Article ID: 1003-6326(2005)03-0697-05
Kinetics of oxidative leaching of ocean polymetallic nodules in molten potassium hydroxide medium
WANG Yun-shan(����ɽ), LI Zuo-hu(������), LI Hao-ran(���Ȼ)
(National Key Laboratory of Biochemical Engineering, Institute of Process Engineering,
Chinese Academy of Sciences, Beijing 100080, China)
Abstract: The kinetics of oxidative leaching of ocean polymetallic nodules in molten potassium hydroxide medium was investigated in terms of the gas-flow rate, stirring rate, reaction time, reaction temperature and partial pressure of oxygen on the ratio of manganese conversion in ocean polymetallic nodules. The category of the rate controlling step was determined. The process of the liquid-phase oxidation reaction can be described by surface chemical reaction-controlled non-reaction shrinking core model(SCM), the apparent activation energy was achieved and the rate equation was put forward. From the equation, it can be concluded that the reaction rate mainly depends on the reaction temperature and the partial pressure of oxygen.
Key words: ocean polymetallic nodule; oxidative leaching; kinetics; reaction CLC
number: TF111.3 Document code: A
1 INTRODUCTION
The finite on-shore mineral resources and their rapid depletion have justifiably directed the attention of international community of scientists to the vast seabed as the future source of supply of minerals and raw materials[1]. It is estimated that till about 2050, the reserves of manganese, copper, cobalt and nickel in land ores all over the world can not meet the ever-growing demand[2]. Ocean polymetallic nodules are the most important submarine mineral resources and contain abundant metallic elements such as manganese, iron, copper, cobalt and nickel, and the relative reserves of manganese, copper, cobalt and nickel in ocean polymetallic nodules are one to three magnitude order larger than those reserved in land mineral deposits[3], so these metallic elements should be extracted and utilized. Because the copper, cobalt and nickel are deposited in manganese and iron oxides(especially in manganese oxide) mine-phase, and there have no separate minerals of themselves, they cannot be enriched or separated by the conventional methods. However, the application of the chemical or metallurgical processing approaches can solve this problem[4]. Since ocean polymetallic nodules have been found, a large number of researches on ocean polymetallic nodules have been carried out both at home and abroad[4], dozens of methods have been proposed, and the new explorations are still going on. However, up to now, each method has its drawbacks and most of the existent processes yet remain at the laboratory minitype experimental study scale[5, 6], and the goal of making the commercial and industrial utilization of the ocean polymetallic nodules is still far away.
ZHANG et al[7, 8] adopted green chemical principle and industry ecological ��3R�� principles and put forward inferior molten salt liquid-oxidation cleaner production process integration technology. Based on this technological platform, the present authors developed the new process for processing ocean polymetallic nodules utilizing liquid-phase molten salt as transforming agent. The manganese in ocean polymetallic nodules is in the form of manganese dioxide, so it can be converted into potassium manganate through oxidation. Many researches of kinetics before were focused on reductive leaching, specially in acid medium[9-14]. Based on the study of oxidative leaching in alkaline medium, the kinetics of the oxidative leaching is discussed and the effects of gas-flow, stirring rate, reaction time, reaction temperature and oxygen partial pressure on the ratio of manganese conversion are considered.
2 EXPERIMENTAL
2.1 Materials
Ocean polymetallic nodules being picked up from the East Pacific Ocean sea basin were crushed and milled to pass a series of sieves to obtain representative samples. The contents of main metal elements are listed in Table 1.
Table 1 Contents of metal elements in ocean polymetallic nodules(mass fraction, %)

2.2 Methods
1L stainless vessel with temperature control instrument and stirring apparatus was used as the reactor. Certain concentration of potassium hydroxide was put into the reactor and temperature was elevated with stirring at certain rate. When the temperature reached the set value, ocean polymetallic nodules with particle size between 74��m and 70��m were fed in the form of slurry, and the air or oxygen being purified and preheated was filled. When the temperature was stable, counting and sampling were picked up at set intervals and the samples were processed with separating, washing, drying and so on, then they were analyzed with atomic absorption or chemical analysis, finally the conversion ratio of manganese in ocean polymetallic nodules was calculated.
