of surface broken bonds follows the order: ![]() >
>![]() >
> ![]() >
>![]() >
>![]() >
>![]() . The
. The ![]() surface with stoichiometry and charge neutrality has the smallest surface broken bonds density and the largest interplanar spacing of the six calculated planes, and this surface can be created readily in the presence of external stress and is the perfect cleavage plane of calcite. For fluorite, the order of broken bond density of three surfaces is as follows: (100)>(110)>(111). The {111} surface, consisting of alternate F―Ca―F ionic layer within which the chemical bonds are very strong but weak between the layers, has the smallest broken bond density and the largest interlayer spacing among the three calculated surfaces and is the perfect cleavage plane of fluorite.
surface with stoichiometry and charge neutrality has the smallest surface broken bonds density and the largest interplanar spacing of the six calculated planes, and this surface can be created readily in the presence of external stress and is the perfect cleavage plane of calcite. For fluorite, the order of broken bond density of three surfaces is as follows: (100)>(110)>(111). The {111} surface, consisting of alternate F―Ca―F ionic layer within which the chemical bonds are very strong but weak between the layers, has the smallest broken bond density and the largest interlayer spacing among the three calculated surfaces and is the perfect cleavage plane of fluorite.
Table 1 Calculation of broken bond population of calcite and fluorite surfaces
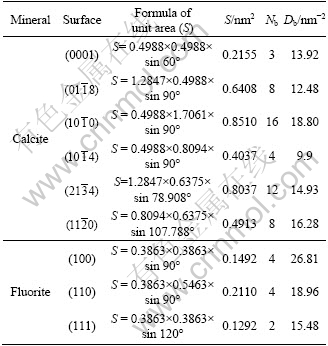
Therefore, ![]() surface of calcite and (111) surface of fluorite with the smallest broken bond density are the perfect cleavage planes and found to be frequently exposed in the morphologies of them [6].
surface of calcite and (111) surface of fluorite with the smallest broken bond density are the perfect cleavage planes and found to be frequently exposed in the morphologies of them [6].
3.3 Relations between surface broken bond density and surface energy of calcite and fluorite crystals
In a certain surface of calcite, calcium ions lose certain number of bonds to oxygen atoms when the surface is created, and oxygen atoms of the carbonate groups are also under-coordinated leaving dangling bonds. As a result, this surface is very reactive. It is well recognized that surface energy (strictly speaking, surface free energy) is one of the most important factors that affect the reactive ability of a surface. Therefore, we predict that there is a direct links between the surface energies and the surface broken bonds densities. The surface energies of different surfaces of calcite and fluorite were calculated employing atomistic simulation technique by LEEUW and PARKER [7] and molecular modeling by PRADIP et al [8], respectively. Not surprisingly, we found that the surface energies are approximately linear with surface broken bond densities for the two minerals, as seen from Fig. 1. The correlation of determination (R2) of the surface energies and surface broken bonds densities for calcite and fluorite are 0.9613 and 0.9969, respectively, indicating that the surface broken bond density determines the surface energy for these two minerals.
