άκΉ”ΫΜΜΜ ς÷§¥”Μ‘νβΩσΥα–‘Ϋΰ≥ω“Κ÷–÷±Ϋ”Χα»ΓMo (VI)ΒΡΨ≤Χ§ΚΆΕ·Χ§ΈϋΗΫ―–ΨΩ
ά¥‘¥ΤΎΩ·ΘΚ÷–Ιζ”–…ΪΫπ τ―ß±®(”ΔΈΡΑφ)2018ΡξΒΎ8ΤΎ
¬έΈΡΉς’ΏΘΚΗΕ‘ΤΖψ –Λ«εΙσ ΗΏ“ψ”± Ρΰ≈σΗη –λΚλ±ρ ’≈ή≤
ΈΡ’¬“≥¬κΘΚ1660 - 1669
ΙΊΦϋ¥ ΘΚνβΘΜΥα–‘Ϋΰ≥ω“ΚΘΜάκΉ”ΫΜΜΜ ς÷§ΘΜΕ·ΝΠ―ßΘΜ…œ÷υΝ§–χΈϋΗΫ
Key wordsΘΚmolybdenum; acidic leach solution; ion exchange resin; kinetics; continuous column adsorption
’Σ “ΣΘΚ―–ΨΩάκΉ”ΫΜΜΜ ς÷§D301‘Ύ«ΩΥα–‘Ϋΰ≥ω“Κ÷–Ε‘ΝυΦένβΒΡΈϋΗΫ––ΈΣΓΘάϊ”Ο…®ΟηΒγΨΒ(SEM)ΓΔΒγΉ”ΡήΤΉ(EDS)ΚΆά≠¬ϋΙβΤΉ(Raman spectra)Ε‘ ς÷§ΒΡΈϋΗΫΡήΝΠΓΔΈϋΗΫΕ·ΝΠ―ßΚΆΈϋΗΫΜζάμΫχ––œξœΗΩΦ≤λΓΘΫαΙϊ±μΟςΘ§D301άκΉ”ΫΜΜΜ ς÷§‘Ύ«ΩΥα–‘Ϋΰ≥ω“Κ÷–ΒΡΕ‘νβΈϋΗΫΝΩ¥οΒΫ463.63 mg/gΘ§ΈϋΗΫΩΊ÷Τ≤Ϋ÷ηΈΣΩ≈ΝΘΡΎά©…ΔΙΐ≥ΧΘ§Β± ς÷§ΝΘΕ»¥”0.9~1.2 mmΦθ–ΓΒΫ0.6~0.9 mm ±Θ§ΜνΜ·Ρή‘ρ¥”25.47 kJ/molΫΒΒΆΒΫ20.38 kJ/molΓΘΫβΈω Β―ι±μΟςΘ§2 mol/LΒΡΑ±Υ°Ω…“‘ΉςΈΣD301 ς÷§ΒΡΫβΈϋ“ΚΓΘΕ·Χ§…œ÷υ Β―ι―ι÷ΛάκΉ”ΫΜΜΜ ς÷§D301‘Ύ«ΩΥα–‘Ϋΰ≥ω“ΚΕ‘νβ÷±Ϋ”Χα»ΓΒΡΩ…–––‘ΓΘ¥ΥΆβΘ§ΡφΝςΈϋΗΫΩ…“‘ΗΡ…ΤΕ·Χ§Ν§–χΈϋΗΫ–ßΙϊΓΘ
Abstract: The adsorption behavior of ion exchange resin D301 in the extraction of hexavalent molybdenum from high acidic leach solution was investigated. SEM, EDS and Raman spectra analyses were applied to studying the adsorption capacity, reaction kinetics and possible adsorption mechanism in detail. Results showed that the adsorption capacity of D301 resin for molybdenum from high acidic leach solution was up to 463.63 mg/g. Results of the kinetic analysis indicated that the adsorption process was controlled by the particle diffusion with the activation energy 25.47 kJ/mol (0.9-1.2 mm) and 20.38 kJ/mol (0.6-0.9 mm). Furthermore, the molybdenum loaded on the resin could be eluted by using 2 mol/L ammonia hydroxide solution. Besides, dynamic continuous column experiments verified direct extraction of molybdenum from acidic leach solutions by ion exchange resin D301 and the upstream flow improved dynamic continuous absorption.
