
Immobilization of laccase on amine-terminated magnetic nano-composite by glutaraldehyde crosslinking method
XIAO Hai-yan(����), HUANG Jun(�� ��), LIU Cheng(�� ��), JIANG De-sheng(������)
Key Laboratory of Fiber Optic Sensing Technology and Information Processing of Ministry of Education,
Wuhan University of Technology, Wuhan 430070, China
Received 10 April 2006; accepted 25 April 2006
Abstract: The immobilizing procedure has great influence on the activity of the immobilized laccase. The laccase was immobilized on copper tetraaminophthalocyanine (CuTAPc)-Fe3O4 magnetic nano-composite by glutaraldehyde crosslinking method via a two-step reaction. For 1 g nano-composite, the optimum pH value, the quantity, the concentration and reaction time of glutaraldehyde are 7.0, 10 mL, 10% and 4 h, respectively. In the second step the optimum pH value, reaction temperature and reaction time of laccase are 5.0, 0 �� and 2 h, respectively. The average diameter of the immobilized laccase is 78 nm, the saturation magnetization (Ms) of the immobilize laccase is 23.45 A?m2/kg and the surface area of the immobilized laccase is 115.03 m2/g. When 2.0 mg/mL laccase solution is used under optimum conditions and 2, 2��-azino-bis-(3-ethylbenzothiazoling-6-sulphonic acid) is used as substrate, the activity of immobilized laccase reaches 1 430 U/g (nano-composite) and the Michealis-Menten constant(Km) of the immobilized laccase is 2.38��10-5 mol/L.
Key words: immobilization; laccase; glutaraldehyde; copper tetraaminophthalocyanine-Fe3O4; activity
1 Introduction
Laccases (E.C.1.10.3.2) can oxidize many substrates and be applied in various fields such as biotechnology, food and agriculture product processing, health care, medicine, dye bleaching and pollution monitoring[1]. In biosensors based on laccase catalysis, immobilized laccases are often used to improve their stability and isolate them from the substrates and products for reuse[2]. Magnetic supports have been shown wide interest since they have been applied in the fields of protein and enzyme immobilization[3-6]. Due to the small size and excellent magnetic property, the copper tetraaminophthalocyanine (CuTAPc)-Fe3O4 composite was selected as the support for the immobilization of laccase. However, the activity of the immobilized laccase is dependent largely upon the properties of the carrier and the immobilization procedure.
With the terminated amino group outside the composite, the immobilization of CuTAPc-Fe3O4 composite is a two-step reaction by crosslinking with glutaraldehyde. In the first step, the optimum pH value, the quantity, the concentration and reaction time of glutaraldehyde were studied; in the second step, the optimum pH value, reaction temperature and reaction time of laccase were also studied, and thus the optimum conditions of immobilization were achieved. Moreover, the properties of the immobilized laccase such as average diameter, surface area, specific activity, kinetic constants, and operational stability were also investigated. All the results obtained in this paper would provide a sound basis to biological sensing material.
2 Experimental
2.1 Materials and instruments
Pycnoporus sanguineus laccase, a white-rot fungus laccase with a molecular mass of 64 kDa, was provided by the Institute of Microbiology of the Chinese Academy of Sciences and its purity was tested by electrophoresis with a single protein band. The 2, 2��-azino-bis-(3-ethylbenzothiazoline-6-sulphonic acid)(ABTS) was purchased from Sigma Chemical Company (Deisenhofen, Germany). All other chemicals were of analytical grade and were used without further purification.
The particle diameter and size distribution of the immobilized laccase were analyzed by a laser-based particle size analyzer (LB-550, Japan). The magnetic property of the immobilized laccase was determined by Modle 4HF vibrating sample magnetometor(ADE, USA). The surface area was analyzed by Nova instrument (Quantachrome, USA). The activity of laccase was assayed by using a UV-2450 spectrophotometer (Shimadzu, Japan).
