


Trans. Nonferrous Met. Soc. China 22(2012) 1176-1181
Extension of analytical model of solid-state phase transformation
JIANG Yi-hui, LIU Feng, SONG Shao-jie
State Key Laboratory of Solidification Processing, Northwestern Polytechnical University, Xi’an 710072, China
Received 23 September 2011; accepted 5 January 2012
Abstract: Departing from an analytical phase transformation model, a new analytical approach to deduce transformed fraction for non-isothermal phase transformation was developed. In the new approach, the effect of the initial transformation temperature and the accurate “temperature integral” approximations are incorporated to obtain an extended analytical model. Numerical approach demonstrated that the extended analytical model prediction for transformed fraction and transformation rate is in good agreement with the exact numerical calculation. The new model can describe more precisely the kinetic behavior than the original analytical model, especially for transformation with relatively high initial transformation temperature. The kinetic parameters obtained from the new model are more accurate and reasonable than those from the original analytical model.
Key words: non-isothermal transformation; analytical model; initial transformation temperature; approximation
1 Introduction
In studies of solid-state phase transformations involving nucleation and growth, the classical Johnson-Mehl-Avrami (JMA) approach [1,2] or modified JMA approach [3,4] always plays a central role. The JMA equation can only be validated under certain condition, in particular for nucleation. Under these conditions, the kinetic parameters should be constant during the course of the transformation. Recently, a more general modular, analytical phase transformation model [5,6], has been proposed which recognizes the three mechanisms, nucleation, growth, and impingement of growing new phase particles, as entities that can be modeled separately. The model leads to equations for the degree of transformation which have the structure of JMA equation but with variable kinetic parameters. The analytical model has been successfully used for description of the crystallization of some amorphous alloys [7,8].
Actually, an assumption, T>>T0 (T and T0 represent the transformation temperature and the initial transformation temperature, respectively) is made in deducing the analytical model [5] for non-isothermal transformation, so, in the temperature range considered, the values of the terms involving T0 can be neglected in comparison with the corresponding terms involving T. Neglecting terms involving T0 can also be found in many commonly used methods of non-isothermal kinetics analysis [9,10]. During the last decade, some scientists [11,12] realized that in a limited number of cases including low temperature reactions and multi-stage reactions [13] (T0 for the consecutive second or third, etc. transformation is relative high), the effect due to T0 terms cannot be neglected in the kinetic analysis. Furthermore, in deducing the analytical model for non-isothermal transformation, the so-called “temperature integral” [12] or “general temperature integral” [14] cannot be solved analytically and has to be approximated. The approximation applied to analytical model is too coarse to give a sufficiently precise description for non-isothermal transformation.
As the reasons mentioned above, the original analytical model cannot be used widely. In this work, the analytical model is extended to a more general case by using a new analytical approach. The precision of the new model is also evaluated by the numerical calculation. Because it incorporates the effect of T0 and the accurate “temperature integral” approximation, the new model can be applied to describing precisely the kinetic behavior of the transformation with high initial transformation temperature.
2 Theoretical background
2.1 Modes of nucleation
The term “site saturation” is used here for the case of initial nucleation site saturation where all nuclei are present at T0 already. This implies for the nucleation rate at time τ [8]:
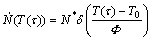 (1)
(1)
with δ denotes Dirac functions; N* is the number of nuclei per unit volume; F (=dT/dτ=dT/dt) is the constant heating rate with T(τ)=T0+Fτ.
The “continuous nucleation” rate per unit volume (i.e. the rate of formation of particles or nuclei of supercritical size) is at a large undercooling only determined by the rate of the jumping of atoms through the interface between the nucleus of critical size and the parent phase, which can be given by an Arrhenius term:
 (2)
(2)
where N0 is a temperature-independent pre-exponential factor, and QN is the temperature-independent activation energy for nucleation.
Here, a short introduction is given for (more general) mixed nucleation in non-isothermal transformation. “Mixed nucleation” represents a combination of site saturation and continuous nucleation modes: the nucleation rate is equal to some weighed sum of the nucleation rates according to continuous nucleation and site saturation [8]. Hence,
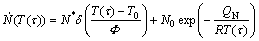 (3)
(3)
where N* and N0 include the relative contributions of the two modes of nucleation.
