
Adhesive strength and structure of micro-arc oxidation ceramic coatings grown in-situ on LY12 aluminum alloy
WU Zhen-dong(吴振东)1, 2, JIANG Zhao-hua(姜兆华) 1, YAO Zhong-ping(姚忠平)1
1. Department of Applied Chemistry, Harbin Institute of Technology Harbin 150001, China;
2. Department of Chemistry, Baicheng Normal College, Baicheng 137000, China
Received 28 July 2006; accepted 15 September 2006
Abstract: The ceramic coatings containing zirconium dioxide were grown in-situ on LY12 aluminium alloy by micro-arc oxidation in mixed zirconate and phosphate solution. The phase composition and morphology of the coatings were studied by XRD and SEM. The adhesive strength of ceramic coatings was assessed by thermal shock test and tensile test. The results show that the coating is composed of m-ZrO2, t-ZrO2, and a little γ-Al2O3. Along the section of the coating, t-ZrO2 is more on both sides than that in the middle, while m-ZrO2 is more in the middle than that on both sides. Meantime the coating is also composed of a dense layer and a loose layer. The coating has excellent thermal shock resistance under 550 ℃ and 600 ℃. And tensile tests show the adhesive strength of the dense layer of the coating with the substrate is more than 17.5 MPa.
Key words: LY12 aluminum alloy; micro-arc oxidation; ZrO2 ceramic coating; adhesive strength
1 Introduction
Micro-arc oxidation is a novel surface treatment technique, by which the ceramic coating is grown in-situ on the surface of valve metals like Al, Ti, Ta, Ni and other valve metals[1-3]. The technique is based on the result of cooperation action of physical discharging, electrochemical oxidation and heat- chemistry and so on, and the produced coatings are of high hardness, good wearing resistance, corrosion resistance, insulation and other fine properties[4-6]. Much research has been fulfilled on micro-arc oxidation of aluminum alloy all the time, and the produced coating in NaAlO2 solution was mainly composed of α-Al2O3 and γ-Al2O3[7-9], while besides Al2O3, mullite was also obtained in the coating produced in the Na2SiO3 solution[10-11].
Zirconium dioxide, which has high strength, fracture toughness property, good wear resistance and heat-insulating property, is an important coating material with great development potential, whether as a structure material or a functional material[12-13]. However, the research on the ceramic coatings containing ZrO2 on aluminum alloy by this technique is seldom reported except that in Ref.[14]. Therefore, we prepared the ceramic coatings containing ZrO2 on LY12 aluminum alloy by micro-arc oxidation in mixed zirconate and phosphate solution, and studied the structure and the composition of the coatings. Besides, the adhesive strength of micro-arc oxidation ceramic coating was assessed by thermal shock test and tensile test
2 Experimental
The samples with the reaction dimension of 15 mm×15 mm×1.5 mm were used as an anode, and the electrolytic cell made of stainless steel was used as a cathode. A homemade pulsed bi-polar electric source of 4 kW was used for MAO process, the current density of both pulses was fixed at 8A/dm2 with the frequency of 50 Hz. The concentration of K2ZrF6 was 8 g/L and that of phosphate 6 g/L. The electrolyte temperature was main- tained less than 30 ℃, using cool water.
The morphology of the coatings was examined by Hitachi S-4700 scanning electron microscopy. Phase composition of the coatings was studied by Japan D/max-rB X-ray diffraction(XRD), with a Cu Kα source. The adhesive strength of ceramic coatings with the substrate was assessed by thermal shock resistance test and tensile test. Thermal shock resistance test was carried out in the muffle under 550 ℃ and 600 ℃ respectively. The samples were kept for 2 min under a fixed temperature then put into the cool water of 25 ℃. Repeating tests and observing the experimental phenomena. The tensile test was carried out through InSTRON-5569 tension instrument.
3 Results and discussion
3.1 Morphology of coating
Fig.1 shows the surface and section images of the coating. As shown in Fig.1(a), there are a lot of sintered particles and apparent tracks of melting and sintering on the surface, which is like the left holes of the volcano after spouting. As shown in Fig.1(b), there are some micro holes on the cross section of the coating and the coating is composed of dense inner layer and loose layer near the surface. There exist no gaps or cracks at the interface between the coating and the substrate. The thickness of the coating is increased linearly with the increasing time as shown in Fig.2. The fitted formula is at thickness of 0.975 μm and time of 4.415 min, with the growing speed of 0.975 μm/ min.
