
Electrochemical performance of Ti4+-doped LiFePO4 synthesized by co-precipitation and post-sintering method
WU Ling(�� ��), WANG Zhi-xing(��־��), LI Xin-hai(���º�), LI Ling-jun(�����),
GUO Hua-jun(������), ZHENG Jun-chao(֣����), WANG Xiao-juan(��С��)
School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China
Received 6 July 2009; accepted 23 December 2009
Abstract: Ti4+-mixed FePO4��xH2O precursor was prepared by co-precipitation method, with which Ti4+ cations were added in the process of preparing FePO4��xH2O to pursue an effective and homogenous doping way. Ti4+-doped LiFePO4 was prepared by an ambient-reduction and post-sintering method using the as-prepared precursor, Li2CO3 and oxalic acid as raw materials. The samples were characterized by scanning electron microscopy (SEM), X-ray diffractometry (XRD), electrochemical impedance spectroscopy (EIS), and electrochemical charge/discharge test. Effects of Ti4+-doping and sintering temperature on the physical and electrochemical performance of LiFePO4 powders were investigated. It is noted that Ti4+-doping can improve the electrochemical performance of LiFePO4 remarkably. The Ti4+-doped sample sintered at 600 �� delivers an initial discharge capacity of 150, 130 and 125 mA��h/g with 0.1C, 1C and 2C rates, respectively, without fading after 40 cycles.
Key words: lithium-ion battery; cathode material; LiFePO4; Ti4+-doping; co-precipitation
1 Introduction
LiFePO4 has been one of the most promising cathode materials for rechargeable lithium-ion batteries because of its low cost, low toxicity, high theoretical capacity (170 mA��h/g), excellent cycling stability and thermal stability, etc[1]. The performance of LiFePO4, however, is limited by its poor electronic conductivity and the inability of lithium ions to diffuse easily through the LiFePO4/FePO4 interface, which can result in a significant loss of capacity at high currents[1-2]. Several effective ways have been proposed to improve the electronic conductivity, including synthesis of LiFePO4/electronic conductor composites (carbon or metal nano-particles)[3-5] and substitution of a small quantity of Li+ by supervalent metal ions[6-10]. To reduce the limitation of lithium-ion diffusion, the main way is to prepare LiFePO4 powders with fine particles[11]. Recently, it is found that the porous structure of powders could also facilitate the lithium-ion diffusion[12].
It was reported that doping a small amount of Ti4+ cations into LiFePO4 could improve its rate performance and cycling stability[6]. Ti4+-doped LiFePO4 powders are usually prepared via conventional solid-state reaction of mechanically mixed lithium compounds (typically Li2CO3 or LiOH��H2O), iron compounds (typically FeC2O4��2H2O or Fe2O3), titanium compounds (typically TiO2 or Ti(OCH3)(CH3OH)2) and phosphates (typically NH4H2PO4 or (NH4)2HPO4)[6, 13-14]. However, the titanium compounds are hard to be mixed homogenously with other starting materials via mechanical mixing, due to the fact that the titanium contents are usually very low (less than 5%, molar fraction). Furthermore, the solid-state route generally contains several grindings, high temperature and long-time calcinations, which usually results in the formation of larger particles with poor electrochemical properties. In order to improve the doping effect, WANG et al[15] prepared Ti4+-doped LiFePO4 via sol-gel method and obtained good results. Nevertheless, the sol-gel method is impractical because of its high cost and complicated synthesis routes.
In this study, we introduced a novel and simple synthesis method for Ti4+-doped LiFePO4. Namely, titanium compound was added to the process of preparing FePO4��xH2O precursor to pursue a homogenous doping way, by which the ingredient Fe3+,Ti4+ and PO43- were mixed on the atomic scale in water-based solution. Therefore, Ti4+ could be evenly brought into FePO4��xH2O particles while they grew. Then, Ti4+-doped LiFePO4 was prepared by an ambient-reduction and post-sintering method using the as-prepared precursor as raw material. The effects of Ti4+-doping and sintering temperature on physical and electrochemical performance of LiFePO4 powders were investigated.