Trans. Nonferrous Met. Soc. China 28(2018) 1660-1669
Yun-feng FU1,2,3, Qing-gui XIAO1,2, Yi-ying GAO1,2, Peng-ge NING1,2, Hong-bin XU1,2, Yi ZHANG1,2
1. Key Laboratory of Green Process and Engineering, Institute of Process Engineering, Chinese Academy of Sciences, Beijing 100190, China;
2. National Engineering Laboratory for Hydrometallurgical Cleaner Production Technology, Institute of Process Engineering, Chinese Academy of Sciences, Beijing 100190, China;
3. University of the Chinese Academy of Sciences, Beijing 100049, China
Received 18 April 2017; accepted 12 December 2017
Abstract: The adsorption behavior of ion exchange resin D301 in the extraction of hexavalent molybdenum from high acidic leach solution was investigated. SEM, EDS and Raman spectra analyses were applied to studying the adsorption capacity, reaction kinetics and possible adsorption mechanism in detail. Results showed that the adsorption capacity of D301 resin for molybdenum from high acidic leach solution was up to 463.63 mg/g. Results of the kinetic analysis indicated that the adsorption process was controlled by the particle diffusion with the activation energy 25.47 kJ/mol (0.9-1.2 mm) and 20.38 kJ/mol (0.6-0.9 mm). Furthermore, the molybdenum loaded on the resin could be eluted by using 2 mol/L ammonia hydroxide solution. Besides, dynamic continuous column experiments verified direct extraction of molybdenum from acidic leach solutions by ion exchange resin D301 and the upstream flow improved dynamic continuous absorption.
Key words: molybdenum; acidic leach solution; ion exchange resin; kinetics; continuous column adsorption
1 Introduction
In industry, molybdenite concentrate (MoS2) is the main mineral sources (approximately 85%) of molybdenum deposit for manufacturing molybdenum downstream products, so it is widely used in the commercial metallurgical processes [1-4]. In traditional treatment of molybdenite, oxidation roasting followed by ammonia leaching is employed to recover molybdenum [4-6]. However, limited by the characteristics of mineral and process themselves, there are still many unavoidable problems: volatilization loss of MoO3, low concentration of SO2 and generation of insoluble molybdate [7,8].
Aqueous pressure leaching of molybdenite con- centrate with oxygen in neutral water and recovery of dissolved molybdenum by ion exchange resin have therefore become important in recent years as a promising industrial process. This method is characterized as simple medium, environment-friendly, high recovery of molybdenum and technical feasibility. In the leaching process, solid leaching residue and residual acid solution are obtained. From the solid residue, molybdenum can be easily separated by subsequently leaching with ammonia solution and precipitating as ammonium molybdate of commercial grade. The residual acid solution produced in leaching process dissolves valuable molybdenum and other non- valuable metals [9-11].
So far, many methods have been developed for the extraction of molybdenum from acidic solution, such as chemical precipitation [12], solvent extraction [13-16] and ion exchange [17-21]. Precipitating molybdenum from acidic solution requires a large amount of alkali to neutralize the acid in solution, which costs much and may introduce other impurity ions. The operation of solvent extraction is inconvenient in the commercial run. During the past decade, the extraction of molybdenum from solution by various ion exchange resins has also been reported and the feasibility of extracting molybdenum from solution has already been confirmed.
NGUYEN and LEE [17] found that the best condition to separate molybdenum and vanadium from sulfuric acid solution was at pH lower than 1.2 and the loading of molybdenum by AG1-x8 resin was nearly 0 at pH lower than -1. According to Ref. [19], the adsorption of molybdenum at pH 7.25 was nearly 200 mg/g by D301 resin. ZHANG et al [20] revealed that the loaded molybdenum on resin could be displaced by tungstate when the molybdenum and tungsten coexisted in the solution at pH 7.25. However, many problems still remain to limit the use of ion exchange resin. The study of molybdenum extraction by various ion exchange resins mainly focused on the pH value from 2 to 7 because molybdenum in this pH scope existed as negative ion with multiple charge [19,20,22]. Although PENG et al [23] proposed that at pH 0.68, molybdenum could combine with  to form heteropoly acid anion in acidic sulfate solution and therefore in this pH range the molybdenum in solution could be recovered by anion resin. But, the actual leach solution of aqueous pressure leaching of molybdenite concentrate with oxygen in neutral water was highly acidic and few studies were conducted on the molybdenum extraction by ion exchange resin in this pH range in actual leach solution. Therefore, it is interesting and practical to study the direct extraction of molybdenum from acidic leach solution of molybdenite ore by ion exchange resin.
to form heteropoly acid anion in acidic sulfate solution and therefore in this pH range the molybdenum in solution could be recovered by anion resin. But, the actual leach solution of aqueous pressure leaching of molybdenite concentrate with oxygen in neutral water was highly acidic and few studies were conducted on the molybdenum extraction by ion exchange resin in this pH range in actual leach solution. Therefore, it is interesting and practical to study the direct extraction of molybdenum from acidic leach solution of molybdenite ore by ion exchange resin.
In this work, the separation of molybdenum and other metals (Cu, Fe, Al and Mg) from the leach residual solution was concerned. The objectives of this research were to 1) find the optimum condition to extract molybdenum from acidic solution in batch experiments; 2) study the adsorption kinetics of extraction of molybdenum from acidic solution; 3) investigate the desorption of molybdenum from the resin; and 4) investigate the adsorption of molybdenum in column experiments.
2 Experimental
2.1 Chemicals and ion exchange resin
The weak basic anion exchanger D301 (Tianjin Nankai HECHENG S&T Co., Ltd., Tianjin, China) with tertiary ammonium functional groups was used. The physicochemical properties of resin are listed in Table 1. Before use, the resin was first soaked in deionized water for 24 h, and then treated with NaOH and HCl, alternately, and rinsed with deionized water until the eluate was at pH=7, finally the resin was dehydrated at 50 ΓψC.
