Isolation and characterization of acidophilic bacterium fromDongxiangshan Mine in Xinjiang Province, China
来源期刊:中南大学学报(英文版)2010年第1期
论文作者:夏金兰 张倩 张瑞永 彭娟花 彭安安 赵小娟 聂珍媛 邱冠周
文章页码:50 - 55
Key words:Acidithiobacillus ferrooxidans; bioleaching; iro and tth gene; 16S rRNA
Abstract: One bioleaching bacterium, named as strain DXS, was isolated from acid mine drainages (AMDs) of Dongxiangshan Mine of Hami, Xinjiang Province, China. The strain DXS is gram-negative and rod-shaped with a size of (0.40±0.05) μm×(1.3±0.5) μm. The optimal temperature and pH for growth are 30 ℃ and pH 2.0, respectively. It can grow autotrophically by using ferrous iron, elemental sulfur and NaS2O3 as sole energy sources. In the phylogenetic tree, strain DXS has similarity with Acidithiobacillus ferrooxidans type strain ATCC 23270 with 99.57% sequence similarity. The cloning and sequencing of Iro protein gene (iro) and tetrathionate hydrolase gene (tth) reveal that strain DXS is completely identical in iro gene sequence to A. ferrooxidans LY (DQ166841), and almost identical in tth gene sequene to A. ferrooxidans (AB259312) (only two nucleotides change). The bioleaching experiments of marmatite and pyrite reveal that the leached zinc and iron concentrations reach 3.01 g/L and 2.75 g/L, respectively. The strain has a well potential application in industry bioleaching.
基金信息:the National Natural Science Foundation of China
the National Basic Research Program of China
J. Cent. South Univ. Technol. (2010) 17: 50-55
DOI: 10.1007/s11771-010-0010-x ![]()
XIA Jin-lan(夏金兰)1, 2, ZHANG Qian(张倩)1, 2, ZHANG Rui-yong(张瑞永)1, 2, PENG Juan-hua(彭娟花)1, 2,
PENG An-an(彭安安)1, 2, ZHAO Xiao-juan(赵小娟)1, 2, NIE Zhen-yuan(聂珍媛)1, 2, QIU Guan-zhou(邱冠周)1, 2
1. Key Laboratory of Biometallurgy of Ministry of Education, Central South University, Changsha 410083, China;
2. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: One bioleaching bacterium, named as strain DXS, was isolated from acid mine drainages (AMDs) of Dongxiangshan Mine of Hami, Xinjiang Province, China. The strain DXS is gram-negative and rod-shaped with a size of (0.40±0.05) μm×(1.3±0.5) μm. The optimal temperature and pH for growth are 30 ℃ and pH 2.0, respectively. It can grow autotrophically by using ferrous iron, elemental sulfur and NaS2O3 as sole energy sources. In the phylogenetic tree, strain DXS has similarity with Acidithiobacillus ferrooxidans type strain ATCC 23270 with 99.57% sequence similarity. The cloning and sequencing of Iro protein gene (iro) and tetrathionate hydrolase gene (tth) reveal that strain DXS is completely identical in iro gene sequence to A. ferrooxidans LY (DQ166841), and almost identical in tth gene sequene to A. ferrooxidans (AB259312) (only two nucleotides change). The bioleaching experiments of marmatite and pyrite reveal that the leached zinc and iron concentrations reach 3.01 g/L and 2.75 g/L, respectively. The strain has a well potential application in industry bioleaching.
Key words: Acidithiobacillus ferrooxidans; bioleaching; iro and tth gene; 16S rRNA
1 Introduction
Bacterial leaching of metal sulfides has developed rapidly in the last decades, and it is often considered more environmentally friendly and economical than other available technologies [1-4]. Recovering metals from sulphide minerals by acidophiles has developed into a successful and expanding area of biotechnology. Up to now, many bioleaching microbes have been found, such as the genus Acidithiobacillus, Leptospirillum, Ferroplasma, Sulfobacillus and Sulfolobus. The classical leaching bacteria now belong to the genus Acidithiobacillus, mainly including A. ferrooxidans, A. thiooxidans, A. albertensis and A. caldus [5-7].
Acidithiobacillus ferrooxidans (A. ferrooxidans, formerly named Thiobacillus ferrooxidans) is an acidophilic chemolithotrophic gram-negative bacterium, which widely exists in mesophilic bioleaching systems. A. ferrooxidans has attracted great interest because of its use in industrial mineral processing and unusual physiology. It can oxidize ferrous iron, elemental sulfur, reduced sulfur compounds and sulfide minerals. This ability makes it suitable for biomining to recover metals such as copper, gold and uranium [8].
