Bone-like apatite formation on HA/316L stainless steelcomposite surface in simulated body fluid
来源期刊:中国有色金属学报(英文版)2009年第2期
论文作者:樊新 陈 剑 邹俭鹏 万千 周忠诚 阮建明
文章页码:347 - 352
Key words:hydroxyapatite; 316L stainless steel; biocomposites; simulated body fluid; bone-like apatite
Abstract: HA/316L stainless steel(316L SS) biocomposites were prepared by hot-pressing technique. The formation of bone-like apatite on the biocomposite surfaces in simulated body fluid(SBF) was analyzed by digital pH meter, plasma emission spectrometer, scanning electron microscope(SEM) and energy dispersive X-ray energy spectrometer(EDX). The results indicate that the pH value in SBF varies slightly during the immersion. It is a dynamic process of dissolution-precipitation for the formation of apatite on the surface. With prolonging immersion time, Ca and P ion concentrations increase gradually, and then approach equilibrium. The bone-like apatite layer forms on the composites surface, which possesses benign bioactivity and favorable biocompatibility and achieves osseointegration, and can provide firm fixation between HA60/316L SS composite implants and human body bone.
基金信息:the National Natural Science Foundation of China
the Postgraduate Education and Innovation Project from Central South University, China
FAN Xin(樊 新), CHEN Jian(陈 剑), ZOU Jian-peng(邹俭鹏),
WAN Qian(万 千), ZHOU Zhong-cheng(周忠诚), RUAN Jian-ming(阮建明)
State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China
Received 4 May 2008; accepted 25 August 2008
Abstract: HA/316L stainless steel(316L SS) biocomposites were prepared by hot-pressing technique. The formation of bone-like apatite on the biocomposite surfaces in simulated body fluid(SBF) was analyzed by digital pH meter, plasma emission spectrometer, scanning electron microscope(SEM) and energy dispersive X-ray energy spectrometer(EDX). The results indicate that the pH value in SBF varies slightly during the immersion. It is a dynamic process of dissolution-precipitation for the formation of apatite on the surface. With prolonging immersion time, Ca and P ion concentrations increase gradually, and then approach equilibrium. The bone-like apatite layer forms on the composites surface, which possesses benign bioactivity and favorable biocompatibility and achieves osseointegration, and can provide firm fixation between HA60/316L SS composite implants and human body bone.
Key words: hydroxyapatite; 316L stainless steel; biocomposites; simulated body fluid; bone-like apatite
1 Introduction
Hydroxyapatite(HA, Ca10(PO4)6(OH)2) is a familiar bioactive material and has excellent properties such as bioactivity, biocompatibility and ability to induce bone tissue growth. After implanation into the body, HA can form strong chemical bonds with natural bone and to promote new bone growth because of its similar chemical and mineralogical composition and crystallographic structure to apatite of human body living bone[1-2]. However, HA suffers from its brittleness and relatively poor mechanical properties, which impede its clinical use as long term load-bearing applications[3]. 316L stainless steel (316L SS) is the most popular surgical metal material as load-bearing implant because of its excellent biocompatibility and favorable mechanical properties[4-6]. However, 316L SS is an inert material, which cannot form strong chemical bonds with natural bone. Therefore, biocomposites containing both bioactive materials and metals, such as 316 SS and 316L SS, are expected to have more broad practical applications than bioactive coatings because each material can compensate for the disadvantages of the other.
RODRIGUEZ et al[7] investigated the in vitro bioactivity assessment of the coatings on 316L SS. LIN et al[8] studied the bone-like apatite layer formed on the 316L SS fiber. The results showed that the composition of apatite was Ca and P, and the Ca/P ratio was similar to that of human body bone. BALAMURUGAN et al[9] investigated the crystallinity, microstructure and morphology of the hydroxyapatite coatings. The results revealed that the formation of hydroxyapatite coating was appropriate for extensive biomedical applications of 316L SS. The biocomposites containing both HA and 316L SS are mechanically stronger and tougher than HA, which can cater for the load-bearing implants[10-11]. Consequently, it is significant to investigate the bioactivity of the HA/316L SS biocomposites.