The actual leach solution of aqueous pressure leaching with oxygen of molybdenum ore was used as the feed solution, which mainly contained Mo, Fe, Mg, Al and Cu. In addition to these metal elements, a large number of non-metallic ions, H+ and  , were also in the solution. The concentrations of these components in solution are shown in Table 2. The high acidic leach solution was used without further treatment. All the chemicals used in this study were of analytical reagent grade.
, were also in the solution. The concentrations of these components in solution are shown in Table 2. The high acidic leach solution was used without further treatment. All the chemicals used in this study were of analytical reagent grade.
Table 1 Properties of D301 resin

Table 2 Concentrations of acidic leach solution of molybdenum ore (g/L)

2.2 Batch adsorption experiments
Batch experiments were carried out to study the effect of liquor-to-resin ratio, grain size and temperature on the adsorption of molybdenum. For each experimental run, the leach solution was used as feed solution with known doses of resin in a water-bath shaker at a constant temperature. After the desired adsorption time, filtration was conducted to separate the resin from solution. The concentrations of molybdenum in the feed solution and adsorbed solution were analyzed.
2.3 Kinetic experiments
For each kinetic experiment, 0.1 g of pretreated resin was stirred in 60 mL of leach solution at a certain temperature. The liquor-to-solid ratio was so large that the molybdenum concentration in the solution could be regarded as constant during adsorption to ensure the infinite solution volume kinetic condition. After the desired adsorption time, filtration was conducted to separate the resin from the solution. Then, the resin was rinsed to remove the ions on the resin surface. The adsorbed molybdenum ions could be completely desorbed by using 2.5 mol/L NaOH solution at 45 ΓψC. Therefore, such solution was used as desorption agent to recovery Mo on the spent resin, and the concentration of molybdenum in desorption solution was analyzed.
2.4 Column experiments
In column experiments, a glass column (d1.5 cm ΓΝ 45 cm) was used. 30 mL (1 bed volume) of the pretreated resin was wet-packed into the glass column. The trials were carried out at 25 ΓψC with a constant flow rate of 30 mL/h. The flow rate of the solution was controlled by a pump. The effluent from the column was collected periodically and the concentration of molybdenum was analyzed.
2.5 Analysis and characterization
The concentration of molybdenum in the solution was analyzed by using inductively coupled plasma- optical emission spectrometer (PE Optima 5300DV, PerkinElmer), and the extraction rate of molybdenum, adsorption rate of molybdenum at a certain time in batch experiments and kinetic experiments could be calculated according to the following equations:
Π«=(C0-Ct)/C0ΓΝ100% (1)
q=(C0-Ce)V/m (for batch experiments) (2)
q=CdVd/m (for kinetic experiments) (3)
where Π« is the extraction rate of molybdenum, %; q is the amount of molybdenum adsorbed by the resin (mg/g); C0 is the initial concentration of molybdenum (mg/mL); Ct is the concentration of molybdenum at time t (mg/mL); Ce is the concentration of molybdenum at equilibrium (mg/mL); V is the initial volume of molybdenum solution (mL); Cd is the concentration of the desorption solution; Vd is the volume of desorption solution (mL); and m is the mass of the resin (g).
In order to explore the process of adsorption, the adsorbed resin particles were dried at 65 ΓψC, and then cut from the middle to achieve symmetrical flat cross surface. The morphology of cross surface and the distribution of molybdenum along the diameter direction were analyzed by a scanning electron microscopy (JSM-7610F) and an X-ray electron dispersive spectroscopy (EDS).
3 Results and discussion
3.1 Selectivity of adsorption
Figure 1 presents the relationship between adsorption amount of metal elements and the adsorption time. From Fig. 1, it could be seen that the molybdenum adsorption increased with the increase of the adsorption time to near 463 mg/g. While the adsorption capacities of Fe, Cu, Al and Mg were as low as 5 mg/g. Besides, the adsorption capacities of Fe, Cu, Al and Mg did not increase with the increase of time. Therefore, results indicated that the resin could be used to recover molybdenum from highly acidic leach solution.
3.2 Batch adsorption
Figure 2 shows the effect of liquor-to-resin ratio on molybdenum extraction rate and adsorption amount of resin. From Fig. 2(a), it was found that the absorption of molybdenum increased with the increase of contact time. In the initial 60 min, molybdenum extraction rate increased sharply because of the large number of blank adsorption sites on resin, after that the extraction rate increased slowly, and became constant after 10 h. Molybdenum extraction rate varied from 67.4% to 97.5% with the increase of liquor-to-resin from 0.6/30 to 1.6/30. Whilst Fig. 2(b) showed that in a certain amount of feed solution, the adsorption amount of resin decreased from 416.25 to 225.72 mg/g with the increase of resin dosage, resulting in the wastage of the resin.

