
Microwave direct synthesis and thermoelectric properties of Mg2Si by solid-state reaction
ZHOU Shu-cai1, 2, BAI Chen-guang1
1. College of Material and Science Engineering, Chongqing University, Chongqing 400044, China;
2. School of Metallurgy and Material Engineering,
Chongqing University of Science and Technology, Chongqing 401331, China
Received 20 August 2010; accepted 14 February 2011
Abstract: In order to reduce the oxidation and volatilization caused by Mg element in the traditional methods for synthesizing Mg2Si compounds, Mg2Si thermoelectric materials were prepared by solid state reaction and microwave radiation techniques. Structure and phase composition of the materials were investigated by X-ray diffraction. The electrical conductivity, Seebeck coefficient and thermal conductivity were measured as a function of temperature from 300 to 700 K. It is found that high purity Mg2Si powders can be obtained with excessive content of 8% Mg from the stoichiometric Mg2Si at 853 K and 2.5 kW for 30 min. A maximum dimensionless figure of merit, ZT, of about 0.13 was obtained for Mg2Si at 600 K.
Key words: Mg2Si; thermoelectric materials; solid state reaction; microwave synthesis
1 Introduction
Magnesium silicide (Mg2Si) with a face-centered- cubic type structure has been identified as a promising thermoelectric material for power generation in a temperature range from 500 to 800 K [1-4]. Because of the environmental-friendly advantages, such as the abundance of its constituent elements in the earth crust and the non-toxicity of processing by-products, more and more researchers have paid much attention to those compounds [5-6].
However, the phase purity and microstructure of the product Mg2Si are difficult to control by conventional techniques because of the easy volatilization and oxidation of Mg and the great discrepancy of melting point between Mg and Si. Many synthesis methods have also been applied to Mg2Si-based compounds to improve their thermoelectric properties, such as vacuum melting [4], solid state reaction [7], mechanical alloying [8-10] and spark plasma sintering (SPS) [3, 11]. Mechanical alloying (MA) or ball milling usually takes a long time and the samples may be contaminated and oxidized even in a protective atmosphere during the milling process. Vacuum melting is difficult to control the synthesis and constituents, the adjustment of structure and performance of the materials because of some problems such as the evaporation and oxidation of Mg, the unevenness or segregation caused by different melting points and different density of Mg from Si. However, the issues above have not been solved radically yet.
These problems mentioned above motivate the development of a new synthetic method that overcomes these problems and facilitates product formation. The microwave-assisted synthesis method is presented. This method is a very promising preparation method for many materials because it is fast, clean, energy efficient and does not suffer from the disadvantages of the classical preparation technique. Many materials have been synthesized by the microwave radiation method at a considerably lower temperature and in a shorter time than the conventional methods [12-14]. The rapid synthesis may lead to smaller grain size and consequently better mechanical properties to improve the product uniformity. Until recently, microwave processing has been mostly restricted to ceramics, cemented carbides and ferrites [13]. Applicability of microwave sintering to metals was ignored due to the fact that they reflect microwaves. ROY et al [12] reported that particulate metals can be heated rapidly in microwaves.
This led to the use of microwaves to consolidate a range of particulate metals and alloys [15]. This is very interesting from the applicative point of view.
In this work, Mg2Si thermoelectric materials were prepared by microwave-assisted activation synthesis. It is a rapid process and results in a pure phase material. The results obtained seem to be very interesting and stimulate further research in this direction.
2 Experimental
Figure 1 shows a schematic illustration of the experimental equipment, which was used for the production of activated Mg and Si particles. It mainly consisted of a microwave oven and a heat preservation system.