If the linear relation between the anisotropic density of surface broken bonds and surface energy is tenable, the surface energies of ![]() and
and ![]() surfaces can be predicted to be 0.87 and 1.18 J/m2, which are relatively low compared to those of
surfaces can be predicted to be 0.87 and 1.18 J/m2, which are relatively low compared to those of ![]() and
and ![]() surfaces, 1.43 and 1.59 J/m2, respectively. COOPER and LEEUW [12] held that the equilibrium morphology of a crystal is determined by the surface energy and the related growth rate of various surfaces. GIBBS [13] proposed that the energy associated with a crystal surface is known to influence its growth rate, and as the growth rates of each surface can vary, this can have a profound influence on the resulting crystal morphology. More specifically, a surface with a high surface energy is expected to have a large growth rate and this fast growing surface will not be expressed in the resulting crystal morphology. Only surfaces with low surface energies and hence slow growing will be expressed. The
surfaces, 1.43 and 1.59 J/m2, respectively. COOPER and LEEUW [12] held that the equilibrium morphology of a crystal is determined by the surface energy and the related growth rate of various surfaces. GIBBS [13] proposed that the energy associated with a crystal surface is known to influence its growth rate, and as the growth rates of each surface can vary, this can have a profound influence on the resulting crystal morphology. More specifically, a surface with a high surface energy is expected to have a large growth rate and this fast growing surface will not be expressed in the resulting crystal morphology. Only surfaces with low surface energies and hence slow growing will be expressed. The ![]() and
and ![]() surfaces with lower surface energies and smaller anisotropic densities of surface broken bonds are expected to be largely expressed in the calcite crystal morphology. It was reported that ditrigonal scalenohedron
surfaces with lower surface energies and smaller anisotropic densities of surface broken bonds are expected to be largely expressed in the calcite crystal morphology. It was reported that ditrigonal scalenohedron ![]() is one of the most common single forms of calcite crystal, and the
is one of the most common single forms of calcite crystal, and the ![]() surface is also a common polysynthetic twin plane of it [6,14].
surface is also a common polysynthetic twin plane of it [6,14].
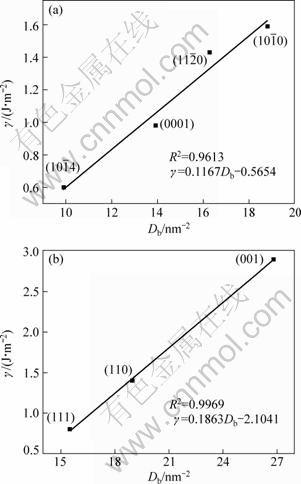
Fig. 1 Relation between surface energy (γ) and surface broken bonds density (Db) of calcite (a) and fluorite (b)
Generally, the most commonly exposed mineral surfaces expected in the flotation slurry will be both cleavage planes and expressed surfaces in the crystal morphology of the mineral [11]. Consequently, the![]() ,
, ![]() and
and ![]() surfaces are usually the three most commonly exposed surfaces in flotation slurry of calcite, which were found to agree well with the experimentally observed morphologies [6,14].
surfaces are usually the three most commonly exposed surfaces in flotation slurry of calcite, which were found to agree well with the experimentally observed morphologies [6,14].
3.4 Relations between surface wettability and broken bond density of calcite and fluorite crystals
As discussed in section 3.3, there is a rectilinear relation between the surface broken bond density and the surface energy. It is anticipated that surface broken bond density determines the surface energy (γSV), which in turn determines the surface wettability. In order to investigate the anisotropic wetting behavior of water at the three commonly exposed surfaces of calcite and two of fluorite, the contact angle measurements were carried out on the surfaces immersed in distilled water. The contact angle measurement results are given in Table 2.
Table 2 Water contact angles of three calcite surfaces and two fluorite surfaces immersed in distilled water

From Table 2, the contact angle or hydrophobicity of three surfaces of calcite and two of fluorite decreases in the order ![]() >
>![]() >
>![]() and (111)>(001), respectively. The results can be explained by the Young’s Equation as follows:
and (111)>(001), respectively. The results can be explained by the Young’s Equation as follows:
γSV=γSL+γLVcos θ (2)
where γLV, γSV and γSL refer to the surface energies of the liquid/vapor, solid/vapor and solid/liquid interfaces, respectively. It needs to be mentioned that after being vacuum dried at 50℃ for around 10 min, there is still a small amount of moisture at the crystal surfaces earlier immersed in distilled water, which may have an effect on the subsequent contact angle measurements in this study. LEEUW and PARKER [7] also calculated the surface energies of four hydrated calcite surfaces at a full monolayer coverage of water molecules and suggested that the surface energies of hydrated surfaces decreased exactly in the same order as that of pure surfaces, i.e. γSV. In this context, the substitution of γSV for surface energies of hydrated surfaces after being vacuum dried may be rational in the following wettability analysis.