Fig. 1 Relationship between adsorption amount of metal elements and adsorption time (Adsorption condition: feed solution 30 mL, resin mass 0.1 g, grain size 0.9 to 1.2 mm, 55 ΓψC and shaking rate 300 r/min)

Fig. 2 Effect of liquor-to-resin ratio on molybdenum extraction rate (a) and adsorption amount of resin (b) (Adsorption condition: solution 30 mL, grain size 0.9-1.2 mm, 25 ΓψC and 270 r/min)
Figure 3 shows the effect of grain size of the resin on molybdenum extraction rate and adsorption amount of resin. As shown in Fig. 3(a), the grain size of the resin significantly influenced the adsorption of molybdenum. It was found that compared to the particle size of resin between 0.9 to 1.2 mm, the molybdenum extraction rate at particle size of resin between 0.6 to 0.9 mm after 10 h adsorption increased from 77.04% to 84.78% when 0.8 g resin was used for 30 mL feed solution. And with the amount of smaller resin for 30 mL solution increasing to 1.2 g, the molybdenum extraction rate could reach 95.76%. Figure 3(b) shows that the adsorption amount of resin was obviously improved by using smaller grain size. It increased from 356.77 to 392.63 mg/g when 0.8 g was used for 30 mL solution.

Fig. 3 Effect of grain size of resin on molybdenum extraction rate (a) and adsorption amount of resin (b) (Adsorption condition: solution 30 mL, 25 ΓψC and 270 r/min)
Figure 4 reveals the influence of temperature on molybdenum adsorption rate and adsorption amount of resin. The results demonstrated that an increase in temperature both increased molybdenum extraction rate and the adsorption amount of resin, and the significant influence was mainly behaved on these two indexes in the initial 3 h. After 10 h, the molybdenum extraction rate and adsorption amount of resin at 25, 35 and 45 ΓψC were 95.76%, 97.47% and 99.3% and 295.66 mg/g, 300.95 mg/g and 306.6 mg/g, respectively. From the data, we could find that the molybdenum extraction rate and adsorption amount of resin at different temperatures did not vary much. Besides, high temperature was not economic to apply in industry. Therefore, room temperature was chosen reasonably for adsorption experiments.

Fig. 4 Effect of temperature on molybdenum extraction rate (a) and adsorption amount of resin (b) (Adsorption condition: solution 30 mL, resin 1.2 g, grain size 0.6-0.9 mm and 270 r/min)
3.3 Adsorption kinetics
Interruption experiment was carried out under conditions of feed solution 40 mL, resin 0.5 g, 35 ΓψC, shaking rate 270 r/min and grain size of resin between 0.9 to 1.2 mm. After adsorption for a time, resins were rapidly separated from the feed solution for a few minutes. Then, the resins were reloaded in the solution and the adsorption was resumed. Figure 5 shows the variation on adsorption rate of molybdenum with time during the interruption experiment. As shown in Fig. 5, the slope of the adsorption curve after interruption was much larger than that before interruption, suggesting that the adsorption rate was faster after interruption. Such a variation in adsorption rate could be attributed to the slow diffusion rate in the resin particle. During the long interruption, the molybdenum in particle diffusion layer gradually further diffused to the new adsorption surface with the slow rate which increased the molybdenum concentration gradient between the surface and the internal of the resin. The acceleration of molybdenum adsorption rate was caused by this increased concentration gradient. Meanwhile, based on this slow diffusion rate, we deduced that the rate-controlling step of the adsorption process was particle diffusion.

Fig. 5 Adsorption curve of resin in interruption experiment (Adsorption condition: solution 40 mL, resin 0.5 g, 35 ΓψC, 270 r/min and grain size of resin 0.9 to 1.2 mm)
It was a well-established fact that this kind of ionic adsorption in aqueous system was a heterogeneous process, and the adsorption occurred on the active adsorption sites of the resin particles. In addition, the shape of resin particles was supposed to be spherical without volume variation. Hence, in the present study, a shrinking particle model can be used to describe the adsorption process. In shrinking particle model, there were three simplified kinetic equations shown as below when the rate-determining steps were different [24-26].
x=kLt (Liquid boundary layer diffusion control) (4)
1-3(1-x)2/3+2(1-x)=kPt (Particle diffusion control) (5)
1-(1-x)1/3=kSt (Surface reaction control) (6)
where x is the adsorption rate of molybdenum; k is the adsorption rate constant; and t is the adsorption time.
Figure 6 presents adsorption curves of the resin with the particles size of 0.9-1.2 mm and 0.6-0.9 mm, respectively. According to Figs. 6(a) and (b), the increase of temperature and the decrease of the resin particle size were conducive to molybdenum adsorption. And to understand the rate-determining steps of the molybdenum adsorption process, the adsorption rates of molybdenum at temperature from 25 to 55 ΓψC were fitted into the kinetic equation, with the results presented in Figs. 6(c), (d) and Table 3. It was found that the data obtained from the experiments fitted Eq. (5) perfectly when the resin particle sizes were 0.9-1.2 mm and 0.6-0.9 mm, respectively. This result indicated that the extraction rate of molybdenum in acidic leach solution by resin was particle diffusion controlled. From Table 3, it could be seen that the reaction rate constant k increased from 0.00147 to 0.00386 min-1, as the temperature increased from 25 to 55 ΓψC. Furthermore, with resin particle size decreasing from 0.9-1.2 to 0.6-0.9 mm, there was an obvious increment of the reaction rate constant k. These kinetic results were consistent with previous batch experimental results. The apparent activation energy was calculated by using Eq. (7), and the results were presented in Figs. 6(e) and (f).
ln k = ln A-Ea/(RT) (7)
where k is the reaction rate constant; Ea is the apparent activation energy; A is the pre-exponential factor; R is the molar gas constant; and T is the temperature.