2.3 Analysis of macro kinetics
The reaction of ocean polymetallic nodules oxidized in molten potassium hydroxide is as follows:
[(Cu, Ni, Co)O��MnO2��2FeO(OH)](s)+
2KOH(l)+[SX(]1[]2[SX)]O2(g)=(Cu, Ni, Co)O(s)+
Fe2O3(s)+K2MnO4(s)+2H2O(g)
It is a typical gas-liquid-solid heterogeneous reaction. Reactions in heterogeneous systems are complex and involve not only chemical reactions but also mass transfer processes[11]. Due to the rapid gas dissolving into liquid, and then the liquid reacting with solid, the process is a liquid-solid reaction substantially. Generally, liquid-solid reactions proceed through taking a number of basic steps and may be controlled by either of these steps[11]. The relationships between conversion ratio and time of each category of controlled step has been given in Refs.[15, 16]. Since iron, copper, nickel and cobalt are released in the form of oxides from ocean polymetallic nodules, the non-reaction shrinking core model(SCM) or non-reaction shrinking particle model(SPM) can be applied to describe the oxidative leaching process[17].
3 RESULTS AND DISCUSSION
3.1 Effects of gas-flow, stirring rate and reaction time
Fig.1 presents the effects of different gas-flow rates, stirring rates and reaction time on the rate of manganese conversion at reaction temperature of 300��, mass ratio of alkali to nodules of 10 and oxygen partial pressure of 2.1��104Pa(pure air).
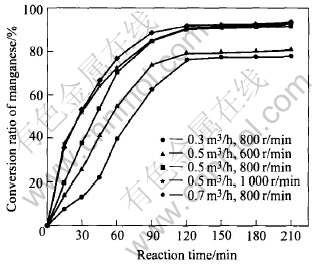
Fig.1 Effects of airflow rate and stirring rate on conversion ratio of manganese
The results show a general increase in the manganese conversion ratio as gas-flow rate, stirring rate and reaction time increase and the reaction rate within 120min is fast. It is obvious that the effective reaction occurs within 120min, and the reaction nearly completes within 120min, thus the reaction kinetics within 120min is mainly studied in this paper.
Fig.2 presents the linear relationships of X(the conversion ratio of manganese), 1-(1-X)1/3 and 1+2(1-X)-3(1-X)2/3 with reaction time, which represents that the process is controlled by external diffusion, surface chemical reaction and internal diffusion, respectively.
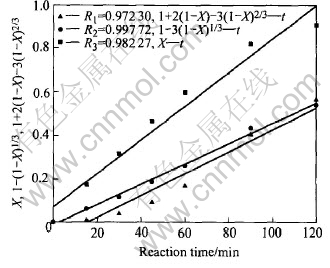
Fig.2 Plot of X, 1-(1-X)1/3 and 1+2(1-X)-3(1-X)2/3 vs reaction time
All the three curves have good linear relationship, and 1-(1-X)1/3 has the best related coefficient, that is to say, either of the models can interpret the reaction, but the best is 1-(1-X)1/3, whereas 1-(1-X)1/3 represents that the process is controlled by surface chemical reaction.
3.2 Effect of temperature
The effect of reaction temperature on the conversion ratio of manganese at mass ratio of alkali to nodules of 10, oxygen partial pressure of 2.1��104Pa, stirring rate of 800r/min, gas-flow rate of 0.5m3/h is presented in Fig.3.
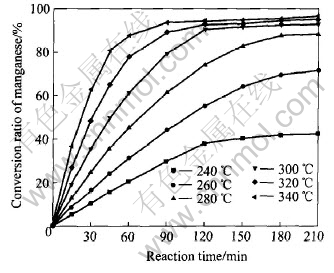
Fig.3 Effect of reaction temperature on conversion ratio of manganese
It can be found that the conversion ratio of manganese increases as the temperature increases. The relationships between 1-(1-X)1/3 and reaction time at different reaction temperatures are shown in Fig.4.
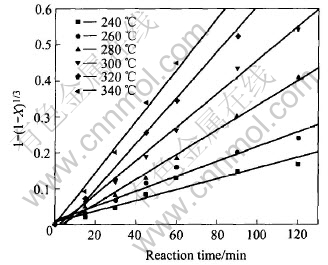
Fig.4 Plot of 1-(1-X)1/3 vs reaction time at various reaction temperatures
Fig.4 shows that there are good linear relationships between 1-(1-X)1/3 and reaction time at different reaction temperatures, and this can also prove that the oxidative leaching process is controlled by surface chemical reaction. The slopes of the lines in Fig.4 represent the reaction rate constants at different reaction temperatures, as listed in Table 2.
