
Comparison of Sb and Sr on modification and refinement of Mg2Si phase in AZ61-0.7Si magnesium alloy
YANG Ming-bo(杨明波)1, 2, PAN Fu-sheng(潘复生)2, SHEN Jia(沈 佳)1, BAI Liang(白 亮)2
1. Materials Science and Engineering College, Chongqing Institute of Technology, Chongqing 400050, China;
2. Materials Science and Engineering College, Chongqing University, Chongqing 400030, China
Received 15 April 2008; accepted 20 June 2008
Abstract: The effects of Sb and Sr on the modification and refinement of Mg2Si phase in an AZ61-0.7Si magnesium alloy were investigated and compared. The results indicate that the difference of Sb and Sr in the modification and refinement of Mg2Si phase is significant. Addition of 0.4%Sb (mass fraction) to AZ61-0.7Si alloy can refine but not modify Mg2Si phase. Oppositely, by addition of 0.12%Sr to AZ61-0.7Si alloy, the Mg2Si phase changes from initial coarse Chinese script shape to fine granule and/or irregular polygonal shape. Accordingly, the Sr-modified AZ61-0.7Si alloy exhibits higher tensile and creep properties than the Sb-modified one. The difference of Sb and Sr in the modification and refinement of Mg2Si phase is possibly related to the following two aspects: 1) the disregistry at (100)Al4Sr//(100)Mg2Si is lower than that at (0001)Mg3Sb2//(111)Mg2Si (0.69% vs 1.72%) and 2) the onset crystallizing temperature of the Sr-modified AZ61-0.7Si alloy is lower than that of the Sb-modified one (603.1 ℃ vs 606.9 ℃).
Key words: magnesium alloy; AZ61-0.7Si alloy; Mg2Si phase; modification
1 Introduction
It is well known that Mg-Al-Si based alloys are potential high temperature lightmass materials[1-2] due to the existence of Mg2Si phase that has high melting point, high hardness, low density, high elastic modulus and low thermal expansion coefficient. Furthermore, the Mg2Si phase is very stable and can impede grain boundary sliding at high temperatures[3-5]. However, under a low solidification rate it is easy to form Mg2Si phase with coarse Chinese script shape and lower the mechanical properties of the alloys. Therefore, the modification and refinement of Mg2Si phase is one of the means to improve the mechanical properties of Mg-Al-Si based alloys[6-8]. Many researches about modification and refinement of Mg2Si phase in Mg-Al-Si based alloys have been carried out. It has been reported that the Chinese script Mg2Si phase in Mg-Al-Si based alloys could be modified and refined by addition of Sb or Sr[9-13]. For example, YUAN et al[9] reported that when 0.5%Sb was added to Mg-5Al-1Zn-1Si alloy, the Mg3Sb2 particles, acting as nuclei for Mg2Si phase, would form in the alloy and change Mg2Si particles from coarse Chinese script shapes to small polygons. SRINIVASAN et al[11] reported that the addition of Sr to a Si-containing AZ91-Mg alloy refined the microstructure by decreasing grain size, and the coarse Chinese script Mg2Si precipitates were smaller and more uniformly distributed. Similar results were obtained by NAM et al[12] and SONG et al[13]. In spite of the abovementioned studies, further investigation about the effects of Sb and Sr on the modification and refinement of Chinese script Mg2Si phase in Mg-Al-Si based alloys are still necessary due to the following reasons. 1) Some disagreement for the Sb modification still exists. For example, QUIMBY et al[14] reported that Sb is not an effective modifier of Mg2Si phase in Si-containing Mg alloys. 2) The mechanism of modification and refinement of Mg2Si phase in Sr-containing Mg-Al-Si based alloys is not completely clear. 3) The comparison of Sb and Sr in the modification and refinement of Mg2Si phase in Mg-Al-Si based alloys is scarce. So, in the present works, the effects of Sb and Sr on the modification and refinement of Mg2Si phase in an AZ61-0.7Si alloy were compared and the corresponding mechanism was analyzed.