The apparent activation energy values for the extraction rate of molybdenum in acidic leach solution by resin were calculated to be 25.47 and 20.38 kJ/mol, when the resin particles sizes were 0.9-1.2 and 0.6-0.9 mm, respectively, indicating that reducing the resin particle sizes could lower the activation energy remarkably.
In order to understand the adsorption process of molybdenum in acidic leach solution by resin intuitively, the mineralogy and component analyses of the resin particles in kinetics experiments were carried out by using SEM and EDS as presented in Fig. 7. It was observed that there were clear boundaries in the regions of loaded, loading and unloaded molybdenum ions. As the adsorption proceeded, the molybdenum ions in solution gradually diffused into the resin particles resulting in that the loaded and loading regions broadened, and the unloaded region diminished. When adsorption was completed, the resin particles were fully loaded by molybdenum ions. Integrating the previous adsorption kinetics study, the SEM and EDS analyses were regarded as an important proof that the adsorption process was particle diffusion controlled.
3.4 Adsorption mechanisms

Fig. 6 Adsorption curves of resin with particle sizes of 0.9-1.2 mm (a) and 0.6-0.9 mm (b) at different temperatures, plot of 1-3(1-x)2/3+2(1-x) vs time at various temperatures for molybdenum adsorption of resin with particle sizes of 0.9-1.2 mm (c) and 0.6-0.9 mm (d), and Arrhenius plots for molybdenum adsorption of resin with particle size 0.9-1.2 mm (e) and 0.6-0.9 mm (f)
Table 3 Kinetic parameters of molybdenum extraction
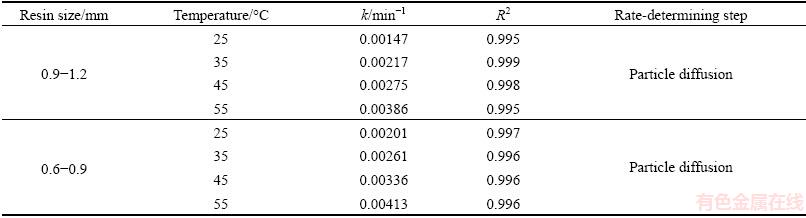

Fig. 7 SEM images (a, c, e) and EDS analyses (b, d, f) of adsorbed D301 particles at different contact time
Previous researches revealed that in strongly acidic solution, molybdenum was in the form of cationic species including  ,
,  and
and  [23,27], while impurity metal elements also existed as cation, such as Fe3+, Mg2+, Al3+ and Cu2+, indicating that molybdenum and other metal elements could not be separated in strongly acidic solution by cation exchanger. Figure 8 shows the Raman spectrum of leach liquor, suggesting that absorption peaks emerged at 915-923, 953-956, 983-985 and 1054 cm-1 confirmed the existence of
[23,27], while impurity metal elements also existed as cation, such as Fe3+, Mg2+, Al3+ and Cu2+, indicating that molybdenum and other metal elements could not be separated in strongly acidic solution by cation exchanger. Figure 8 shows the Raman spectrum of leach liquor, suggesting that absorption peaks emerged at 915-923, 953-956, 983-985 and 1054 cm-1 confirmed the existence of  ,
,  ,
,  and
and  , respectively [28-32]. However, the above batch adsorption experiments showed that molybdenum ions could be completely adsorbed by anion exchanger in acidic solutions, suggesting that molybdenum ions existed in an anionic form. Figure 9 presents the SEM and EDS analyses of adsorbed D301 particles at 600 min. It could be seen that besides the molybdenum ions the sulfur was also adsorbed. Therefore, it was reasonable to deduce that the adsorption of
, respectively [28-32]. However, the above batch adsorption experiments showed that molybdenum ions could be completely adsorbed by anion exchanger in acidic solutions, suggesting that molybdenum ions existed in an anionic form. Figure 9 presents the SEM and EDS analyses of adsorbed D301 particles at 600 min. It could be seen that besides the molybdenum ions the sulfur was also adsorbed. Therefore, it was reasonable to deduce that the adsorption of  was due to the generation of
was due to the generation of  for the combination with
for the combination with  [23].
[23].