2 Experimental
2.1 Sample preparation
Ti4+-mixed FePO4��xH2O precursor was synthesized by the following procedure. 1) FeSO4��7H2O (AR), H3PO4 (AR) and Ti(SO4)2��H2O (CP) in a molar ratio of 1: 1: 0.03 were dissolved in de-ionized water to obtain 0.5 mol/L (Fe) solution; 2) Concentrated hydrogen peroxide (30%, mass fraction) was added to the solution under vigorous stirring; 3) NH3��H2O (2 mol/L, AR) was dropped into the solution to control the pH to be 2.1��0.1; subsequently, a white precipitate formed immediately; 4) After being stirred for 10 min, the precipitates were filtered, washed several times with de-ionized water and dried in an oven at 120 ��. Thus, Ti4+-mixed FePO4��xH2O powders were obtained. For comparison, pure FePO4��xH2O precursor was prepared with the same procedure.
The as-prepared precursors, Li2CO3 (AR) and oxalic acid (AR) in stoichiometric ratio (Fe?C?Li=1?1?(1-4x), where x means titanium content) were mixed by high-energy ball milling. The milling was performed at a speed of 200 r/min for 3 h at ambient temperature. The as-obtained mixtures were sintered at different temperatures (550, 600 and 650 ��) for 12 h with flowing argon. After being cooled to room-temperature, the crystalline LiFePO4 samples were obtained. More details about the ambient-reduction and post-sintering method can be found in Ref.[16].
2.2 Characterization of synthesized samples
The phase structure of LiFePO4 powders was analyzed by X-ray diffractometry (XRD, Rint-2000, Rigaku) using Cu K�� radiation, and the crystal cell parameters were calculated by WINPLOTR. The powder morphology was observed by scanning electron microscopy (SEM, JEOL, JSM-5600LV). The metal content of FePO4��xH2O powders was analyzed using inductively coupled plasma emission spectroscopy (ICP, IRIS intrepid XSP, Thermo Electron Corporation).
2.3 Electrochemical measurement
The electrochemical performance was performed using a two-electrode coin-type cell (CR2025) of Li��LiPF6 (EC?EMC?DMC=1?1?1 in volume ratio)��LiFePO4. The working cathode is composed of 80% LiFePO4 powders, 10% acetylene black as conducting agent, and 10% poly (vinylidene fluoride) as binder. After being blended in N-methyl pyrrolidinone, the mixed slurry was spread uniformly on a thin aluminum foil and dried in vacuum for 12 h at 120 ��. A metal lithium foil was used as anode. Electrodes were punched in the form of disks with 14 mm in diameter. A polypropylene micro-porous film was used as the separator. The assembly of the cells was carried out in a dry argon-filled glove box. The cells were charged and discharged over a voltage range of 2.5-4.1 V versus Li/Li+ electrode at room temperature. Electrochemical impedance measurements were conducted on a CHI660A Electrochemical Workstation (0.01 Hz-100 kHz, 5 mV).
3 Results and discussion
3.1 Structure and morphology
ICP results show that the x(Ti)/x(Fe) value of Ti4+-mixed FePO4��xH2O precursor, 2.97%, is highly close to the target value, 3.0%. This demonstrates that depositing Ti4+ in FePO4��xH2O powders by the co-precipitation method is very efficient.
X-ray diffraction (XRD) patterns of LiFePO4 and Ti4+-doped LiFePO4 synthesized at 600 �� are shown in Fig.1. All diffraction peaks are indexed to an orthorhombic crystal structure (space group Pnma), and no impurity phases are detected. Previous studies[6, 8, 13] found that Ti4+ tend to occupy M1 sites to form solid solutions without any impurity phase, owing to the fact that the ionic radius of Ti4+ in octahedral coordination (0.061 nm) is quite consistent with the radius of Li+ (0.068 nm). In contrast, if Ti4+ cations occupy M2 sites, some impurities such as Li3PO4, Fe3P and Li3Fe2(PO4)3 would be detected[14-15]. Hence, we believe that Ti4+ cations have successfully doped into M1 sites. The lattice
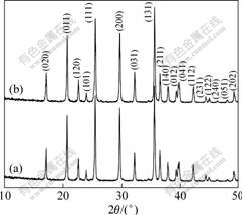
Fig.1 XRD patterns of LiFePO4 (a) and Ti4+-doped LiFePO4 (b) synthesized at 600 ��
parameters of LiFePO4 samples were calculated: a=1.032 2 nm, b=0.600 2 nm, c=0.469 5 nm, and V=0.290 9 nm3, while the Ti4+-doped LiFePO4 has slightly varied lattice parameters: a=1.031 7 nm, b=0.600 0 nm, c=0.469 3 nm, and V=0.290 5 nm3. The unit cell of the crystal lattice was slightly diminished along x, y, and z directions, which should be attributed to the existence of Li+ vacancies caused by Ti4+ doping.