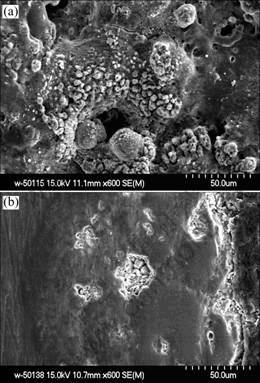
Fig.1 Cross-section image (a) and surface image (b) of ceramic coating for 150 min
3.2 Elemental distribution in coating
Fig.3 shows the EDS analyses of the section image of the coating. The coating mainly contains Zr, Al, O and P, of which Zr is the most while P is the least in the coating. The element analyses are consistent with the phase composition of the coating. Along the coating from the inner to the outer, the content of Zr was increased while the content of Al was decreased. At the beginning of the reaction, because the coating was thin, Al from the substrate easily joined the reaction. Some of Al came into the coating and formed the coating, the other part was reacted with F- to form [AlF6]3- and came into the solution. Extending the reaction time, the coating thickness was increased, the diffusion of Al became more and more difficult, and therefore the content of Al was decreased gradually from the inner to the outer of the coating. While Zr was from the electrolyte, and its diffusion to the inner was gradually decreased with the increase of the thickness, which led to the decrease of the content of Zr from the outer to the inner of the coating.
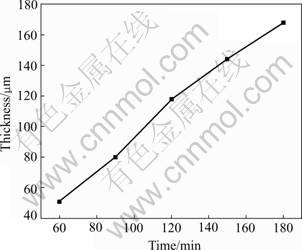
Fig.2 Curve of coating thickness vs time

Fig.3 Elemental distribution of coating
3.3 XRD analysis of coating
Fig.4 shows the XRD patterns of the coating. The coating was composed of t-ZrO2, m-ZrO2 and a little amount of γ-Al2O3, of which t-ZrO2 is the main crystalline. The ratio of t-ZrO2 to m-ZrO2 was estimated by XRD with the result shown in Fig.5. Along the section of the coating, t-ZrO2 was more on both sides than that in the middle; quite oppositely, m-ZrO2 was more in the middle than that on both sides. Because of the high temperature and high pressure within the discharging channels during MAO process and the quick-cooling effect of the solution[3], t-ZrO2 was formed first at the beginning. However, the quick-cooling effect of the solution acted greater on the both sides of the coating than in the middle because of the increase of the thickness of the coating and the good heat-shield property of ZrO2. Therefore, more t-ZrO2 was formed on both sides, while phase transformation from t-ZrO2 to m-ZrO2 mainly happened in the middle part.
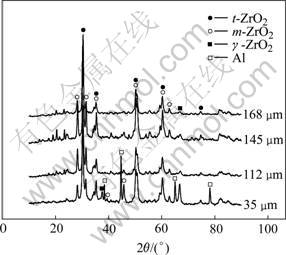
Fig.4 XRD patterns of coatings
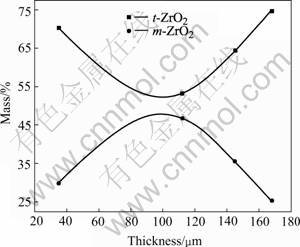
Fig.5 ZrO2 relative content of coating
3.4 Thermal shock resistance test
Under 550-25 ℃, the coating did not spall off the substrate after cycling 50 times. Under 600-25 ℃, the small part of the coating spalled off after cycling for 7 times. And most of the coating was spalled off after cycling for 13 times. The linear expansion coefficient of ZrO2 is approaching that of the metal materials; and 550℃ is far from the melting point of aluminum alloy. Therefore, the expansion deformation of the coating and the substrate was similar, and the produced stress was very small, which guaranteed that the coating did not spall off the substrate, i.e. the coating has excellent thermal shock resistance. The better the thermal shock resistance, the higher the adhesive strength of the ceramic coating with the substrate is. As for 600 ℃, near the melting point of aluminum alloy, the differences between the expansion deformation of the aluminum alloy and the coating are increased so greatly that the much higher stress is produced, which leads to the spallation of the coating from the substrate.
3.5 Tensile test
Fig.6 shows the surface morphology of the tensile specimen. Fig.6(a) shows the morphology of the tensile specimen without abrading, it can be seen that partial loose layer of the coating is spalled off, and the adhesive strength is 9.5 MPa or so. Fig.6(b) shows the morphology of the tensile specimen after abrading the loose layer of the coating, in which the epoxide resin is scattered on the dense layer uniformly and the abruption morphology of the epoxide resin can be seen clearly. Only a small part of the dense layer emerges in the position where the epoxide resin is pull out. The dense layer does not spall off the substrate and the adhesive strength is 17.5 MPa, which means that the adhesive strength of the dense layer with the substrate is more than 17.5 MPa. It is known that the expansion in bulk due to transformation from t-ZrO2 to m-ZrO2 leads to the formation of inner stress; therefore the stress in the inner layer is less because of the high content of t- ZrO2, which guarantees the good adhesive strength of the coating with the substrate.