3.5 Resin desorption
Generally, it was known that ammonium molybdate was one of the most important molybdenum chemical products. Thus, ammonia hydroxide was chosen as the eluant for molybdenum from the loaded resin. The concentration of ammonia hydroxide was varied to 2, 3, 4, 5 and 6 mol/L and the mass ratio of the loaded resin to ammonia hydroxide solution was kept at 2:3.15. The temperature was kept at 30 ΓψC. Figure 10 showed that the desorption rate of molybdenum reached 96% as the concentration of ammonia hydroxide varied from 2 to 6 mol/L. By combining the SEM images of resin before (Figs. 7(e) and (f)) and after (Fig. 11) elution, it could be concluded that it was easy to elute from the resin with ammonia hydroxide.

Fig. 8 Raman spectrum of leach liquor

Fig. 9 SEM image (a) and EDS analyses (b, c) of adsorbed D301 particles at 600 min

Fig. 10 Desorption curve of loaded resin

Fig. 11 SEM image (a) and EDS analysis (b) of resin after elution
3.6 Column adsorption experiments
Continuous column experiments were carried out with the acidic leach solution. Figure 12 shows the variation in the concentrations of metals with time. From Fig. 12(a) it could be seen that in downstream experiment, Fe, Mg, Al and Cu were detected in the effluent after 1 bed volume, the concentrations of Fe, Mg, Al and Cu increased speedily in the effluent and this was soon the same as that in the feed solution, indicating that the loading of Fe, Mg, Al and Cu was nearly zero and these metals would remain in the effluent. While, the breakthrough point for molybdenum occurred at 3 bed volumes, which indicated that molybdenum could separate from other metals in the solution by using this ion exchange method. And according to this curve, the loading capacity of the resin for molybdenum in this condition was 142.83 mg/g. This value was much lower than that of the batch experiment due to the short residence time between the resin and the feed solution. Therefore, upstream flow was employed to prolong the residence time of the feed solution in the column. From Fig. 12(b), it could be seen that the variation in the concentrations of Fe, Mg, Al and Cu in the effluent was the same as that of the downstream experiment, suggesting that the resin did not adsorb Fe, Mg, Al and Cu. Moreover, the load of molybdenum was improved dramatically. The breakthrough point occurred after 6 bed volumes of feed solution, and the loading capacity of the resin for molybdenum in this condition was 285.66 mg/g.

Fig. 12 Column adsorption curves in downstream (a) and upstream (b) experiments of resin for Mo, Fe, Mg, Al and Cu
4 Conclusions
1) The loading capacity of D301 resin for molybdenum from high acidic leach solution was up to 463.63 mg/g.
2) The adsorption process was controlled by the particle diffusion, and the activation energy decreased from 25.47 to 20.38 kJ/mol, with the resin particle sizes reducing from 0.9-1.2 mm to 0.6-0.9 mm.
3) The molybdenum in the loaded resin could be eluted by the use of ammonia solution.
4) Continuous column experiments verified direct extraction of molybdenum from acidic leach solutions by ion exchange resin D301 and the upstream flow was improved absorption.
Acknowledgements
The authors will extend our sincere thanks to Dr. Peng ZHOU for his revision in our manuscript.
References
[1] LESSARD J D, SHEKHTER L N. Thermodynamic modeling of atmospheric hydrometallurgical removal of chalcopyrite from molybdenite concentrates [J]. Hydrometallurgy, 2014, 150(1): 9-13.
[2] LASKOWSKI J S, CASTRO S, RAMOS O. Effect of seawater main components on frothability in the flotation of Cu-Mo sulfide ore [J]. Physicochemical Problems of Mineral Processing, 2014, 50(1): 17-29.
[3] HADI A, ZAHRA M, ZIAEDIN S S, NOAPARAST M, NAVID M M. Removal of copper from molybdenite concentrate by mesophilic and extreme thermophilic microorganisms [J]. International Journal of Mining Science and Technology, 2013, 23(6): 827-834.
[4] WANG Lu, ZHANG Guo-hua, DANG Jie, CHOU Kuo-chi. Oxidation roasting of molybdenite concentrate [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(12): 4167-4174.
[5] KHOLMOGOROV A G, KONONOVA O N, PASHKOV G L, KACHIN S V, PANCHENKO O N, KALAKINA O P. Molybdenum recovery from mineral raw materials by hydrometallurgical methods [J]. The European Journal of Mineral Processing and Environmental Protection, 2002, 2(2): 82-93.
[6] ZHOU Qiu-sheng, YUN Wei-tao, XI Jun-tao, LI Xiao-bin, QI Tian-gui, LIU Gui-hua, PENG Zhi-hong. Molybdenite®Climestone oxidizing roasting followed by calcine leaching with ammonium carbonate solution [J]. Transactions of Nonferrous Metals Society of China, 2017, 27(7): 1618-1626.
[7] TRIPATHY P K, RAKHASIA R H. Chemical processing of a low grade molybdenite concentrate to recover molybdenum [J]. Mineral Processing and Extractive Metallurgy, 2013, 115(1): 8-14.
[8] ZHAO Zhong-wei, LI Jiang-tao, CAO Cai-fang, HUO Guang-sheng, ZHANG Gang, LI Hong-gui. Recovery and purification of molybdenum from Ni-Mo ore by direct air oxidation in alkaline solution [J]. Hydrometallurgy, 2010, 103(1): 68-73.