Table 2 Reaction rate constants at various temperatures
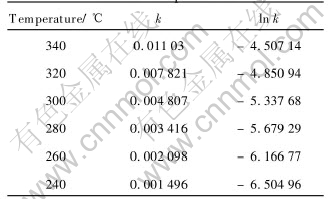
According to the Arrhenius equation: k=A��exp (-E/RT), lnk=lnA-E/RT. The pre-exponential factor A includes the effects of gas-flow, stirring rate, nodules�� size, mass ratio of alkali to nodules and oxygen partial pressure. Fig.5 presents the relationship of lnk with 1/T.
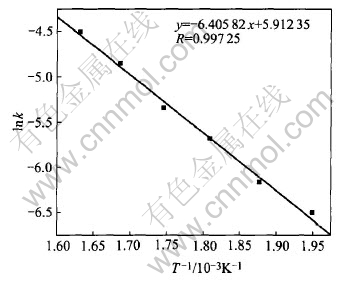
Fig.5 Arrhenius plot showing relationship between lnk and 1/T
A straight line is achieved and the apparent activation energy of the reaction under experimental conditions can be acquired from the slope. This activation energy includes effects of intrinsic chemical reaction and mass transfer. The activation energy can be calculated as E=53.26kJ/mol. Generally, when the activation energy is above 41.8kJ/mol, the process is controlled by chemical reaction[15, 16], thus, it can be proved that the liquid-phase oxidation process is chemical reaction-controlled.
3.3 Effect of oxygen partial pressure
The effect of oxygen partial pressure on manganese conversion rate at mass ratio of alkali to nodules of 10, reaction temperature of 300��, gas-flow rate of 0.5m3/h and stirring rate of 800r/min is presented in Fig.6.
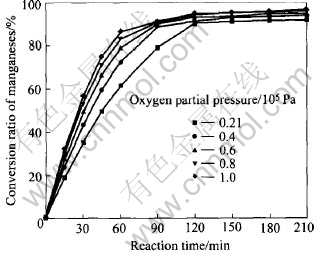
Fig.6 Effect of oxygen partial pressure on conversion ratio of manganese
Fig.6 shows an increase of manganese conversion ratio as oxygen partial pressure increases. Oxygen partial pressure affects mass transfer across the boundary layer between liquid and gas and thus affects the reaction rate. Experimental results indicate that oxygen partial pressure above 2��104Pa can make most of manganese convert. The plots of 1-(1-X)1/3 vs reaction time at various oxygen partial pressures are presented in Fig.7.
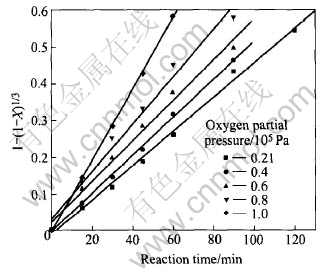
Fig.7 Plots of 1-(1-X)1/3 vs reaction time at various oxygen partial pressures
It can be found that under different oxygen partial pressures, the relationship between 1- (1-X)1/3 and reaction time is still in linear form, and this also manifests the fact that the liquid-phase oxidation process of ocean polymetallic nodules at various oxygen partial pressures is chemical reaction-controlled.
From the slope of each line, the reaction rate constants at various oxygen partial pressures are calculated and presented in Table 3. Based on the data, the relationship between lnk and lnp(O2) is shown in Fig.8.
Table 3 Reaction rate constants at various oxygen partial pressures
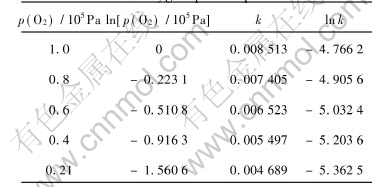
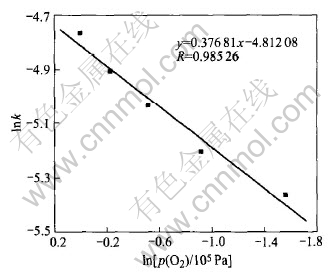
Fig.8 Relationship between natural logarithm of rate constant and natural logarithm of oxygen partial pressure
Fig.8 shows that the relationship between lnk and lnp(O2) appears in linear form approximately, and the slope is 0.37681. So the relationship of reaction rate with oxygen partial pressure is k=kpp(O2)0.37681. According to the relationship of reaction rate equation with reaction temperature, the expression of reaction rate constant can therefore be expressed in the form of k=k1exp(-5.326��104/RT)��p(O2)0.3768. By 1-(1-X)1/3=kt and manganese conversion rate at certain conditions, the value of k1 can be calculated to be 0.584��103min-1. Thus, at reaction temperature of 240-340��, oxygen partial pressure of 2.1��104Pa-105Pa, gas-flow rate above 0.5m3/h, stirring rate above 800r/min and particle size of ocean polymetallic nodules of 70-74��m, the macrokinetics rate equation of ocean polymetallic nodules oxidative leaching in molten potassium hydroxide can be written as
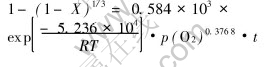
4 CONCLUSIONS
1) The rate of manganese extraction is mainly dependent on the reaction temperature and oxygen partial pressure.