The remarkable feature of the species A. ferrooxidans is that it derives energy from the oxidation of ferrous iron or reduced sulfur compounds. This special metabolism is important in highly acidic environments and metal bioleaching.
Based on the great importance of A. ferrooxidans in hydrometallurgy, the isolation of more efficient strains for the practical applications is urgent affair especially for complicated metal sulfide minerals. A number of A. ferrooxidans were isolated from acidic water samples such as acidic mine drainages and acidic hot springs all over the world, and these, as a result, improved leaching rate of metal sulfides [9-11].
In present work, a study was undertaken to identify a newly isolated A. ferrooxidans strain DXS from the acid mine drainages of Dongxiangshan Mine of Hami, Xinjiang Province, China. A series of morphological and biochemical characterizations as well as the analysis of 16S rRNA sequences were done.
2 Experimental
2.1 Strain isolation
A water sample was collected from acid mine drainages of copper mines with a temperature of 20 ℃and pH 3.0, located at Hami, Xinjiang Province China. Isolation and purification were done as follows: the sample was inoculated onto the ferrous-agarose solid medium plate, and incubated at 30 ℃ for 8-10 d until the colonies appeared. A single colony was then picked out for next inoculation. The transfer was repeated until the pure culture was obtained. Their identities were confirmed based on their phenotypic characteristics and 16S rRNA sequences.
2.2 Growth conditions
If not stated otherwise, strain DXS was cultivated in 250 mL flasks with 100 mL modified medium 9K. 9K medium was prepared as follows: solution A (700 mL): 3 g/L (NH4)2SO4, 0.5 g/L K2HPO4, 0.5 g/L MgSO4, 0.1 g/L KCl, 0.01 g/L Ca(NO3)2; solution B (300 mL): 44.7 g/L FeSO4・7H2O or 5.0 g/L S0. Solutions A and B were mixed after being autoclaved separately. The initial pH of the medium was adjusted to 2.0 and 2.5. Ferrous- agarose solid plates were prepared by adding 1% agar powder to the liquid 9K medium above. When a parameter that influenced the growth of the A. ferrooxidans was changed, other parameters were kept at optimum. Strains were cultivated in 250 mL flasks containing 100 mL of 9K medium on a shaker at 180 r/min and 30 ℃. The inoculum size comprised 2% (volume fraction) of total culture and the bacteria were cultivated to the stationary growth phase.
2.3 Microscopic studies
Cell motile behavior was observed using Olympus CX 31 optical microscope, and Gram staining was performed using a Gram stain reagent kit (Haitai Biotech, China). Fine morphological features were revealed by scanning electron microscope (SEM, JEOL JSM-6360 LV) as described previously [12].
2.4 Biochemical and physiological experiments
The optimum pH and temperature of the strain DXS were determined in pH- and temperature-controlled cultures, respectively. The isolated DXS grew in 9K medium with the culture maintained at pH 2.0 (to determine the optimum temperature) or 30 ℃ (to determine the optimum pH). The following organic compounds were tested as possible substrates at different concentrations with or without ferrous iron. Growth was estimated after incubation for 72 h. The possible substrates used were as follows: peptone (0.1%), glucose (0.1%), sodium thiosulfate (1.0%), sulphur powder (5%), FeSO4・7H2O (14.7%), FeSO4・7H2O (14.7%)+peptone (1.0%), FeSO4・7H2O (14.7%)+glucose (0.1%), pyrite (0.5%) and marmatite (0.5%), respectively. All supplements were asepticized.
2.5 Sulfur or ferrous iron oxidizing activity
The oxidizing activity of ferrous iron and elemental sulfur was comparatively studied with the A. ferrooxidans ATCC 23270. When ferrous iron was used as the sole energy source, the content of Fe3+ in the medium was determined as described by YANG et al [13]. When elemental sulfur was used as the sole energy source, the concentration of sulfate of the medium was measured as described by ZHANG et al [14].