2.2 Modes of growth
The “diffusion-controlled” and the “interface- controlled” growth modes can be given in a compact form. At time t (i.e. temperature T(t)=T0+Φ(t)) the volume Y of a particle nucleated at time τ (i.e. temperature T(τ)) is given by [8]:
 (4)
(4)
where g is a particle-geometry factor; ν is the growth velocity; m is the growth mode parameter (m=1 for interface-controlled growth; m=2 for volume diffusion- controlled growth); d is the dimensionality of the growth (d=1, 2, 3) [8].
For a large undercooling, ν=ν0exp{-QG/[RT(t)]} with QG as the temperature-independent activation energy for growth. For interface-controlled growth, n0 is a temperature-independent pre-exponential factor and QG represents the energy barrier at the interface. For volume diffusion-controlled growth, ν0 equals the pre- exponential factor for diffusion D0 and QG represents the activation energy for diffusion QD [8].
2.3 Impingement
The number of supercritical nuclei formed in a unit volume at time τ during a time lapse, dT(τ)/Φ, is calculated by Eqs. (1)-(3) for different modes. The volume of each of these nuclei grows from τ (i.e. T(τ)) to t (i.e. T(t)) according to Eq. (4) where it is supposed that every particle grows into an infinitely large parent phase, in the absence of other growing nuclei. In this hypothetical case, the volume of all particles at temperature T, called the extended transformed volume, Ve, can be described as:
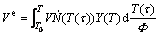 (5)
(5)
with V as the sample volume, which is supposed to be constant throughout the transformation. Accordingly, the extended transformed fraction, xe, is defined as xe=Ve/V.
In reality, the particles do not grow individually into an infinitely large parent phase. A relationship between the real transformed fraction, f, and the extended transformed fraction, xe, is required. Several equations for different types of hard impingement were summarized in Ref. [8]. In this study only the mode of random dispersed nuclei is taken into consideration [2]:
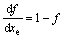 and
and 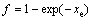 (6)
(6)
3 New analytical approach
Upon deriving the original model, the terms involving T0 are assumed as negligible, and the “temperature integral” and the “general temperature integral” are doing a coarse approximation. In this section, an analytical approach incorporating the effect of T0, the more accurate and recently proposed “temperature integral” approximation will be shown.
Assuming mixed nucleation, interface-controlled growth, the extended volume can be given as (see Eqs. (3), (4) and (5)):
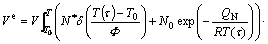
 (7)
(7)
To avoid unnecessary, nonessential complications in the following formulas, set d/m=1 (the analytical process for other value of d/m is similar). Hence, Eq. (7) can be rewritten as:

 (8)
(8)
To derive the analytical expression of Eq. (8), the so-called “temperature integral” and/or “general temperature integral” must be approximated. Recently, it has been found that the approximations in the analytical model could be replaced by the high accuracy approximations [15]. The high accuracy approximations, which guarantee the accuracy of the analytical description, for the “general temperature integral”, can be expressed as [14]:
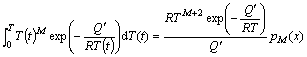 (9)
(9)
where x=Q′/(RT) (e.g. xG=QG/(RT), xN,iG=(QN+iQG)/(RT), i=0, 1, 2, 3…); Q′ is the constant activation energy; M is a constant; pM(x) represents the function of M and x. For M=0, the left-hand side of Eq. (9) becomes the “temperature integral” [12]. It can be expressed as:
 (10)
(10)
where q(x) represents the function of x. The formulas for pM(x) and q(x) are different according to the different approximate methods [15]. Applying the approximation for “temperature integral” and “general temperature integral”, Eq. (8) becomes,





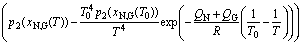 (11)
(11)
To derive the original model, the terms involving T0 in Eq. (11) are neglected. Actually, this is not the indispensable process to obtain the analytical solution. In this paper, these terms are reserved. Analogous to the treatment in the appendix A of Ref. [5], the explicit analytical expression for xe for non-isothermal transformation is given as:
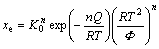 (12)
(12)
with the temperature-dependent expressions for n, Q and K0 gathered in Table 1. In combination with different modes of impingement, the real transformed fraction can be given. Such as the impingement during randomly dispersed nuclei, it can be given as:
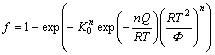 (13)
(13)
Clearly, the current model possesses an analogous form to the original model. The only different between these two models is the expression for Cs and Cc (see Ref. [5] and Table 1). As shown in Eq. (13) and Table 1, the original model results provided that the T0 terms are neglected, and coarse approximations are used (i.e. q(x)=1 and pM(x)=1) in the current model.
