
Corrosion behavior of B30 Cu-Ni alloy and anti-corrosion coating in
marine environment
LI Qing-fen(�����)1 , LI Chun-hui(���)1 , XU Li-kun (������) 2��LIU Guang-zhou(������)2
1. College of Mechanical and Electrical Engineering, Harbin Engineering University, Harbin 150001, China;
2. State Key Laboratory for Marine Corrosion and Protection, Qingdao 266071, China
Received 15 July 2007; accepted 10 September 2007
Abstract: The corrosion behavior of B30 Cu-Ni alloy in a sterile seawater and a SRB solution was investigated. The results show that the corrosion potential of specimen in the SRB solution is much lower than that in the sterile seawater. The polarization resistance of specimen in the SRB solution decreases quickly after a period immersion and becomes much lower than that in the sterile seawater. It is concluded that the SRB accelerates the corrosion process of B30 Cu-Ni alloy greatly. An anti-corrosion electroless Ni-P coating was produced and applied to the alloy. The results show that specimens coated with Ni-P plating exhibit favorable corrosion resistance property in SRB solution. Severe pitting corrosion appears on the uncoated specimens in the SRB solution when the coated specimens are still in good condition. The anti-corrosion mechanism of Ni-P plating was analyzed. It is concluded that coating the B30 Cu-Ni alloy with electroless Ni-P plating is an effective technique against the attack of SRB in marine environment.
Key words: Cu-Ni alloy; microbiological influenced corrosion(MIC); sulfate-reducing bacteria (SRB); anti-corrosion coating; corrosion behavior
1 Introduction
A variety of microorganisms in the marine environment may cause microbiological influenced corrosion (MIC), one of the most serious forms of degradation. The MIC failures exit in many industries and the cost of such damage may reach billions of dollars annually[1-3]. Among them, the sulfate-reducing bacteria (SRB) influenced corrosion is one of the most severe one in marine environment as it is rapid and complex[4-5].
Copper alloys have been applied in marine environment for a long time due to their excellent properties. Cu-Ni alloy is widely used in the marine pipeline system which is often attacked by the SRB influenced corrosion[6-7]. Some techniques for controlling such corrosion are used, such as biocides, UV irradiation, corrosion inhibitor, and coating. Among them, the electroless Ni �CP plating is known as a useful method and it has been applied over various materials in many fields[8-13]. The anti-corrosion performance of electroless plating Ni-P coatings has been investigated extensively[14-18]. However, few corrosion data for the Ni-P coating used in the marine microbiological environment is available.
In the present work, the corrosion behavior of marine pipeline material B30 Cu-30% Ni-base alloy, and the protection efficiency of electroless Ni-P coating in the sterile seawater and SRB solution was investigated.
2 Experimental
The chemical composition of the B30 Cu-Ni alloy used in the present investigation is shown in Table 1. The size of uncoated test sample is d 11.3 mm�� 6 mm with working area of 1 cm2. It is 20 mm��15 mm��2 mm with working area of 2.4 cm2 for sample coated with electroless Ni-P plating. A copper wire was soldered to the lower surface of the test sample for potential measurements. Samples were polished up to grit 400 and then cleaned with deionized water. All samples were sterilized under UV for 3 h before test.
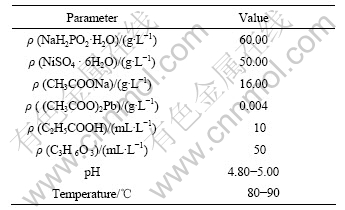
Table 2 lists the plating solution composition and the plating condition for preparing the electroless Ni-P coating.
Table 1 Chemical composition of Cu-Ni alloy(mass fraction, %)

Table 2 Bath composition and operating condition for electroless Ni�CP
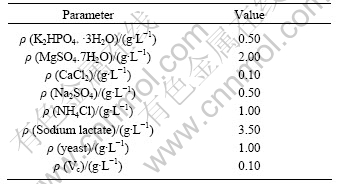
Tests were carried out in two solutions: the sterilized seawater and the SRB solution. The SRB strains were isolated from the sea-mud in Qingdao seashore. The composition of the SRB solution is listed in Table 3.
MIC behavior of sample in different media was investigated by electrochemical impedance spectroscopy (EIS), energy dispersive spectroscopy (EDS), X-ray diffractometry (XRD) and scanning electron microscopy (SEM). The corrosion potential and impedance measurements were performed using the IM6 electrochemical workstation of ZAHNER. Tests were carried out at ambient temperature using a three- electrode system where the working electrode was the tested sample. A platinum niobium wire was used as the counter electrode, and a saturated calomel electrode (SCE) was the reference electrode. The applied voltage amplitude of 20 mV was used at frequencies between 0.005 Hz and 105 Hz.