2.6 Genomic DNA extraction and phylogenetic analysis of 16S rRNA
Genomic DNA was extracted using an EZ-10 Spin Column Genomic DNA Minipreps Kit (Bio Basic Inc., Canada) according to the introduction of the kit. The primers for amplification of 16S rRNA were designed based on the previous report [15]. The PCR amplification was carried out according to the method described by PENG et al [16]. When the PCR program was finished, the PCR product was separated by gel electrophoresis on a 1.0% agar gel in tris/acetate-buffer and analyzed by staining with ethidium bromide (EB) under UV light. The band of the expected size (approximately 1 400 bp) was cut off and purified with a commercial kit (Gel Extraction Kit, Promega, USA). The purified PCR product was ligated into pGM-T vector(Tiangen Biotech, China). The positive clone containing insert was screened out by colony PCR and sequenced by Sunbiotech Company (Beijing, China).
The nucleotide sequence was searched for homology by BlastN at the NCBI server (http:// ww.ncbi. nlm.nih.gov/BLAST/Blast) and submitted to Genbank (Table 1). All available subsets of 16S rRNA gene sequences were selected, analyzed and aligned with CLUSTALX 1.8 [17], and the final phylogenetic tree was generated by MEGA 4.0.
2.7 Cloning and sequencing of iro and tth genes
The iro and tth genes were amplified by PCR. Two pairs of primers were designed based on the nucleotide sequences of iro and tth genes of type strain ATCC 23270 (Table 1). The PCR program consisted of one cycle of DNA denaturation for 3 min at 95 ℃, and then 35 cycles were performed as follows: 45 s at 94℃ to denature, 30 s at 55 ℃ to anneal and 60 or 90 s at 72 ℃ to extend. The PCR products were purified and sequenced as described above. The Genbank accession number of each sequence is shown in Table 1.
Table 1 Primers used in this work

2.8 Bioleaching of marmatite and pyrite
Marmatite and chalcopyrite used in this experiment were provided by Institute of Minerals Processing Engineering, Central South University, China. The marmatite contains 48.4% of Zn, 20.2% of S0 and 1.1% of Fe, and the pyrite contains 44.8% of Fe, 47.8% of S0, and 0.3% of Cu.
Bioleaching tests were carried out in 250 mL flasks containing 100 mL 9K medium. The mineral concentration was 3% (mass fraction). The inoculum of strain DXS culture was 10% (volume fraction), and all the experiments were carried out in triplicate. Abiotic controls were also designed by replacing the bacterial inoculum by an equal volume of related medium. Aliquots of leachate were sampled, and the concentrations of zinc and iron were determined by atomic absorption spectrometry (Hitachi Z-8000) within 28 d of incubation. The lost water in the medium was supplemented with sterilized deionized water after sampling each time.
3 Results and discussion
3.1 Isolation and morphological observation
By streak plate method, a mesophilic acidophilic bacterium named strain DXS was isolated from Dongxiangshan Mine, Xinjiang Province, China. Single colonies appear within 10 d at 30 ℃. Colonies of the isolate are circular, convex and maroon (Fig.1(a)). It is gram-negative, long rod-shaped. The cell of strain DXS is 0.8-1.5 μm in length and about 0.4 μm in diameter. The morphological character is shown in Fig.1(b).

Fig.1 Colonies (a) and scanning electron micrograph (b) of strain DXS
3.2 Biochemical and physiological characteristics
3.2.1 Temperature and pH
The growth status of strain DXS at different temperatures is shown in Fig.2. It is indicated that the strain is mesophilic and able to grow in a temperature range from 20 to 35 ℃, with an optimum at 30 ℃. No growth is detected at 45 ℃ or at the temperature higher than 45 ℃ after 7 d of inoculation. Strain DXS grows in a pH range from 1.0 to 3.0, with optimal growth at pH 2.0 (Fig.3), but no growth is detected at pH lower than 0.5 or higher than 3.5.
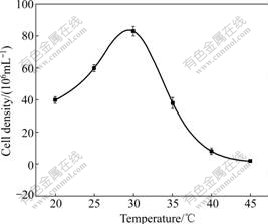
Fig.2 Effect of temperature on growth of strain DXS
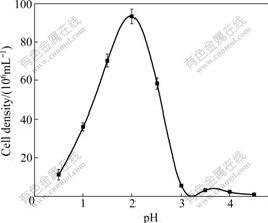
Fig.3 Effect of pH on growth of strain DXS
According to the optimum growth parameters as mentioned above, experiment was carried out in the optimal growth condition of strain DXS. The experiment performed at 30 ℃, pH 2.0 and 180 r/min, 5% of the seed culture of stable growth stage was inoculated to medium with addition of 14.7% FeSO4・7H2O or 0.5% S0. The growth curves of isolate DXS are shown in Fig.4. The data show that the strain can be activated in a short time, and the number of cells reaches the maximum (about 5.2×107 mL-1) after 40 h when grown on FeSO4・7H2O. When grown on S0, the strain requires 160 h to reach the maximum, however, the bacterial density reaches 10.1×107 mL-1, which is much higher than that grown on FeSO4・7H2O.