4 Evaluation of extended analytical model
The precision of different types of approximations without considering the effect of T0 has been evaluated in Ref. [15]. In this section, the most accuracy combination of “temperature integral” and “general temperature integral” approximation (i.e. the combination of CAI et al [14] as pM(x) and ?RF?O as q(x) [12]) will be used to illustrate the effect of T0.
The transformed fraction and transformation rate can be numerically calculated exactly, as a function of temperature, on the basis of Eq. (6) by numerical integration of Eq. (5) for Ve. These exact results can then be compared with corresponding results from the analytical approaches. Thereby, the accuracy of different models is tested. Using model parameters given in Table 2, in cases of the mixed nucleation, interface-controlled growth and impingement due to random nuclei dispersion, the transformed fraction can be predicted at different values of T0 by the original analytical model (i.e. Ref. [5]) and the current model (i.e., Eq. (13) with Table 1) (see Figs. 1(a)-(c)). Accordingly, the transformation rate can be obtained for different models (see Figs. 1(e)-(f)). Using the same values for the model parameters, the numerical calculations are performed.
Table 1 Expressions for growth exponent n, overall effective activation energy Q, and pre-exponential factor of rate constant K0

Table 2 Values of parameters used for numerical approach for non-isothermal transformations

As shown in Fig.1, the original analytical model prediction deviates largely from exact values, and it becomes worse and worse with increasing T0. However, the current model prediction is always in good agreement with the numerical calculation even if for the transformation with high T0. It can also be seen from Fig.1 that the model predications without considering T0 (i.e. the original analytical model) become better and better as Φ increases for a certain T0. It can be explained that, with increasing Φ, the temperature range for overall transformation increases, accordingly, the strengthen of T0 effect decreases.
Evolutions of n and Q with f are shown in Fig. 2. For clarity, only the values for n and Q of the transformation with T0=1000 K and Φ=5 K/min (see Fig. 1(c)) are shown. It can be seen that the kinetic parameters for different models vary considerably. The deviation of transformed fraction and transformation rate for different models from the exact values must be derived from the inaccurate prediction of the kinetic parameters. This implies that the kinetic parameters due to the current analytical model should be more precise.
5 Conclusions
The analytical process incorporating the effect of the initial transformation temperature and the accurate “temperature integral” approximations is presented. The newly derived analytical model possesses an analogous form to the original analytical model. If the T0 terms are neglected and the coarse approximations for “temperature integral” are used in the newly derived analytical model, it leads to the original analytical model. Numerical approach has demonstrated that, once T0, as compared with the temperature range considered, is so high that cannot be neglected, the newly derived model prediction for transformed fraction and transformation rate is in better agreement with the exact numerical calculation than the original model prediction. And the kinetic parameters due to the newly derived analytical model are more precise.
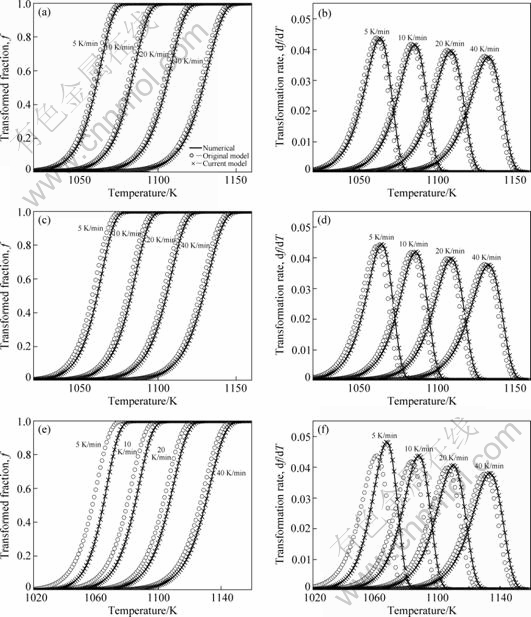
Fig. 1 Comparison of transformed fraction and transformation rate between model predictions, at T0=900 K (a, b), 950 K (c, d), and 1000 K(e, f), in cases of mixed nucleation, interface-controlled growth and impingement due to random nuclei dispersion

Fig. 2 Evolution of growth exponent n (a) and overall effective activation energy Q (b), with transformed fraction calculated by different models at T0=1000 K and Ф=5 K/min
References
[1] CHRISTIAN J W. The theory of transformation in metals and alloys, Part1: Equilibrium and general kinetics theory [M]. 2nd ed. Oxford: Pergamon Press, 1975: 1-22.
[2] AVRAMI M. Kinetics of phase change. II Transformation―time relations for random distribution of nuclei [J]. J Chem Phys, 1940, 8: 212-224.