2 Experimental
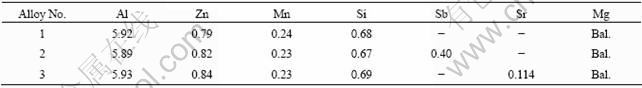
The Sb and Sr containing AZ61-0.7Si alloys were prepared by adding following materials: commercial AM60 alloy, pure Mg, Al, Zn and Sb (>99.9%), Al-30%Si and Al-10%Sr master alloys. The studied alloys were melted in a crucible resistance furnace and protected by a flux addition. When the melt temperature was around 740 ℃, the 0.4%Sb and 0.12%Sr were added into the melt of studied alloys, respectively. After being held at 740 ℃ for 60 min, the melts were homogenized by mechanical stirring and then poured into a preheated permanent mould. As reference, the AZ61-0.7Si alloy without Sb and Sr modification was also cast and analyzed under the same conditions. The actual chemical compositions of the studied alloys are listed in Table 1.
Table 1 Compositions of studied alloys (mass fraction, %)
The samples were etched with 8% nitric acid distilled water solution, and then examined by JOEL/ JSM-6460LV type scanning electron microscope equipped with Oxford energy dispersive spectrometer. The phases in the studied alloys were analyzed by D/Max-1200X type X-ray diffractometer operated at 40 kV and 30 mA. The differential scanning calorimetry (DSC) testing was carried out using a NETZSCH STA 449C system. Samples weighted around 30 mg were heated in a flowing argon atmosphere from room temperature to 700 ℃ and held for 5 min before being cold down to 100 ℃. The cooling curve was recorded at a controlling rate of 15 ℃/min.
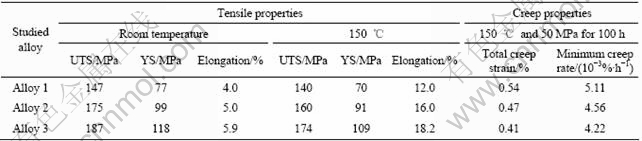
In addition, the tensile and creep properties of studied alloys were also tested. The tensile properties of studied alloys at room temperature and 150 ℃ were determined from a complete stress―strain curve. The 0.2% yield strength(YS), ultimate tensile strength(UTS) and elongation to failure (elongation) were obtained based on the average of three tests. The constant-load tensile creep tests were performed at 150 ℃ and 50 MPa for creep extension up to 100 h. The total creep strain and minimum creep rate were measured from each elongation―time curve and averaged over three tests respectively.
3 Results and discussion
3.1 As-cast microstructures of studied alloys
Fig.1 shows the XRD patterns of alloys 2 and 3. The alloy 1 is composed of α-Mg, M17Al12 and Mg2Si. According to Fig.1, adding 0.4%Sb and 0.12%Sr to AZ61-0.7Si alloy will cause the formation of small amounts of Mg3Sb2 and Al4Sr, respectively.
