������ս����������Ȼ��صĸ�����̬����ˮ������ѧ����
��Դ�ڿ����й���ɫ����ѧ����Ӣ�İ棩2011���8��
�������ߣ����� ��ռ�� �Ÿ���
����ҳ�룺1847 - 1854
�ؼ��ʣ��Ȼ��أ��ս�۳�������������ѧ��������¶
Key words��potassium chloride; sintering dust; leaching; kinetics; surface exposure
ժ Ҫ��
Ϊ�����Ȼ��ش��ս���������ˮ�������ʼ���������ѧ��ʹ��ɨ��羵-��ɨ������Ȼ������ս��������еĸ���״̬��������������֮��ĸ����ϵ��ʵ�鷢���ս�������ƽ������С��10 ��m��������ž���ʽ���ڣ��Ȼ��غ��Ȼ��ƿ������ս������еIJ��ֱ����������۳����������������þ���������ճ�����ӡ��Ȼ������ս������еĴ�����ʽ������ˮ�������̿���Ϊ����ˮ�����ʵ��ܽ���̡����������ߵ絼�ʲ����ֶη����ս������ҵ�ˮ������ѧ�����֤���÷۳���ˮ�����̷�����ɢ���Ƶ��ܽ����ģ�͡�����ƽ����5 min֮�ڼ��ɴﵽ��ͬʱ�Ȼ��صĽ����ʴﵽ95%���ϡ�
Abstract: In order to examine the leaching rate of potassium chloride from the sintering dust by water, surface morphology and inner structure of the dust, especially the existing state of potassium chloride, were observed by scanning electron microscopy (SEM) and linear scanning technique via energy dispersive spectroscopy (EDS). The characterization shows that the sintering dusts are mostly porous composites or agglomerates of the fine dust particles with size less than 10 ��m, and the potassium chloride and sodium chloride particles are partly covered by other water insoluble matters in the dust which consist of elements iron, calcium and etc. Exposure of potassium chloride in the agglomerated dust matrix of this kind suggests that the leaching can be simply perceived as the dissolution of water soluble matters in the dust. On-line monitor of specific electrical conductivity of the leaching system verifies the prediction that leaching kinetics of potassium chloride from the sintering dust fits dissolution model well. Leaching equilibrium can be reached within 5 min with potassium leaching ratio more than 95%.

PENG Cui, GUO Zhan-cheng, ZHANG Fu-li
State Key Laboratory of Advanced Metallurgy, University of Science and Technology Beijing, Beijing 100083, China
Received 16 September 2010; accepted 19 January 2011
Abstract: In order to examine the leaching rate of potassium chloride from the sintering dust by water, surface morphology and inner structure of the dust, especially the existing state of potassium chloride, were observed by scanning electron microscopy (SEM) and linear scanning technique via energy dispersive spectroscopy (EDS). The characterization shows that the sintering dusts are mostly porous composites or agglomerates of the fine dust particles with size less than 10 ��m, and the potassium chloride and sodium chloride particles are partly covered by other water insoluble matters in the dust which consist of elements iron, calcium and etc. Exposure of potassium chloride in the agglomerated dust matrix of this kind suggests that the leaching can be simply perceived as the dissolution of water soluble matters in the dust. On-line monitor of specific electrical conductivity of the leaching system verifies the prediction that leaching kinetics of potassium chloride from the sintering dust fits dissolution model well. Leaching equilibrium can be reached within 5 min with potassium leaching ratio more than 95%.
Key words: potassium chloride; sintering dust; leaching; kinetics; surface exposure
1 Introduction
Sintering dust, here referring in particular to the dust arrested by electrostatic precipitation in sinter plant, as well as other dusts produced in iron and steel companies, is a kind of solid waste which needs proper disposal, not only because of a great variety of hazardous components contained, but a lot of valuable elements such as Fe and C left. At present, there are several typical and classical ways to deal with the dusts, such as Inmetco, Fastmet and Comet [1-4]. Most of the techniques only concentrate on the reuse of Fe and C, but neglect the enrichment of some hazardous ones such as Zn, Pb, K and Na. Since 1960s, researchers have noticed the destructive impact of alkali metals to the blast furnace [5]. Operational experience for many years has also shown that volatility and accumulation of alkali metals always affect the convulsion ability of sintering [6]. In recent years, the extraction of Zn from the dusts is becoming a hot point for many researchers [7-10]. However, the extraction of K from the dusts has rarely been reported.