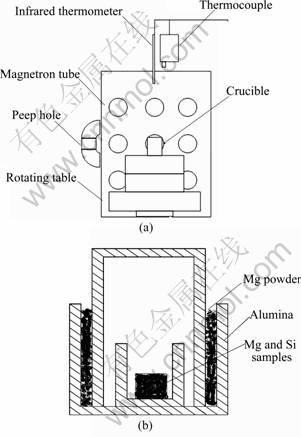
Fig. 1 Schematic diagrams of microwave system setup (a) and crucible (b)
Due to the volatility of Mg during the synthesis of this compound, the excessive content of Mg is indispensable. In this study, samples with nominal formula Mg2.08Si were prepared. According to the stoichiometric proportion of Mg2Si, Mg (>99.5% purity, 74 μm) and Si (>99.5% purity, 44 μm) with molar ratio of 2.08:1 were used as raw materials. The required amount of these materials was well mixed by an ultrasonic homogenizer in ethanol for 45 min. After being completely dried in the cabinet, they were compressed to form pellets with dimensions of d20 mm×10 mm under appropriate pressure. The samples were then placed in an alumina crucible in the center of the microwave oven and the solid-state reaction was carried out under the protection of high-purity Ar (99.9%). The microwave oven used in the present work consists of a 2.45 GHz microwave generator with continuous adjustable power output of 0-15 kW. An infrared pyrometer was used (Raytek, Marathon series) for temperature measurement. Microwave heating used in the present work was carried out at power level of 2.0-3.5 kW and heated to a designed temperature (580-600 °C), then kept at this temperature (with power level of 1.0-1.5 kW) for 20-40 min. The samples were cooled to room temperature for further control experiment.
Phases of the fabricated materials were detected by X-ray diffraction (D/Max-IIIA) with Cu Kα radiation (λ =0.154 06 nm). The electrical conductivity (σ) and Seebeck coefficient (α) were simultaneously measured by the four-probe dc method in helium atmosphere from room temperature to 800 K using a computer-assisted device. The errors of the Seebeck coefficient and the electrical resistivity measurements were estimated to be less than ±5%. The thermal diffusivity (D) and the specific heat capacity (cp) of the samples were measured by a laser flash apparatus (Netzsch LFA 457) and a thermal analyzer (Netzsch DSC 404), respectively. The thermal conductivity k was calculated from the relationship k=ρDcp, where ρ is the density of the material, measured using Archimedes method at room temperature. Relative density of the compacts measured by Archimedes method was about 96% of the theoretical value.
3 Results and discussion
3.1 Effect of microwave power on Mg2Si compounds
In order to find out the complete reaction temperature of Mg and Si to form Mg2Si in microwave chamber, the raw mixed powders are sintered by microwave at temperature from 573 to 903 K. When the input power is kept constant at 2.5 kW and 853 K, Mg2Si single phase is formed, and no obvious Mg, Si, MgO and SiO2 peaks are detected from the XRD patterns, as shown in Fig. 2. Phases of microwave sintered samples at 3.5 kW and 903 K show that some MgO and SiO2 peaks appear, as shown in Fig. 3. It can be attributed to the easier oxidation of Mg at higher temperature. Thus, the optimum temperature of solid-state synthesis by microwave is about 853 K, at which Mg and Si react completely and nearly no obvious oxidation is detected.
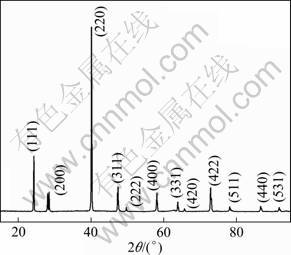
Fig. 2 XRD pattern of Mg2Si sample sintered at microwave power of 2.5 kW

Fig. 3 XRD pattern of Mg2Si sample sintered at microwave power of 3.5 kW
3.2 Effect of isothermal holding time on Mg2Si compounds
Figure 4 shows the XRD patterns of Mg and Si powders on stoichiometric proportion at 853 K in microwave oven for various holding time. The phase analysis of the XRD patterns reveals that Mg2Si is the major phase in the sample, and complete reaction takes place when the holding time is only 30 min. The peaks are very sharp at this situation and no obvious MgO peak appears. However, MgO and SiO2 impurity peaks appear when the holding time is 40 min. When the holding time is only 20 min, there are a small amount of Mg peaks of raw powder. Thus, the optimum holding time is 30 min and the excessive content of Mg is essential to compensate the volatilization of Mg during processing.
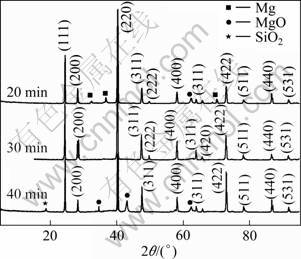
Fig. 4 XRD patterns of Mg2Si samples with different holding time during synthesis processing
3.3 Thermoelectric properties of Mg2Si compound
The temperature dependence of electrical conductivity of the samples are shown in Fig. 5. In all the samples, the electrical conductivity increases monotonically with temperature in a range of 300-700 K, indicating semiconduction behavior [16].