The primary particle size, d, was calculated from the XRD pattern using the Scherrer formula. The d value of Ti4+-doped LiFePO4, 44.1 nm, is smaller than that of the undoped sample (46.3 nm), indicating that dopant Ti4+ could restrain the growth of LiFePO4 crystal.
Fig.2 shows the influence of Ti4+-doping on the morphologies of LiFePO4 powders. Primary particles in the size of 200-800 nm can be observed in both samples, but the Ti4+-doped sample owns less agglomeration and scatters more uniformly than the undoped one. This demonstrates that the dopant Ti4+ could effectively inhibit the particles aggregation. Reducing the particle size could enhance the lithium-ion diffusion speed cross the LiFePO4/FePO4 interface and, consequently, would improve the electrochemical properties of LiFePO4 at high current rates[11].

Fig.2 SEM images of LiFePO4 (a) and Ti4+-doped LiFePO4 (b) synthesized at 600 ��
Fig.3 represents the XRD patterns of Ti4+-doped LiFePO4 synthesized at different temperatures. The intensity of diffraction peaks of LiFePO4 powders are gradually enhanced with the increase of sintering temperature. It is clear that increasing sintering temperature could improve the crystallinity of LiFePO4; nevertheless, high temperature also results in the increase of particle size. The primary particle sizes of Ti4+-doped LiFePO4 synthesized at 550, 600 and 650 �� are 41.0, 44.1 and 49.4 nm, respectively.
SEM images of Ti4+-doped LiFePO4 synthesized at various temperatures are shown in Fig.4. The sample
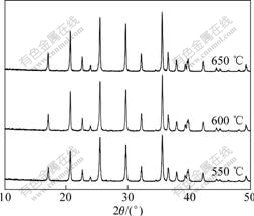
Fig.3 XRD patterns of Ti4+-doped LiFePO4 synthesized at different temperatures

Fig.4 SEM images of Ti4+-doped LiFePO4 synthesized at different sintering temperatures: (a) 550 ��; (b) 600 ��; (c) 650 ��
synthesized at 550 �� exhibits a uniform fine-grained microstructure with secondary particle size of 100-500 nm. As mentioned above, the small particle size could improve the electrochemical performance of LiFePO4. However, this sample also exhibits a relatively low crystallinity (Fig.3), which would probably make against its electrochemical properties. On the contrary, the material synthesized at 650 �� is well crystallized but serious aggregation exists. In contrast, the sample synthesized at 600 �� shows not only little aggregation but also good crystallinity; therefore, a better electrochemical performance should be expected.
3.2 Electrochemical characteristics
Fig.5 exhibits the Nyquist plots of LiFePO4 samples synthesized at 600 ��. Both Nyquist plots are comprised of a depressed semicircle in high frequency region and a straight line in low frequency region. An intercept at Z��-axis in the very high frequency region identifies the ohmic resistance (Rs) of the electrolyte and electrodes. The radius of the semicircle at high frequency region on the Z��-axis is related to the charge transfer resistance (Rct). The slope of inclined line in low frequency represents the Warburg impedance (W), which is associated with lithium-ion diffusion in LiFePO4 cathode. It is clear that the charge transfer resistance of Ti4+-doped LiFePO4 cathode (130 ��) is much smaller than the undoped one (830 ��), which should be attributed to the Ti4+ doping. Furthermore, Ti4+-doped LiFePO4 cathode has a higher slope of the inclined line in low frequency, indicating lower Warburg impedance.