2.2 Crosslinking reaction with glutaraldehyde
The CuTAPc-Fe3O4 composite was prepared according to the method in Ref.[7]. 1 g CuTAPC-Fe3O4 composite crosslinked with a certain amount of glutaraldehyde solution with stirring and the excess glutaraldehyde was washed off with distilled water in the presence of a magnetic field. Then the treated CuTAPc�CFe3O4 composites were added to 0.1 mol/L phosphate buffer solution (PBS) containing a certain amount of laccase with continuous shaking. In each case, the immobilized laccase was washed with PBS (pH 7.0) until no protein was detected in the eluate and was then lyophilized and stored at 4 ��. In the first step crosslin-
king reaction of composite with glutaraldehyde, in order to evaluate the effect of pH value, different pH conditions in the range from 6.0 to 8.0 were used; the influence of the amount of glutaraldehyde was examined varying from 0.5 to 4 g; to determine the effect of concentration of glutaraldehyde, the composite reacted with different concentrations of glutaraldehyde ranging from 2% to 25%; to determine the effect of reaction time, values from 1 to 16 h were tested. In the second step crosslinking reaction of glutaraldehyde with laccase, in order to evaluate the effect of pH value on laccase immobilization, different pH conditions in the range from 4.8 to 8.0 were used; to determine the effect of temperature on laccase immobilization, values from 0 �� to 55 �� were tested; the influence of immobilization time was examined varying from 0.5 to 16 h.
2.3 Analysis of laccase activity
Activities of immobilized laccase were assayed by using 4 mg/mL ABTS in distilled water as a substrate. The reaction mixture consisting of 3 mL of 0.1 mol/L tartaric acid buffer (pH 3.0) containing 15 mg of immobilized laccase was initiated by adding 20 ��L of substrate and reacted at 25 �� for 5 min. During the process, the increase in the absorbance at 420 nm was measured by a UV-2450 spectrophotometer. The molar extinction coefficient of ABTS is 36 dm3/(mol?cm-1). One activity unit was defined as the amount of enzyme required to catalyze 1 ��mol of substrate per min [8]. Kinetic tests were performed by measuring the initial reaction rates of the immobilized laccase using different concentrations of ABTS as substrate in 0.1 mol/L tartaric acid buffer (pH 3.0) at 25 ��. The Michealis-Menten constant(Km) values were calculated by linear regression according to the lineweaver-burk relationship. In order to assess the operating stability of immobilized laccase, several consecutive operating cycles were performed by oxidizing ABTS, as described by DAVIS et al[9].
3 Results and discussion
The crosslinking of glutaraldehyde is a two-step reaction as shown in Fig.1. First, the terminated amino group of CuTAPc in the composite reacts with glutaral-
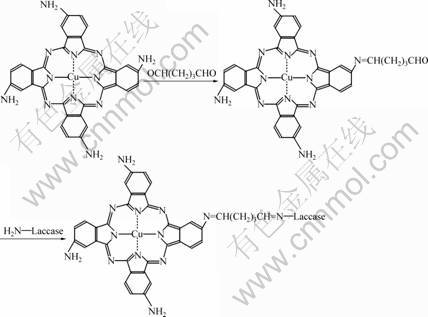
Fig.1 Schematic illustration of laccase immobilized on CuTaPc-Fe3O4 nanoparticles by crosslinking with glutaraldehyde
dehyde to form a Schiff-base linkage and provide a terminal aldehyde, which is then condensed with the amino group in laccase to form a second Schiff-base linkage. The immobilizing procedure has great influence on the activities of the immobilized laccase. For the purpose of calculation, the activity observed in optimal conditions was taken as 100%.
3.1 Optimal reactive conditions of amine-terminated composite with glutaraldehyde
The crosslinking reaction of glutaraldehyde with the terminated amino group of CuTAPc involves the nucleophilic addition between the amino groups and carbonyl of glutaraldehyde. Protonation of carbonyl oxygen makes carbonyl carbon more susceptible to nucleophilic attack. This means that the environment with high acidity is very helpful for addition reaction. Nevertheless, the amino group can also undergo protonation to form +H3N ion, which lacks unshared electrons and is no longer nucleophilic[10]. Thus, the favored environment for addition reaction will be low acidity. Therefore, the optimal pH value for the addition reaction depends on the balance of these two kinds of affecting factors. In the range from 6.0 to 8.0, the optimal pH value is 7.0 and the relatively activities decrease slowly as the pH value changed, as shown in Fig. 2. When CuTAPc is a cyclic flat conjugated molecule, the amino groups are very active in attacking the aldehyde group to form carbon-nitrogen double bonds and the conjugated chain is increased, the reaction is not sensitive to the pH value.
