
J. Cent. South Univ. (2019) 26: 2681-2687
DOI: https://doi.org/10.1007/s11771-019-4205-5
Preparation of wettable TiB2-TiB/Ti cathode by electrolytic boronizing for aluminum electrolytic
HUANG You-guo(黄有国)1, WANG Yi(王益)1, ZHANG Xiao-hui(张晓辉)2,WANG Hong-qiang(王红强)1, LI Qing-yu(李庆余)1
1. Guangxi Key Laboratory of Low Carbon Energy Materials, School of Chemistry and Pharmaceutical Sciences, Guangxi Normal University, Guilin 541004, China;
2. Guangxi Key Laboratory of Comprehensive Utilization of Calcium Carbonate Resources, College of Materials and Environmental Engineering, Hezhou University, Hezhou 542899, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Abstract: According to the problems of short life and low strength of TiB2 coating cathode for current technology in aluminium electrolysis industry, this work synthesized TiB2-TiB/Ti gradient composite with TiB2 coating and TiB whiskers in metallic Ti matrix by a electrolytic boronizing method based on similar density and thermal expansivity of the three materials. The phase composition and morphology of the cross-section were determined by X-ray diffraction (XRD), scanning electronic microscope (SEM) and X-ray energy dispersive spectrum (EDS). The results show that uniform TiB2 layer with a thickness of 8-10 μm is continuously coated on the surface while the TiB whisker connected with TiB2 layer was embedded dispersedly into the matrix. The TiB crystal whisker has a maximum length of about 220 μm. The growth rate of TiB2 and TiB is enhanced by the strong reduction of B4C. The novel gradient design of the composite helps to extend life and improve strength of the TiB2 cathode in aluminium electrolysis.
Key words: aluminium electrolysis; electrolytic boronizing; titanium diboride; gradient materials
Cite this article as: HUANG You-guo, WANG Yi, ZHANG Xiao-hui, WANG Hong-qiang, LI Qing-yu. Preparation of wettable TiB2-TiB/Ti cathode by electrolytic boronizing for aluminum electrolytic [J]. Journal of Central South University, 2019, 26(10): 2681-2687. DOI: https://doi.org/10.1007/s11771-019-4205-5.
1 Introduction
Aluminium electrolysis is a high energy consumption and serious evironment-pollution industry. New green aluminium electrolysis technology based on inert wettable cathode and inert anode can solve the problem and save energy [1, 2]. TiB2 is considered as a near-perfect cathode material due to the advantages of high melting temperature, hardness and corrosion resistance, especially good wattability with liquid metal aluminium [3]. However, some problems should be solved that TiB2 has high production cost [4-6]. The poor thermal shock resistance is another issue [7, 8], due to a strong covalent bond and ionic bond in TiB2 crystal [9, 10]. The composite method with other components, such as TiB2-TiC [11], TiB2-SiC [12], TiB2-Al2O3 [13] and TiB2-AlN [14], improves the sintering performance and mechanical properties of TiB2 to some extent. However, the conductivity of the TiB2 composite is greatly reduced and the composite elements introduced tend to cause intergranular corrosion. The TiB2/C composite enhances the conductivity and improves the machining property, but the sodium penetration property and wattability can not reach requirement in a low content of TiB2 in TiB2/C composite [15].The cost increases as the TiB2 content increases. In addition, the reaction of carbon with the molten aluminum leads to chemical damage to the cathode material.
In addition, TiB2 is generally deposited on the surface of the conductive substrate by molten salt electrodeposition which has the advantages of low cost, wide raw material resources, and lower preparation temperature. The molten salt electrodeposition is also suitable to coat a base material with a complex surface structure, so it is particularly suitable for large-scale industrial production. Moreover, the coating layer by molten salt electrodeposition exhibit smooth surface and less impurity content. The size, shape and deposition rate of the coated particles can be easily controlled by changing process parameters. However, these conductive substrates are usually made of carbon materials, the molybdenum and steel are also selected in rare case [16-18]. The above materials have big difference in thermal expansion coefficient and density with TiB2. Under the condition of high temperature in aluminum electrolysis, the binding force of TiB2 layer is weakened. As a result, TiB2 layer is broken away and even slipps off from substrate, leading to a shorten service life. Therefore, it is a developing topic to explore new TiB2-based composite cathodes with more stable structure design. Metallic Ti has the physical properties of good plasticity, machenical property and crrosion stability, combined with superior electric connectivity. All the advantages above make Ti become a better matrix for the TiB2. Moreover, Ti and TiB2 have similar density of 4.57 and 4.52 g/cm2 and thermal expansion coefficient of 8.6×10-6, and 6.2×10-6 K-1 [19], suggesting Ti is introduced as matrix of TiB2 should exhibit good sintering performance and mechanical properties, combined with better connection of electrode with current collector.
