J. Cent. South Univ. Technol. (2008) 15: 29-33
DOI: 10.1007/s11771-008-0007-x

Preparation of spherical and dense LiNi0.8Co0.2O2 lithium-ion battery particles by spray pyrolysis
HU Guo-rong(胡国荣), DENG Xin-rong(邓新荣), PENG Zhong-dong(彭忠东),
CAO Yan-bin(曹雁冰), LIU Zhi-min(刘智敏), LIU Ye-xiang(刘业翔)
(School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China )
Abstract:With citric acid as a polymeric agent layered LiNi0.8Co0.2O2 materials were synthesized by a spray pyrolysis method. The LiNi0.8Co0.2O2 particles were characterized by means of XRD, SEM and TEM. The electrochemical performances of LiNi0.8Co0.2O2 particles were studied in a voltage window of 3.00-4.35 V and at a current density of 30 mA/g. The results show that in the pilot-scale spray pyrolysis process, the morphology of particles is dependent upon the precursor concentration and flux of carrier gas. The initial discharge capacity of the LiNi0.8Co0.2O2 particles at 720 ℃ for 12 h is 187.3 mA?h/g, and the capacity remains 96.8 % with excellent cycleability after 30 cycles. The LiNi0.8Co0.2O2 samples synthesized under the optimized conditions by the spray pyrolysis method shows a good electrochemical performance.
Key words: LiNi0.8Co0.2O2; lithium-ion battery; cathode; spray pyrolysis; preparation
1 Introduction
Lithium-ion battery technology has been focusing extensively on lithiated transition metal oxides as positive electrodes because they possess the best electrochemical performances such as high energy density and good cycle stability. The major positive electrode materials currently are LiCoO2, LiMn2O4, and LiNi1-yCoyO2 (y<0.3)[1-3]. The LiCoO2 particle possesses many disadvantages in terms of cost, toxicity and electrochemical capacity.
Therefore, other materials such as LiNiO2, LiMn2O4 and LiNi0.8Co0.2O2 have been widely studied as lower cost substitutes with higher electrochemical capacity for LiCoO2. However, among these materials, a solid solution of LiNi0.8Co0.2O2 has been found to be the best because it decreases the cost of the battery by appreciably lowering the amount of expensive cobalt to 80%[4-5]. In addition, the capacity retained by LiNi0.8Co0.2O2 is higher than that of LiCoO2.
It should be noted that LiNi0.8Co0.2O2 is usually made by solid state reaction, which involves the mechanical mixing of oxides or carbonates. These reactions usually require a long heating time and several grinding during the heating process, moreover, it is difficult to control the particle size of the product. In recent years, there has been considerable interest in producing materials with various particle morphologies, bulk density, and stoichiometry by the liquid-phase reaction. It is well known that the sol-gel technique has been widely used for a homogeneous process. However, sol-gel method for preparing LiNi0.8Co0.2O2 is also not an economical one because it involves long heating time and complex synthesis process to get the final particle.
Compared with the particles prepared by solid state reaction, the size distribution of particles by ultrasonic spray pyrolysis is narrow and controllable from micrometer to sub-micrometer, the purity of the products is high and it is easy to control the composition and morphology of particles[6-9]. By controlling the process parameters, such as concentration, homogeneity of initial solution and type of precursor, it is possible to control the morphology, size distribution, homogeneity and phase composition of as-prepared particles. Post heating would also influence the nanostructure and crystallization of obtained pyrolysis particles[10].
In this work, the LiNi0.8Co0.2O2 lithium-ion battery cathode materials were prepared by the ultrasonic spray pyrolysis method. The particle characteristics, such as morphology and particle size, and electrochemical properties of the compounds were investigated.
2 Experimental
2.1 Experimental apparatus
The spray equipment consisted of an ultrasonic aerosol generator with six vibrators(1.7 MHz), and a
quartz tube(length of 1 000 mm and diameter of 50 mm), and a particle collector. The flow rate of air used as a carrier gas was 20-45 L/min. The residence time of droplets inside the reactor was about 0.6 s.
2.2 Sample preparation
In this experiment, the ultrasonic spray pyrolysis method was adopted to prepare spherical LiNi0.8Co0.2O2 lithium-ion battery cathode materials. The starting materials were prepared with lithium nitrate(LiNO3), cobalt nitrate hexahydrate[Co(NO3)2・6H2O] and nickel nitrate hexahydrate [Ni(NO3)2・6H2O]. All of these with stoichiometric molar ratio(n(Li)?n(Ni)?n(Co)=1.06? 0.80? 0.20) were dissolved in distilled water. The solution was added into a continuously agitated aqueous citric acid solution. The molar ratio of total metal ions to citric acid was fixed at 1:5. The citric acid was used as a polymeric agent for the reaction[11-12]. The prepared spray solutions were atomized by the ultrasonic nebulizer to produce droplets, which were carried into the quartz tube maintained at 700 ℃. The produced LiNi0.8Co0.2O2 lithium-ion battery cathode materials were collected by a special collector.
2.3 Characterization
The crystal structure of LiNi0.8Co0.2O2 cathode material was identified by an X-ray diffraction (XRD) pattern obtained by diffractometer (Rigaku D/max 2550 VB+). The morphology and EDS were observed by scanning electron microscope (SEM, JEOL JSM-6360LV). TEM were performed on Tecnai G220 operated at 200 kV. Electrochemical charge-discharge tests were performed using the cathode with the mixture of 80%(mass fraction) LiNi0.8Co0.2O2, 10% acetylene black(AB) and 10% polyvinylidene fluoride(PVDF). The composite cathode was prepared by mixing the active material with carbon black and PVDF into a slurry in n-methyl-2 pyrrolidone(NMP) that was coated onto aluminum foil using a doctor blade technique. The coated aluminum foil was allowed to dry overnight at 120 ℃ in an oven. The coated foil was roller pressed to better adhere the material to the current collector. The cathodes were punched as circular discs from the foil. The separator was a Celgard 2400 microporous polylene membrane. The lithium metal was used as anode in this study. The electrolyte was 1.0 mol/L LiPF6/EC+DEC (1:1,volum ratio). The cells were assembled in a glove box filled with ultra-pure argon gas. The charge-discharge cycling and cycling performances of the cells were glavanostatically performed at constant current density of 30 mA/g with a voltage window of 3.00-4.35 V(vs Li/Li+) using a LAND CT2001A computer-controlling battery testing system at 25 ℃.
3 Results and discussion
The as-prepared particles prepared by the spray pyrolysis are of spherical shape, smooth surface and non-aggregation characteristics. Fig.1 shows SEM images for as-prepared particles at different concentrations of precursor solution. The as-prepared particles obtained by spray pyrolysis at 700 ℃ in different spray solutions have different morphologies. The particles prepared at concentration of the precursor solution(c0= 0.05 mol/L) are spherical in shape, and it can be seen that the size of the particles ranges from 0.5 to 1.5 μm. However, the particles with irregular morphology were produced at concentrations of the precursor solution(c0= 0.01 mol/L or 0.1 mol/L). The viscosity of the solution follows the concentration of precursor solution. Too high or too low concentration of precursor solution forms the irregular droplets during the spraying, which can produce the particles with irregular morphology.

