
Influence of calcination atmosphere on photocatalytic reactivity of K2La2Ti3O10 for water splitting
YANG Ya-hui(杨亚辉)1, 2, QIU Guan-zhou(邱冠周)2,
CHEN Qi-yuan(陈启元)1, FENG Qi-ming(冯其明)2, YIN Zhou-lan(尹周澜)1
1. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
2. School of Mineral Processing and Bioengineering, Central South University, Changsha 410083, China
Received 28 September 2006; accepted 15 January 2007
Abstract: The layered perovskite type oxide K2La2Ti3O10 powders were prepared under air, Ar and H2 calcination atmospheres by sol-gel method and characterized by X-ray diffractometry, UV-Vis diffuse reflectance and X-ray photoelectron spectroscopy. The influence of the calcination atmosphere on the photocatalytic reactivity of K2La2Ti3O10 for hydrogen production was investigated. The photocatalytic reactivity of K2La2Ti3O10 prepared under air, Ar and H2 atmospheres was compared with that prepared under ultraviolet and visible light radiation using I- as electronic donor. The results show that K2La2Ti3O10 prepared under Ar and H2 atmospheres has higher photocatalytic activity for hydrogen production than that prepared under air atmosphere. The hydrogen production rates under ultraviolet irradiation are 127.5, 81.3 and 57.0 μmol/(L?h) and those under visible light irradiation are 40.2, 30.2 and 16.5 μmol/(L?h) respectively when K2La2Ti3O10 is prepared under Ar, H2 and air atmospheres.
Key words: K2La2Ti3O10; calcination atmosphere; photocatalytic activity; hydrogen
1 Introduction
Because of its high chemical stability and favorable energy band structure, the layered perovskite photocatalyst K2La2Ti3O10 has drawn much attention for its applications in water photocatalytic splitting for H2 and O2 production from potassium hydroxide aqueous solution[1-3]. The photocatalyst K2La2Ti3O10 has one interlayer and this structure is favorable for the improvement of its photocatalytic reactivity for water splitting. Under light irradiation, the light-generated electrons move from the interlayer of K2La2Ti3O10 to external surface and light-generated holes oxidize the water to produce O2 in the interlayer. The photocatalytic reactivity of semiconductors is well known to depend not only on their bulk energy band structure, but also to a great extent, on the point deficiency[4-5]. The metallic vacancy and oxygen vacancy are the common point defects and are affected by the preparation process. K2La2Ti3O10 is usually calcined and/or crystallized in oxidizing atmospheres, such as air and oxygen. The effect of inert atmosphere, such as Ar, and reductive atmosphere, such as H2, has been overlooked. In this study, the effect of calcination atmospheres, including Ar, H2 and air on the photocatalytic reactivity of K2La2Ti3O10 for H2 production from potassium hydroxide aqueous solution under ultraviolet light and visible light irradiation was studied, respectively. The relations between the photocatalytic reactivity and calcination atmospheres were also investigated by X-ray photoelectron spectroscopy(XPS) and ultraviolet-visible (UV-Vis) spectroscopy.
2 Experimental
2.1 Main apparatus and reagents
D/max2250 power X-ray diffraction analyzer was employed to measure the crystal structure of K2La2Ti3O10. Lambda900 UV-Vis spectrometer (Labsphere integrating sphere) was used to measure the UV-Vis diffuse reflectance spectrum of K2La2Ti3O10. X-ray photoelectron spectrometer (Mg Kα X-ray source, 1253.6 eV, 16 mA×12 kV) was used to analyze XPS of K2La2Ti3O10. SP-2305 gas phase chromatography instrument (thermal conductivity detector, Ar carrier, molecular sieve 5 A column) was adopted to determine the evolved hydrogen.
Analytical reagents of titanium tetrabutyl (C16H36O4Ti), KNO3, La(NO3)3?nH2O, KI, KOH, RuCl3? xH2O were used in the experiments.
2.2 Preparation of photocatalyst K2La2Ti3O10
The photocatalyst K2La2Ti3O10 powders were prepared by sol-gel method[6-7]. Solution A was obtained by dissolving the starting materials of 0.02 mol La(NO3)3?nH2O and 0.04 mol KNO3 in 22 mL water, and solution B was obtained by dissolving the starting material of 0.03 mol C16H36O4Ti in 80 mL isopropanol. Solution A was dropwise added to solution B with severe agitation for 5-10 min to get the gelatinous solution. The resulted gelatinous solution was dried and aged under infrared lamp irradiation for 3-4 h when water and isopropanol were separated from the gel. The gel was then crushed and calcined under Ar, H2 and air atmospheres at 950 ℃ for 4 h resptectively, and K2La2- Ti3O10 powders were gained.