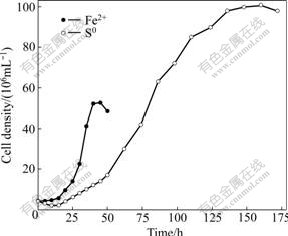
Fig.4 Growth curves of strain DXS using energy source of Fe2+ or S0
3.2.2 Utilization of substrates
Strain DXS can grow autotrophically by using ferrous iron or elemental sulfur as sole energy source. The strain is sensitive to organic acids and other small molecular organic compounds, which can inhibit the growth of these microbes. In addition, strain DXS can grow by oxidizing pyrite and marmatite.
3.2.3 Sulfur and ferrous iron oxidizing activity
The oxidizing activities of ferrous iron and sulfur of strain DXS are shown in Fig.5 and Fig.6, respectively. This indicates that strain DXS shows a higher oxidation activity than the A. ferrooxidans ATCC 23270 when using sulfur or ferrous iron, especially ferrous iron.
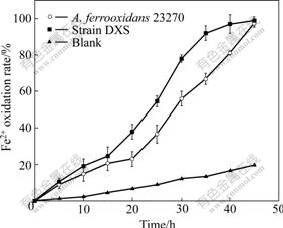
Fig.5 Ferrous iron oxidation of strain DXS
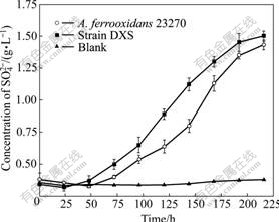
Fig.6 Sulfur oxidation of strain DXS
3.3 Phylogenetic analysis
The nearly complete sequence consisting of 1 395 bases of the amplified 16S rRNA of strain DXS was determined. 16S rRNA sequence analysis indicates that the closest relative of strain DXS is A. ferrooxidans, with a 16S rRNA sequence similarity over 99%. Phylogenetic tree based on 16S rRNA sequences of DXS and the species within genus Acidithiobacillus was constructed using softwares of Clustal X1.8 and MEGA 4.0 (Fig.7). The results also clearly show that strain DXS is most closely related to A. ferrooxidans.

Fig.7 Phylogenetic tree based on 16 rRNA gene sequences of strain DXS and species within Acidithiobacillus (GenBank accession numbers are shown in parentheses and numbers depict bootstrap values obtained for a bootstrap sampling of 1 000)
3.4 Cloning and sequencing of iro and tth genes
The protein encoded by the iro gene (Iro protein) is considered to be a member of the high-potential iron- sulfur protein (HiPIP) family. The Iro protein has been proposed to be the first electron carrier in the ferrous iron respiratory chain between ferrous iron and oxygen [18]. However, recent studies have suggested that the Iro protein is involved in the electron transfer chain by transferring electrons from a cytochrome bc1 complex to a terminal oxidase [19]. These studies suggested an important role for the Iro protein in the oxidation of Fe2+, although its exact physiological role has not been determined [18-19].
Tetrathionate is one of the most important intermediates in dissimilatory sulfur oxidation and can itself be utilized as a sole energy source by some sulfur- oxidizing microorganisms. Tetrathionate hydrolase plays a significant role in tetrathionate oxidation and should catalyze the initial step in the oxidative dissimilation when sulfur-oxidizing bacteria grow on tetrathionate [20]. Investigations of the detailed regulatory mechanism for the expression of Af-tth and the synthesis of recombinant Tth enzyme as an active form are now in progress to clarify the mechanism of dissimilatory sulfur oxidation in A. ferrooxidans.
In this work, PCR amplification of iro and tth genes was performed with the gene specific primers. The results reveal that iro and tth gene fragments of the expected size can be amplified from the genomic DNA of strain DXS, and the nucleotide sequence of the iro gene from strain DXS is completely identical to that of strain LY (DQ166841). The nucleotide sequence of the tth gene of strain DXS is almost identical (only two nucleotides changed) to that of A. ferrooxidans (AB259312). The nucleotide sequences of the iro and tth genes were submitted to Genbank (Table 1).