Fig. 5 Variation of electrical conductivity with holding temperature for Mg2Si compounds
Figure 6 shows the variation of Seebeck coefficient with holding temperature of Mg2Si. The Seebeck coefficient for Mg2Si compound increases with temperature, and reaches the lowest value of -440 μV/K at about 550 K, and then decreases at higher temperature mainly due to the occurrence of an increasing number of thermally excited minority carriers. A negative Seebeck coefficient suggests that the obtained materials exhibit n-type conductivity.
The variation of thermal conductivity for Mg2Si compound k with the holding temperature is also depicted in Fig. 7. The overall thermal conductivity k is given by k=ke+kph, where ke is the electronic thermal conductivity and kph is the lattice thermal conductivity. ke is related to electrical conductivity by Wiedemann-Franz law as:
ke=L0σT
where L0 is Lorenz number. For the degenerate conduction, L0 = 2.45×10-8 V2/K2 is used. kph can be obtained by subtracting ke from the total thermal conductivity. For Mg2Si compounds, kph is 75%-96% of k. kph gives essential contribution, indicating phonon- phonon interactions are the primary sources of thermal resistance [17].
The temperature dependence of the dimensionless figure of merit 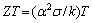 of Mg2Si compound is presented in Fig. 8. With the increase of temperature, the ZT value of Mg2Si compounds increases. A maximum figure of merit ZT = 0.13 is obtained for Mg2Si at about 600 K.
of Mg2Si compound is presented in Fig. 8. With the increase of temperature, the ZT value of Mg2Si compounds increases. A maximum figure of merit ZT = 0.13 is obtained for Mg2Si at about 600 K.
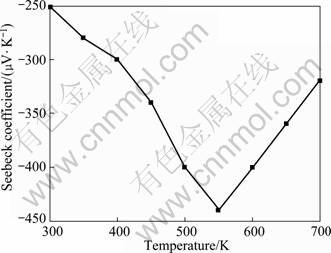
Fig. 6 Variation of Seebeck coefficient with holding temperature for Mg2Si compounds
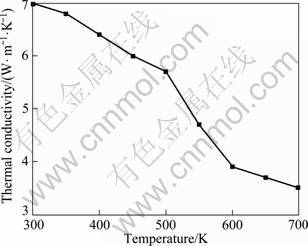
Fig. 7 Variation of thermal conductivity with holding temperature for Mg2Si compound
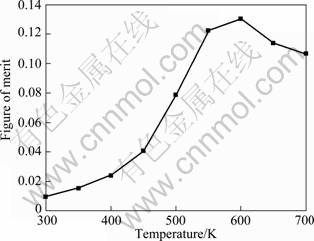
Fig. 8 Temperature dependency of figure of merit, ZT, for Mg2Si compound
4 Conclusions
1) The optimum parameters of microwave heating from Mg and Si raw powders to synthesize Mg2Si are heating temperature of 853 K, holding time of 30 min with an excessive content of 8% Mg from the stoichiometric ratio.
2) With the electrical conductivity, Seebeck coefficient and thermal conductivity are measured from room temperature to 700 K, the dimensionless figure of merit is calculated and discussed. The highest ZT value is 0.13 at 600 K for Mg2Si compound. It is also demonstrated that microwave-assisted synthesis technique is a suitable method for preparing Mg2Si thermoelectric materials.
References
[1] YOSHINAGA M, IIDA T. Bulk crystal growth of Mg2Si by the vertical bridgmam method [J]. Thin Solid Film, 2004, 461(1): 86-89.
[2] MORRIS R G, REDIN R D, DANIELSON G C. Semiconducting properties of Mg2Ge single crystals [J]. Physical Review, 1958, 109(6): 1916-1920.
[3] JUN-ICHI T, HIROYASU K. Thermoelectric properties of Sb-doped Mg2Si semiconductors [J]. Intermetallics, 2007, 15(9): 1202-1207.
[4] ZAITSEV V K, FEDOROV M I, GURIEVA E A, EREMINI S, KONSTANTINOV P P, SAMUNIN A Y, UEDERNIKOV M V. Highly effective Mg2Si1-xSnx thermoelectrics[J]. Physical Review B, 2006, 74(4): 045207-1- 045207-5.