Bioactivity, as an important factor for evaluating the properties of biomaterials, is the ability to form the chemical bonds between biomaterials and human body living bone tissue. The formation of bone-like apatite or Ca/P layer was regarded as an essential factor for biomaterials with bioactivity[12-15]. In the present work, the simulated body fluid(SBF) was used to evaluate the in vitro bioactivity of HA/316L SS biocomposites. The biocomposite containing 60% HA (volume fraction) and 40% 316L SS(HA60/316L SS) was used to investigate the bone-like apatite formation on HA/316L SS biocomposite surfaces in SBF because of favorable mechanical properties and appropriate biological performance.
2 Experimental
2.1 Preparation of biocomposites
The biocomposites using HA and 316L SS as starting materials were fabricated by hot-pressing technique[16-17]. The biocomposite samples with a size of 10 mm×5 mm×2 mm were prepared by line-incision technology. The samples as prepared were polished by rough sand-paper and rinsed ultrasonically in acetone, absolute alcohol and deionized water in turn for several times. Finally, they were dried in an oven at 80 ℃ for 12 h. The samples treated for investigation in SBF were obtained.
2.2 Preparation of SBF
KOKUBO et al[18] investigated the in vitro bioactivity of biocomposites using SBF. The results were well consistent with those in vivo. At the present time, SBF is a standard method by which the bioactivity of biomaterials is investigated. In the present work, SBF, of which chemical components are similar to the inorganic mineral ions of human blood plasma and of which ion concentrations are also similar to those of human blood plasma, was prepared (as listed in Table 1).
Table 1 Ion concentrations of SBF and human blood plasma(mmol/L)
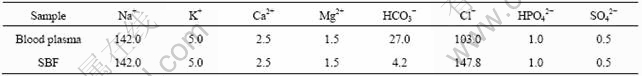
The special preparation process of SBF was as follows. The solution was prepared by dissolving reagent-grade sodium chloride (NaCl, 7.995 g), potassium chloride (KCl, 0.224 g), calcium chloride dihydrate (CaCl2・2H2O, 0.277 g), magnesium chloride hexahydrate (MgCl2・6H2O, 0.305 g), sodium hydrogen carbonate (NaHCO3, 0.353 g), dipotassium hydrogen phosphate trihydrate (K2HPO4・3H2O, 0.228 g), sodium sulphate (Na2SO4, 0.071 g) in deionized water. Then the solution above was buffered to physiological pH7.33 at 37 ℃ by both hydrochloric acid (HCl) and tris- (hydroxymethyl)-aminomethane ((CH2OH)3CNH2)[19]. The pH value of SBF as obtained was similar to that of human blood plasma.
2.3 In vitro test
The HA/316L SS biocomposite samples were immersed into SBF(placed in 100 mL conical flask) for in vitro test. The conical flasks were sealed and then incubated in constant temperature water bath for 1, 4, 7 and 14 d at (37±1) ℃, respectively. The specimens were taken out from SBF, and rinsed with acetone, absolute alcohol and deionized water in turn for several times so that the surface reaction was prevented and the residual elements on the surface were removed. Finally, the treated specimens were maintained at 40 ℃for 5 h in an oven.
2.4 Analysis and characterization
The tests of pH value of SBF were obtained by pHS-25C digital pH meter(Shanghai LI DA Instrument Factory, China).
Ca and P ion concentrations in SBF after removing the biocomposites from SBF were performed on IRIS advantage 1000 plasma emission spectrometer.
The surface morphologies were observed using scanning electron microscope(model KYKY2800 from JEOL Company).
The energy dispersive X-ray energy spectroscope (EDX) (model Finder 1000) was used to analyze the element composition and element content of microregion on the biocomposite surfaces.