It is obtained that the mass contribution of KCl is up to 30%, some even up to 40% in the dust. Statistically, there are approximate 4 kg of this kind of dust produced per ton steel. Yet even in the year of 2007 in China, the steel yield was about 490 million ton. So it is a considerable number of the total amount of this dust, which contains 0.77 million ton KCl per year (calculating with KCl content of 30%). As known, the production of potassium fertilizer in China is far below the consumption with a foreign dependency degree of 80%, so it is undoubtedly necessary to recover KCl concentrated in the sintering dust. If KCl in the sintering dust can be recovered completely and converted to available potassium fertilizer such as K2SO4, great contribution would be done to Chinese agriculture. In this work, some works were done to the sintering dust and it was found that potassium enriches to a startling extent in some sintering processes. Furthermore, it presents in a simple form of KCl in the sintering dust, which is easy to be separated from other components.
A processing route for recycling potassium chloride from the sintering dust was proposed. Firstly, the dust was water leached to separate potassium chloride and iron oxides, then sulfide precipitated for removing the impurities in the leaching solution, and finally fractional crystallized to obtain product potassium chloride [11-12].
In order to check the leaching rate of potassium chloride from the sintering dust by water, find out the determinant factors influencing the leaching rate of potassium chloride and further analyze the leaching kinetics, surface morphology and inner structure of the sintering dust, especially surface exposure of potassium chloride which directly determines the degree of its leaching facility, were examined in this work. In addition, effects of dust particle size, leaching temperature and stirring speed on the leaching rate of water-soluble matters were examined by in-situ monitor of specific electrical conductivity of the leaching system. The leaching recovery rate of potassium chloride was also measured when different liquid to solid ratios were taken. These analysis results are expected to provide some hints for scale-up design of extractors.
2 Experimental
2.1 Sampling and component analysis
Sintering dust sampled from several iron and steel companies were subjected to chemical analysis to determine the mass contribution of KCl and NaCl. In this study, five selected companies were represented by letters A, B, C, D and E, and ordinary numbers 1st, 2nd, 3rd and 4th were used to refer to four steps of electrostatic precipitation for each sintering in these five companies. So a combination of a letter A and an ordinary number 3rd, A3rd, means the third step electrostatic precipitation of the sintering in company A.
In addition, one of the samples mentioned above was used as the research material to determine the chemical composition of the sintering dust and existing state of potassium element.
For composition analysis, a proper amount of the dust sample was mixed with acid mixture (HNO3: HClO4: HF=5:3:2, volume ratio)in a Teflon-lined closed vessel and placed in a high-pressure digestion oven at 170 ��C for 5 h. The digested acid mixture was analyzed using a Perkin-Elmer OPTIMA-3000 ICP-AES to determine the concentrations of Al, Ca, Fe, K, Mg, Na, Pb, Cu, Ni and Zn. In addition, a carbon and sulfur combined tester was used to determine the concentration of elements C and S, and the concentration of element Si was determined using the molybdenum blue photometric method with a spectrophotometer UNICO-2000.
2.2 Existing state of KCl in dust
The morphology of the dust and exposure of KCl particles in the dust matrix were analyzed by SEM with line scanning technique. For morphology analysis, the drying dust samples were directly stuck on a small piece of conductive resin and put in the specimen chamber of SEM for imaging after carbon coating. For surface area BET measurement, the dust sample was firstly sent to the sample chamber, heated, vacuumized for degasification, weighted and cooled into the liquid nitrogen, and then the nitrogen adsorption amount at low temperature was recorded at preset pressure. For exposure of KCl in the agglomerated dust matrix, the dust sample was pressed in an organic resin by cold cure, ground with silicon carbide paper and polished with diamond suspension, and then the prepared sample was also carbon coated for imaging and EDS analysis.
2.3 Leaching experiments
The leaching experiments were conducted in two forms: 1) the values of specific electrical conductivity were measured throughout the experiments directly in the bottles; 2) the release of major ions was measured by sampling during the course of the experiment.
