
Behavior of titanium dioxide in alumina carbothermic reduction-chlorination process in vacuum
ZHU Fu-long1, 2, 3, YUAN Hai-bin1, 2, 3, YU Qing-chun1, 2, YANG Bin1, 2,
XU Bao-qiang1, 2, DAI Yong-nian1, 2
1. National Engineering Laboratory of Vacuum Metallurgy, Kunming University of Science and Technology, Kunming 650093, China;
2. Key Laboratory of Nonferrous Metals Vacuum Metallurgy of Yunnan Province, Kunming University of Science and Technology, Kunming 650093, China;
3. Faculty of Materials Science and Engineering, Kunming University of Science and Technology, Kunming 650093, China
Received 15 September 2010; accepted 6 April 2011
Abstract: Behaviors of TiO2 in the alumina carbothermic reduction and chlorination process in vacuum at different temperatures were investigated experimentally by means of XRD, SEM and EDS. In the preparation of materials, the molar ratio of Al2O3 to C was 1:4, and 10% TiO2 and excess AlCl3 were added. The results show that TiC is produced by C and TiO2 after TiO2 transforms from anatase into rutile gradually. In the temperature range of 1 763-1 783 K, the compounds of Ti and Al are not found in slags and condensate. The purity of aluminum reaches 98.35%, and TiO2 does not participate in alumina carbothermic reduction process and chlorination process in vacuum.
Key words: titanium dioxide; alumina; carbothermic reduction; chlorination
1 Introduction
It is known that aluminum is not produced from bauxite directly. The process producing aluminum industrially includes alumina refining and aluminum electrolysis. But the development of electrolysis process is limited by some factors such as expensive raw materials, serious environmental pollution and large electrical energy consumption [1]. In recent years, extracting aluminum from alumina or bauxite by subhalide decompositions process has been studied [2-4]. The mechanism of this process includes carbothermic reduction, chlorination and subhalide decomposition [5]. The recovery rate and the content of aluminum may change obviously when bauxite is used as material [6]. Because of the presence of impurities such as silica, iron oxide and titanium dioxide, the content of alumina in bauxite is different (25%-80%). The content of titanium dioxide is less than 11% roughly [7]. The disproportionations of titanium dioxide in the aluminium extraction with subfluorides in vacuum have been studied [8]. If titanium dioxide less than 10% (mass fraction) was added in raw materials, the recovery rate of aluminum increases with increasing the content of titanium dioxide [6]. On the other hand, it is con?rmed that the reactions of TiO2 and C could occur as [9]: TiO2→TinO2n-1(n>4), TinO2n-1→Ti4O7, Ti4O7→Ti3O5, Ti3O5→Ti2O3, Ti2O3→TiCxOy and TiCxOy→TiC. Ti3AlC2, TiAl3, Ti3Al, Ti2AlC, Ti3AlC and Ti3AlC2 could be successfully obtained by the reaction of TiC, Ti, Al4C3 and Al [10-12]. But specific effects of titanium dioxide in alumina carbothermic reduction process at different temperatures in chlorination process have not been studied systematically.
The aim of the present work is to investigate the definite behavior of TiO2 in alumina carbothermic reduction and chlorination process. The behaviors of TiO2 at different temperatures were studied by XRD analysis. The surface morphology and composition of condensation product were examined by SEM and EDS.
2 Experimental
2.1 Raw material
Analytical grade alumina, graphite, titanium dioxide and anhydrous aluminum chloride were used as raw materials in the experiments.