In this work, a novel gradient composite is designed and prepared by molten salt electrolytic boronizing on metallic Ti substrate. In the designed TiB2-TiB/Ti composite structure, the formed TiB2 layer on the surface is connected by TiB whiskers which are embedded into Ti bulk. More precisely, TiB also have similar density and thermal expansion coefficient with Ti and TiB2, so it is better at avoiding the weak binding problem resulted from different mechanical stress in synthesis and application. The TiB2 coating and embedded TiB enhances the wettability and serve life of the electrode for green aluminum electrolysis. The morphology was determined by SEM and the phase structure was detected by XRD. The distribution of elements of boride and the interface between molten aluminum and TB2 were analyzed by EDS, through which evaluates the wettability of TiB2-TiB/Ti composite by molten aluminum. The effect of B4C on boronizing was analyzed by thermodynamics.
2 Experimental
2.1 Materials and reagents
The titanium metal used in the experiment was commercial pure titanium with a industrial purity (impurity content: Fe≤0.3%, Si≤0.15%, C≤0.1%, N≤0.05%, O≤0.2%, H≤0.02%). Na2B4O7 and K2CO3 were analytical pure. The purity of B4C is 99%. All reagents were kept at 150 °C for 24 h in a vacuum drying oven before use.
2.2 Electrolytic boriding
The Na2B4O7, K2CO3 and B4C with a percentage of 75%, 20% and 5% in a total mass of 200 mg were mixed and then transferred into a graphite crucible. Then, the mixture is heated in a resistance furnace. The Ti sample serves as cathode while the graphite crucible serves as anode. The temperature of melts was monitored by Pt/Pt-10%Rh thermocouple. The current density is 200 mA/cm2. The boriding treatment was conducted at 950 °C for 3 h. The resulted sample was obtained after washing in boiling water to remove the solid electrolyte.
2.3 Characterization
A thin specimen of 0.3 mm was sectioned from the body by a linear cutting longitudinally. The exposed section was then polished and etched by Kroll reagent to show the microstructure. The microstructure analysing was conducted using X-ray diffraction (XRD, Rigku D/max 2500v/pc). The cross-section morphology of the sample was investigated by scanning electronic microscope (SEM, FEI Quanta 200 FEG). The element content was measured by X-ray energy dispersive spectrum (EDS, EDAX JENSIS60S).
3 Result and discussion
3.1 Phase analysis of boride surface
The crystalline structure and chemical composition of the resultant TiB2-TiB/Ti sample was studied by X-ray diffraction (XRD). Figure 1 shows the diffraction patterns of the pure titanium and borided samples respectively. Compared with the XRD pattern of the pure Ti, the peaks of the prepared composite at 950 °C shows characteristic diffraction peaks indexed to TiB2 and TiB phases. The sharp shape indicates a well crystalline structure and no impurity can be found, suggesting this boronizing process successfully synthesized TiB2-TiB/Ti composite. All the samples prepared at different temperatures above 850 °C showed the characteristic diffraction peaks of Ti, TIB and TiB2 phases. The peaks become sharper as the temperature increases due to increased degree of crystallinity.
3.2 Morphology analysis
In order to further explore the composition of the boride coating, the cross-section of the boride coating was etched by Kroll reagent. As shown in Figure 2, the different locations along the depth were analyzed with EDS. At points A and C located at the outermost layer and the inside layer of the TiB2-TiB/Ti respectively, point B is located between A and C. The results are listed in Table 1, the content of boron at point A, B and C is 47.50%, 63.03% and 22.11% respectively, implying that the distribution of boron element is irregular from the outside to inside. Moreover, the boron element is a light element whose uncertainty of amount degree is 5%-10% for EDS test. The distribution of titanium element can reflect the distribution of the whole boride. As seen from Table 1, the titanium content at point A, B and C is 19.75%, 36.97% and 77.89%, respectively with the ratios of B/Ti of 2.41, 1.70, and 0.28 for the three points. As well known, the component ratio of B/Ti decreases from surface to depth of the resultant TiB2-TiB/Ti sample. The result is in agreement with the boronizing process, which is a solid diffusion of B element in Ti base. In this process, active boron atoms diffuse from surface to inside of the Ti matrix, then the TiB crystals grow vertically and horizontally and produce whisker structure. In order to investigate the distribution of the boride element, EDS line- scan is tested in cross-section of the boride coating. Because the boron element is very light the uncertainty of EDS is very high. Only the titanium element was analyzed. As seen in Figure 3, the titanium content increases from the boride surface to the inside of boride, which agrees with the EDS results for the different location in a boride. However, the titanium content steeply increases in the TiB2 layer at some area. The increasing distance corresponds with the thickness of TiB2. The thickness of the TiB2 layer is 8-10 μm by measuring in Figure 4.