A proper amount of dried sintering dust was poured into a stirred vessel with 1 000 mL distilled water placed in advance, as shown in Fig.1. The vessel was jacketed for temperature control, and stirred by a digital stirrer engine equipped with a two-bladed impeller. For the first form of leaching experiments, electrode of the electric conductivity meter was inserted into the solution directly during the leaching process; for the second form, 5 mL samples were collected with a plastic syringe with increasing time intervals for a period of 30 min, and then filtered through 0.22 ��m membrane filters. The leading percentage of K+ and Na2+ was calculated by analyzing their concentration in the filtrate using flame photometer FP640.
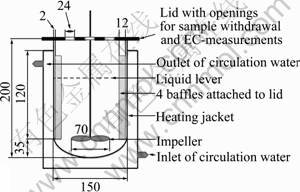
Fig. 1 Schematic diagram of jacketed PMMA reactor with four baffles (unit: mm)
3 Results and discussion
3.1 Mass contribution of KCl and composition analysis of dust
To discover the possible potassium resources in the sintering dust of iron and steel companies in China, sintering dusts sampled from several iron and steel companies were subjected to KCl and NaCl contents analysis, the results are shown in Fig. 2, letters A, B, C, D and E represent five iron and steel companies, and ordinary numbers 1st, 2nd, 3rd and 4th refer to four steps of electrostatic precipitation.
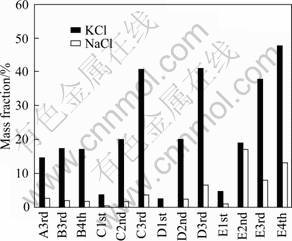
Fig. 2 Mass contribution of KCl and NaCl in sintering dusts sampled from several iron and steel companies
It is clear seen that most of the sintering dust samples contain more than 20% KCl, some up to 30%, and even 40% in the 3rd and 4th electrostatic precipitation step. However, NaCl contribution is far lower than that of KCl, although the same distribution rule is presented as that of KCl. In view of the total amount of the sintering dust production, it is worthy to note that there is a great amount of potassium resource stored in the sintering dust.
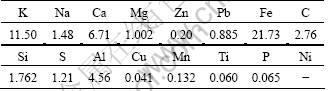
Sintering converts the powdered iron ores into agglomerates, which posses necessary strengh and gas permeability for efficient high-productivity blast furnace operation. The process is carried out by wet mixing a carefully blended mixture of fine iron ores with 3%-5% (mass fraction) coke breeze and other additives, such as limestone, lime chloride, olivine and recycled ferrugininous dusts from upstream processes, the complexity of material composition determines the complexity of the dust composition. Table 1 lists the elemental contribution of one sintering dust sample. Potassium compounds are volatile at sintering temperatures, so they vaporize and precipitate in cooler area as particles [13]. In addition, there are many raw materials, i.e., blast furnace slag and slurry containing K2O. The contribution of K and Pb is higher in the sintering process than in the other processes in particulate emissions from an integrated iron and steel facility [13]. This complicated composition of the dust determines the separation difficulty of K from other elements, and correct disposal essentially considers high emissions of Pb and Zn [14-16].
Table 1 Composition of sintering dust (mass fraction, %)
3.2 Surface exposure of KCl in agglomerated dust
Since potassium exists in the sintering dust in the form of KCl [11], it is natural to choose water leaching as the separation method to recover these potassium resources from the sintering dust which contains high level of potassium chloride. Meanwhile, the other two major constituents in the sintering dust, iron oxide and carbon cannot be leached into the solution, but left and enriched in the leaching residual. Thus, the leaching residual can be returned for sintering to reuse the iron oxides and carbon.
It is well known that the leaching rate of potassium chloride from the sintering dust mainly depends upon the surface area and exposure of KCl in the dust matrix. So it is necessary to clarify the existing state of KCl in the dust.
At first, the dust samples were stuck on a small piece of electronically-conductive tape, the morphology was observed by SEM as shown in Fig. 3.