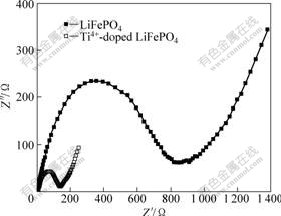
Fig.5 EIS spectra of LiFePO4 and Ti4+-doped LiFePO4 synthesized at 600 ��
Fig.6 presents the initial discharge curves and cycling performance of LiFePO4 samples at various current rates. As shown in Fig.6(a), at the discharge rate of 0.1C, 1C and 2C, LiFePO4 shows a capacity of 144, 113 and 98 mA��h/g, respectively, while the Ti4+-doped sample delivers a capacity of 150, 130 and 125 mA��h/g,
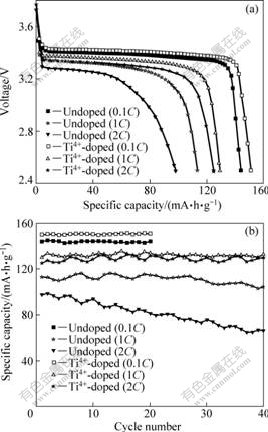
Fig.6 Initial discharge curves (a) and cycling performance (b) of LiFePO4 and Ti4+-doped LiFePO4 synthesized at 600 ��
respectively. From Fig.6(b), it is clear that both samples exhibit excellent cyclic stability at 0.1C rate, but at higher current rates, the Ti4+-doped sample shows much better cycling performance. After 40 cycles, Ti4+-doped LiFePO4 shows a capacity of 133 and 130 mA��h/g at 1C and 2C rate, respectively, and retains 102.3% and 104.0% of its initial discharge capacity, while LiFePO4 only maintains 92.0% (104 mA��h/g) and 67.3% (66 mA��h/g) of its initial discharge capacity. From analysis above, Ti4+-doped LiFePO4 with better electrochemical properties can be attributed to the following reasons: 1) Ti4+ doping intrinsically improves the bulk electronic conductivity of LiFePO4 material by inducing an increased p-type semi-conductivity[6]; 2) Ti4+ doping effectively inhibits the particles aggregation and, consequently, enhances the lithium-ion diffusion speed cross the LiFePO4/FePO4 interface; 3) Ti4+ doping reduces the charge transfer resistance (Rct) and Warburg impedance (W) of LiFePO4 cathode.
The influence of sintering temperature on electrochemical performance of Ti4+-doped LiFePO4 is shown in Fig.7. The samples synthesized at 550, 600 and 650 �� deliver initial discharge capacities of 107, 130 and 121 mA��h/g, respectively, and maintains a capacity of 100 (93.5% of its initial value), 132 (101.5% of its initial value) and 116 mA��h/g (95.9% of its initial value) after 40 cycles, respectively. The sample synthesized at 550 �� exhibits the lowest discharge capacity and the worst cyclic stability, which are ascribed to the poor crystallinity. Also, the electrochemical properties of Ti4+-doped LiFePO4 synthesized at 650 �� are not satisfactory, due to the serious particles aggregation. The sample synthesized at 600 �� shows the most impressive performance, owing to its good crystallinity and little aggregation.
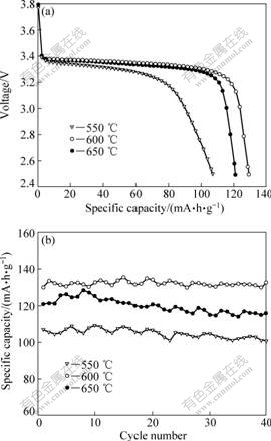
Fig.7 Initial discharge curves (a) and cycling performance (b) of Ti4+-doped LiFePO4 synthesized at different temperatures (Charge/discharge at 1C rate)
4 Conclusions
1) Olive-type Ti4+-doped LiFePO4 is synthesized by co-precipitation and post-sintering method. XRD results indicate that Ti4+ ions enter into the lattices of LiFePO4 crystal and do not obviously change its structure.
2) Ti4+ doping can effectively inhibit the LiFePO4 particles aggregation and consequently enhances the Li+ diffusion speed cross the LiFePO4/FePO4 interface. And Ti4+ doping can reduce the charge transfer resistance and Warburg impedance of LiFePO4 electrode.
3) The Ti4+-doped LiFePO4 synthesized at 600 �� shows the most impressive electrochemical performance with the initial discharge capacity of 150, 130 and 125 mA��h/g at 0.1C, 1C and 2C rate, respectively, and without capacity fading after 40 cycles.
References
[1] PADHI A K, NANJUNDASWAMY K S, GOODENOUGH G B. Phospho-olivines as positive-electrode materials for rechargeable lithium batteries [J]. J Electrochem Soc, 1997, 144(4): 1188-1194.