At pH value of 7.0, the influence of the amount of glutaraldehyde was examined varying from 0.5 g to 4.0 g. The results show that for 1 g nano-composite, the acti-
vity of immobilized enzyme increases first and reaches a maximum at 2 g, thereafter the activity decreases with increasing the amount of glutaraldehyde. This could be presumed that high amount of glutaraldehyd leads to a high amount of protein loading and relative activity,
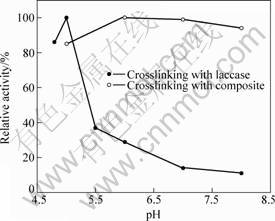
Fig.2 Effect of pH value on crosslinking reaction
whereas more enzyme molecules go into the carriers�� inner after the active sites of the carriers are saturated and the enzyme molecules couldn��t efficiently perform the catalysis capability for the limitation of substrate diffusion. Meanwhile, enzyme can be hidden and some active sites damaged at high immobilization yield system.
To determine the effect of concentration of glutaraldehyde, the composite reacted with different concentrations of glutaraldehyde ranging from 2% to 25%. The results show that 10% is the optimal concentration. Lower concentration will decrease the reactive rate while higher concentration could denature laccase, which leads to the decrease of relative activities.
To determine the effect of reactive time, the values from 1 to 16 h were tested. After 4 h, the immobilized laccase shows that the maximum activity and the relative activities of immobilized laccase decrease with further prolonging time as shown in Fig. 3. Due to their large surface area and excess glutaraldehyde, the CuTAPc�CFe3O4 composites may first adsorb glutaraldehyde on their surface, and then the amino groups of the composite react quickly with nearby aldehyde groups.
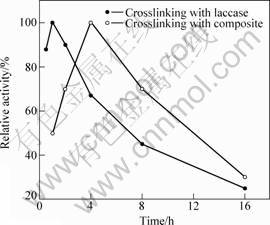
Fig.3 Effect of time on crosslinking reaction
In the first step crosslinking reaction, excess glutaraldehyde was used to completely activate all amino groups on the surface of the composite to provide a terminal aldehyde for laccase, avoiding the intermolecular crosslinking among CuTAPc.
3.2 Optimal reactive condition of glutaraldehyde with laccase
The laccase crosslinking reaction is another nucleophilic addition, so it is often necessary to adjust the reaction medium to the right acidity. The optimal pH value is 5.0 and the relative activity will drastically decrease when the pH value deviates from 5.0. Laccases are made up of peptide chains. Some side chains of the peptide chains contain basic groups amino (��NH2), other side chains contain acidic groups carboxyl(��COOH). Because of these acidic and basic side chains, there are positively and negatively charged groups along the peptide chain. Side chains affect the properties of proteins not only by their acidity or basicity, but also by their chemical properties. So the laccase crosslinking reaction is more sensitive to the pH value as can be seen in Fig.2.
Laccase was crosslinked at different temperatures ranging from 0 to 55 ��. In the present study, the optimal temperature is 0 �� and the activity of the immobilized laccase seems to decrease with the temperature increasing. Usually when the reactive temperature raises the rate of addition reaction, the amount of bound enzyme will increase, which leads to relatively high catalytic activity. However, the free pycnoporus sanguineus laccase is often easily inactivated in PBS (pH 5.0.) at high temperature. So the crosslinking reaction preferred anging e oobilizationisting of to be carried out at 0 �� to prevent the denaturalization caused by the environmental factors.
At 0 �� and pH 5.0, laccase was crosslinked with glutaraldehyde on the CuTAPc-Fe3O4 composite using different time durations, varying from 0.5 to 24 h. At first the relative activity of the immobilized laccase increases with the increase of immobilization time. The immobilized laccase exhibits the maximum activity after 1 h immobilization, and then the relative activities of immobilized enzymes decrease with further prolonging immobilization time as shown in Fig.3. With an increase of immobilization time, some of the laccases are denaturalized in the acidic solution, leading to the decrease of relative activities.
From the observed results, it is evident that the activity of the immobilized enzyme is dependent largely upon the reactive conditions and the stability of the free laccase.
3.3 Properties of immobilized laccase by glutaralde- hyde crosslinking method
Size analysis shows that the average diameter of the immobilized laccase is 78 nm with a size distribution (52% of the total immobilized laccase has diameter with the range from 60 to 90 nm), which could give less restriction for diffusion in biological reaction[11]. The virgin curve (Fig.4) shows the Ms of the immobilized laccase is 23.45 A?m2/kg, which shows the excellent magnetic property is very helpful for the reuse of laccase in the magnetic field. The surface area was calculated according to multi-point BET as shown in Fig.5, where w represents the mass of adsorbate gas at the relative pressure. The surface area of the immobilized laccase is 115.03 m2/g, which indicates that the immobilized laccase has large surface area for the laccase catalysis reaction. The optimal conditions avoid the intermolecular crossinglink among CuTAPc, so laccase immobilized on CuTAPc-Fe3O4 composite by glutaraldehyde crosslinking method has the virtue of the carrier.
