
Electrochemical process of titanium extraction
CH. RVS. NAGESH, C. S. RAMACHANDRAN
Defence Metallurgical Research Laboratory, Hyderabad, A.P. India-500058
Received 20 November 2005; accepted 9 January 2006
Abstract: A wide variety of processes are being pursued by researchers for cost effective extraction of titanium metal. Electrochemical processes are promising due to simplicity and being less capital intensive. Some of the promising electrochemical processes of titanium extraction were reviewed and the results of laboratory scale experiments on electrochemical reduction of TiO2 granules were brought out. Some of the kinetic parameters of the reduction process were discussed while presenting the quality improvements achieved in the experimentation.
Key words: electrochemical reduction; titanium dioxide; titanium; kinetic parameters
1 Introduction
Titanium metal has enormous potential for a wide variety of applications but its usage and consumption have been limited to aerospace and few other areas mainly because of the high cost of metal. Currently titanium is extracted from titanium oxide ore by a series of process steps involving: 1) chlorination of oxide concentrate to prepare titanium tetrachloride (TiCl4), 2) reduction of TiCl4 by magnesium metal (Kroll process), 3) vacuum distillation/leaching of reduced mass. Electrolysis of the by-product MgCl2 is a parallel process carried out for recovering magnesium metal and chlorine. Sodium reduction of TiCl4 (Hunter process) had also been operated till early 1990s but its commercial application at present is limited. Fused salt electrolysis of TiCl4 in alkali chloride mixtures has been extensively pursued in Dow-Howmet, USA and Ginatta, Italy. But the process could not be commercialized due to several problems such as multi-valency of the metal, selection of materials of construction of the cell and other techno-economic factors.
Extraction of titanium directly from titanium dioxide has been proven to be futile due to high affinity of the metal towards oxygen and high process temperatures. Conventional carbothermic reduction and metallothermic reduction methods did not yield titanium of required purity owing to limiting thermodynamic constraint and high chemical reactivity of the metal. Fused salt electrolysis of TiO2 in floride/phosphate baths to prepare titanium metal was not successful due to problems such as complexities in the cell design, poor solubility of oxide in the melts, recombination of metal, problems associated with purity of the metal.
2 Electrochemical reduction processes
The current practice of Kroll process for titanium sponge production has undergone tremendous improvements and advancements, resulting in significant improvement in energy savings as well as quality of the product[1]. Apparently the Kroll technology has assumed stagnation and there is hardly any scope existing for bringing down the cost of titanium metal further by this method. There have been a wide variety of efforts to develop an alternate process of titanium extraction to produce the metal at a cheaper price. Extensive study on electrochemical de-oxidation of titanium in molten calcium chloride electrolyte led to the development of electrochemical reduction processes. The following is a brief description of important electrochemical/ electrometallurgical reduction processes that are being developed.
2.1 FCC process
The process patented by FRAY, FARTHING and CHEN of Cambridge University, UK[2-3], involves removal of oxygen from solid titanium dioxide by subjecting it to electrolysis in molten calcium chloride bath using graphite as anode. The process involves preparation of TiO2 compacts/pellets from the oxide powder that are sintered and taken as cathode. In this process it is understood that, oxygen diffuses out of the oxide through crystal defects under the conditions of applied DC voltage that is also aided by the formation of electrically conductive ��magnelli�� phase at high temperature. Oxygen ions pass through molten CaCl2 electrolyte to form CO/CO2 gas at the graphite anode. The process is vigorously being pursued by TIMET, USA at California University, Berkeley for scale up and commercial adaptation[4].
2.2 OS process
The calciothermic reduction of TiO2 and in-situ electrolysis of CaO in molten calcium chloride was extensively studied by ONO and SUZUKI[5] for preparing titanium sponge granules. The OS process, thus combines the calciothermic reduction at about 900�� and fused salt electrolysis of CaO in CaCl2 at DC voltage in the range 2.7-3.2 V in a single set up comprising a reduction zone and an electrolytic zone. In a conceptual cell design proposed by this process, titanium dioxide powder is continuously fed into a titanium crucible and from the bottom of the crucible molten CaCl2 with dissolved CaO is taken out and fed into the electrolytic compartment. Calcium metal is fed into the crucible from the electrolytic chamber. The process is under investigation for improvements and scale up.
2.3 OIT liquid titanium process
Quebec Iron and Titanium(QIT), Canada has recently patented a new titanium extraction process[6], in which titania slag as obtained by beneficiation of ilmenite can be treated in an electric arc furnace to remove impurity oxides and then subsequently subjected to electrolysis using molten CaF2 as electrolyte to electrowin titanium that can be tapped out in liquid condition. A few other processes for titanium extraction based on electrometallurgy that are being tried on laboratory scale are given in Table 1.
Table 1 Other electrochemical processes of titanium extraction under study

3 Experimental
At the Defence Metallurgical Research Laboratory, Hyderabad(DMRL) experimental work on laboratory scale has been carried out to study the electrochemical reduction of TiO2 in molten calcium chloride bath (Fig.1). Studies are also under way at DMRL to explore the kinetics of the process. The following is a description of experimental work and discussion of the results.