Fig.6 Morphologies of tensile samples: (a) Specimen without abrading; (b) Tensile specimen after abrading loose layer of coating
4 Conclusions
1) The coating is composed of m-ZrO2, t-ZrO2, and a little γ-Al2O3; along the section of the coating, t-ZrO2 is more on both sides than that in the middle, while m-ZrO2 is more in the middle than that on both sides.
2) The thickness of the coating is increased linearly with the increasing reaction time. There are micro holes in the coating, and the coating adheres to the substrate well without any gaps or cracks at the interface.
3) The coating has excellent thermal shock resistance under 550 ℃, while the thermal shock resistance of the coating turned a little worse under 600℃. Besides, tensile tests show the adhesive strength of dense layer with the substrate is more than 17.5 MPa.
References
[1] VAN T B, BROWN S D, WIRTZ G P. Mechanism of anodic spark deposition [J]. American Ceramic Society Bulletin, 1977, 56(6): 563-566.
[2] WIRTZ G P, BROWN S D, KRIVEN W M, WI. Ceramic coatings by anodic spark deposition [J]. Materials and Manufacturing Processes, 1991, 6(1): 87-115.
[3] YEROHIN A L, NIE X, LEYLAND A, MATTHEWS A, DOWEY S J. Plasma electrolysis for surface engineering [J]. Surface and Coatings Technology, 1999, 122: 73-93.
[4] SUNDARARAJAN G, RAMA KRISHNA L Mechanisms underlying the formation of thick alumina coatings through the MAO coating technology [J]. Surf Coat Technol, 2003, 167: 269-277.
[5] BUTYAGIN P I, KHOKHRYAOV Y E V, MAMAEV A I. Microplasma systems for creating coatings on aluminium alloys [J]. Mater Letters, 2003, 57: 1748-1751.
[6] RAMA KRISHNA L, SOMARAJU K R C, SUNDARARAJAN G. The tribological performance of ultra-hard ceramic composite coatings obtained through micro-arc oxidation [J]. Surf Coat Technol, 2003, 163: 184-490.
[7] LUO Sheng-lian, ZHOU Hai-hui, ZHEN Jin-hua, KUANG Ya-fei. Microarc oxidation of ZL cast aluminum alloys [J]. The Chinese Journal of Nonferrous Metals, 2002, 12(3): 491-495.(in Chinese)
[8] JIANG Bai-ling, BAI Li-jing, JIANG Yong-feng. Growth of alumina ceramic coatings on aluminum matrix material surface [J]. The Chinese Journal of Nonferrous Metals. 2001, 11(2): 186-189. (in Chinese)
[9] GU Wei-chao, SHEN De-jiu, WANG Yu-lin, CHEN Guang-liang, WEN-ran, ZHANG Gu-ling, FAN Song-hua, LIU Chi-zi, YANG Si-ze. Deposition of duplex Al2O3/aluminum coatings on steel using a combined technique of arc spraying and plasma electrolytic oxidation [J]. Applied Surface Science, 2006, 252: 2927-2932.
[10] XIN Shi-gang, Song Li-xin, Zhao Rong-gen, HU Xing-fang. Microstructure and strength of Al-Si-O micro-arc oxidation coating [J]. Journal of Inorganic Materials, 2006, 21(2): 493-498. (in Chinese)
[11] XUE Wen-bin, DENG Zhi-wei, CHEN Ru-Yi, ZHANG Tong-he. Growth regularity of ceramic coating formed by microarc oxidation on Al-Cu-Mg alloy [J]. Thin Solid Films, 2000, 372(1): 114-117.
[12] LIANG Bo, CHEN Huang. Overview of the application and development of ZrO2 coatings [J]. Bulletin of the Chinese Ceramic Society, 2003(6): 63-68. (in Chinese)
[13] HU Chuan-shun, WANG Fu-hui, WU Wei. Review of progress in thermal barrier coatings [J]. Corrosion Science and Protection Technology, 2000, 12(3): 160-163. (in Chinese)
[14] SCHUKIN G L, BELANOVICH A L, SAVENKO V P, IVASHKEVICH L S, SVIRIDOV V V. Micro-plasma anodic oxidation of aluminum and its alloy containing Cu in K2ZrF6 solution [J]. J Appl Chem, 1996, 69(6): 939-941. (in Russian)
(Edited by HE Xue-feng)
Foundation item: Project( 50171026) supported by the National Natural Science Foundation of China; Project (SKLT04-08) supported by the State Key Laboratory of Tribology, Tsinghua University, China
Corresponding author: JIANG Zhao-hua; Tel: +86-451-86413710; E-mail: jiangzhaohua@hit.edu.cn