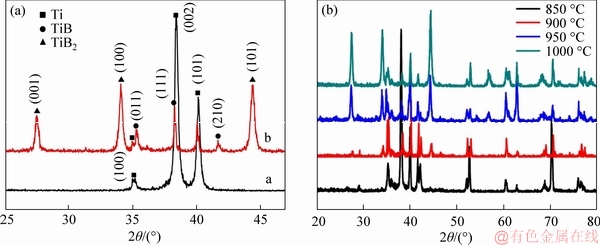
Figure 1 XRD pattern of pure Ti and TiB2-TiB/Ti sample at 950 °C (a) and samples prepared at different boriding temperatures (b)
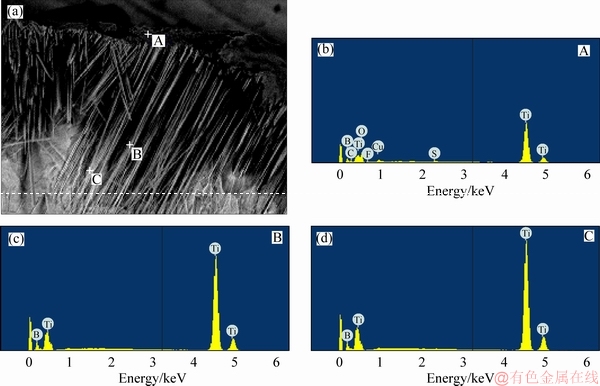
Figure 2 EDS patterns of cross-section of boride coating for different locations
Table 1 Contents of B and Ti at different locations of cross-section

The SEM images of the cross-section of the TiB2-TiB/Ti are shown in Figure 4. The boride is a gradient distribution on the surface of the sample. The outermost layer is a continuous compact layer, the next layer is consisted of disperse whiskers. Combined with the XRD result, the compacted coating layer is TiB2 and the related literature revealed the sample conclusion [20-22], while the whiskers layer is TiB phase. As shown in Figure 4(a), the thickness of TiB2 layer is about 10 μm. The TiB whiskers clearly exposed after etching by Kroll reagent show dense array. It is interesting to note that the end of the TiB whiskers firmly insert into the Ti matrix, as shown in Figure 4(c). The longest TiB whisker is about 220 μm and the diameter is about 2.08 μm, as shown in Figures 4(b) and (c), with an aspect ratio of 106, which is higher than that reported in Ref. [8, 9]. The high aspect ratio and the deeply embedded whiskers Ti matrix is beneficial to increase the adhesion between the coating layer and the base. In addition, with help of the B4C additive in this work, the thickness of TiB2 synthesized in Na2B4O7-K2CO3 for 3 h was about 10 μm with a growth rate of 3.33 μm/h, which is enhanced by the previous sample of 2.16 m/h prepared without B4C [20].
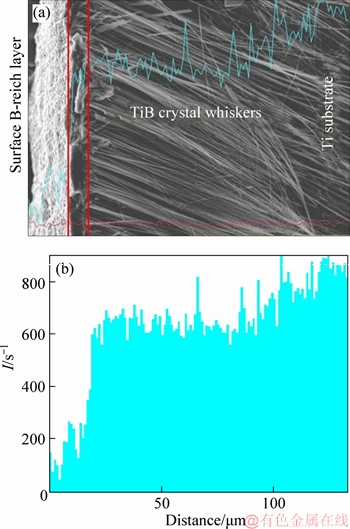
Figure 3 EDS line-scan of cross-section of TiB2-TiB/Ti sample
3.3 Electrolytic boronizing mechanism of enhance rate
The growth rate of TiB2 was largely enhanced by addition of B4C and the mechanism is discussed as follows. During electrolytic boronizing in the molten salt of Na2B4O7-K2CO3, the alkali ions in the molten salt is reduced on the cathode to form alkali metals which owns a strong reducing activity:
Na+→Na (1)
K+→K (2)
Borax is partially ionized into B2O3 in molten salt, and the alkali metals react with B2O3 and forms active boron atoms. The reactions are expressed as follows:
 (3)
(3)
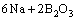 =
=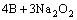 (4)
(4)
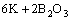 =
=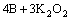 (5)
(5)
The prodeced B atoms with high activity diffuse to the titanium substrate, adsorbed on the surface, and diffuse into the lattice of Ti by solid phase diffusion. Finally, the boron reacts with Ti to produce titanium borides:
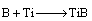 (6)
(6)
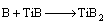 (7)
(7)
In the process above, with the addition of B4C into Na2B4O7-K2CO3 system, B4C with a certain degree of reductivity like alkali metal react with B2O3 to form active boron atoms:
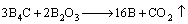 (8)
(8)
As a result, the B4C added in molten salt increases the concentration of active boron atoms. Then the increased boron potential on the surface of the metallic Ti speeds up the growth speed of crystals. However, it is noted that the growth speed does not increases unlimitedly along with increased additive amount of B4C, because the viscosity of molten salt is increased with the continuous addition of B4C. The too large viscosity seriously hinders the diffusion of activity boron atoms in molten salts.