In order to get high photocatalytic reactivity, RuO2 was loaded on the photocatalyst K2La2Ti3O10 powders using impregnation method[8-9]. The K2La2Ti3O10 powders were dispersed in RuCl3 aqueous solution by a magnetic stirrer and were heated until water was completely volatilized. The K2La2Ti3O10 powders were ground and oxidized at 500 ℃ for 5 h in air to make RuO2 loaded K2La2Ti3O10.
2.3 Photocatalytic water splitting reaction
Photocatalytic water splitting reaction was carried out in a home-made gas-closed inner irradiation cell made of quartz (see Fig.1). The photocatalyst powder was dispersed in 650 mL potassium iodide aqueous solution (a given amount of KI was added to pure water after boiling for 30 min to remove the dissolved oxygen). KOH was added to control the pH value of solution. The light source was a 250 W high-pressure Hg lamp, of which the radiative wavelength was about 300-400 nm, the intensity of illumination was 15×103 μW/cm2 and the average luminous flux was 75×104 lx. During the photocatalytic reaction, the infrared wave was removed by cooling water in the jacket of inner irradiation quartz cell. The evolved gases were determined with TCD gas chromatograph and its volume was collected and confirmed by drainage method.
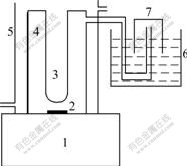
Fig.1 Device for photocatalytic water splitting: 1 Magnetic stirrer; 2 Magnetic bar; 3 250 W high-pressure Hg lamp or xenon lamp; 4 Gas-closed inner irradiation cell; 5 Cooling jacket; 6 Water channel; 7 Gas collector
The 250 W high-pressure Hg lamp was substituted by a 250 W xenon lamp whose average luminous flux was 15×104 lx. The xenon lamp had about the same characteristic spectrum with sunlight. The same experimental procedure described above was carried out to measure photocatalytic reactivity of K2La2Ti3O10 under visible light irradiation.
3 Results and discussion
3.1 XRD patterns of K2La2Ti3O10 powders
Fig.2 shows the XRD patterns of K2La2Ti3O10 powders prepared under Ar, H2 and air atmospheres respectively, which shows that all the powders have perfect crystal structure and remain almost the same under different atmospheres.
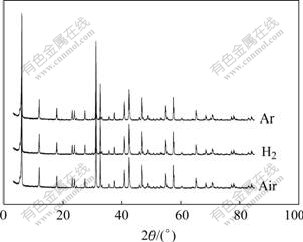
Fig.2 XRD patterns of K2La2Ti3O10 photocatalyst calcined under different atmospheres
3.2 Influence of calcination atmosphere on photo- catalytic reactivity of K2La2Ti3O10
The photocatalytic reactivity of K2La2Ti3O10 prepared in air, Ar and H2 atmospheres was compared under ultraviolet and visible light radiation using I- as electron donor. Fig.3, Fig.4 and Table 1 illustrate that K2La2Ti3O10 prepared under Ar and H2 atmosphere has higher photocatalytic activity for hydrogen production than that prepared under air atmospheres. The photocatalyst calcined under Ar atmosphere exhibits the highest hydrogen production rate. The hydrogen production rates under ultraviolet irradiation are 127.5, 81.3 and 57.0 μmol/(L?h) and those under visible light irradiation are 40.2, 30.2 and 16.5 μmol/(L?h) respectively when K2La2Ti3O10 is prepared under Ar, H2 and air atmospheres.
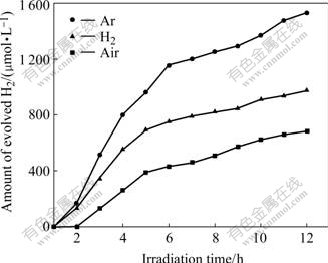
Fig.3 Dependence of photocatalytic reactivity of K2La2Ti3O10 on calcination atmosphere for hydrogen production under ultraviolet radiation
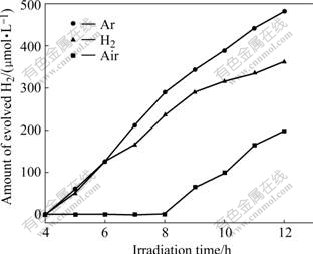
Fig.4 Dependence of photocatalytic reactivity of K2La2Ti3O10 on calcination atmosphere for hydrogen production under visible radiation
Table1Rates of hydrogen evolution of K2La2Ti3O10 calcined under different atmospheres (μmol?L-1?h-1)

Fig.5 shows the diffuse reflectance spectra of K2La2Ti3O10 photocatalysts calcined under different atmospheres. All the photocatalysts show sharp UV absorptions near 300 nm, which are consistent with the theoretical band gap energy. Their visible light absorption behavior at 400-500 nm is different. The K2La2Ti3O10 powders calcined under Ar and H2 show large visible-light adsorption correlated consistently with their activities.