2.2 Experimental method
Alumina (15 g) and graphite (6 g) as starting materials were mixed on a molar ratio of 1?4 of Al2O3to C with addition of 10% TiO2. They were then made into pieces of d 20 mm × 5 mm under pressure of 2-6 MPa. The pieces and AlCl3 were put in different graphite crucibles (shown in Fig. 1). The heating rate of the system was about 20 K/min. In the alumina carbothermic reduction process, the system needs to be kept at 993, 1 193, 1 393, 1 493, 1 593, 1 713 and 1 763 K for 30 min, respectively. Afterward, AlCl3 sublimated at 373-403 K and was transported to the carbothermic reduction slag for 40-90 min, then chlorination process started. At high temperature, AlCl(g) was produced and moved into upper condensing towers. Meanwhile, AlCl(g) decomposed into AlCl3(g) and Al(l) at low temperature. The schematic diagram of vacuum furnace is shown in Fig. 1.
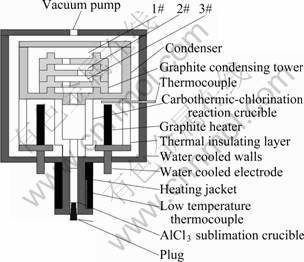
Fig. 1 Schematic diagram of vacuum furnace
To identify the effect of TiO2 on carbothermic reduction, another powder mixture without TiO2 powders was prepared and kept at 1 763 K for 30 min in the same way. The molar ratio of Al2O3 to C in the mixture remained 1:4.
2.3 Analysis methods
The crystalline phase of the products was identified by X-ray diffraction instrument (Rigaku Corporation, D/max-3B). The structural features and chemical composition were characterized by scanning electron microscopy and EDAX (Philips, Holland), respectively.
3 Results and discussion
3.1 Behavioral analysis of TiO2 in alumina carbothermic reduction process at different temperatures
The XRD patterns of the slags after alumina carbothermic reduction process at different temperature are shown in Fig. 2. Because the location and intensity of diffraction peaks of γ-Al2O3, η-Al2O3, θ-Al2O3, κ-Al2O3 and δ-Al2O3 are similar, it is difficult to determine the specific crystalline forms of alumina. But there is obvious difference between corundum (α-Al2O3) and other alumina [13]. Only Al2O3 and α-Al2O3 are marked in Fig. 2.
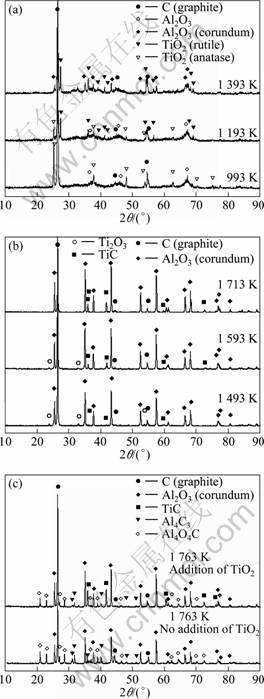
Fig. 2 XRD patterns of slag after carbothermic reduction at different temperatures
It can be seen from Fig. 2(a) that the slag contains alumina, graphite and anatase at 993 K. Diffraction peaks of rutile already appear at 1 193 K. The experimental result is consistent with that in Ref. [14], which indicates that the phase transformation of TiO2 occurs at 1 193 K or below. With increasing temperature from 1 193 to 1 393 K, the intensity of diffraction peaks of rutile increases, while that of significantly anatase becomes weaker than that at 993 K and finally disappears.
Besides, it is observed that the diffraction peak of α-Al2O3 appears and original alumina is consumed completely at 1 393 K. The experimental result is consistent with that in Ref. [15], which reaches the changing conditions of α-Al2O3. It was reported that the crystalline transformations of many materials may be affected by some elements and compounds [16-17]. But in this process, the crystalline transformations of TiO2 and Al2O3 do not involve with each other.
In Fig. 2(b), graphite and α-Al2O3 remain unchanged at 1 493-1 713 K. Ti2O3 and TiC are generated, while rutile disappears at 1 493 K. The intensity of TiC increases with increasing temperature from 1 493 to 1 713 K. Ti2O3 reduces and disappears gradually. Only Ti2O3 and TiC are detected while other intermediates fail to be detected, according to Ref. [8], because the reactions of these intermediate products may be too fast, and the interval of temperature range of experimental research is too broad and other factors.