Fig.1 Steps in electrochemical reduction process for titanium sponge production
Initially bench scale experimental work was conducted in which different pre-forms of titanium dioxide were subjected to electrochemical reduction in calcium chloride bath at a temperature range of 800-900 �� to study the important parameters of the process, and the details of which were presented elsewhere[7]. This has led to the current activity of cell operation on a batch size of 100-200 g of sponge.
The schematic of the cell is shown in Fig.2 that consists of a stainless steel retort heated by an electrical resistance furnace. The retort is closed by a lid that has provision of nozzles for electrode leads, argon gas supply and vent gas outlet. TiO2 granules prepared from the oxide powder are sintered and taken into a stainless steel perforated basket that is inserted into a graphite crucible placed in the retort with the help of a steel rod. The steel rod and graphite crucible are connected to the two terminals of a DC power source. Typical experimental procedure involves: 1) loading of weighed quantity of calcium chloride into the graphite crucible and melting; 2) loading of weighed quantity of oxide granules into the steel basket and assembly of the system so that the basket with granules lies well above the melt level; 3) melting of salt and lowering of the basket to a pre-determined level; and 4) application of DC voltage and continuation of electrolysis for selected time. During the electrolysis argon gas is purged into the system and vent gases are allowed to pass through a bubbler filled with baryta solution. At the end of the electrolysis, the basket is lifted up above the melt level and the system is cooled to room temperature under argon gas cover. At room temperature the basket with metallized granules is thoroughly washed with water, acetic acid and dilute HCl to remove the adhered salt completely. The metallized granules are then dried and taken for characterization.
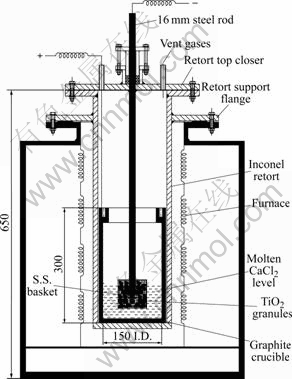
Fig.2 Schematic of experimental set-up
A number of experiments were conducted to study various parameters such as bath temperature, inter-electrode distance, DC voltage. Some improvements in the cell configuration could be incorporated for achieving better results.
In an electrochemical reduction process, the overall reaction can be described as: O(Metal)+e=O2- (Molten salt). At graphite anode, O2-(Molten salt)+C=CO/CO2. The reduction process involving complete removal of oxygen from the oxide is viewed to take place by the following steps: 1) diffusion of oxygen from bulk titanium dioxide to the oxide-salt interface; 2) transport of oxygen ions through molten electrolyte towards graphite anode; and 3) liberation of CO/CO2 gas at the anode. Among these steps oxygen diffusion in solid titanium dioxide is the slowest and becomes the rate controlling step. Diffusion of oxygen in the oxide depends on parameters such as defects concentration, temperature, and electrical conductivity. The ionic mobility may be computed employing a simple model[8] such as ?= , where
, where  is vibration frequency, q is charge of ion and �� is mean free path. Electrical conductivity of titanium dioxide was measured in the temperature range of 25-1 000 ��. The conductivity measurement method followed the standards of setup as well as procedure listed in the ASTM standard D257-99. Typical measurement involves placing of the oxide specimen in a tubular furnace and heating to high temperature and application of DC voltage of the order of milli volts and measuring current response (in milli amps) employing Ketheley instrument continuously every few seconds. It is found that there is a sharp increase in conductivity (Fig.3) at a temperature of about 930 ��, which is explained by the formation of highly conductive ��magnelli�� phase[3]. The rate of oxygen ion removal from the oxide-salt interface might also influence the bulk diffusion of oxygen within the oxide.
is vibration frequency, q is charge of ion and �� is mean free path. Electrical conductivity of titanium dioxide was measured in the temperature range of 25-1 000 ��. The conductivity measurement method followed the standards of setup as well as procedure listed in the ASTM standard D257-99. Typical measurement involves placing of the oxide specimen in a tubular furnace and heating to high temperature and application of DC voltage of the order of milli volts and measuring current response (in milli amps) employing Ketheley instrument continuously every few seconds. It is found that there is a sharp increase in conductivity (Fig.3) at a temperature of about 930 ��, which is explained by the formation of highly conductive ��magnelli�� phase[3]. The rate of oxygen ion removal from the oxide-salt interface might also influence the bulk diffusion of oxygen within the oxide.