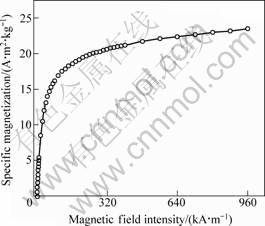
Fig.4 Virgin curve of immobilized laccase
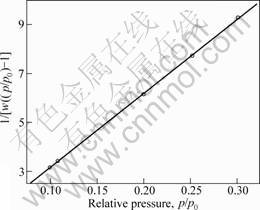
Fig.5 Multi-point BET plot of immobilized laccase
When 2.0 mg/mL laccase solution is used under optimum conditions and ABTS is used as substrate, the activity of immobilized laccase reaches 1 430 U/g (nano- composite) and the Km of the immobilized laccase is 2.38��10-5 mol/L. After 5 consecutive operations, the immobilized laccase could still retain 80 % of its initial activity. The catalytic properties of the immobilized laccase provide a useful indication for a practical application.
4 Conclusions
In the crosslinking reaction, for 1 g nano-composite, the optimum pH value, the quantity, the concentration and reaction time of glutaraldehyde are 7.0, 10 mL, 10% and 4 h, respectively; in the second step the optimum pH, reaction temperature and reaction time of laccase are 5.0, 0 �� and 2 h, respectively. The immobilized laccase has small particle size, large surface area and excellent magnetic property. When 2.0 mg/mL laccase solution is used under optimum conditions and ABTS as substrate, the activity of immobilized laccase arrives 1 430 U/g (nano-composite) and the Km of the immobilized laccase is 2.38��10-5 mol/L. After 5 consecutive operations, the immobilized laccase could still retain 80 % of its initial activity. The results indicate that this kind of immobilized laccase might be a good candidate for the biological sensing material of fiber optic biosensors.
References
[1] ALFRED M M, RICHARD C S. Laccase: new functions for an old enzyme[J]. Phytochemistry, 2002, 60: 551-565.
[2] NELSON D, MARIA A R, ALESSANDRO D, LILIANA G. Applications of laccases and tyrosinases (phenoloxidases) immobilized on different supports: a review[J]. Enzyme Microb Tech, 2002, 31: 907-931.
[3] YAKUP A M, HANDAN Y, SULEYMAN P, ADIL D. Immobilization of glucoamylase onto spacer-arm attached magnetic poly-methylmethacrylate microspheres: characterization and application to a continuous flow reactor[J]. J Molec Catal B: Enzymatic, 2000, 11: 127-138.
[4] CHEN D H, LIAO M H. Preparation and characterization of YADH-bound magnetic nanoparticles[J]. J Molec Catal B: Enzymatic, 2002, 16: 283-291.
[5] EVEREET E C. Iron nanoparticles as potential magnetic carriers[J]. J Magnetism and Magnetic Materials, 2001, 225: 17-20.
[6] ZHENG G, SHU B, YAN S. Preparation and characterization of immobilized lipase on magnetic hydrophobic microspheres[J]. Enzyme and Microbial Technology, 2003, 32: 776-782.
[7] HUANG J, ZHOU J Y, XIAO H Y, LONG S Y, WANG J T. Study of CuTAPc-Fe3O4 nanoparticles and their laccase immobilization[J]. Acta Chimica Sinica, 2005, 63(14): 1343-1347.
[8] JIANG D S, LONG S Y, HUANG J, XIAO H Y. Immobilization of pycnoporus sanguineus laccase on magnetic chitosan microspheres[J]. Biochemical Engineering, 2005, 25: 15-24.
[9] DAVIS S, BURNS R G. Covalent immobilization of laccase on activated carbon for phenolic effluent treatment[J]. Appl Microbiol Biotechnol, 1992, 37: 474-479.
[10] ROBERT T, ROBERT N B. Organic Chemistry(4th ed)[M]. Massachusetts: Allyn and Bacon Press, 1983. 757.
[11] OH J T, Kim J H. Preparation and properties of immobilized amyloglucosidase on nonporous PS/PNaSS microspheres[J]. Enzyme Microb Tech, 2000, 27: 356-361.
(Edited by CHEN Wei-ping)
Foundation item: Projects (60377032, 60537050) supported by the National Natural Science Foundation of China
Corresponding author: HUANG Jun; Tel: +86-27-87651850-8005; Fax: +86-27-87665287; E-mail: fosrcwut@public.wh.hb.cn