Table 3 Composition of SRB solution
3 Results and discussion
Fig.1 shows the variation of corrosion potential over time of uncoated B30 Cu-Ni alloy samples in different test solutions and Fig.2 shows the variation of charge reaction resistance which came from the data of equivalent electrical circuits (EEC) in the electrochemical impedance Nyquist plots (Figs.3 and 4). The results of EDS of samples original and after 30 d exposure in SRB solution are given in Table 4. Fig.5 shows the XRD of coated samples original and after 30 d immersion in SRB solution. Fig.6 and Fig.7 show the surface micrographs of samples.
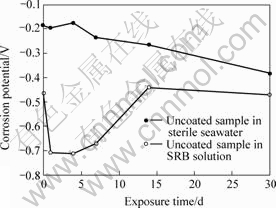
Fig.1 Variation of corrosion potential with time
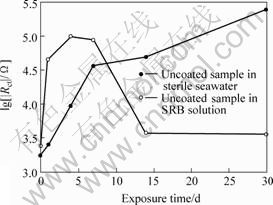
Fig.2 Variation of charge reaction resistance over time
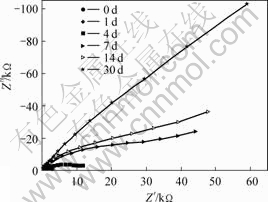
Fig.3 Nyquist plots for sterilized seawater
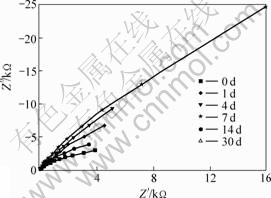
Fig.4 Nyquist plots for SRB solution
Table 4 EDS results of specimens original and after 30 d immersion in SRB solution (mass fraction, %)
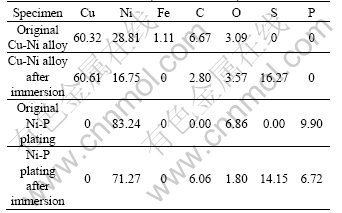
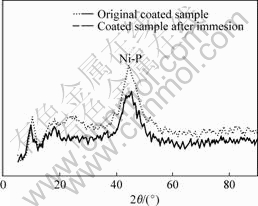
Fig.5 XRD pattern of coated samples original and after 30 d immersion in SRB solution
From Figs.1 and 2, we see that the corrosion potential of uncoated sample in the SRB solution is much lower than that in the sterile seawater. The polarization resistance of specimen at the first several days in the SRB solution is higher than that in the sterile seawater, and then it decreases quickly after a period immersion and becomes much lower than that in the sterile seawater in which the polarization resistance of specimen increases continuously. It is concluded that the SRB accelerates the process of corrosion greatly for the uncoated B30 Cu-Ni alloy samples.
Results of energy dispersive spectroscopy (EDS) gave the same conclusion. From Table 4, we can see that, compared with the original uncoated sample, the content of element Ni and Fe of the Cu-Ni alloy decreases greatly and the high content of element S appears after 30 d immersion in the SRB solution. It suggests that the SRB promotes the dissolution of B30 Cu-Ni alloy and the de-alloy corrosion may have happened in SRB solution.

Fig.6 Surface micrographs of uncoated samples: (a) Original sample; (b) In sterile seawater; (c) In SRB solution

Fig.7 Surface micrographs of samples: (a) Original sample; (b) In steril seawater; (c) In SRB solution
However, experimental results show that samples coated with Ni-P plating in SRB solution exhibits favorable corrosion resistance property.
From Table 4, we see that the main elements of the original Ni-P plating are Ni, P, and O. After 30 d exposure in the SRB solution, the content of these elements decreased obviously and the element S appeared and reached to 14.15% in mass fraction. It means that the SRB also promoted the dissolution of the Ni-P plating. However, the lower potential of Ni-P plating may provide cathodic protection to the B30 copper-based alloy at first when exposed to the corrosive environment. After a period immersion, the corrosion products of Ni-P plating formed, and barrier type protection will therefore be offered by the plating. With increasing nickel consumption and corrosion product depositions, the base B30 alloy is protected with improved physical barrier.
From Fig.5, we may see that there is no crystalline peak value presented in the XRD for the coated sample after 30 d immersion in the SRB solution. It suggests that the structure of the Ni-P plating still kept un-crystalline, and that the B30 Cu-Ni alloy under the Ni-P plating was not corroded for the structure of the base material is crystalline.
The micrographs of samples (Figs.6 and 7) show that severe pitting corrosion appears on the uncoated specimens in the SRB solution when the coated specimens in both the sterile seawater and SRB solution were still in good condition. This confirmed that the Ni-P plating protected the B30 copper alloy effectively in the microbiological environment.
The anti-corrosion mechanism of Ni-P plating is that besides it may offer both barrier and cathodic protection to the base metal as we analyzed above, the structure of Ni-P plating is high uniform and amorphous without the structure defects. The passive film on the surface of the Ni-P plating is also high uniform and non-crystalline structure[16]. The non-crystalline structure will therefore improve the corrosion resistance because it does not have crystalline defects such as dislocation, grain boundary, twin and so on which may cause corrosion easily. The P element in the plating also plays an important role by improving the stable potential of the plating, and reduces the risk of being corroded.