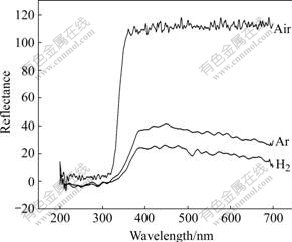
Fig.5 Dependence of diffuse reflectance spectrum of K2La2Ti3O10 photocatalyst on calcination atmosphere
Of the transition metal (composite) oxide, the presence of oxygen defects must be considered in the [TiO6] octahedron. The divalent oxygen ion cannot illustrate the existing state of oxygen in the transition metal (composite) oxide. The oxygen defects will affect the photocatalytic reactivity of K2La2Ti3O10 for water splitting. O(1s) XPS studies (see Fig.6 and Table 2) indicate that there exist mainly two types of surface oxygen, i.e. lattice oxygen(α) whose binding energy is 529.3 eV and adsorption oxygen(β) whose binding energy is 530.9 eV. The binding energies of Ti 2p (457.8 eV for Ti 2p1/2 and 463.6 eV for Ti 2p3/2) for the three samples are typical of Ti4+ (see Fig.7 and Table 3).
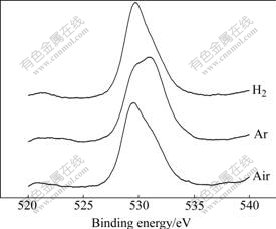
Fig.6 O1s-XPS spectra of K2La2Ti3O10 prepared under different calcination atmospheres
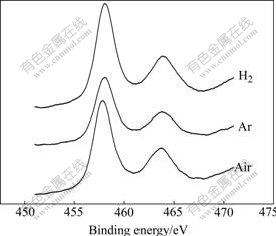
Fig.7 Ti2p-XPS spectra of K2La2Ti3O10 prepared under different calcination atmospheres
Table 2 Molar ratios of lattice and adsorption oxygen of K2La2Ti3O10 under different calcination atmospheres (%)
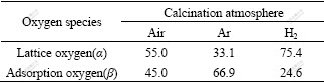
Table 3 Molar ratios of Ti 2p1/2 and Ti 2p3/2 of K2La2Ti3O10 under different calcination atmospheres (%)
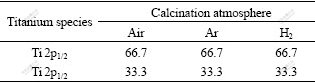
The ratio of adsorption oxygen(β) of K2La2Ti3O10 calcined under Ar is 66.9%, which is higher than that of K2La2Ti3O10 calcined under H2 and air. This indicates that K2La2Ti3O10 calcined under Ar has higher oxygen vacancy ratio and less metal vacancy ratio[10]. The non-stoichiometry oxygen due to thermal treatment in reductive H2 atmosphere leads to lower valence of Ti ions, most likely Ti3+. The typical XPS peak of Ti3+ is not obvious because of its low content. The migration of oxygen in K2La2Ti3O10 semiconductor will produce oxygen vacancy when being calcined under Ar atmosphere. The oxygen vacancy can not only trap the photo-generated electrons but also increase the ratio of adsorption oxygen(β) on the surface of K2La2Ti3O10 semiconductor[11].
When K2La2Ti3O10 powders are calcined under H2 atmosphere, the possible reactions may be described as follows[12]:
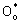 →1/2O2(g)+
→1/2O2(g)+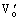 +e- (1)
+e- (1)
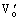 →
→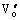 +e- (2)
+e- (2)
1/2O2(g)→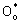 +
+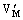 +h+ (3)
+h+ (3)
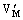 →
→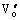 +h+ (4)
+h+ (4)
In the above equations, 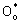 is the lattice oxygen (O2-),
is the lattice oxygen (O2-), 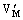 is the metal vacancy and
is the metal vacancy and 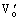 and
and 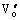 are the oxygen vacancies. Both oxygen vacancies belong to positive centers and are easily excited to the conduction band of K2La2Ti3O10 semiconductor and act as donor. The donor band level(ED) is at the bottom of conduction band(EC). But the metal vacancy belongs to negative center and is easily excited to the valence band of K2La2Ti3O10 semiconductor as acceptor. The acceptor band level(ED) is at the top of valence band(EV) (see Fig.8).
are the oxygen vacancies. Both oxygen vacancies belong to positive centers and are easily excited to the conduction band of K2La2Ti3O10 semiconductor and act as donor. The donor band level(ED) is at the bottom of conduction band(EC). But the metal vacancy belongs to negative center and is easily excited to the valence band of K2La2Ti3O10 semiconductor as acceptor. The acceptor band level(ED) is at the top of valence band(EV) (see Fig.8).