2) The liquid-phase oxidation process of ocean polymetallic nodules can be expressed by non-reaction shrinking core model(SCM). The apparent activation energy is 53.26kJ/mol, and the rate of manganese extraction process is controlled by surface chemical reaction.
3) The reaction rate of manganese extraction process is best described by the rate equation as follows:
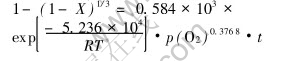
REFERENCES
[1]Singh T R P. Deep seabed minerals and India��s plans in relation to future metal demands [J]. Journal of Mines, Metals & Fuels, 1989, 37(12): 608-610.
[2]SHENG Gui-nong. Exploitation of ocean polymetallic nodules resources [J]. China��s Manganese Industry, 1992, 10(2): 54-56.
[3]HOU Hui-fen. Complex utilization of ocean manganese nodules [J]. Shanghai Nonferrous Metals, 1999, 20(3): 143-147.
[4]WANG Dian-zuo, ZHANG Ya-hui, SUN Chuan-yao. Comment of processing technologies for ocean polymetallic nodules [J]. Metallic Ore Dressing Abroad, 1996, 33(9): 3-13.
[5]YIN Cai-qiao. Achievement and prospects of ocean polymetallic nodules�� metallurgy [J]. Colored Smelting, 2000, 29(2): 11-14.
[6]XU Chuan-hua. Research on technical economy model of ocean polymetallic nodules resource exploitation [J]. Mining & Metallurgy, 2000, 9(1): 59-64.
[7]ZHANG Yi. Green process engineering [J]. Journal of Process Engineering, 2001, 1(1): 10-15.
[8]ZHANG Yi, LI Zuo-hu, QI Tao, et al. Green chemistry of chromate cleaner production [J]. Chinese Journal of Chemistry, 1999, 17(3): 258-266.
[9]CHEN hsiaohong, FU Chong-yue, ZHENG Di-ji. Reduction leaching of manganese nodules by nickel matte in hydrochloric acid solution [J]. Hydrometallurgy, 1992, 28(2): 269-275.
[10]Kanungo S B. Rate process of the reduction leaching of manganese nodules in dilute HCl in presence of pyrite [J]. Hydrometallurgy, 1999, 52(3): 313-330.
[11]Acharya R, Ghosh M K, Anand S, et al. Leaching of metals from indian ocean nodules in SO2-H2O-H2SO4-(NH4)2SO4 medium [J]. Hydrometallurgy, 1999, 53(2): 169-175.
[12]Momade F W Y, Momade Z G. A study of kinetics of reductive leaching of manganese oxide ore in aqueous methanol-sulphuric acid medium [J]. Hydrometallurgy, 1999, 54(1): 25-39.
[13]CHI Ru-an, ZHU Guo-cai, TIAN Jun. Leaching kinetics of rare earth from black weathering mud with hydrochloric acid [J]. Trans Nonferrous Met Soc China, 2000, 10(4): 531-533.
[14]ZHANG Ya-hui, QI Liu, SUN Chuan-yao. Sulfuric acid leaching of ocean manganese nodules using phenols as reducing agents [J]. Minerals Engineering, 2001, 14(5): 525-537.
[15]YANG Xian-wan, QIU Ding-fan. Hydrometallurgy [M]. Beijing: Metallurgical Industry Press, 1998: 154-174.
[16]LI Hong-gui. Hydrometallurgy [M]. Changsha: Central South University Press, 2002. 69-79.
[17]Sohn H Y, Wadsworth M E. ZHENG Di-ji Tr. Extraction Metallurgy Rate Process [M]. Beijing: Metallurgical Industry Press, 1984. 1-46.
(Edited by YANG Bing)
Foundation item: Project(DY 105-04-01-05) supported by the Long-range Development Special Term of China
Received date: 2004-10-10; Accepted date: 2005-01-27
Correspondence: WANG Yun-shan, PhD candidate; Tel: +86-010-82627064; Fax: +86-10-62561822; E-mail: wangys@home.ipe.ac.cn