A graph of Gibbs free energy change (ΔGT) related to temperature (T) under 120 Pa is drawn to clarify these processes (Fig. 3). Because of being short of thermo- dynamic data on TinO2n-1 (n>10) and TiCxOy, only TiO2→Ti4O7, Ti4O7→Ti3O5, Ti3O5→Ti2O3, Ti2O3→TiC are analyzed in these processes:
4TiO2+C=Ti4O7+CO(g) (1)
3Ti4O7+C=4Ti3O5+CO(g) (2)
2Ti3O5+C=3Ti2O3+CO(g) (3)
Ti2O3+5C=2TiC+3CO(g) (4)
As shown in Fig. 3, these process reactions begin at 1 250 K or below, but the experiments show that all these begin above 1 393 K. This is because the reaction rate of solid-solid phase reaction is not only relative with temperature, but also with activation energy, distribution of grain, contact area of reactant and etc [18].
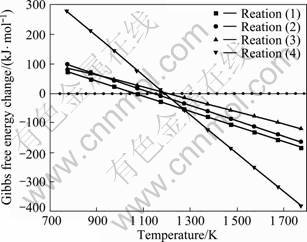
Fig. 3 Relationship between Gibbs free energy change and temperature under 120 Pa
The XRD patterns of the slags kept at 1 763 K for 30 min with and without the addition of TiO2 are shown in Fig. 2(c). The intermediates (Al4C3 and Al4O4C) which are very important to the chlorination process [6] are generated by reactions between graphite and alumina at 1 763-1 783 K in vacuum (15-120 Pa). As seen in Fig. 2(c), the intermediates are produced by graphite and alumina whether adding TiO2 or not. No compounds of Ti and Al are detected (Ti3AlC2, Al3Ti, Ti2AlC and so on).
Although TiC is produced by the reaction between graphite and TiO2, this process is completed before alumina carbonthermic reduction. The intermediates (Al4C3 and Al4O4C) generated in alumina carbothermic reduction process are not apparently different with or without the addition of TiO2. Compounds of Ti and Al are not detected in alumina carbothermic reduction process. Titanium-containing materials are not involved in this process.
3.2 Behavioral analysis of TiO2 in chlorination process
Intermediates could be participated in chlorination process at 1 763-1 783 K in vacuum (15-120 Pa) [6]. Figure 4 shows the XRD pattern of the slags obtained after chlorination process during which TiO2 is added. The materials contained in slags are the same as the materials after carbothermic reduction. The compounds of Ti and Al are also not detected after chlorination process, which implicates that TiC is not involved in chlorination process.
During the chlorination process, AlCl(g) is generated, which would decompose at low temperature, then aluminum is produced [6]. As shown in Fig. 5, aluminum appears in every condensing tower. Element C is detected in the condensate because the condensing towers are made from graphite. Al4C3 in condensing towers may be produced by the reaction between Al and C as:
4Al+3C=Al4C3 (5)
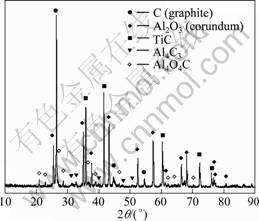
Fig. 4 XRD pattern of slag after carbothermic reduction- chlorination process
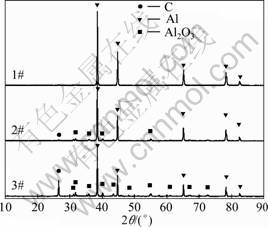
Fig. 5 XRD patterns of condensates from different condensing tower
As shown in Fig. 6, the Gibbs free energy change (ΔGT) of reaction (5) is below zero when the system temperature is below 1 773 K. Below and above the melting point of aluminum (993.4 K), the Gibbs free energy change of reaction (5) is evident. Because the temperature gradient in the condenser is obvious, the condensates in 2# condensing tower contain less Al4C3 than those in 3#. Aluminum in 1# condensing tower is pure.