It was concluded that coating the alloy with electroless Ni-P plating is an effective technique against the attack of SRB in marine environment. It is obviously valuable for the marine pipeline system material used in the marine microbiological environment.
4 Conclusions
1) The specimens in the sterile seawater and SRB solution show that the SRB accelerates the corrosion process of B30 Cu-Ni alloy greatly in marine environment.
2) An anti-corrosion electroless Ni-P coating is produced and applied to the B30 Cu-N alloy. The coated specimens exhibit favorable corrosion resistance property in SRB solution. It is therefore concluded that coating the B30 Cu-Ni alloy with electroless Ni-P plating is an effective technique against the attack of SRB. This method is obviously valuable for the marine pipeline system material used in the marine microbiological environment.
References
[1] CHEN Liu-ping. Economic loss caused by MIC and the countermeasure[J]. Corrosion & Protection��1996��17(6): 248-251.
[2] WU Jian-hua, LIU Guang-zhou, YU Hui. Research method of marine microbiological influenced corrosion[J]. Corrosion & Protection��1999, 5(2): 231-237.
[3] ZHU Su-lan, HOU Bao-rong, ZHANG Jing-lei. Research of microorganisms and metal corrosion[J]. Marine Environment Science, 2000, 19(4): 27-30.
[4] BOOTH G H, TILLER A K. Cathodic characteristic of mild steel in suspensions of sulfate-reducing bacteria[J]. Corrosion Science, 1968, 8: 583-600.
[5] HALUK B, ZBIGNIEW L. Dynamics of lead immobilization in sulfate reducing biofilms[J]. Water Research, 2004, 38: 2726�C2736.
[6] SONG Shi-zhe, YIN Li-hui, WU Jie. Electrochemical corrosion of brass pipeline in circular cooling system[J]. Journal of Chemical Industry and Engineering, 2005, 56(1): 121-125.
[7] CUI Guo-feng, LI Ning, LI De-yu, ZHENG Jian, WU Qing-long. The physical and electrochemical properties of electroless deposited nickel�Cphosphorus black coatings[J]. Surface & Coatings Technology, 2006(200): 6808�C6814.
[8] LI Zhen-gang, XU Jing-sheng. Forming mechanism of electroless deposited Ni-P non-crystalline structure[J]. Journal of Daqing Petroleum Institute, 1993, 17(2): 32-38.
[9] FUNDO A M, ABRANTES L M. The electrocatalytic behaviour of electroless Ni�CP alloys[J]. Journal of Electroanalytical Chemistry, 2007(600): 63�C79.
[10] PEETERS P, HOOM G V D, DAENENA T, KUROWSKI A, STAIKOV G. Properties of electroless and electroplated Ni�CP and its application in microgalvanics[J]. Electrochimica Acta, 2001, 47: 161�C169.
[11] BALARAJU J N, NARAYANAN T S, SESHADRI S K. Structure and phase transformation behaviour of electroless Ni-P composite coatings[J]. Materials Research Bulletin, 2006, 41: 847�C860.
[12] BASKARAN I, NARAYANAN T S, STEPHEN A. Effect of accelerators and stabilizers on the formation and characteristics of electroless Ni-P deposits[J]. Materials Chemistry and Physics, 2006, 99: 117�C126.
[13] CHU Guang, LIU Chang-sheng. Preparation of Ni-Cu composite coating by composite electroplating[J]. Journal of Central South University: Science and Technology, 2007, 38(3): 474-479.
[14] HU Guang-hui, WU Hui-huang, YANG Fang-zu. The relation of anti-corrosion property and the P content of electroless Ni-P coating[J]. Acta Phys Chem Science, 2005, 21(11): 1299-1302.
[15] FANG Qi-xian, LIU Xin-kuan, MA Ming-liang, WANG Yi-qing. Research on the anti-corrosion performance of electroless plating Ni-P based alloy[J]. Corrosion & Protection, 1998, 19(2): 67-68.
[16] HU Yong-jun, XIONG Ling, MENG Ji-long. Electron microscopic study on interfacial characterization of electroless Ni-W-P plating on aluminium alloy[J]. Applied Surface Science, 2007, 253: 5029�C5034.
[17] SINGH D D N, GHOSH R. Electroless nickel-phosphorus coatings to protect steel reinforcement bars from chloride induced corrosion[J]. Surface & Coatings Technology, 2006, 201: 90 �C101.
[18] XU Li-jian, GONG Zhu-qing, TANG Jian-xin, HE Quan-guo, HE Nong-yue, DU Jing-jing. Ni-Cr alloy electrodepositing technology on Fe substrate and coating performance[J]. Journal of Central South University of Technology, 2007, 14(2): 181-185.
Foundation item: Project (2005JS5144904.01) supported by the State Key Laboratory for Marine Corrosion and Protection, China
Correspondence: LI Qing-fen, Professor; Tel: +86-451-82589056; E-mail: qingfli@yahoo.com.cn
(Edited by YANG Hua)