[3] V?ZQUEZ J, WAGNER C, VILLARES P, JIM?NEZ-GARAY R. A theoretical method for determining the crystallized fraction and kinetic parameters by DSC, using non-isothermal techniques [J]. Acta Mater, 1996, 44: 4807-4813.
[4] FARJAS J, ROURA P. Modification of the Kolmogorov-Johnson- Mehl-Avrami rate equation for non-isothermal experiments and its analytical solution [J]. Acta Mater, 2006, 54: 5573-5579.
[5] LIU F, SOMMER F, MITTEMEIJER E J. An analytical model for isothermal and isochronal transformation kinetics [J]. J Mater Sci, 2004, 39: 1621-1634.
[6] LIU F, SOMMER F, MITTEMEIJER E J. Analysis of the kinetics of phase transformations, roles of nucleation index and temperature dependent site saturation, and recipes for the extraction of kinetic parameters [J]. J Mater Sci, 2007, 42: 573-587.
[7] KEMPEN A T W, SOMMER F, MITTEMEIJER E J. Determination and interpretation of isothermal and non-isothermal transformation kinetics: The effective activation energies in terms of nucleation and growth [J]. J Mater Sci, 2002, 37: 1321-1332.
[8] LIU F, SOMMER F, BOS C, MITTEMEIJER E J. Analysis of solid state phase transformation kinetics: Models and recipes [J]. Int Mater Rev, 2007, 52: 193-212.
[9] STARINK M J. The determination of activation energy from linear heating rate experiments: A comparison of the accuracy of isoconversion methods [J]. Thermochim Acta, 2003, 404: 163-176.
[10] VYAZOVKIN S, GORIYACHKO V. Potentialities of software for kinetic processing of thermoanalytical data by the isoconversion method [J]. Thermochim Acta, 1992, 194: 221-230.
[11] STARINK M J. Activation energy determination for linear heating experiments: Deviations due to neglecting the low temperature end of the temperature integral [J]. J Mater Sci, 2007, 42: 483-489.
[12] ?RF?O J J M. Review and evaluation of the approximations to the temperature integral [J]. AIChE J, 2007, 53: 2905-2915.
[13] WANG J, KOU H C, LI J S, GU X F, ZHONG H, CHANG H, ZHOU L. An integral fitting method for analyzing the isochronal transformation kinetics: Application to the crystallization of a Ti-based amorphous alloy [J]. J Phys Chem Solids, 2009, 70: 1448-1453.
[14] CAI J M, LIU R H, WANG Y Y. Kinetic analysis of solid-state reactions: A new integral method for nonisothermal kinetics with the dependence of the preexponential factor on the temperature (A=A0Tn) [J]. Solid State Sci, 2007, 9: 421-428.
[15] JIANG Y H, LIU F, SONG S J. Improved analytical description for non-isothermal solid-state transformation [J]. Thermochim. Acta, 2011, 515: 51-57.
固态相变解析模型的扩展
姜伊辉,刘 峰,宋韶杰
西北工业大学 凝固技术国家重点实验室,西安 710072
摘 要:基于固态相变解析模型,提出推导非等温固态相变的转变分数的新解析方法。该方法将转变起始温度和精确的温度积分引入到解析模型,从而得到扩展的解析模型。计算表明,扩展的解析模型能够准确地预测计算得到的转变分数与转变速率。扩展的解析模型与原有解析模型相比,它能够更加准确地描述相变过程的动力学行为。在转变初始温度较高的情况下,扩展的解析模型具有优越性。同时,从扩展的解析模型得到的动力学参数比原有解析模型的更合理、准确。
关键词:非等温转变;解析模型;转变初始温度;近似方法
(Edited by YANG Hua)
Foundation item: Projects (09-QZ-2008, 24-TZ-2009) supported by the Free Research Fund of State Key Laboratory of Solidification Processing, China; Project (B08040) supported by the Program of Introducing Talents of Discipline to Universities, China; Projects (51071127, 51134011) supported by the National Natural Science Foundation of China; Project (JC200801) supported by the Fundamental Research Fund of Northwestern Polytechnical University, China; Project (51125002) supported by the National Science Foundation for Distinguished Young Scholars, China; Project (2011CB610403) supported by the National Basic Research Program of China
Corresponding author: LIU Feng; Tel: +86-29-88460374; Fax: +86-29-88491000; E-mail: liufeng@nwpu.edu.cn
DOI: 10.1016/S1003-6326(11)61302-2