Figure 4 SEM images of cross-section of TiB2-TiB/Ti sample:
4 Conclusions
Gradient TiB2-TiB/Ti cathode used in aluminium electrolysis was prepared by an electrolytic boronizing on titanium substrate in Na2B4O7-K2CO3-B4C melt. In the gradient composite structure, the continuous TiB2 layer has a growth rate of 3.33 μm/h and a thickness of 8-10 μm coating on the surface of the electrode, while TiB crystal whiskers grow into the Ti body with a maximum length of about 220 μm and a diameter of 2.08 μm. Boronizing rate is accelerated by the addition of B4C. Meanwhile, TiB whisker is deeply embedded in titanium substrate, which is very beneficial to increase the strength of the composite. The design of wettable TiB2 coating layer fixed by TiB whiskers in Ti matrix lays a theoretical foundation and guides the synthesis of new cathode with long life and high property in green aluminum electrolysis.
References
[1] DING Ji-lin, LI Jie, ZHANG Hong-liang, XU Yu-jie, YANG Shuai, LIU Ye-xiang. Comparison of structure and physical fields in 400 kA aluminum reduction cells [J]. Journal of Central South University, 2014, 21(11): 4097-4103.
[2] SONG Y, PENG J P, DI Y Z, WANG Y W, LI B K, FENG N X. Modified cathode with protrusions in aluminum reduction cell [J]. Metallurgical Research & Technology, 2016, 113(13): 306-312.
[3] BLOKHINA I A, IVANOV V V, KIRIK S D, NIKOLAEVA N S. Carbothermal synthesis of TiB2 powders of micron size [J]. Inorganic Materials, 2016, 9(4): 767-780.
[4] TALLON C, FRANKS G V. Exploring inexpensive processing routes to prepare dense TiB2 components [J]. Advances in Applied Ceramics, 2016, 52(6): 550-557.
[5] AN J, SONG J P, LIANG G X, GAO J J, XIE J C, CAO L, WANG S Y, LV M. Effects of HfB2 and HfN additions on the microstructures and mechanical properties of TiB2-based ceramic tool materials [J]. Materials, 2017, 10(5): 461.
[6] ZHAO K, NIU B, ZHANG F, ZHANG J. Microstructure and mechanical properties of spark plasma sintered TiB2 ceramics combined with a high-entropy alloy sintering aid [J]. Advances in Applied Ceramics, 2017, 116(1): 19-23.
[7] LIU X, PEI J H, LIU M J, WANG Z, LIU L X, JING L, WU Z J. Microstructure and mechanical properties of textured TiB2 ceramic fabricated by combination of catalyst and hot-forging [J]. Materials Chemistry and Physics, 2017, 200(1): 217-222.
[8] BASU B, RAJU G B, SURI A K. Processing and properties of monolithic TiB2 based materials [J]. International Materials Reviews, 2006, 51(6): 352-374.
[9] PANDA K B, RAVI CHANDRAN K S. Determination of elastic constants of titanium diboride (TiB2) from first principles using FLAPW implementation of the density functional theory [J]. Computational Materials Science, 2006, 35(2): 045115.
[10] FARHADI K, NAMINI A S, ASL M S, MOHAMMADZADEH A, KAKROUDI M G. Characterization of hot pressed SiC whisker reinforced TiB2 based composites [J]. International of Journal of Refractory Metals and Hard Materials, 2016, 61: 84-90.
[11] NIE J F, WANG F, LI Y S, CAO Y, LIU X F, ZHAO Y H, ZHU Y T. Microstructure evolution and mechanical properties of Al-TiB2/TiC in situ aluminum-based composites during accumulative roll bonding (ARB) process [J]. Materials, 2017, 10(2): 109.