Fig.1 SEM images of particles synthesized with different precursor concentrations: (a) 0.01 mol/L; (b) 0.05 mol/L; (c) 0.10 mol/L
The flux of carrier gas plays an important role in determining the particle morphology[13-14]. Fig.2 shows SEM images of LiNi0.8Co0.2O2 particles prepared at different flow rates of carrier gas. The as-prepared particles prepared at the flow rate of carrier gas(v0=20 L/min) have spherical and regular morphology. However, the as-prepared particles obtained at low or high flow rate of carrier gas(v0=10 L/min or v0=45 L/min) have non-spherical and fractured morphology. The regularly spherical shape of particle was favored under the condition of a certain gas flow rate[15].

Fig.2 SEM images of particles synthesized at different flow rates of carrier gas: (a) 10 L/min; (b) 20 L/min; (c) 45 L/min
Fig.3 shows TEM image of the LiNi0.8Co0.2O2 particles prepared at 20 L/min of flow rate of carrier gas and 0.05 mol/L of precursor concentration. The droplet-to-particle conversion process of spray pyrolysis consists of some complicated transport phenomena such as evaporation of solvent, crystallization of solute, drying, thermal decomposition, and sintering. However, the particles morphology is mainly controlled by the evaporation[15]. At the constant profile, a certain concentration of the precursor solution and rate of carrier gas could make evaporation of solvent gradually progress and the precipitation of solute occur on the interior of the droplet. Thus, when the precursor concentration is 0.05 mol/L and the flow rate of carrier gas is 20 L/min, a dense and spherical particle can be prepared.