As mentioned in Refs. [11-12], sizing analysis of the sintering dust shows that many very fine individual dust particles with size of about 1.29 ��m are present, at the same time, some larger particles with size of 40 ��m are also observed. These larger particles are composites or agglomerates of particles in size ranging from 1 to 10 ��m with various shapes, as illustrated in Fig. 3. It also can be seen that the composites or agglomerates are porous, as shown in Fig. 3(b). It is easy to understand that the porous structure and the multipoint BET surface area of 4.58 m2/g indicates a moderate surface area with quite high pore volume, which results in low bulk density and high porosity. Whereas, the skeletal density is higher than 2.52 g/cm3 as reported in the prior work [12]. In addition, it is found some crystals with regular shape partly embedded in the agglomerates, it is doubted that potassium chloride may be included in the exposed crystals.
In order to further clarify the exposure extent of potassium chloride in the sintering dust, the dust samples were pressed in an organic resin by cold cure, ground with silicon carbide paper and polished with diamond suspension. Then the prepared samples were carbon coated for imaging and EDS analysis. The results are shown in Figs.4 and 5.
As illustrated in Figs.4 and 5, potassium chloride and sodium chloride particles are not wholly independent on other dust particles or surrounded by other particles, they are partly connected with some dust particles consisting of elements Fe, Ca and etc.
According to the analysis of inner structure of the dust particles above, it is concluded that the sintering dusts are mostly porous composites and agglomerates composed of many small size particles, and the potassium chloride and sodium chloride in the dust are possibly partly covered by other dust particles which are mainly iron oxides or salts containing calcium. Therefore, it can be concluded that the leaching rate of potassium chloride, sodium chloride and other water-soluble constituents in the dust are mainly controlled by diffusion, and the leaching process might fit the dissolution kinetics model. In the following part of this work, the leaching kinetics is investigated and this inference is justified.

Fig. 3 SEM images of sintering dust particle under different magnifications
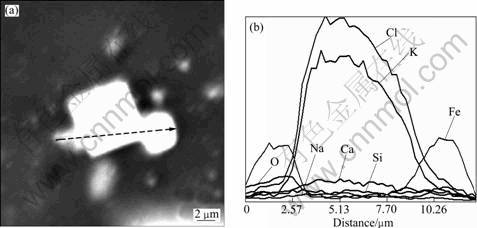
Fig.4 SEM image (a) and line scanning result (b) of sintering dust particle containing high content KCl
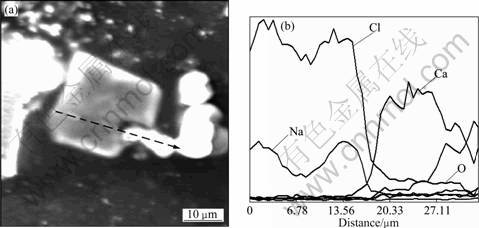
Fig.4 SEM image (a) and line scanning result (b) of sintering dust particle containing high content NaCl
3.3 Leaching kinetics analysis
Due to the hydrophobicity of the insoluble dust particles and small size less than 10 ��m of the dust, new aggregates are easily formed once being in touch with water, which is observed in the experiment. Then the aggregates gradually shrink because of dissolution of some water-soluble particles with diffusion of water from the particle pore space into the bulk solution, and the distance from particle surface to the bulk solution is called the diffusion layer. Under powerful stirring, water-soluble particles disappear at last, and the insoluble residues left, as illustrated in Fig. 6.
Here the rate of diffusion is described as a linear function of the difference between the volume-averaged concentration in the particles and the surface concentration. Early work concluded that the approximation was acceptable in many cases [17-18]. To avoid complex mathematical formulations, the simplification attracts a lot of interest when developing approximate models for diffusion and reaction in porous particles.
The approximate diffusion model is written as:
![]() (1)
(1)
where �� is the mass transfer coefficient that governs the rate of mass transport, Cs is the interfacial concentration of the particle and C is the particle concentration in the solution [20].
According to the Faraday��s law, the relation between specific electrical conductivity �� and concentration Ci is
![]() (2)
(2)
where Ci, Zi, Ui and F refers to the concentration, charge number, mobility of ion i and a constant related to Faraday constant, respectively.
Apparently, there is a linear relationship between specific electrical conductivity �� and concentration Ci. Therefore, for dissolution, the kinetics model can also be written as:
![]() (3)
(3)
where ��s and �� refer to the specific electrical conductivity of the particles in the interfacial and solution, respectively. The mass transfer coefficient �� is expressed as:
![]() (4)
(4)
where D, A, V and �� are the diffusion coefficient, surface area, volume of the reactants and the thickness of diffusion layer, respectively [19].