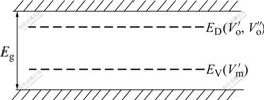
Fig.8 Band structure of K2La2Ti3O10 with oxygen vacancy and metal vacancy
Because of the formation of acceptor band level and donor band level of K2La2Ti3O10 calcined under Ar and H2 atmospheres, the photo-generated electrons can jump step by step and reduce the energy gap compared with K2La2Ti3O10 calcined under air. Though the ratio of adsorption oxygen(β) of K2La2Ti3O10 calcined under H2 is low(24.6%) compared with that of K2La2Ti3O10 calcined Ar and air (see Table 2), the metal vacancy plays the same part in the reduction of energy gap as the oxygen vacancy[13]. This may explain the observation that there is an optimum visible-light absorption for higher hydrogen rate.
4 Conclusions
1) K2La2Ti3O10 has higher photocatalytic activity for hydrogen production when being calcined under Ar and H2 atmospheres than under air atmosphere.
2) The hydrogen production rates under ultraviolet irradiation are 127.5, 81.3 and 57.0 μmol/(L?h) respectively when K2La2Ti3O10 is prepared under Ar, H2 and air atmospheres.
3) The hydrogen production rates under visible light irradiation are 40.2, 30.2 and 16.5 μmol/(L?h) respectively when K2La2Ti3O10 is prepared under Ar, H2 and air atmospheres.
References
[1] TAKATA T, SHINOHARA K, TANAKA A, HARA M, KONDO J N, DOMEN K. A highly active photocatalyst for overall water splitting with a hydrated layered perovskite structure[J]. Journal of Photochemistry and Photobiology A: Chemistry, 1997, 106(1/3): 45-49.
[2] THAMINIMULLA C T K, TAKATA T, HARA M, KONDO J N, DOMEN K. Effect of chromium addition for photocatalytic overall water splitting on Ni-K2La2Ti3O10 [J]. Journal of Catalysis, 2000, 196(2): 362-365.
[3] IKEDA S, HARA M, KONDO J N, DOMEN K, TAKAHASHI H, OKUBO T, KAKIHANA M. Preparation of K2La2Ti3O10 by polymerized complex method and photocatalytic decomposition of water [J]. Chemistry of Materials,1998, 10(1): 72-77.
[4] GAO You-liang, CHEN Qi-yuan, YIN Zhou-lan, HU Hui-ping, LI Jie. Influence of oxygen vacancies of WO3 on photocatalytic activity for O2 evolution [J]. Chinese Journal of Inorganic Chemistry, 2005, 21(10): 1510-1514. (in Chinese)
[5] WU N L, LEE M S, PON Z J, HSU J Z. Effect of calcination atmosphere on TiO2 photocatalysis in hydrogen production from methanol/water solution[J]. Journal of Photochemistry and Photobiology A: Chemistry, 2004, 163(1/2): 277-280.
[6] CHEN Zhen-liang, WANG Zheng-cun, SHEN Cheng-min, JIA Yong-zhong, YANG Jin-xian. Preparation of nanocrystalline strontium titanate by the sol-gel process [J]. Journal of Functional Materials, 1999, 30(6): 633-635. (in Chinese)
[7] LI Kun, CHEN-WANG Li-hua, CAI Zhong-long, LI Jin-hua. Preparation and properties of (Sr0.12Ba0.18)TiO3 solution [J]. Journal of Jiangsu Institute of Petrochemaical Technology, 2000, 12(1): 1-4. (in Chinese)
[8] WANG Chuan-yi, LIU Chun-yan, SHEN Tao. Surface modification of semiconductor photocatalyst [J]. Chemical Journal of Chinese University, 1998, 19(12): 2013-2019. (in Chinese)
[9] AMY L L, LU G Q, JOHN T Y. Photocatalysis on TiO2 surfaces: Principles, mechanisms, and selected results [J]. Chemical Reviews, 1995, 95(3): 735-758.
[10] PENA M A, FIERRO J L G. Chemical structures and performance of perovskite oxides [J]. Chemical Reviews, 2001, 101(7): 1981-2017.
[11] SANG Li-xia, ZHONG Shun-he, FU Xi-xian. Oxygen migration and photocatalystic activity of pervoskite-type oxides [J]. Chemical Journal of Chinese University, 2003, 24(2): 320-323. (in Chinese)
[12] XU Yu-long. Semiconductor basis on oxides and compounds [M]. Xi’an: Xi’an University of Electronic Science and Technology Press, 1991: 49-55. (in Chinese)
[13] FU Xi-xian, YANG Qiu-hua, SANG Li-xia. Studies on photocata lytic activity of perovskite type LaFe1-xCuxO3 [J]. Chemical Journal of Chinese University, 2002, 23(2): 283-286. (in Chinese)
Foundation item: Project(2002AA327140) supported by the National High-Tech Research and Development Program of China
Corresponding author: YANG Ya-hui; Tel: +86-731-8877364; E-mail: yangyh@mail.csu.edu.cn
(Edited by CHEN Wei-ping)