[9] PARSONS G J, BRIMACOMBE J K, PETERS E. Computer simulation of a molybdenite leaching process using dilute nitric acid [J]. Hydrometallurgy, 1987, 17(1): 133-154.
[10] MEHDI M G, BAHRAM R, ANAHITA S. Leaching and kinetic modelling of molybdenite concentrate using hydrogen peroxide in sulfuric acid solution [J]. Iran J Chem Chem Eng, 2014, 33(2): 73-85.
[11] SMIRNOV K M, RASPOPOV N A, SHNEERSON Y M, LAPIN A Y, BITKOV G A, MENSHIKOV Y A, PASKHIN P N, KIRICHENKO V P. Autoclave leaching of molybdenite concentrates with catalytic additives of nitric acid [J]. Russian Metallurgy (Metally), 2010, 2010(7): 588-595.
[12] BARRY H F, HALLADA C J. Liquid phase oxidation process: U.S. patent, 3656888 [P]. 1972-04-18.
[13] ZENG Li, LIAO Xiao-li, SUN Ya-hui, XIAO Lian-sheng. Direct extraction of molybdenum from high acid leach solutions of Ni-Mo ore using an oxime extractant of HBL101 [J]. International Journal of Refractory Metals and Hard Materials, 2015, 51(1): 14-18.
[14] WU Peng-hui, FENG Guang-fen. The processes of strong acid leaching-Solvent extraction of Ni-Mo ore: China patent, CN1033784A [P]. 1989-07-12. (in Chinese)
[15] COCA J, DIEZ F V, MORIS M A. Solvent extraction of molybdenum and tungsten by Alamine 336 and DEHPA [J]. Hydrometallurgy, 1990, 25(1): 125-135.
[16] PARHI P K, PARK K H, KIM H I, PARK J T. Recovery of molybdenum from the sea nodule leach liquor by solvent extraction using Alamine 304-I [J]. Hydrometallurgy, 2011, 105(3): 195-200.
[17] NGUYEN T H, LEE M. Separation of molybdenum and vanadium from acid solutions by ion exchange [J]. Hydrometallurgy, 2013, 136(1): 65-70.
[18] KEKES T, TOROK T I, ISSHIK M. Anion exchange of chromium, molybdenum and tungsten species of various oxidation states, providing the basis for separation and purification in HCl solutions [J]. Hydrometallurgy, 2005, 77(1): 81-88.
[19] ZHAO Zhong-wei, ZHANG Jia-liang, CHEN Xing-yu, LIU Xu-heng, LI Jiang-tao, ZHANG Wei-guang. Separation of tungsten and molybdenum using macroporous resin: Equilibrium adsorption for single and binary systems [J]. Hydrometallurgy, 2013, 140(1): 120-127.
[20] ZHANG Jia-liang, LIU Xu-heng, CHEN Xing-yu, LI Jiang-tao, ZHAO Zhong-wei. Separation of tungsten and molybdenum using macroporous resin: Competitive adsorption kinetics in binary system [J]. Hydrometallurgy, 2014, 144(1): 77-85.
[21] LI Yan-chun, YUAN Ying-zhen, LIU Hui, PENG Bing, LIU Zhi-lou. Iron extraction from lead slag by bath smelting [J]. Transactions of Nonferrous Metals Society of China, 2017, 27(8): 1862-1869.
[22] KIRIYAMA T, KURODA R. Application of anion-exchange techniques to the determination of traces of molybdenum in sea water [J]. Talanta, 1984, 31(6): 472-474.
[23] PENG Jun, WANG Xue-wen, JIANG Chang-jun, WANG Ming-yu, MA Yi-qian, XIANG Xiao-yan. Separation of Mo(VI) and Fe(III) from the acid leaching solution of carbonaceous Ni-Mo ore by ion exchange [J]. Hydrometallurgy, 2014, 142(1): 116-120.
[24] LEVENSPIEL O. Chemical reaction engineering [M]. 3rd ed. New York: John Wiley and Sons, Inc, 1999.
[25] ZHANG Hai, XU Hong-bin, ZHANG Xiao-fei, ZHANG Yang, ZHANG Yi. Pressure oxidative leaching of Indian chromite ore in concentrated NaOH solution [J]. Hydrometallurgy, 2014, 142(1): 47-55.
[26] MA Jia-yu, DU Xue-lan, QIN Yuan-hang, WU Zai-kun, CHI Ru-an, WANG Cun-wen. Kinetics on leaching of potassium from phosphorus®Cpotassium associated ore in HCl-H3PO4 media [J]. Transactions of Nonferrous Metals Society of China, 2017, 27(8): 1870-1877.
[27] OLAZABAL M A, ORIVE M M, FERNANDEZ L A, MADARIAGA J M. Selective extraction of vanadium (V) from solutions containing molybdenum (Vi) by ammonium salts dissolved in toluene [J]. Solvent Extraction and Ion Exchange, 2007, 10(4): 623-635.