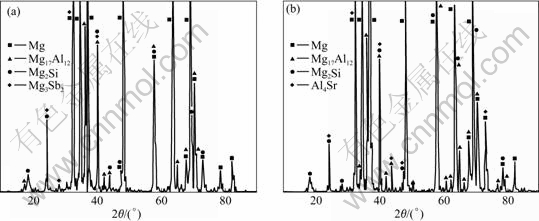
Fig.1 XRD patterns of studied alloys: (a) Alloy 2; (b) Alloy 3
Fig.2 shows the SEM images of studied alloys. Combined with the EDS results of experimental alloys (Table 2), it is found that the Mg2Si phase in the AZ61-0.7Si alloy without Sb or Sr modification exhibits coarse Chinese script morphology. However, after adding 0.4%Sb to AZ61-0.7Si alloy, the Mg2Si phase in the alloy becomes relatively fine although the Chinese script morphology is still observable (Fig.2(b)). The above results indicate that adding 0.4%Sb to AZ61-0.7Si alloy can refine the Chinese script shaped Mg2Si phase in the
alloy. Furthermore, by comparing Figs.2(b) with (c), it is interesting to observe that after adding 0.12%Sr to AZ61-0.7Si alloy, the Mg2Si phase in the alloy becomes very fine, and its morphology changes from initial Chinese script shape to granule and/or irregular polygon, indicating that Sr can effectively modify and refine the Chinese script Mg2Si phase in the AZ61-0.7Si alloy. The coarse Chinese script shaped Mg2Si phase will cause detrimental effect on the mechanical properties of Si-containing Mg alloys where long cracks can easily nucleate along the interface between Chinese script Mg2Si particles and α-Mg matrix[15]. Therefore, it is inferred that the difference of Sb and Sr in the modification and refinement of Mg2Si phase in AZ61-0.7Si alloy results in different tensile and creep properties of experimental alloys. Table 3 lists the tensile and creep properties of studied alloys. It is observed from Table 3 that the tensile and creep properties of alloys 2 and 3 are higher than those of the alloy 1, and that the alloy 3 exhibits higher tensile and creep properties than alloy 2, especially for the elongation. Apparently, the testing results of tensile and creep properties of studied alloys are consistent with the above microstructural analysis.

Fig.2 SEM images of studied alloys: (a) Alloy 1; (b) Alloy 2; (c) Alloy 3; (d) Local magnification in (c)
Table 2 EDS results of studied alloys (atomic fraction, %)

Table 3 Tensile and creep properties of studied alloys
 3.2 Discussion
3.2 Discussion
In general, in the process of nucleus formation, the boundary energy of heterogeneous nucleus with the crystallization phase has an effect on nucleus formation and the effect depends on the structures of the two contacting crystalline faces. One criterion of heterogeneous nucleation is that the disregistry of nucleant planes is less than 6%[9]. According to the report of YUAN et al[9], the two-dimensional lattice misfit mathematical model is given as:
 (1)
(1)
where (hkl)s and (hkl)n are the low index planes of the substrate and nucleated solid respectively; [uvw]s and [uvw]n are the low index directions in (hkl)s and (hkl)n, respectively; d[uvw]s and d[uvw]n are the atomic spacing along [uvw]s and the [uvw]n, respectively; θ is the angle between [uvw]s and [uvw]n.
Figs.3 and 4 show the relationship of some possible crystal faces of Mg3Sb2 to Mg2Si and Al4Sr to Mg2Si, respectively. According to Figs.3 and 4 and Eq.(1), the planar mismatch of some possible crystallographic orientations for Mg2Si nucleation on the Mg3Sb2 or Al4Sr particles were calculated, and the results are listed in Tables 4 and 5. It is found from Table 4 that, when the orientation relationship between Mg3Sb2 and Mg2Si is 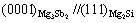 , the disregistry is the lowest (1.72%). Furthermore, it is found from Table 5 that the orientation relationship between Al4Sr and Mg2Si has the lowest disregistry value of 0.69% at
, the disregistry is the lowest (1.72%). Furthermore, it is found from Table 5 that the orientation relationship between Al4Sr and Mg2Si has the lowest disregistry value of 0.69% at 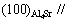
 Although the disregistry at
Although the disregistry at 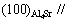
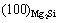 is lower than that at
is lower than that at 
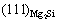 (0.69% vs 1.72%), the Mg3Sb2 is also possible to act as the heterogeneous nucleus for the Mg2Si particles according to the above-mentioned disregistry mechanism. Therefore, it is inferred that the disregistry is possible one of the reasons for the difference of Sb and Sr in the modification and refinement of Mg2Si phase in the AZ61-0.7Si alloy. The difference of Sb and Sr in the modification and refinement of Mg2Si phase in the AZ61-0.7Si alloy may be related to other reasons.
(0.69% vs 1.72%), the Mg3Sb2 is also possible to act as the heterogeneous nucleus for the Mg2Si particles according to the above-mentioned disregistry mechanism. Therefore, it is inferred that the disregistry is possible one of the reasons for the difference of Sb and Sr in the modification and refinement of Mg2Si phase in the AZ61-0.7Si alloy. The difference of Sb and Sr in the modification and refinement of Mg2Si phase in the AZ61-0.7Si alloy may be related to other reasons.