[2] PROSINI P P, LISI M, ZANE D, PASQUALI M. Determination of the chemical diffusion coefficient of lithium in LiFePO4 [J]. Solid State Ionics, 2002, 148: 45-51.
[3] LIU H P, WANG Z X, LI X H, GUO H J, PENG W J, ZHANG Y H, HU Q Y. Synthesis and electrochemical properties of olivine LiFePO4 prepared by a carbothermal reduction method [J]. J Power Sources, 2008, 184: 469-472.
[4] CROCE F, EPIFANIO A D, HASSOUN J, DEPTULA A, OLCZAC T. A novel concept for the synthesis of an improved LiFePO4 lithium battery cathode [J]. Electrochem Solid-State Lett, 2002, 5(3): A47-A50.
[5] MI C H, CAO Y X, ZHANG X G, ZHAO X B, LI H L. Synthesis and characterization of LiFePO4/(Ag+C) composite cathodes with nano-carbon webs [J]. Powder Technol, 2007, 179: 171-176.
[6] CHUNG S Y, BLOKING J T, CHIANG Y M. Electronically conductive phospho-olivines as lithium storage electrodes [J]. Nat Mater, 2002, 1(2): 123-128.
[7] ROBERTS M R, VITINS G, OWEN J R. High-throughput studies of Li1-xMgx/2FePO4 and LiFe1-yMgyPO4 and the effect of carbon coating [J]. J Power Sources, 2008, 179: 754-762.
[8] HU Y, DOEFF M M, KOSTECKI R, FINONES R. Electrochemical performance of sol-gel synthesized LiFePO4 in lithium batteries [J]. J Electrochem Soc, 2004, 151(8): A1279-A1285.
[9] WU L, LI X H, WANG Z X, LI L J, ZHENG J C, GUO H J, HU Q Y, FANG J. Synthesis and electrochemical properties of metals-doped LiFePO4 prepared from the FeSO4��7H2O waste slag [J]. J Power Sources, 2009, 189: 681-684.
[10] WANG Xiao-qiong, LI Xin-hai, WANG Zhi-xing, GUO Hua-jun, FENG Wen-jie, LIU Feng-ju. Synthesis and performance of LiFe0.9Ni0.1PO4 as cathode material in lithium-ion battery [J]. The Chinese Journal of Nonferrous Metals, 2006, 16(4): 739-745. (in Chinese)
[11] YAMADA A, CHUNG S C, HINOKUMA K. Optimized LiFePO4 for lithium battery cathodes [J]. J Electrochem Soc, 2001, 148(3): A224-A229.
[12] YU F, ZHANG J, YANG Y, SONG G. Preparation and characterization of mesoporous LiFePO4/C microsphere by spray drying assisted template method [J]. J Power Sources, 2009, 189: 794-797.
[13] HU Guo-rong, GAO Xu-guang, PENG Zhong-dong, DU Ke, TAN Xian-yan, LIU Yan-jun. Influence of Ti4+ doping on electrochemical properties of LiFePO4/C cathode material for lithium-ion batteries [J]. Trans Nonferrous Met Soc China, 2007, 17: 296-300.
[14] SUN Y H, LIU X Q. Preparation and characterization of novel Ti-doped M-site deficient olivine LiFePO4 [J]. Chin Chem Lett, 2006, 17(8): 1093-1096.
[15] WANG G X, BEWLAY S, NEEDHAM S A, LIU H K, LIU R S, DROZD V A, LEE J F, CHEN J M. Synthesis and characterization of LiFePO4 and LiTi0.01Fe0.99PO4 cathode materials [J]. J Electrochem Soc, 2006, 153(1): A25-A31.
[16] ZHENG J C, LI X H, WANG Z X, GUO H J, ZHOU S Y. LiFePO4 with enhanced performance synthesized by a novel synthetic route [J]. J Power Sources, 2008, 184: 574-577.
(Edited by YANG Bing)
Foundation item: Project(2007CB613607) supported by the National Basic Research Program of China
Corresponding author: WANG Zhi-xing; Tel: +86-731-88836633; E-mail: wuling19840404@163.com
DOI: 10.1016/S1003-6326(09)60219-3