[28] KNOPF D A, LUO B P, KRIEGER U K, KOOP T. Thermodynamic dissociation constant of the bisulfate ion from Raman and ion interaction modeling studies of aqueous sulfuric acid at low temperatures [J]. The Journal of Physical Chemistry A, 2003, 107(1): 4322-4332.
[29] NAM Y I, SEO S Y, KANG Y C, KIM M J, SENANAYAKE G, TRAN T. Purification of molybdenum trioxide calcine by selective leaching of copper with HCl-NH4Cl [J]. Hydrometallurgy, 2011, 109(1-2): 9-17.
[30] YUEN K A, SIMON N C, GULARI E. Spectroscopic and scattering investigation of isopoly-molybdate and tungstate solutions [J]. Polyhedron, 1984, 3(8): 1001-1011.
[31] HIMENO S, ISHII N, HASEGAWA M, SAITO A. Spectrophotometric studies on the hexamolybdate ion in aqueous organic solutions [J]. Inorganica Chimica Acta, 1987, 131(1): 11-13.
[32] JENTZSCH P V, KAMPE B, ROSCH P, POPP J. Raman spectroscopic study of crystallization from solutions containing MgSO4 and Na2SO4: Raman spectra of double salts [J]. J Phys Chem A, 2011, 115(22): 5540-5546.
ΗΕ‘ΤΖψ1,2,3Θ§–Λ«εΙσ1,2Θ§ΗΏ“ψ”±1,2Θ§Ρΰ≈σΗη1,2Θ§–λΚλ±ρ1,2Θ§’≈ ή≤1,2
1. ÷–ΙζΩΤ―ß‘ΚΙΐ≥ΧΙΛ≥Χ―–ΨΩΥυ ¬Χ…ΪΙΐ≥Χ”κΙΛ≥Χ÷ΊΒψ Β―ι “Θ§±±Ψ© 100190ΘΜ
2. ÷–ΙζΩΤ―ß‘ΚΙΐ≥ΧΙΛ≥Χ―–ΨΩΥυ ΣΖ®“±Ϋπ«εΫύ…ζ≤ζΦΦ θΙζΦ“ΙΛ≥Χ Β―ι “Θ§±±Ψ©100190ΘΜ
3. ÷–ΙζΩΤ―ß‘Κ¥σ―ßΘ§±±Ψ© 100049
’Σ “ΣΘΚ―–ΨΩάκΉ”ΫΜΜΜ ς÷§D301‘Ύ«ΩΥα–‘Ϋΰ≥ω“Κ÷–Ε‘ΝυΦένβΒΡΈϋΗΫ––ΈΣΓΘάϊ”Ο…®ΟηΒγΨΒ(SEM)ΓΔΒγΉ”ΡήΤΉ(EDS)ΚΆά≠¬ϋΙβΤΉ(Raman spectra)Ε‘ ς÷§ΒΡΈϋΗΫΡήΝΠΓΔΈϋΗΫΕ·ΝΠ―ßΚΆΈϋΗΫΜζάμΫχ––œξœΗΩΦ≤λΓΘΫαΙϊ±μΟςΘ§D301άκΉ”ΫΜΜΜ ς÷§‘Ύ«ΩΥα–‘Ϋΰ≥ω“Κ÷–ΒΡΕ‘νβΈϋΗΫΝΩ¥οΒΫ463.63 mg/gΘ§ΈϋΗΫΩΊ÷Τ≤Ϋ÷ηΈΣΩ≈ΝΘΡΎά©…ΔΙΐ≥ΧΘ§Β± ς÷§ΝΘΕ»¥”0.9~ 1.2 mmΦθ–ΓΒΫ0.6~0.9 mm ±Θ§ΜνΜ·Ρή‘ρ¥”25.47 kJ/molΫΒΒΆΒΫ20.38 kJ/molΓΘΫβΈω Β―ι±μΟςΘ§2 mol/LΒΡΑ±Υ°Ω…“‘ΉςΈΣD301 ς÷§ΒΡΫβΈϋ“ΚΓΘΕ·Χ§…œ÷υ Β―ι―ι÷ΛάκΉ”ΫΜΜΜ ς÷§D301‘Ύ«ΩΥα–‘Ϋΰ≥ω“ΚΕ‘νβ÷±Ϋ”Χα»ΓΒΡΩ…–––‘ΓΘ¥ΥΆβΘ§ΡφΝςΈϋΗΫΩ…“‘ΗΡ…ΤΕ·Χ§Ν§–χΈϋΗΫ–ßΙϊΓΘ
ΙΊΦϋ¥ ΘΚνβΘΜΥα–‘Ϋΰ≥ω“ΚΘΜάκΉ”ΫΜΜΜ ς÷§ΘΜΕ·ΝΠ―ßΘΜ…œ÷υΝ§–χΈϋΗΫ
(Edited by Wei-ping CHEN)
Foundation item: Projects (21376251, 21406233) supported by the National Natural Science Foundation of China
Corresponding author: Hong-bin XU; E-mail: hbxu@ipe.ac.cn
DOI: 10.1016/S1003-6326(18)64809-5