Fig.3 TEM image of particles synthesized at 0.05 mol/L of precursor concentration and 20 L/min of flow rate of carrier gas
XRD patterns of the LiNi0.8Co0.2O2 precursor obtained at 700 ℃ are shown in Fig.4. The as-prepared particles by the spray pyrolysis at 700 ℃ have pure LiNi0.8Co0.2O2 phase though crystallzability is not fully developed because of short residence time of particles inside tubular reactor for about 0.6 s. Fig.5 shows XRD patterns of the LiNi0.8Co0.2O2 particles, which were spray-pyrolysis-synthesized at 700 ℃ and subsequently heated at different temperatures for 12 h in oxygen. As can be seen, layered structure was observed exclusively in the particles without other phase. It is found that the intensity of the diffraction peaks increases with increasing heat-treatment temperature. The integrated intensity ratios of the (003) to (104) peaks, R, equal 0.89, 1.27, 1.40 and 1.68 for samples prepared at 680, 700, 720 and 750 ℃ for 12 h, when R is more than 1.2, it can be free of cation mixing[16-17]. So these samples synthesized over 680 ℃ show acceptable structure stabilization.
The surface composition of the particles was analyzed by using scanning electronic microscope energy spectra, as shown in Fig.6. The atomic ratios of prepared particles are in good agreement with the ones for the starting solution under the optimized condition. From the composition and the XRD pattern of the particles, it can be concluded that the stoichiometric LiNi0.8Co0.2O2 particles can be synthesized by spray pyrolysis successfully.
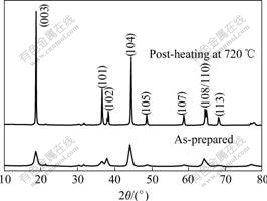
Fig.4 XRD patterns of particles obtained before and after post- heating

Fig.5 XRD patterns of LiNi0.8Co0.2O2 particles obtained at different post-heating temperatures

Fig.6 Scanning electronic microscope energy spectrum of LiNi0.8Co0.2O2 sample
Fig.7 shows curves of the discharge capacity vs cell voltage of LiNi0.8Co0.2O2 particles after post heat-treatment at 720 ℃ for different time such as 6, 12 and 24 h. The electrochemical performance of LiNi0.8Co0.2O2 particles was tested in Li half-cell. In Fig.7, the cell with 12 h heat-treatment particle exhibits the highest initial discharge capacity of 187.3 mA?h/g.

Fig.7 Initial discharge curves of LiNi0.8Co0.2O2 samples at different post-heating time
Fig.8 shows the initial discharge voltage profiles of LiNi0.8Co0.2O2 particles with 12 h (680-750 ℃) at 3.00- 4.35 V. The initial specific discharge capacity improves by rising the temperature of heat-treatment from 680 to 720 ℃, though the sample prepared at 750 ℃ reveals higher R than others, with low initial specific discharge capacity of 171.1 mA?h/g, which may be attributed to that the large particle size and the long diffusion distance of Li+ ion make the intercalation/de-intercalation difficult.

Fig.8 Initial discharge curves of LiNi0.8Co0.2O2 samples at different post-heating temperatures for 12 h
The cycle performance of LiNi0.8Co0.2O2 particles is shown in Fig.9. It can be seen that at the current density of 30 mA/g, the total discharge capacity of LiNi0.8Co0.2O2 is over 181.4 mA?h/g, and the capacity retain rate is 96.8 % after 30 cycles.