In a contact angle measurement, γLV is fixed while the γSV decreases in the calculated order of Db. The interaction of a solid with a liquid often reduces the interfacial energy in a reverse order of γSV [15]. As a result, the contact angle of different surfaces reverses the order of γSV. It was also reported that hydrophilicity is indicated by smaller contact angles and higher surface energy [16]. Therefore, the (001) surface of fluorite has a larger broken bond density or higher surface energy and hence a smaller contact angle than (111) surface. The anisotropic wetting behavior of water on the three calcite surfaces can also be explained in the same way.
3.5 Anisotropic wetting behavior of sodium oleate at commonly exposed calcite surfaces
In order to investigate the anisotropic wetting behavior of oleate on the three commonly exposed surfaces of calcite, the contact angle measurements were carried out on the surfaces immersed in sodium oleate solutions with a pH range of 8.5-9.0. The contact angle measurement results are presented in Fig. 2.
The presence of sodium oleate makes all the three surfaces hydrophobic, which may result from the organization of oleat molecule on calcite surface in such a way that their hydrocarbon chains are directed outwards, and their polar heads (carboxyl groups) towards the calcite surface. From Fig. 2, at a low concentration of sodium oleate (<5×10-6 mol/L), the contact angle values are relatively small and oscillate, which may indicate that the oleate molecules on calcite surfaces are not closely packed and some regions of uncovered mineral surface may occur. The contact angle increases steadily with the sodium oleate concentration for immersing increasing from 5×10-6 to 5×10-5 mol/L. The maximum contact angle for the three surfaces was obtained at a sodium oleate concentration of above 5×10-5 mol/L, which is in good agreement with the flotation experiment. Our flotation results showed that the calcite has a high recovery of 90%-95% when using sodium oleate as collector at a concentration of over 5×10-5 mol/L and pH of 9.0. The contact angle values at a given NaOl concentration in the range of 1×10-7-5×10-3 mol/L decrease in the order of ![]() >
>![]() >
>![]() .
.
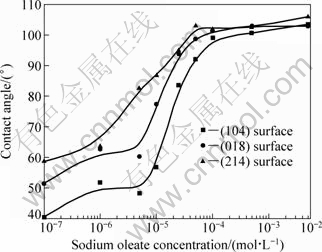
Fig. 2 Contact angle measured with sessile-drop method for water drop (pH 6.8) placed on calcite surface as a function of sodium oleate concentration for immersing (pH=8.5-9.0)
The anisotropic wetting behavior of oleate on three calcite surfaces can be explained by the anisotropic surface chemistry of calcite, since![]() ,
,![]() and
and![]() surfaces have different surface structures. The previous studies indicated that the extent of surface adsorption and the rate of adsorption are influenced by the nature of the surface cations, since oleate adsorption takes place at the cationic sites of mineral surface [17,18]. We hold that spacing or structural factors, such as the cross-sectional area per oleate molecule vertically adsorbed on the surface and the number and the distribution of Ca active sites on a certain surface, can remarkably affect the adsorption behavior of oleate molecules on this mineral surface and hence the wettability of the surface.
surfaces have different surface structures. The previous studies indicated that the extent of surface adsorption and the rate of adsorption are influenced by the nature of the surface cations, since oleate adsorption takes place at the cationic sites of mineral surface [17,18]. We hold that spacing or structural factors, such as the cross-sectional area per oleate molecule vertically adsorbed on the surface and the number and the distribution of Ca active sites on a certain surface, can remarkably affect the adsorption behavior of oleate molecules on this mineral surface and hence the wettability of the surface.
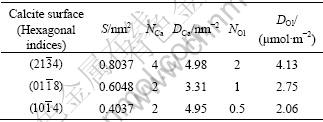
The densities of Ca active sites (DCa) and the adsorption densities of oleate molecule (DOl) adsorbed at a completed monolayer on different calcite surfaces were calculated and the results are summarized in Table 3, while the cross-sectional area per oleate molecule vertically adsorbed on the surface is considered to be 0.32 nm2 [19].