[12] ZHAO G L, HUANG C Z, HE N, LIU H L, ZOU B. Effects of sintering conditions on microstructure and mechanical properties of reactive hot pressed TiB2-SiC ceramic composites [J]. Ceramics-Silikáty, 2016, 60(3): 226-233.
[13] POPOV A Y, SIVAK A A, BORODIANSKA H Y, SHABALIN I L. High toughness TiB2-Al2O3 composite ceramics produced by reactive hot pressing with fusible components [J]. Advances in Applied Ceramics, 2015, 114(3): 178-182.
[14] BUNIN V A, KARPOV A V, SENKOVENKO M Y. Fabrication, structure, and properties of TiB2-AlN ceramics [J]. Inorganic Materials, 2002, 38(7): 746-749.
[15] LV X J, HU L Y, SHUANG Y J, LIU J H, LAI Y Q, JIANG L X, LI J. The growth behavior of titanium boride layers in α and β phase fields of titanium [J]. Metallurgical and Materials Transactions A, 2016, 47(7): 3573-3579.
[16] RYBAKOVA N, BABUSHKINA O, ARTNER W, NAUER G E. Electrochemical synthesis of TiB2 layers out of FLiNaK electrolyte in the presence of TaCl5 additive [J]. Journal of the Electrochemical Society, 2010, 157(12): D593-D599.
[17] FASTNER U, STECK T, PASCUAL A, FAFILEK G, NAUER G E. Electrochemical deposition of TiB2 in high temperature molten salts [J]. Journal of Alloys and Compounds, 2008, 452: 32-35.
[18] KRENDELSBERGER R, SOUTO MF, SYTCHEV J, BESENHARD J O, FAFILEK G, KRONBERGER H, NAUER G E. Texture effects in TiB2 coatings electrodeposited from a NaCl-KCl-K2TiF6-NaF-NaBF4 melt at 700 °C [J]. Journal of Mining and Metallurgy, 2003, 39(1, 2): 269-274.
[19] HUANG Y G, CHEN J R, ZHANG M L, ZHONG X X, WANG H Q, LI Q Y. Electrolytic boronizing of titanium in Na2B4O7-20%K2CO3 [J]. Materials and Manufacturing Processes, 2013, 28(12): 1310-1313.
[20] TAAZIM N T, JAUHARI I, MIYASHITA Y, SABRI M F M. Development and kinetics of TiB2 layers on the surface of titanium alloy by superplastic boronizing [J]. Metallurgical and Materials Transactions A, 2016, 47A(5): 2217-2222.
[21] HUANG Y G, CHEN J R, ZHANG M L, ZHONG X X, LI Q Y, WANG H Q. Effects of M2CO3 (M=Li, Na, K) on electrolytic boronising of titanium in Na2B4O7 melts [J]. Surface Engineering, 2014, 30(2): 134-137.
[22] HUANG Y G, CHEN J R, ZHANG X H, WANG H Q, FANG Z, LI Q Y. Effects of La2O3 on electrolytic boronising of titanium [J]. Surface Engineering, 2015, 31(8): 570-574.
(Edited by HE Yun-bin)
中文导读
通过铝电解的电解渗硼制备可湿性的TiB2-TiB/Ti阴极
摘要:针对铝电解工业现有技术中TiB2涂层阴极寿命短、强度低的问题,采用密度和热膨胀率相近的三种材料的电解渗硼方法,合成了TiB2涂层和TiB晶须在金属Ti基体中的TiB2-TiB/Ti梯度复合材料。采用X射线衍射(XRD)、扫描电子显微镜(SEM)和X射线能谱(EDS)等分析手段测定了材料的相组成和截面形貌。结果表明,表面连续涂覆厚度为8~10 μm的均匀TiB2层,而与TiB2层连接的TiB晶须分散嵌入基体中。TiB晶须的最大长度约为220 μm,通过B4C的强烈还原,TiB2和TiB的生长速率得到提高,这种复合材料的新型梯度设计有助于延长TiB2阴极在铝电解中的寿命和提高其强度。
关键词:铝电解;电解渗硼;二硼化钛;梯度材料
Foundation item: Project(21473042) supported by the National Natural Science Foundation of China; Project(GUIKE-AD18126001) supported by the Guangxi Science and Technology Special Project on Bases and Talents, China
Received date: 2018-01-28; Accepted date: 2019-04-17
Corresponding author: LI Qing-yu, PhD, Professor; Tel: +86-18877313663; E-mail: liqingyu62@126.com; ZHANG Xiao-hui, PhD, Research Assistant; Tel: +86-13978324757; E-mail: zxhui017@163.com