3 Results and discussion
3.1 Analysis of pH value of SBF
Fig.1 shows the influences of the immersion time (from 0 d to 14 d) of HA60/316L SS biocomposites in SBF on the pH value of SBF. It can be seen that the pH value was 7.33 before immersion in SBF, which is similar to that of human blood plasma. The pH value of SBF increases slightly as the immersion time increases during early stage. The initial increase of pH value results from the accumulation of OH- on the biocomposite surfaces. Then the pH value reaches its maximum (approximately 7.55) at the 7th day, which is a little higher than the normal value of human blood plasma but it does not do harm to human body. However, with further prolonging immersion time, the pH value decreases gradually. After immersion for 14 d, the pH value of SBF becomes 7.4 which is equivalent to the normal value of human blood plasma.
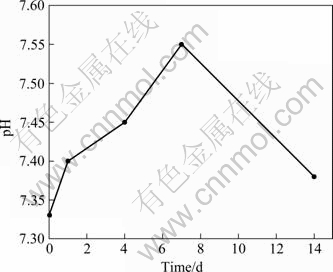
Fig.1 Effects of immersion time of HA60/316L SS biocomposites in SBF on pH value of SBF
When HA60/316L SS biocomposites were immersed in SBF, Ca ions in SBF were received by the dissolving of composite surfaces; and then OH- was
accumulated gradually because of the exchange of Ca2+ and H+, which results in the increase of pH value. The accumulation of OH- on the surface is believed necessary for apatite nucleation[20]. It is a dynamic process for apatite formation on the composite surfaces that the material surfaces are dissolved firstly and then the apatite layers are formed on the surfaces. As Ca and P ion concentrations reach their maximum, the pH value increases to its maximum correspondingly. According to Ref.[21], Ca-P compounds were precipitated spontaneously when the pH value of the physiological solution was over 7.4. During the late stage, Ca and P ions are consumed largely due to the formation of abundant apatite; and simultaneously, OH- is consumed correspondingly. Thus, the pH value of SBF decreases. When the equilibrium of dissolution and precipitation is achieved, the pH value becomes invariable.
3.2 Testing of concentrations of Ca and P ions in SBF
Changes of Ca and P ion concentrations with immersion time(from 0 to 14 d) of HA60/316L SS biocomposites in SBF are shown in Fig.2.

Fig.2 Changes of Ca and P ion concentrations with immersion time of HA60/316L SS biocomposites in SBF: (a) Ca ion con- centration; (b) P ion concentration
It is a dynamic process for the formation and growth of apatite on the HA60/316L SS biocomposite surfaces, in which the material surfaces are dissolved firstly and then the apatite layers are precipitated on the surfaces. The formation of apatite in SBF is
10Ca2++(6-x)PO43-+2OH-+xCO32-→
Ca10(PO4)6-x(CO3)x(OH)2 (1)
On one hand, the dissolving of the surface layer makes the Ca ion concentration in SBF increase. On the other hand, Ca ions and P ions are consumed because of nucleation and growth of apatite. During early stage, the dissolving rate of Ca ions on the surface outclasses the precipitation rate. With prolonging immersion time, the
dissolving rate and the precipitation rate of Ca ions in SBF approach equilibrium, so the increase of Ca ion concentration becomes slower and slower (Fig.2(a)). However, the P ion concentration decreases in some sort during early stage, which indicates that the precipitation rate of P ions in SBF is higher than the dissolving rate because of being effected by the dissolving of Ca ions. During late stage, the P ion concentration increases continuously due to dissolving of P ions on the surface (Fig.2(b)).
3.3 SEM characterization and component analysis
Fig.3 shows the surface morphologies of HA60/ 316L SS biocomposites after immersion in SBF for various periods of time. EDX spectra of HA60/316L SS biocomposites after soaking in SBF for various periods of time are shown in Fig.4.