In this work, the effects of particle size of the dust, stirring speed and temperature on the leaching results of the water-soluble materials in the sintering dust are monitored by measuring the variation of specific electrical conductivity of the solution during the leaching process.
3.3.1 Effect of particle size
To investigate the effect of particle size on the particle concentration in the leachate, a grinder was used to mill the sintering dust for different periods. When the dust was ground for 0, 2, 8 and 15 min, the average particle sizes (50% of normal distribution) were 8.30, 6.25, 6.16 and 4.79 ��m, respectively. These four dusts with different size distribution were used as materials and water was used as leaching reagent, the specific electrical conductivity variation during the leaching process was measured, the result is shown in Fig. 7(a). It can be seen that the leaching equilibrium is achieved after about 300 s for different size distributed dusts. Furthermore, the bigger the particle size, the smaller the specific electrical conductivity, and the slower the leaching equilibrium achieved.
A linear relationship is obtained when d��/dt is plotted with leaching time t (Fig. 7(b)), suggesting that this leaching process can be perceived as dissolution of the dissolvable constituents in the sintering dust, such as KCl and NaCl, and it is diffusion controlled. In the following paragraphs, the same results were obtained on the investigation of the effect of stirring speed and temperature.
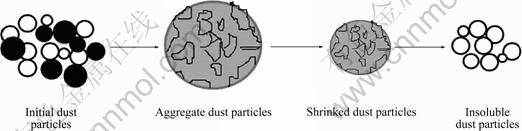
Fig. 6 Dissolution mechanism of sintering dust particles during water leaching process
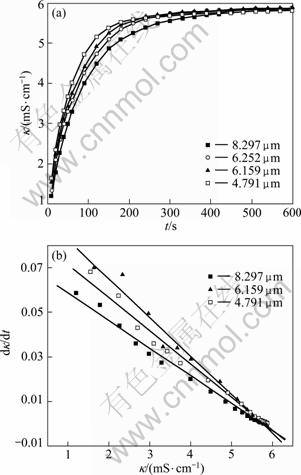
Fig. 7 Effect of particle size on specific electrical conductivity of leaching solution at 303 K with L/S ratio (ratio of water volume to mass of dust, mL/g) of 20 and stirring speed of 550 r/min (a) and d��/dt as function of �� for different particle sizes (b)
3.3.2 Effect of temperature
As shown in Fig. 8(a), specific electrical conductivity of the leaching solution increases with increasing temperature, and the leaching equilibrium is achieved within 200 s. Still good linear relationship is obtained when plotting d��/dt is taken as a function of �� at different temperatures (Fig. 8(b)), further indicating the leaching process is controlled by diffusion.
3.3.3 Effect of stirring speed
For dissolution, stirring speed is also one of the important factors influencing the leaching process. Figure 9(a) shows the specific electrical conductivity of the leaching solution variation in leaching process at different stirring speeds. Apparently, it is seen that stirring speed has noticeable effect on the increasing leaching rate. At stirring speed of 250 r/min, electrical conductivity of the leaching solution increases much slower compared with those at stirring speed of 350, 450 and 550 r/min. As illustrated in Fig. 9(b), the linear relationship between d��/dt and �� further proves the presumed kinetic model to be true.

Fig. 8 Effect of temperature on specific electrical conductivity of leaching solution with L/S ratio of 20, stirring speed of 550 r/min and particle size of 8.297 ��m (a) and d��/dt as function of �� for different temperatures (b)
All the above leaching experiments are carried out at L/S ratio of 20. By multiple linear regression, the leaching rate can be obtained as
![]()
![]() (5)
(5)
where r, d and T refer to the stirring speed, particle size of the dust and leaching temperature, respectively.
3.3.4 Effect of L/S ratio
The leaching experiments above monitor the specific electrical conductivity variation during the leaching process, which reflects the physical performance of all the dissolvable materials in the dust. However, the leaching rate and recovery rate of KCl in the sintering dust are also concerned, so it is necessary to sample from the leaching solution during the leaching process and analyze the corresponding concentration of potassium ion. Figure 10 shows the results of L/S ratio effect on the leaching recovery rate of potassium. As expected, almost all the potassium in the dust can be recovered by water leaching when the L/S ratio is big enough. In addition, the leaching recovery rate of potassium decreases to 89% when the L/S ratio decreases to 2. So it is important to wash the filter cake adequately in order to assure the recovery of KCl in the dust as high as possible.