Fig.3 Variation of electrical conductivity of TiO2 with temperature
4 Discussion
During the reduction process the cell current is found to be gradually increasing while the cell voltage is kept constant, which may be explained by the fall in resistance of the oxide granules due to metallization. The cell current comprises current due to ionic conductivity and current due to electronic conductivity of the oxide. The ionic currents are expected to reduce as the metallization progresses. The electronic current is expected to increase as the electrical resistance of the system falls at high temperature. However, detailed study of these aspects only can bring out the current patterns. As the electrolysis is initiated, formation of white precipitate is noticed in the baryta bubbler indicating the occurrence of desired de-oxidation reactions. On the basis of conversion of oxygen removed from the oxide to CO/CO2 that is absorbed in baryta and change in rate of formation of carbonate further insight into the kinetics of the process may be determined.
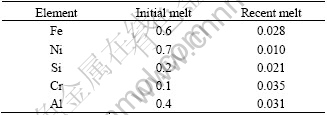
SEM and EDAX characterization of the washed and dried granules show a network of metallic species, i.e. titanium particulate with very low levels of oxygen as seen from Figs.4 and 5. Photographs of initial oxide granules and metallized species generated after the experiment and a button (measuring approximately 50 mm in diameter and 5 mm in thickness) melted out of the sponge by non-consumable arc melting are shown in Fig.6. Characterization of the button by hardness testing, chemical analysis and metallography reveals good metallization in the samples. The hardness of the button is found to be as high as BHN 270, which must be due to higher oxygen and carbon contents that were analyzed to be about 1% and 0.6% respectively. However, higher oxygen content in the button could be due to the fact that it is very difficult to separate partially metallized granules from the fully metallized granules which are melted into button. The degree of metallization is found to be improved with increased electrolyte temperature. However, at higher temperatures it is found that the carbon content in the metal increases. It should be noted that pick up of carbon is noticed even in the early experimental work reported on de-oxidation of titanium by electrochemical means[9]. Some improvements in cell parameters could lead to reduced carbon impurity content of the metal as seen from Fig.7, which shows carbon X-ray images taken by EPMA of initial melts and recent melts with improvements. There are some improvements with respect to other impurity elements as seen from Table 2 that shows the chemical analysis of the button samples of the initial and recent melts.

Fig.4 SEM photographs of sponge formed by electrochemical reduction of TiO2
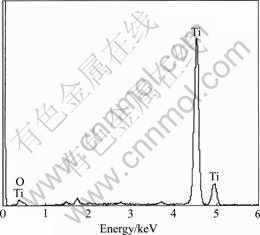
Fig.5 Typical EDAX profile of sponge granule

Fig.6 Photographs of sintered oxide granules(a), titanium sponge granules(b) and button melted out of sponge granules(c)

Fig.7 Carbon X-ray images of titanium samples: (a) Initial melts; (b) Recent melts
Table 2 Chemical analysis of titanium button pieces (mass fraction, %) m
5 Conclusions
The results obtained on electrochemical reduction process have been very encouraging. There is a lot of scope to improve the process and quality of the product. Various fundamental aspects of the process including kinetics are being explored for further understanding of the process.
Acknowledgements
The authors are thankful to Dr. A. M. Sriram Murthy, Director, DMRL for permitting to publish this work. They are grateful to Defence Research & Development Organization(DRDO) for initiating this activity and extending financial support. They also wish to acknowledge the help received from Dr. V. V. Bhanu Prasad and Sir V. V. Rama Rao of CCG & EPMA groups of DMRL. Technical support to carry out the resistance measurement of TiO2 received from Dr. A. K. Suri, BARC is gratefully acknowledged.
References
[1] HYADO T, ICHIHASH H. Establishment of the manufacture of 5N super purity titanium billets by Kroll process [A]. LUTJERING G, ALBRECHT J. Titanium��2003 Science and Technology [C]. Wiley-Vch Verald GmbH, 2004: 141-148.
[2] FRAY, FARTHING, CHEN. Removal of oxygen from metal oxides and solid solutions by electrolysis in a fused salt [P]. International Patent, WO 99/64638, 1999.
[3] CHEN, FRAY, FARTHING. Direct electrochemical reduction of titanium dioxide to titanium in molten calcium chloride [J]. Nature, 2000, 407: 361-364.
[4] ��Switched �Con titanium��, MBM July 2003, 20.
[5] SUZUKI R O, TERANUMA K O H, ONO K. Calciothermic reduction of titanium oxide and in-situ electrolysis in molten CaCl2 [J]. Met Mat Trans B, 2003, 34B: 287-295.
[6] KRAFT E H. Summary of emerging titanium cost reduction technologies [R]. Report by EHK technologies. Vancouver, WA 2004.
[7] RAMMOHAN RAO A V, NARASIMHA RAO R V L, NAGESH C R, BHANU PRASAD V V, RAMACHANDRAN C S. Titanium sponge production by a novel electrochemical reduction of TiO2 [R]. DMRL technical report, DMR TR 378, 2005-07.
[8] SEITZ F. The Modern Theory of Solids [M]. McGraw Hill Book Co, 1940.
[9] OKABE T H, NAKAMURA M, OISHI T, ONO K. Electrochemical de-oxidation of titanium [J]. Met Trans B, 1993, 24B: 449-454.
(Edited by YUAN Sai-qian)
Corresponding author: CH. RVS. NAGESH; E-mail: nagesh_chrvs@rediffmail.com