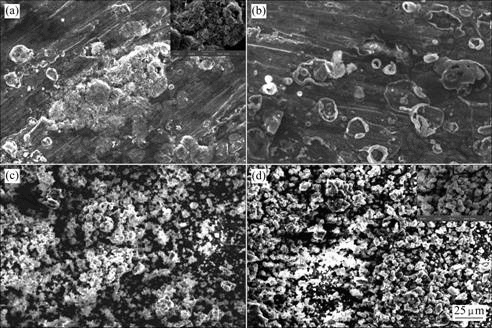
Fig.3 Surface morphologies of HA60/316L SS biocomposites after soaking in SBF for various periods of time: (a) 1 d; (b) 4 d; (c) 7 d; (d) 14 d
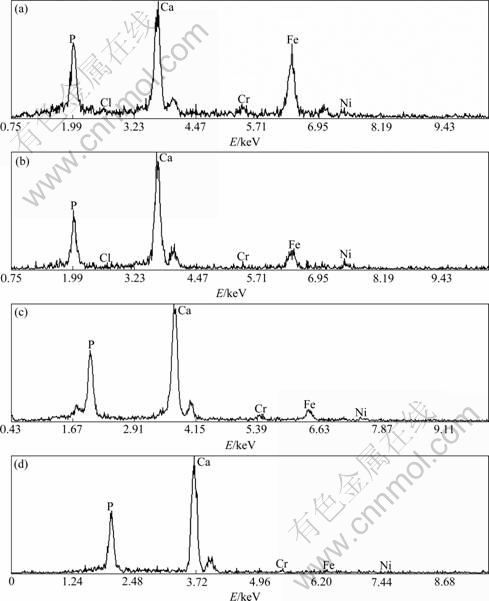
Fig.4 EDX spectra of HA60/316L SS biocomposites after soaking in SBF for various periods of time: (a) 1 d; (b) 4 d; (c) 7 d; (d) 14 d
After soaking for 1 d in SBF for HA60/316L SS biocomposites, there are leaf-like apatite crystals on the surfaces of HA60/316L SS biocomposites (inset in Fig.3(a)) and the crystals develop spherical particles gradually (Fig.3(a)). With prolonging immersion time, the spherical particles accumulate continuously and the
whole surface is covered with apatite layer, what is more, the layer becomes thicker and thicker increasingly. During the late immersion time, no leaf-like apatite crystal is seen on the surfaces even in the newly precipitated apatite crystals(Figs.3(b)-(d)).
According to the mechanics of formation of bone-like apatite[22-23], in the initial immersion in SBF, Ca and P ions on the biocomposite surfaces are dissolved into SBF; however, the supersaturation of Ca and P ions is low. Therefore, the apatite layer is nucleated where the local ion concentrations are high comparatively and few crystal nuclei are formed (Figs.3(a) and (b)). The crystal nuclei adsorb the ions from their own surrounding environment and develop in their growth process. Because the adsorption on the biocomposite surfaces competes with each other, the growth way of apatite is preferential orientation. All these lead to the formation of leaf-like apatite in the initial stage of immersion. But as the immersion time prolongs, more and more Ca and P ions in SBF are dissolved and the growth of apatite is out of restriction gradually. At the co-effect of surface tension and adhesion force around crystals and the lowest energy principle, the spherical apatite particles are obtained. With further prolonging immersion time, Ca and P ion concentrations continue to increase and the nucleation on surface becomes much rapid. Therefore, the preferential orientation of crystal growth disappears, and the leaf-like crystals are transited to polycrystals, namely, it is polycrystal nucleation mechanism during the late stage and the leaf-like crystals cannot be observed in the late stage of immersion[24-25]. Thus, it can be seen that existence of local Ca and P ion concentrations is not only important because of apatite formation but also a key factor with relation to crystal morphology.
The EDX spectrum in Fig.4(a) shows that the dominating elements on the biocomposite surfaces are Ca, P, Fe, Cr and Ni after immersion in SBF. With prolonging immersion time, Fe, Cr and Ni on the surface decrease gradually before 7 d. The content of 316L SS is scarcely observed on the surface and only Ca and P can be examined on the biocomposite surfaces after immersion for 14 d (Figs.4(b)-(d)). It reveals that the apatite layer containing rich Ca and P on the biocomposite surfaces is formed. However, the apatite layer as formed on the biocomposite surfaces is different from the HA of biocomposites. Based on the calculation results of EDX spectra in Figs.4(a)-(d), the ratio of Ca and P is small (only 0.95) for 1 d immersion in SBF because of the influence of the substrate materials. The ratio of Ca and P increases with prolonging immersion time and it maintains a constant, about 1.5, when the thickness of apatite layer increases to a certain extent. The gained maximal ratio is lower than the theoretical value (1.67), which shows that the newly formed apatite layer is a Ca-deficient apatite layer that is consistent with the properties of bone-like apatite[26] and with no relation to the substrate. That is to say, the Ca-deficient apatite is bone-like apatite. The formation of such bone-like apatite eventually achieves osseointegration. Osseointegration provides firm fixation between HA60/316L SS biocomposite implants and human body
bone[27]. Accordingly, HA60/316L SS biocomposites are those which possess benign bioactivity and favorable biocompatibility and accord with the requirement of bioactivity and biocompatibility for biomedical materials.