Fig.9 Capacity vs cycle number for LiNi0.8Co0.2O2 sample synthesized at 720 ℃ for 12 h
4 Conclusions
1) Spherical and dense LiNi0.8Co0.2O2 lithium-ion battery cathode particles are prepared by pilot-scale spray pyrolysis.
2) The morphologies of the products are related to the precursor concentration and flux of carrier gas. When the concentration of the precursor solution is 0.05 m/L and the flow rate of carrier gas is 20 L/min, the as-prepared particles are of spherical and regular morphology.
3) The optimal post-heating condition of the as- prepared particles is 720 ℃ for 12 h. Under the optimized conditions, the initial discharge capacity of the LiNi0.8Co0.2O2 materials is 187.3 mA・h/g, and the discharge capacity remains 96.8% after 30 cycles at current density of 30 mA/g.
References
[1] ONDA K, OHSHIMA T, NAKAYAMA M, FUKUDA K. Thermal behavior of small lithium-ion secondary battery during rapid charge and discharge cycles[J]. Journal of Power Sources, 2006, 158(1): 535-542.
[2] GUO Hua-jun, LI Xin-hai, ZHANG Xin-ming, ZENG Su-ming, WANG Zhi-xing. Characteristics of LiCoO2, LiMn2O4 and LiNi0.45Co0.1Mn0.45O2 as cathodes of lithium in batteries[J]. Journal of Central South University of Technology, 2005, 12(1): 44-49.
[3] PENG Zhong-dong, HU Guo-rong, LIU Ye-xiang. Influence on performance and structure of spinel LiMn2O4 for lithium-ion batteries by doping rare-earth Sm[J]. Journal of Central South University of Technology, 2005, 12(1): 28-32.
[4] WANG Miao-jun, NAVROTSKY A. Enthalpy of formation of LiNiO2, LiCoO2 and their solid solution, LiNi1-xCoxO2[J]. Solid State Ionics, 2004, 166(1/2): 167-173.
[5] LIU Han-san, LI Jie, ZHANG Zhong-ru, GONG Zheng-liang, YANG Yong. Structural, electrochemical and thermal properties of LiNi0.8-yTiyCo0.2O2 as cathode materials for lithium ion battery[J]. Electrochimica Acta, 2004, 49(7): 1151-1159.
[6] GURMEN S, STOPIC S, FRIEDRICH B. Synthesis of nanosized spherical cobalt powder by ultrasonic spray pyrolysis[J]. Materials Research Bulletin, 2006, 41(10): 1882-1890.
[7] JUNG D S, HONG S K, LEE H J, KANGY C. Gd2O3: Eu phosphor particles prepared from spray solution containing boric acid flux and polymeric precursor by spray pyrolysis[J]. Optical Materials, 2006, 28(5): 530-535.
[8] KRUNKS M, DEDOVA T, OJA ACIK I. Spray pyrolysis deposition of zinc oxide nanostructured layers[J]. Thin Solid Films, 2006, 515(3): 1157-1160.
[9] ESLAMIAN M, ASHGRIZ N. Effect of precursor, ambient pressure, and temperature on the morphology, crystallinity, and decomposition of powders prepared by spray pyrolysis and drying[J]. Powder Technology, 2006, 167(3): 149-159.
[10] SHIMOMURA Y, KIJIMA N. Effect of ammonium chloride addition on spray pyrolysis synthesis of BaMgAl10O17:Eu2+ phosphor without post-heating[J]. Journal of the Electrochemical Society, 2004, 151(8): H192-H197.
[11] PARK S H, OH S W, MYUNG S T, KANG Y C, SUN Y K. Effects of synthesis condition on LiNi1/2Mn3/2O4 cathode material prepared by ultrasonic spray pyrolysis method[J]. Solid State Ionics, 2005, 176(5/6): 481-486.
[12] JUNG K Y, HAN K H. Densification and photoluminescence improvement of Y2O3 phosphor particles prepared by spray pyrolysis[J]. Electrochemical and Solid-State Letters, 2005, 8(2): 17-20.
[13] GARDNER T J, MESSING G L. Preparation of MgO powder by evaporative decomposition of solutions[J]. American Ceramic Society Bulletin, 1984, 63(12): 1498-1501, 1504.
[14] LIN Jui-chen. Control of particle morphology and homogeneity during spray pyrolysis: diffusion drying stage[D]. University of Maryland, USA, 1999.
[15] MESSING G L, ZHANG S C, JAYANTHI G V. Ceramic powder synthesis by spray pyrolysis[J]. Journal of the American Ceramic Society, 1993, 76(11): 2707-2726.
[16] YANG Chi-wei. The preparation and characterization of LiNi0.8Co0.2O2 via a novel Co-precipitation[D]. Taiwan, China: Tatung University, 2004.
[17] GAO Yuan, YAKOVLEVA M V, EBNER W B. Novel LiNi1-xTix/2Mgx/2O2 compounds as cathode materials for safer lithium-ion batteries[J]. Electrochemical and Solid-State Letters, 1998, 1(3): 117-119.
(Edited by YANG Hua)
Foundation item: Project(50604018) supported by the National Natural Science Foundation of China
Received date: 2007-03-02; Accepted date: 2007-04-18
Corresponding author: DENG Xin-rong, Doctoral candidate; Tel: +86-731-8830474; E-mail: csudxr@mail.csu.edu.cn