Figure 7 shows the SEM image and EDS analysis of condensates obtained from 1# condensing tower. The product aluminum has a filar structure. As shown in the EDS image, it is obvious that the contents of aluminum and oxygen in condensates are 98.35% and 1.65% (mass fraction), respectively. The titanium-containing compound is not detected in the condensate, which indicates that it is not involved in the subhalide decomposition process.
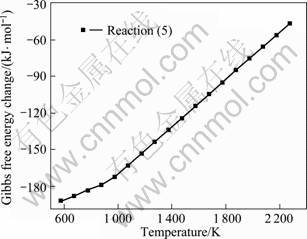
Fig. 6 Relationships between Gibbs free energy change and temperature

Fig. 7 SEM image and EDS analysis of condensation product
4 Conclusions
1) In the carbothermic reduction process, after TiO2 transforms from anatase into rutile gradually, TiC is produced by the reaction between TiO2 and C. In the range of 1 763-1 783 K, Al3C4 and Al4O4C are generated by alumina and graphite with TiC. The compounds of Ti and Al in slag are not detected.
2) In chloride reaction process at 1 763-1 783 K, TiC remains unchanged and the intermediates participate in the reaction. Aluminum is generated and its purity is 98.35%. Compounds of Ti and Al in slag or titanium- containing materials in the condensate are not found.
3) The experimental results indicate that TiO2 is not involved in the alumina carbothermic reduction- chlorination process.
References
[1] FANG Qi-xue, HUANG Guo-zhi, GE Chang-li, LIAO Xin-qin. Overview of world bauxite resource [J]. Light Metals, 2000(10): 8-11. (in Chinese)
[2] WANG Ping-yan, LIU Mou-sheng, DAI Yong-nian. Vacuum metallurgy of Al from bauxite by carbothermic reduction-chlorination [J]. Chinese Journal of Vacuum Science and Technology, 2006, 26(5): 377-380. (in Chinese)
[3] LI Qiu-xia, CHEN Wei-liang, DAI Yong-nian. Preparation of aluminum by disproportion of sub-fluoride in vacuum [J]. The Chinese Journal of Nonferrous Metals, 2008, 18(8): 1550-1554. (in Chinese)
[4] WU Gou-yuan, LIU Da-chun, DAI Yong-nian. Dynamics study of Al extraction from Al2O3 by Al sub-sulphidization [J]. Chinese Journal of Vacuum Science and Technology, 2004, 24(4): 263-270. (in Chinese)
[5] YUAN Hai-bin, YANG Bin, XU Bao-qiang, YU Qing-chun, FENG Yue-bin, DAI Yong-nian. Aluminum production by carbothermo-chlorination reduction of alumina in vacuum [J]. Transactions of Nonferrous Metals Society of China, 2010, 20(8): 1505-1510.
[6] YUAN Hai-bin, ZHU Fu-long, YANG Bin, YU Qing-chun, XU Bao-qiang, DAI Yong-nian. Process of aluminum production by alumina carbothermic reduction-chlorination in vacuum[J]. Chinese Journal of Vacuum Science and Technology, 2010, 30(6): 528-533. (in Chinese)
[7] LIU Zhong-fan. Overview of world bauxite resource [J]. Light Metals, 2001(5): 7-12. (in Chinese)
[8] LI Qiu-xia, JIN Bi, LIU Yong-cheng, DAI Yong-nian. Dispropor-tionation of titanium dioxide in preparation process aluminum with subfluoride in vacuum[J]. Vacuum, 2008, 45(4): 20-23. (in Chinese)
[9] GAO Yun-ming, LI Ci-ying, LI Ya-wei, LI Yuan-bing, NIE Jian-hua, YANG Da-bing. Analysis of carbothermal reduction of TiO2 and extraction of titanium carbonitride from the blast furnace slag bearing titania[J]. Journal of Wuhan University of Science & Technology: Natural Science Edition, 2007, 30(1): 5-9. (in Chinese)
[10] SONGA I H, KIMB D K, HAHNA Y D, KIMA H D. Investigation of Ti3AlC2 in the in situ TiC-Al composite prepared by the exothermic reaction process in liquid aluminum [J]. Materials Letters, 2004, 58(5): 593-597.