The analysis results above indicate that the bioactive bone-like apatite can be formed on the biocomposite surfaces during the immersion in SBF. Therefore, the prepared HA/316L SS biocomposites are bioactive materials for biomedical applications.
4 Conclusions
1) The apatite layer formation on the surface of HA/316L SS biocomposites is investigated by SEM and EDX for the analysis of components and ion concentrations of apatite layers. The results show that the Ca-deficient apatite is bone-like apatite with excellent bioactivity.
2) The HA/316L SS biocomposites are excellent bioactive materials and possess promising applications as biomedical materials.
References
[1] DERAM V, MINICHIELLO C, VANNIER R N, LE MAGUER A, PAWLOWSKI L, MURANO D. Microstructural characterizations of plasma sprayed hydroxyapatite coatings [J]. Surface and Coatings Technology, 2003, 166(2/3): 153-159.
[2] WANG Xue-jiang, WANG Jian-xin, LI Yu-bao, FENG Jian-qing, YAN Yong-gang, HUANG Mei, YANG Ai-ping. Preparation of nanograde hydroxyapatite needle-like crystals under normal atmospheric pressure [J]. High Technology Letters, 2001(11): 92-94. (in Chinese)
[3] CIZEK J, KHOR K A, PROCHAZKA Z. Influence of spraying conditions on thermal and velocity properties of plasma sprayed hydroxyapatite [J]. Mater Sci Eng C, 2007, 27(2): 340-344.
[4] MORRA M, CASSINELLI C. Organic surface chemistry on titanium surfaces via thin film deposition [J]. Journal of Biomedical Materials Research, 1997, 37(2): 198-206.
[5] MONTASSER M. DEWIDAR, KHALIL A, LIM J K. Processing and mechanical properties of porous 316L stainless steel for biomedical applications [J]. Trans Nonferrous Met Soc China,2007, 17(3): 468-473.
[6] XU Rui-dong, GUO Zhong-cheng, PAN Jun-yi. Corrosion resistance of electrodeposited RE-Ni-W-P-SiC-PTFE composite coating in phosphoric and ferric chloride [J]. Trans Nonferrous Met Soc China, 2006, 16(3): 666-670.
[7] RODRIGUEZ H H, VARGAS G, CORTES D A. Electrophoretic deposition of bioactive wollastonite and porcelain-wollastonite coatings on 316L stainless steel [J]. Ceramics International, 2008, 34(5): 1303-1307.
[8] LIN J H, LOUB C W, CHANGA C H, CHENC Y S, LIN G T, LEE C H. In vitro study of bone-like apatite coatings on metallic fiber braids [J]. Journal of Materials Processing Technology, 2007, 192/193: 97-100.
[9] BALAMURUGAN A, BALOSSIER G, KANNAN S, RAJESWARI S. Elaboration of sol-gel derived apatite films on surgical grade stainless steel for biomedical applications [J]. Materials Letters, 2006, 60(17/18): 2288-2293.
[10] NOTINGHER I, BOCCACCINI A R, JONES J, MAQUET V, HENCH L L. Application of Raman micro spectroscopy to the characterization of bioactive materials [J]. Materials Characterization, 2002, 49(3): 255-260.
[11] ZHANG Yu-jun, YIN Yan-sheng, WANG Ying-jun. Advancement of hydroxyapatite-based bioceramic composites [J]. Journal of Biomedical Engineering, 1999, 16(S): 37-39. (in Chinese)
[12] HENCH L L. Biomaterials: a forecast for the future [J]. Biomaterials, 1998, 19(16): 1419-1423.