Fig.3 Crystallographic relationship at interface between Mg3Sb2 and Mg2Si phases[12]: (a)  (b)
(b) 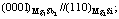 (c)
(c) 

Fig.4 Crystallographic relationship at interface between Al4Sr and Mg2Si phases: (a) (b)
(b) (c)
(c) 
Table 4 Calculated values of planar mismatch between Mg3Sb2 and Mg2Si[12]

Table 5 Calculated values of planar mismatch between Al4Sr and Mg2Si

The degree of undercooling during solidification process is a very pivotal factor for the microstructural
refinement of engineering alloys. According to the classic solidification theory, the relationship between critical nucleus radius and the degree of undercooling is given as follows[16]:
 (2)
(2)
where r* is the critical nucleus radius; ΔGr is the variation of volume free energy; σ is the interfacial energy of unit surface area; Tm is the equilibrium crystallizing temperature; Lm is the crystallizing latent heat; ΔT is the degree of undercooling; Tl is the onset crystallizing temperature. According to Eq.(2), the critical nucleus radius decreases with the decreasing of Tl, then the nucleation energy of crystal nucleus reduces and the nucleation probability increases. Fig.5 shows the DSC cooling curves of studied alloys. It is observed from Fig.5 that after adding 0.4%Sb and 0.12%Sr to AZ61- 0.7Si alloy, the onset crystallizing temperatures of the alloy decreases from 608.8 ℃ to 606.9 ℃ and 603.1 ℃, respectively. Obviously, according to the under- cooling degree mechanism, the nucleation probability of Mg2Si phase in the Sr-modified AZ61-0.7Si alloy is higher than that of the Mg2Si phase in the Sb-modified AZ61-0.7Si alloy. Accordingly, as shown in Fig.2(c), the Mg2Si phases in the Sr-modified AZ61-0.7Si alloy are effectively modified and refined. Oppositely, the Mg2Si phases in the Sb-modified AZ61-0.7Si alloy is only refined, but its modification is not obvious. Actually, the above-mentioned situation is similar to the modification and refinement of Chinese script shaped Mg2Si phases in Si-containing Mg alloys under a fast cooling condition such as die casting that has larger undercooling degree [9, 14].

Fig.5 DSC cooling curves of studied alloys: (a) Alloy 1; (b) Alloy 2; (c) Alloy 3
4 Conclusions
1) The difference of Sb and Sr in the modification and refinement of Mg2Si phase in an AZ61-0.7Si Mg alloy is significant. Adding 0.4%Sb to AZ61-0.7Si alloy can refine the Chinese script Mg2Si phase. However, after adding 0.12%Sr to AZ61-0.7Si alloy, the Mg2Si phase changes from initial coarse Chinese script shape to fine granule and/or irregular polygon. Accordingly, the Sr-modified AZ61-0.7Si alloy exhibits higher tensile and creep properties than the Sb-modified one.
2) The difference of Sb and Sr in the modification and refinement of Mg2Si phase in AZ61-0.7Si alloy is possibly related to the following two aspects: the disregistry at (100)Al4Sr//(100)Mg2Si is lower than that at (0001)Mg3Sb2//(111)Mg2Si (0.69% vs 1.72%) and the onset crystallizing temperature of the Sr-treated AZ61-0.7Si alloy is lower than that of the Sb-modified one (603.1 ℃ vs 606.9 ℃).
References
[1] LUO A, PEKGULERYUZ M Z. Review: cast magnesium alloys for elevated temperature applications [J]. J Mater Sci, 1994, 29: 5259-5271.
[2] DARGUSCH M S, BOWLES A L, PETTERSEN K, BAKKE P, DUNLOP G L. The effect of silicon content on the microstructure and creep behavior in die-cast magnesium AS alloys [J]. Metal Mater Trans A, 2004, 35: 1905-1909.