Fig. 9 Effect of stirring speed on specific electrical conductivity of leaching solution at 303 K with L/S ratio of 20 and particle size of 8.297 ��m (a) and d��/dt as function of �� for different stirring speeds (b)
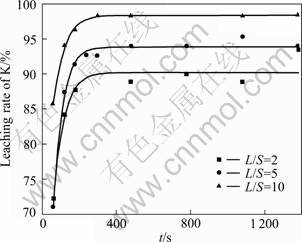
Fig. 10 Effect of L/S ratio on leaching rate of KCl at 303 K with particle size of 8.297 ��m and stirring speed of 550 r/min
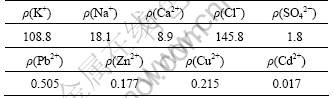
Actually, leaching L/S ratio in the range of 1-2 is advised to the recover potassium chloride from the sintering dust with mass fraction of potassium chloride of about 20%. Proper adjustment to the ratio should be made according to the variation of mass contribution of potassium chloride. Table 2 lists the concentration of the leaching solution of the sintering dust shown in Table 1 with leaching L/S ratio of 1.
As seen from Table 2, the concentration of potassium ion is much higher than that of sodium and calcium, and the chloride ion is the major anion. However, some heavy metal ions are also leached into the solution, which should be paid attention to. In this work, potassium chloride product with about 96% purity is obtained by fractional crystallization from quaternary system of KCl-NaCl-CaCl2-H2O with the concentration of the latter four elements in Table 2 decreasing to 2.9, 0.9, 0.3 and 0.2 mg/L, respectively, by sulfide precipitation. The test-work about potassium recovery from the dust was discussed in detail in Refs. [11-12].
Table 2 Concentration of leaching solution of sintering dust shown in Table 1 with L/S ratio of 1 (g��L-1)
4 Conclusions
1) SEM analyses on morphology and inner structure of the sintering dust show that most of the dust particles are porous composites or agglomerates.
2) Line scanning technique of SEM indicates potassium chloride and sodium chloride particles are probably partly covered by other dust particles which contain elements calcium, iron and so on.
3) In-situ monitor of specific electrical conductivity during the leaching process of the sintering dust confirms that the leaching kinetics fits dissolution model well, and the leaching equilibrium with the potassium chloride leaching recovery rate over 95% can be obtained within 5 min with leaching liquid to solid ratio no smaller than 1 and stirring speed no lower than 450 r/min.
References
[1] ODA H, IBARAKI T, ABE Y. Dust recycling system by the rotary hearth furnace [J]. Nippon Steel Technical Report, 2006, 94: 147-152.
[2] HUANG Jie. A review of development of rotary hearth furnace [J]. Chinese Metallurgy, 2007, 17(4): 23-25. (in Chinese)
[3] SHA Yong-zhi, WANG Feng-qi, ZHOU Yu-sheng. Comment on fastmet process [J]. Journal of Iron and Steel Research, 1996, 8(3): 56-59. (in Chinese)
[4] SHI Zhan-biao. Processing of the dust with ferri contained of iron and steel plant by rotary kiln direct reduction [J]. Iron Smelting, 1985, 6: 45-50. (in Chinese)
[5] MU Ji-yao. Alkali metals in the blast furnace [M]. Beijing: Metallurgical Industry Press, 1992: 12-15. (in Chinese)
[6] WANG Cheng-li, LV Qing, GU Lin-na, LI Fu-min, ZHANG Yong. Reaction and distribution of alkali metals in the blast furnace [J]. Journal of Steel and Ion Research, 2006, 18(6): 121-125. (in Chinese)
[7] ZHAO You-cai, ROBERT S. Integrated hydrometallurgical process for production of zinc from electronic arc furnace dust in alkaline medium [J]. Journal of Hazardous Materials B, 2000, 80(1-3): 223-228.