[13] KOKUBO T. Apatite formation on surfaces of ceramics, metals polymers in body environment [J]. Acta Mater, 1998, 46(7): 2519-2527.
[14] BRUIJN J D, BOVELL Y P, BLITTERSWIJK C A. Structural arrangements at the interface between plasma sprayed calcium phosphates and bone [J]. Biomaterials, 1994, 15(7): 543-550.
[15] KOKUBO T, TAKADAMA H. How useful is SBF in predicting in vivo bone bioactivity [J]. Biomaterials, 2006, 27(15): 2907-2915.
[16] MIAO X. Observation of microcracks formed in HA-316L composites [J]. Materials Letters, 2003, 57(12): 1848-1853.
[17] HE Z M, PLATZEK D, STIEWE C, CHEN H Y, KARPINSKI G, M?LLER E. Thermoelectric properties of hot-pressed Al- and Co-doped iron disilicide materials [J]. Journal of Alloys and Compounds, 2007, 438(1/2): 303-309.
[18] KOKUBO T, KIM H M, MIYAJI F, TAKADAMA H, MIYAZAKI T. Ceramic-metal and ceramic-polymer composites prepared by a biomimetic process [J]. Composites part A: Applied Science and Manufacturing, 1999, 30(4): 405-409.
[19] KOKUBO T, KUSHITANI H, SAKKA S, KITSUGI T, YAMAMURO T. Solution able to reproduce in vivo surface- structure changes in bioactive glass-ceramic A-W [J]. Journal of Biomedical Materials Research, 1990, 24(6): 721-734.
[20] LI P, OHTSUKI C, KOKUBO T, NAKANISHI K, SOGA N, de GROOT K. The role of hydrated silica, titania and alumina in inducing apatite on implants [J]. Journal of Biomedical Materials Research, 1994, 28(1): 7-15.
[21] ZHANG Xiao-kai, LIU Wei, CHEN Xiao-feng. Morphology character of the sol-gel derived bioactive glass in SBF solution [J]. Chinese Journal of Chemical Physics, 2004, 17(4): 495-498. (in Chinese)
[22] PRAMATAROVA L, PECHEVA E. Modified inorganic surfaces as a model for hydroxyapatite growth [C]// MARPLE B R, MOREAU C. Materials Science Foundations. Switzerland: Trans Tech Publications, 2006: 118-122.
[23] EICHERT D, COMBES C, DROUET C, REY C. Formation and evolution of hydrated surface layers of apatites [J]. Key Engineering Materials, 2005, 284(3/6): 105-108.
[24] KOKUBO T, HANAKAWA M, KAWASHITA M, MINODA M, BEPPU T, MIYAMOTO T, NAKAMURA T. Apatite formation on non-woven fabric of carboxymethylated chitin in SBF [J]. Biomaterials, 2004, 25(18): 4485-4488.
[25] ZHANG Qi-yi, CHEN Ji-yong, CAO Yang, FENG Jia-ming, ZHANG Xing-dong. Comparative study of bone-like apatite formation on plasma-sprayed coatings in static and flowing simulated body fluids(SBF) [J]. Journal of Sichuan University: Natural Science Edition, 2002, 39(6): 1085-1088. (in Chinese)
[26] FU Tao, HUANG Ping, HAN Yong, XU Ke-wei. Apatite formation induced by alkaline treatment on titanium alloy substrate [J]. Rare Metal Materials and Engineering, 2000, 29(3): 168-171. (in Chinese)
[27] HAZAN R, BRENER R, ORON U. Bone growth to metal implants is regulated by their surface chemical properties [J]. Biomaterials, 1993, 14(8): 570-574.
Foundation item: Project(50774096) and project (50604017) supported by the National Natural Science Foundation of China; Project(1343-74334000011) supported by the Postgraduate Education and Innovation Project from Central South University, China
Corresponding author: RUAN Jian-ming; Tel: +86-731-8836827; E-mail: jianming@mail.csu.edu.cn
DOI: 10.1016/S1003-6326(08)60276-9