[11] KENNEDY A R, WESTON D P, JONES M I. Reaction in Al-TiC metal matrix composites [J]. Materials Science and Engineering A, 2001, 316(1-2): 32-38.
[12] GAUTHIER-BRUNET V, CABIOC’H T, CHARTIER P, JAOUEN M, DUBOIS S. Reaction synthesis of layered ternary Ti2AlC ceramic [J]. Journal of the European Ceramic Society, 2009, 29(1): 187-194.
[13] SHANG Lian-di. Research and identification of eight kinds of crystalline alumina [J]. Chemistry World, 1994(7): 346-350. (in Chinese)
[14] YIN Li-song, SHEN Hui. Research progress and application of photocatalysis of TiO2 [J]. Material Herald, 2000, 14(12): 23-25. (in Chinese)
[15] LIU Zhi-qiang. Preparation of high purity ultrafine aluminum oxide(II)―Calcination influence on the property of aluminum oxide powder[J]. Journal of Guangdong Non-ferrous Metals, 2002, 12(2): 99-101. (in Chinese)
[16] CAO Hong-shi, CHEN Sha-ou, SHAO WEI-quan, LI Da, SUN Rui-feng, ZHANG Sha-sha. Effect of rare earth dopant on phase transformation and sintering behavior of γ-Al2O3 [J]. Bulletin of the Chinese Ceramic Society, 2008, 27(5): 924-932. (in Chinese)
[17] WANG Li-jian, ZHENG Shui-lin, TIAN Wen-jie. Effects of carrier on phase transformation and crystallite growth of titania in TiO2/diatomite [J]. Journal of the Chinese Ceramic Society, 2008, 36(11): 1644-1648. (in Chinese)
[18] HUA Yi-xin. Kinetics of metallurgical process[M]. Beijing: Metallurgy Industry Press, 2004: 84-86. (in Chinese)
氧化铝真空碳热还原-氯化工艺中
二氧化钛的行为分析
朱富龙1, 2, 3, 袁海滨1, 2, 3, 郁青春1, 2, 杨 斌1, 2, 徐宝强1, 2, 戴永年1, 2
1. 昆明理工大学 真空冶金国家工程实验室,昆明 650093;
2. 昆明理工大学 云南省有色金属真空冶金重点实验室,昆明 650093;
3. 昆明理工大学 材料科学与工程学院,昆明 650093
摘 要:在不同温度下氧化铝真空碳热还原和氯化反应的过程中,利用XRD、SEM 和EDS检测手段分析TiO2的行为。在制备材料时,Al2O3和C的摩尔比为1:4,并添加10% TiO2和过量的AlCl3。结果表明,TiO2 从锐钛矿型转化为金红石型后与C反应生成TiC。在1 763-1 783 K的温度区间,在残渣和冷凝物中没有发现Ti和Al的化合物。生成铝的纯度达到98.35%,且TiO2不参与氧化铝真空碳热还原和氯化过程。
关键词:二氧化钛;氧化铝;碳热还原;氯化反应
(Edited by FANG Jing-hua)
Foundation item: Project (u0837604) supported by the Joint Funds of the National Natural Science Foundation of China and Yunnan Province; Project (20095314110003) supported by the Special Research Funds of the Doctor Subject of Higher School, China
Corresponding author: YANG Bin; Tel: +86-871-5161583; E-mail: kgyb2005@126.com, zfl279@163.com
DOI: 10.1016/S1003-6326(11)60941-2