[8] TOMAS H, BRUNA V S, ANDREA M B, IVO A H S, ANDREA M. Hydrometallurgical processing of carbon steel EAF dust [J]. Journal of Hazardous Materials B, 2006, 135(1-3): 311-318.
[9] MASUD A, ABDEL L. Fundamentals of zinc recovery from metallurgical wastes in the enviroplas process [J]. Minerals Engineering, 2002, 15(11): 945-952.
[10] ORHAN G. Leaching and cementation of heavy metals from electronic arc furnace in alkaline medium [J]. Hydrometallurgy, 2005, 78: 236-248.
[11] PENG Cui, GUO Zhan-cheng, ZHANG Fu-li. Discovery of potassium chloride in the sintering dust by chemical and physical characterization [J]. ISIJ International, 2008, 48(10): 1398-1403.
[12] PENG Cui, ZHANG Fu-li, GUO Zhan-cheng. Separation and recovery of potassium chloride from sintering dust of ironmaking works [J]. ISIJ International, 2009, 49(5): 735-742.
[13] TSAI J H, LIN K H., CHEN C Y, DING J Y, CHOA C G, CHIANG H L. Chemical constituents in particulate emissions from an integrated iron and steel facility [J]. Journal of Hazardous Materials, 2007, 147(1-2): 111-119.
[14] CHEN Wei-qing, WANG Dong-yan, ZHOU Rong-zhang, LIN Zong-cai. Kinetics of evaporation of Zn and Pb from carbon-bearing pellets made of dust containing Zn-Pb-Fe oxides [J]. Journal of University of Science Technology Beijing, 2000, 7(3): 178-183.
[15] DAS B, PRAKASH S, REDDY P S R, MISRA V N. An overview of utilization of slag and sludge from steel industries [J]. Resources Conservation & Recycling, 2007, 50(1): 40-57.
[16] MENAD N, TAYIBI H, CARCEDO F G, HERNANAEZ A. Minimization methods for emissions generated from sinter stands: A review [J]. Journal of Cleaner Production, 2006, 14(8): 740-747.
[17] RAO P S C, JESSUP R E, ROLSTON D E, DAVIDSON J M, KILCREASE D P. Solute transport in aggregated porous media: theoretical and experimental evaluation[J]. Soil Science Society of America Journal, 1980, 44(6): 1139-1146.
[18] GERKE H H, VAN GENUCHTEN M T. Dual�Cporosity model for simulating the preferential movement of water and solutes in structured porous media [J]. Water Resources Research, 1993, 29(2): 305-319.
[19] JIANG Han-ying. Physical chemistry of hydrometallurgy [M]. Beijing: Metallurgical Industry Press, 1984: 117-119. (in Chinese)
�� ��, ��ռ��, �Ÿ���
�����Ƽ���ѧ ��̬��ѭ��ұ��������ص�ʵ���ң����� 100083
ժ Ҫ��Ϊ�����Ȼ��ش��ս���������ˮ�������ʼ���������ѧ��ʹ��ɨ��羵-��ɨ������Ȼ������ս��������еĸ���״̬��������������֮��ĸ����ϵ��ʵ�鷢���ս�������ƽ������С��10 ��m��������ž���ʽ���ڣ��Ȼ��غ��Ȼ��ƿ������ս������еIJ��ֱ����������۳����������������þ���������ճ�����ӡ��Ȼ������ս������еĴ�����ʽ������ˮ�������̿���Ϊ����ˮ�����ʵ��ܽ���̡����������ߵ絼�ʲ����ֶη����ս������ҵ�ˮ������ѧ�����֤���÷۳���ˮ�����̷�����ɢ���Ƶ��ܽ����ģ�͡�����ƽ����5 min֮�ڼ��ɴﵽ��ͬʱ�Ȼ��صĽ����ʴﵽ95%���ϡ�
�ؼ��ʣ��Ȼ��أ��ս�۳�������������ѧ��������¶
(Edited by FANG Jing-hua)
Foundation item: Project (50974018) supported by the National Natural Science Foundation of China; Project (108007) supported by the Science Foundation of Ministry of Education of China
Corresponding author: GUO Zhan-cheng; Tel/Fax: +86-10-82375042; E-mail: zcguo@metall.ustb.edu.cn
DOI: 10.1016/S1003-6326(11)60940-0