3.5 Bioleaching of marmatite and pyrite
Bioleaching of marmatite with A. ferrooxidans DXS is shown in Fig.8. In the whole process, zinc extraction rate continuously increases. In the first 18 d, zinc extraction rate increases quickly and after then zinc concentration reaches 2.81 g/L, However, zinc concentration leached by A. ferrooxidans ATCC 23270 reaches 1.85 g/L. From the 20th day to the 28th day, zinc extraction becomes slow. The final zinc concentration is 3.01 g/L.
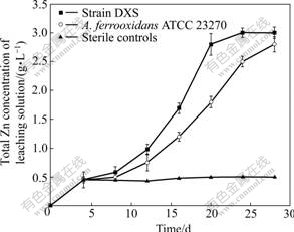
Fig.8 Zinc extraction from marmatite by A. ferrooxidans DXS
The bioleaching results of pyrite by A. ferrooxidans DXS are shown in Fig.9. In the leaching process, total iron extraction continuously increases and finally reaches 2.75 g/L after 28 d. Comparatively, iron concentration leached by A. ferrooxidans ATCC 23270 is 2.41 g/L, which is less than that by A. ferrooxidans DXS. Almost no soluble iron is detected in sterile controls all the time. The data indicate that A. ferrooxidans DXS has great capacity of pyrite leaching. The leaching result is consistent with its high oxidizing activity of sulfur and ferrous iron.
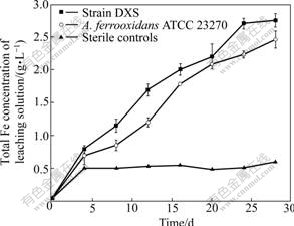
Fig.9 Iron extraction from pyrite by A. ferrooxidans DXS
4 Conclusions
(1) Acidithiobacillus ferrooxidans DXS, an acidophilic bacterium from acidic mine drainages from Dongxiangshan Mine of Hami, Xinjiang Province, China is isolated. The strain grows at pH value of 1.0-3.0 and temperature of 20-35 ℃, with optimal pH and temperature at 2.0 and 30 ℃, respectively. The cells of the strain are in shape of short rod, with (1.3±0.5) μm in length and (0.40±0.05) μm in diameter. The optimum temperature and pH for strain DXS growth are 30 ℃ and 2.0, respectively. It can use ferrous iron, elemental sulfur and NaS2O3 as energy sources. It cannot, however, grow on substrate of glucose, yeast extract, peptone or other alternative organic energy sources.
(2) 16S rRNA sequence analysis indicates that the closest relative of strain A. ferrooxidans DXS is the type strain ATCC 23270, with a 16S rRNA sequence similarity over 99.57%. Strain DXS is completely identical in Iro protein gene (iro) to A. ferrooxidans LY (DQ166841), and almost identical in tth gene to Acidithiobacillus ferrooxidans (AB259312) (only two nucleotides changed).
(3) A. ferrooxidans DXS can oxidize Fe2+ and S0. Bioleaching of marmatite and pyrite with strain DXS can attain good performance.
References
[1] Krebs W, Brombacher C, Bosshard P P, Bachofen R, Brandl H. Microbial recovery of metals from solids [J]. FEMS Microbiology Reviews, 1997, 20(3/4): 605-617.
[2] GOMEZ E, BALLESTER A, BLAZQUEZ M L, GONZALEZ F. Silver catalysed bioleaching of a chalcopyrite concentrate with mixed cultures of moderately thermophilic microorganisms [J]. Hydrometallurgy, 1999, 51(1): 37-46.
[3] Witne J Y, Phillips C V. Bioleaching of Ok Tedi copper concentrate in oxygen- and carbon dioxide-enriched air [J]. Minerals Engineering, 2001, 14(1): 25-48.
[4] BRIERLEY C L. How will biomining be applied in future? [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(6): 1302-1310.
[5] LEDUC L G, FERRONI G D. The chemolithotrophic bacterium Thiobacillus ferrooxidans [J]. FEMS Microbiology Reviews, 1994, 14(2): 103-119.
[6] KELLY D P, WOOD A P. Reclassification of some species of Thiobacillus to the newly designated genera Acidithiobacillus gen. nov., Halothiobacillus gen. nov. and Thermithiobacillus gen.nov. [J]. International Journal of Systematic and Evolutionary Microbiology, 2000, 50(2): 511-516.
[7] ROHWERDER T, GEHRKE T, KINZLER K. Bioleaching review A: Progress in bioleaching fundamentals and mechanisms of bacterial sulfide oxidation [J]. Applied and Environmental Microbiology, 2003, 63(3): 239-248.