[3] YOO M S, SHIN K S, KIM N J. Effect of Mg2Si particles on the elevated temperature tensile properties of squeeze-cast Mg-Al alloys [J]. Metal Mater Trans A, 2004, 35: 1629-1632.
[4] LU Y Z, WANG Q D, ZENG X Q, DING W J, ZHU Y P. Effects of silicon on microstructure, fluidity, mechanical properties, and fracture behaviour of Mg-6Al alloy [J]. Mater Sci Technol, 2001, 17: 207-214.
[5] YANG Ming-bo, PAN Fu-sheng, BAI Liang, TANG Li-wen. Sb modification on the heat-treated microstructure and mechanical properties of Mg-6A1-1Zn-0.7Si magnesium alloy [J]. The Chinese Journal of Nonferrous Metals, 2007, 17(12): 2010-2016. (in Chinese)
[6] JIANG Q C, WANG HY, WANG Y, MA B X, WANG J G. Modification of Mg2Si in Mg-Si alloys with yttrium [J]. Mater Sci Eng A, 2005, 392: 130-135.
[7] LU Y Z, WANG Q D, ZENG X Q, ZHU Y P, DING W J. Behavior of Mg-6Al-xSi alloys during solution heat treatment at 420 ℃ [J]. Mater Sci Eng A, 2001, 301: 255-258.
[8] BARBAGALLO S. Microstructural evolution of AS21X HPDC alloy during thermal treatment [J]. Inter J Cast Metals Research, 2004, 17(6): 370-375.
[9] YUAN G Y, LIU Z L, WANG Q D, DING W J. Microstructure refinement of Mg-Al-Zn-Si alloys [J]. Mater Letters, 2002, 56: 53-58.
[10] SRINIVASAN A, PILLAI U T S, PAI B C. Microstructure and mechanical properties of Si and Sb added AZ91 magnesium alloy [J]. Metal Mater Trans A, 2005, 36: 2235-2243.
[11] SRINIVASAN A, PILLAI U T S, SWAMINATHAN J. Observations of microstructural refinement in Mg-Al-Si alloys containing strontium [J]. J Mater Sci, 2006, 41: 6087-6089.
[12] NAM K Y, SONG D H, LEE C W, LEE S W, PARK Y H, CHO K M, PARK I M. Modification of Mg2Si morphology in as-cast Mg-Al-Si alloys with strontium and antimony [J]. Mater Sci Forum, 2006, 510/511: 238-241.
[13] SONG D H, LEE C W, NAM K Y, LEE S W, PARK Y H, CHO K M, PARK I M. Microstructural characteristics and creep properties of Mg-5Al-2Si alloys modified with Sn and Sr [J]. Mater Sci Forum, 2007, 539/543: 1784-1789.
[14] QUIMBY P D, LU S Z, PLICHTA R, VISSER D K, JACOBE K P. Effects of minor addition and cooling rate on the microstructure of cast magnesium-silicon alloys [C]// LUO A, NEELAMEGGHAM N, BEALS R. Magnesium Technology. San Antonio, Texas: TMS, 2006: 535-538.
[15] KIM J J, KIM D H, SHIN K S, KIM N J. Modification of Mg2Si morphology in squeeze cast Mg-Al-Zn-Si alloys by Ca or P addition [J]. Scripta Mater, 1999, 41: 333-340.
[16] PAN Y C, LIU X F, YANG H. Sr microalloying for refining grain size of AZ91D magnesium alloy [J]. J Wuhan Univer Technology: Mater Sci, 2007, 22(1): 74-76.
Foundation item: Project(50725413) supported by the National Science Fund for Distinguished Young Scholar in China; Project(2007CB613704) supported by the National Basic Research Program of China; Projects(2006AA4012-9-6, 2007BB4400) supported by the Chongqing Science and Technology Commission of China
Corresponding author: YANG Ming-bo; Tel: +86-23-68667140; E-mail: yangmingbo@cqit.edu.cn
DOI: 10.1016/S1003-6326(08)60266-6
(Edited by YANG Hua)