[5] CAILLAT T, BORSHCHEVSKY A, FLEURIAL J P. Properties of single crystalline semiconducting CoSb3 [J]. Journal of Applied Physics, 1996, 80(8): 4442-4449.
[6] BOSE S, ACHARYA H N, BANERJEE H D. Electrocal, thermal, thermoelectric and related properties of magnesium silicide semiconductor prepared from rice husk [J]. Journal of Materials Science, 1993, 28(20): 5461-5468.
[7] JIANG Hong-yi, LONG Hai-shan, ZHANG Lian-meng. Effects of solid-state reaction synthesis processing parameters on thermoelectric properties of Mg2Si [J]. Journal of Wuhan University of Technology: Materials Science, 2004, 19(2): 55-56.
[8] ZHANG Qian, HE Jian, ZHU Tie-jun. High figures of merit and natural nanostructures in Mg2Si0.4Sn0.6 based thermoelectric materials[J]. Applied Physics Letters, 2008, 93(10): 102109.
[9] XIONG Wei, QIN Xiao-ying, WANG Li. Preparation and microstructural characterization of nanocrystalline Mg2Si intermetallic compound bulk [J]. The Chinese Journal of Nonferrous Metals, 2005, 15(3): 380-384. (in Chinese)
[10] NIU Xiao-ping, LI Lu. Formation of magnesium silicide by mechanical alloying [J]. Advanced Performance Materials, 1997, 4(3): 275-283.
[11] HAN Li-qin, YANG Mei-jun, SHEN Qiang, ZHANG Lian-meng. Reaction sintering of magnesium silicide thermoelectric material by the spark plasma sintering technique[J]. Journal of the Chinese Ceramic Society, 2008, 36(8): 337-340. (in Chinese)
[12] ROY R, AGRAWAL D, CHENG J P, GEDEVANISHVILI S. Full sintering of powdered metal bodies in a microwave field [J]. Nature, 1999, 399(17): 668-670.
[13] LEKSE J W, STAGGER T J, AITKEN J A. Microwave metallurgy: Synthesis of intermetallic compounds via microwave irradiation [J]. Chemistry of Materials, 2007, 19(15): 3601-3603.
[14] CLARK D E, SUTTON W H. Microwave processing of materials [J]. Annual Review of Materials Science, 1996, 26(8): 299-331.
[15] PANDA S S, SINGH V, UPADHYAYA A, AGRAWAL D. Sintering response of austenitic (316) and ferritic (434L) stainless steel consolidated in conventional and microwave furnaces [J]. Scripta Mater, 2006, 54(12): 2179-2183.
[16] CLARK C R, WRIGHT C, SURYANARAYANA C, BABURAJ E G, FROES F H. Synthesis of Mg2X (X = Si, Ge, or Sn) intermetallics by mechanical alloying[J]. Materials Letters, 1997, 33(1-2): 71-75.
[17] LABOTZ R J, MASON D R, KANE D F O. The thermal conductivities of Mg2Si and Mg2Ge [J]. Journal of the Electrochemical Society, 1963, 110(2): 127-134.
Mg2Si的微波固相合成及其热电性能
周书才1, 2, 白晨光2
1. 重庆大学 材料科学与工程学院,重庆 400044;
2. 重庆科技学院 冶金与材料工程学院,重庆 401331
摘 要:为了解决Mg2Si 传统制备方法中Mg的氧化、挥发等问题,采用微波低温固相反应法合成Mg2Si热电材料。用XRD分析手段研究合成产物的结构及相组成。在300到700 K的温度范围内,对材料的电导率、Seebeck系数和热导率随温度的变化进行测量。 结果表明,当Mg过量8%、加热功率为2.5 kW时,于853 K保温30 min,可以得到单相Mg2Si热电化合物。在测试温度范围内,Mg2Si具有较高的品质因数ZT值,在600 K温度下达 到0.13。
关键词: Mg2Si;热电材料;固相反应;微波合成
(Edited by FANG Jing-hua)
Foundation item: Project (2009BB4228) supported by the Natural Science Foundation Project of Chongqing Science and Technology Commission, China; Project (CK2010Z09) supported by the Research Foundation of Chongqing University of Science and Technology, China
Corresponding author: ZHOU Shu-cai; Tel: +86-23-65023706; E-mail: zhoushucai71@126.com
DOI: 10.1016/S1003-6326(11)60931-X