From Table 3, the adsorption density of oleate molecules (DOl) on calcite surfaces at monolayer coverage is in the order of ![]() >
>![]() >
>![]() . A higher adsorption density implies that the absorbed oleate molecules are arranged more compactly on the calcite surface and less mineral surface is exposed, resulting in a more hydrophobic surface and hence larger water contact angle of the surface. This accounts for the increased order of hydrophobicity from
. A higher adsorption density implies that the absorbed oleate molecules are arranged more compactly on the calcite surface and less mineral surface is exposed, resulting in a more hydrophobic surface and hence larger water contact angle of the surface. This accounts for the increased order of hydrophobicity from ![]() to
to ![]() and
and ![]() surface of calcite in oleate solutions.
surface of calcite in oleate solutions.
Table 3 Density of Ca sites (DCa), adsorption density of oleate molecule (DOl) at monolayer coverage by oleate molecules at three different calcite surfaces
4 Conclusions
1) The cleavage planes and commonly exposed surfaces of calcite and fluorite crystals were predicted by calculating the anisotropic densities of surface broken bonds. The prediction was consistent with the experimental observation.
2) The relation between the surface broken bond densities and the surface energies was established, and there was a good rectilinear relation between them with correlation determination (R2) of 0.9613 and 0.9969, respectively.
3) The anisotropic wettability of three most commonly exposed surfaces of calcite and two of fluorite in distilled water and sodium oleate solutions with different concentrations was observed through contact angle measurements. The anisotropic wettability of different surfaces after immersing in distilled water and sodium oleate solutions can be explained by anisotropic surface broken bond density and active Ca site density, respectively.
References
[1] SUN G, FANG Y, CONG Q, REN L. Anisotropism of the non-smooth surface of butterfly wing [J]. Journal of Bionic Engineering, 2009, 5-6(1): 71-76.
[2] ZHAO Y, LU Q, LI M, LI X. Anisotropic wetting characteristics on submicrometer-scale periodic grooved surface [J]. Langmuir, 2007, 23(11): 6212-6217.
[3] SOMMERS A D, JACOBI A M. Creating micro-scale surface topology to achieve anisotropic wettability on an aluminum surface [J]. Journal of Micromechanics and Microengineering, 2006, 16(8): 1571-1578.
[4] HENG J Y, BISMARCK A, WILLIAMS D R. Anisotropic surface chemistryof crystalline pharmaceutical solids [J]. Pharmaceutical Science and Technology, 2006, 7(4): E1-E9.
[5] PRADIP, BEENA R , RAO T K, KRISHNAMURTHY S, VETRIVEL R, MIELCZARSKI J, CASES J M. Molecular modeling of interactions of diphosphonic acid based surfactants with calcium minerals [J]. Langmuir, 2002, 18(3): 932-940.
[6] WANG Pu, PAN Zhao-lu, WENG Ling-bao. Systematic mineralogy [M]. Beijing: Geological Press, 1987: 355-357. (in Chinese).
[7] LEEUW N H, PARKER S C. Atomistic simulation of the effect of molecular adsorption of water on the surface structure and energies of calcite surfaces [J]. J Chem Soc, Faraday Trans, 1997, 93(3): 467-475.
[8] PRADIP, BEENA R, RAO T K, KRISHNAMURTHY S, VETRIVEL R, MIELCZARSKI J, CASES J M. Molecular modeling of interactions of alkyl hydroxamates with calcium minerals [J]. Journal of Colloid and Interface Science, 2002, 256(1): 106-113.
[9] CHICHAGOV A V, BELONOZHKO A B, LOPATIN A L. Information-calculating system on crystal structure data of minerals [J]. Crystallography Reports, 1990, 35(3): 610-616.