[8] MERROUN M L, GEIPEL G, NICOLAI R, HEISE K H, SELENSKA-POBELL S. Complexation of uranium (VI) by three ecotypes of Acidithiobacillus ferrooxidans studied using time-resolved laser-induced fluorescence spectroscopy and infrared spectroscopy [J]. Biometals, 2003, 16: 331-339.
[9] WU Xue-ling, JIANG Ying, QIU Guan-zhou, LIU Xin-xing, WENG Wen-yun. Isolation of Acidithiobacillus ferrooxidans strains and effect of anions on their ferrous irons oxidation capacities [J]. The Chinese Journal of Nonferrous Metals, 2008, 18(2): 349-355. (in Chinese)
[10] SUGIO T, WAKABAYASHI M, KANAO T, TAKEUCHI F. Isolation and characterization of Acidithiobacillus ferrooxidans strain D3-2 active in copper bioleaching from a copper mine [J]. Bioscience, Biotechnology and Biochemistry, 2008, 72(4): 998-1004.
[11] YANG Yu, PENG Hong, WAN Min-xi, QIU Guan-zhou, HUANG Ju-fang, HU Yue-hua. Identification of new-subspecies Acidithiobacillus ferrooxidans strain from complex sulfide mines [J]. Transactions of Nonferrous Metals Society of China, 2006, 16(6): 1094-1099.
[12] HIRAISHI A, URATA K, SATOH T. A new genus of marine budding phototrophic bacteria, Rhodobium gen. nov., which includes Rhodobium orientis sp. nov., and Rhodobium marinum comb. nov [J]. International Journal of Systematic and Evolutionary Microbiology, 1995, 45(2): 226-234.
[13] YANG Yu, WAN Min-xi, PENG Hong, QIU Guan-zhou, HUANG Ju-fang, HU Yue-hua. Isolation and charaterization of the bacterium for chalcopyrite bioleaching [J]. Journal of Central South University: Science and Technology, 2007, 38(4): 639-644. (in Chinese)
[14] ZHANG Cheng-gui, XIA Jin-lan, ZHANG Rui-yong, PENG An-an, NIE Zhen-yuan, QIU Guan-zhou. Comparative study on effects of Tween-80 and sodium isobutyl-xanthate on growth and sulfur-oxidizing activities of Acidithiobacillus albertensis BY-05 [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(4): 1003-1007.
[15] DING Jian-nan, HE Huan, ZHANG Cheng-gui, YU Yi-zun, QIU Guan-zhou. Isolation and characterization of YNTC-1, a novel Alicyclobacillus sendaiensis strain [J]. Journal of Central South University of Technology, 2008, 15(4): 508-514.
[16] PENG Juan-hua, ZHANG Rui-yong, ZHANG Qian, ZHANG Li-min, ZHOU Hong-bo. Screening and characterization of Acidiphilium sp. PJH and its role in bioleaching [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(6): 1443-1449.
[17] THOMPSON J D, GIBSON T J, PLEWNIAK F, JEANMOUGIN F, HIGGINS D G. The CLUSTAL_X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools [J]. Nucleic Acids Research, 1997, 25(24): 4876-4882.
[18] YAMANAKA T, FUKUMORI Y. Molecular aspects of the electron transfer system which participates in the oxidation of ferrous iron by Thiobacillus ferrooxidans [J]. FEMS Microbiology Letters, 1995, 17(4): 401-413.
[19] BRUSCELLA P, CASSAGNAUD L, RATOUCHNIAK J, BRASSEUR G, LOJOU E, AMILS R, BONNEFOY V. The HiPIP from the acidophilic Acidithiobacillus ferrooxidans is correctly processed and translocated in Escherichia coli, in spite of the periplasm pH difference between these two microorganisms [J]. Microbiology, 2005, 151(5): 1421-1431.
[20] KANAO T, KAMIMURA K, SUGIO T. Identification of a gene encoding a tetrathionate hydrolase in Acidithiobacillus ferrooxidans [J]. Journal of Biotechnology, 2007, 132(1): 16-22.
Foundation item: Projects(50974140, 50674101) supported by the National Natural Science Foundation of China; Project(2010CB630902) supported by the National Basic Research Program of China
Received date: 2009-01-18; Accepted date: 2009-04-20
Corresponding author: XIA Jin-lan, PhD, Professor; Tel: +86-731-88836944; Email: jlxia@mail.csu.edu.cn
(Edited by YANG You-ping)