[10] CRAWFORD R J, MAINWARING D E. The influence of surfactant adsorption on the surface characterization of Australian coals [J]. Fuel, 2001, 80(3): 313-320.
[11] COOPER T G, LEEUW N H. A computer modeling study of the competitive adsorption of water and organic surfactants at surfaces of the mineral scheelite [J]. Langmuir, 2004, 20(10): 3984-3994.
[12] COOPER T G, LEEUW N H. A combined ab initio and atomistic simulation study of the surface and interfacial structures and energies of hydrated scheelite: Introducing CaWO4 potential model [J]. Surface Science, 2003, 53(2): 1159-1176.
[13] GIBBS J W. The collected works of J W Gibbs [M]. New York: Longmans, 1928.
[14] STIPP S L, HOCHELLA M F. Structure and bonding environments at the calcite surface as observed with X-ray photoelectron spectroscopy (XPS) and low energy electron diffraction (LEED) [J]. Geochim Cosmochim Acta, 1991, 55(6): 1723-1736.
[15] FOWKES F M. Attractive forces at interfaces [J]. Ind Eng Chem, 1964, 56(12): 40-52.
[16] GENNES P. Surface energy [EB/OL]. [2011-05-01]. http://en.wikipedia.org/wiki/Surface_energy.
[17] MOON K S, FUERSTENAU D W. Surface crystal chemistry in selective flotation of spodumene from other aluminosilicates [J]. International Journal of Mineral Processing, 2003, 72(1-4): 11-24.
[18] ANANTHAPADRNANABHAN K P, SOMASUNDARAN P. Oleate chemistry and hematite flotation [M]//YARAR B, SPOTTISWOOD D J. Interfacial Phenomena in Mineral Processing. New York: Engineering Foundation, 1981: 207-227.
[19] REZAEI K A, HAMOUDA A A, DENOYEL R. Influence of sulfate ions on the interaction between fatty acids and calcite surface [J]. Colloids and Surfaces A, 2006, 287(1-3): 29-35.
高志勇,孙 伟,胡岳华,刘晓文
中南大学 资源加工与生物工程学院,长沙 410083
摘 要:计算方解石6个晶面和萤石3个晶面的表面断裂键密度,借助各向异性的表面断裂键密度预测两种含钙矿物的常见暴露面,分析表面断裂键密度与表面能之间的关系,利用接触角实验研究两种矿物常见暴露面的润湿性。表面断裂键密度计算分析表明, ![]() 、
、![]() 和
和![]() 面及 (111) 面分别是方解石和萤石最常见的暴露面;两种矿物表面断裂键密度与表面能之间存在线性关系,其相关系数R2分别为0.9613和0.9969。接触角测量实验表明,两种矿物常见暴露面和水作用后的润湿性与表面断裂键密度有关,表面断裂键密度越大,亲水性越强,接触角越小;和油酸钠作用后的润湿性与表面Ca活性质点密度及质点的空间分布有关。
面及 (111) 面分别是方解石和萤石最常见的暴露面;两种矿物表面断裂键密度与表面能之间存在线性关系,其相关系数R2分别为0.9613和0.9969。接触角测量实验表明,两种矿物常见暴露面和水作用后的润湿性与表面断裂键密度有关,表面断裂键密度越大,亲水性越强,接触角越小;和油酸钠作用后的润湿性与表面Ca活性质点密度及质点的空间分布有关。
关键词:方解石;萤石;表面断裂键;解理;表面能;接触角;润湿性
(Edited by YUAN Sai-qian)
Foundation item: Project (50834006) supported by the National Natural Science Foundation of China; Project (CX2011B122) supported by Hunan Provincial Innovation Foundation for Postgraduate; Project (2011ybjz045) supported by Graduate Degree Thesis Innovation Foundation of Central South University
Corresponding author: HU Yue-hua; Tel: +86-731-88879299; E-mail: HYH@mail.csu.edu.cn
DOI: 10.1016/S1003-6326(11)61306-X

