Porous layered La0.6Sr0.4Co0.2Fe0.8O3 perovskite with enhanced catalytic activities for oxygen reduction
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2021���5��
�������ߣ������� ��λ�� ��־�� ������ ������ л���� лС�� ������ ������
����ҳ�룺1305 - 1315
Key words��oxygen reduction reaction; electrocatalyst; perovskite; layered structure
Abstract: Low-cost catalysts with high activity are in immediate demand for energy storage and conversion devices. In this study, polyvinyl pyrrolidone was used as a complexing agent to synthesize La0.6Sr0.4Co0.2Fe0.8O3 (LSCF) perovskite oxide. The obtained porous layered LSCF has a large specific surface area of 23.74 m2/g, four times higher than that prepared by the traditional sol-gel method (5.08 m2/g). The oxygen reduction reaction activity of the oxide in 0.1 mol/L KOH solution was studied using a rotating ring-disk electrode. In the tests, the initial potential of 0.88 V (vs. reversible hydrogen electrode) and the limiting diffusion current density of 5.02 mA/cm2 were obtained at 1600 r/min. Therefore, higher catalytic activity and stability were demonstrated, compared with the preparation of LSCF perovskite oxide by the traditional method.
Cite this article as: XU Wei-lai, XU Zhi-feng, CHEN Tian-yu, ZHONG Xiao-cong, XIE Yong-min, XIE Xiao-yun, CHEN Zhe-qin, LIU Jia-ming, WANG Rui-xiang. Porous layered La0.6Sr0.4Co0.2Fe0.8O3 perovskite with enhanced catalytic activities for oxygen reduction [J]. Journal of Central South University, 2021, 28(5): 1305-1315. DOI: https://doi.org/10.1007/s11771-021-4698-6.

J. Cent. South Univ. (2021) 28: 1305-1315
DOI: https://doi.org/10.1007/s11771-021-4698-6
XU Wei-lai(��λ��)1, XU Zhi-feng(��־��)2, CHEN Tian-yu(������)1,
ZHONG Xiao-cong(������)1, XIE Yong-min(л����)1, XIE Xiao-yun(лС��)3,
CHEN Zhe-qin(������)1, LIU Jia-ming(������)1, WANG Rui-xiang(������)1
1. Faculty of Materials Metallurgy and Chemistry, Jiangxi University of Science and Technology,Ganzhou 341000, China;
2. Insitute of Green Metallurgy and Process Intensification, Jiangxi University of Science and Technology,Ganzhou 341000, China;
3. School of Applied Science, Jiangxi University of Science and Technology, Ganzhou 341000, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2021
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2021
Abstract: Low-cost catalysts with high activity are in immediate demand for energy storage and conversion devices. In this study, polyvinyl pyrrolidone was used as a complexing agent to synthesize La0.6Sr0.4Co0.2Fe0.8O3 (LSCF) perovskite oxide. The obtained porous layered LSCF has a large specific surface area of 23.74 m2/g, four times higher than that prepared by the traditional sol-gel method (5.08 m2/g). The oxygen reduction reaction activity of the oxide in 0.1 mol/L KOH solution was studied using a rotating ring-disk electrode. In the tests, the initial potential of 0.88 V (vs. reversible hydrogen electrode) and the limiting diffusion current density of 5.02 mA/cm2 were obtained at 1600 r/min. Therefore, higher catalytic activity and stability were demonstrated, compared with the preparation of LSCF perovskite oxide by the traditional method.
Key words: oxygen reduction reaction; electrocatalyst; perovskite; layered structure
Cite this article as: XU Wei-lai, XU Zhi-feng, CHEN Tian-yu, ZHONG Xiao-cong, XIE Yong-min, XIE Xiao-yun, CHEN Zhe-qin, LIU Jia-ming, WANG Rui-xiang. Porous layered La0.6Sr0.4Co0.2Fe0.8O3 perovskite with enhanced catalytic activities for oxygen reduction [J]. Journal of Central South University, 2021, 28(5): 1305-1315. DOI: https://doi.org/10.1007/s11771-021-4698-6.
1 Introduction
Recently, metal-air batteries (e.g., Li-air battery) have been characterized by their high energy density, low cost, and environmental favorability, and are expected to be applied to emerging room-temperature electrification and energy storage [1, 2]. Noble metals and their alloys, such as Pt and Pt alloys, have been extensively studied as efficient electrocatalysts for oxygen reduction reaction (ORR) [3, 4]. However, their relatively high cost hinders their large-scale application [5]. The synthesis of non-noble metal electrocatalysts with high catalytic activity is a compelling method for improving the commercial applicability of metal-air batteries [6]. As a type of non-noble metal catalyst, perovskite oxides have the characteristics of low cost, high catalytic activity, and have been extensively researched [7, 8].
La0.6Sr0.4Co0.2Fe0.8O3 (LSCF) perovskite is the most representative mixed ionic and electronic conducting electrode material, and has been demonstrated to exhibit excellent catalytic activity for oxygen reduction [9, 10]. The commonly used preparation methods of perovskite-type oxides are the solid phase method, combustion method, and sol-gel method [11]. Among these, the sol-gel method has many unique characteristics, such as simplicity, short preparation, and simple control of the reaction process. In addition, the prepared catalyst component has good uniformity and fine grain [12]. However, the traditional sol-gel method commonly uses ethylenediamine tetraacetic acid (EDTA) and citric acid (CA) as complexing agents [13, 14]. After the last step of high-temperature calcination, the catalyst particles easily agglomerate, and the prepared catalyst has a poor specific surface area. Because the ORR is an interface reaction, a small surface area limits the distribution of active sites on catalyst surface; thus, it shows poor apparent oxygen reduction activity [15]. Many strategies have been used to increase the specific surface area, including film deposition, porous structure construction, and nanoparticle growth. These strategies have been proven to work, but they are complicated and costly [16]. Therefore, the development of a simple method for synthesizing a perovskite catalyst with high specific surface area maintains an area of significant interest.
Polyvinyl pyrrolidone (PVP) has the general properties of a water-soluble polymer including protective colloids, film-forming capabilities, and coacervation [17, 18]. When PVP dissolves in water, an entire molecular framework forms with many active groups, whereby the collective interaction occurs simultaneously at each PVP molecular frame unit. This is the advantage of PVP, by which many types of metal nanoparticles can be stabilized [19]. Meanwhile, PVP is also a type of complexing agent and can be decomposed and removed at high temperatures [20, 21]. In this study, we attempted to use a PVP system instead of an EDTA-CA system in the traditional sol-gel method. Porous layered La0.6Sr0.4Co0.2Fe0.8O3 (layered-LSCF) perovskite catalyst with high specific surface area was first synthesized by sol-gel method with PVP as the complexing agent. The catalytic activity of porous layered LSCF for the ORR in 0.1 mol/L KOH solution was characterized using rotating ring-disk electrode (RRDE) testing.
2 Experimental
2.1 Synthesis of La0.6Sr0.4Co0.2Fe0.8O3
Layered LSCF was prepared through a sol-gel method with PVP as the complexing agent. Stoichiometric amounts of analytical grade La(NO3)3��6H2O (99.0%, Aladdin Industrial Corporation), Sr(NO3)2 (99.5%, Aladdin Industrial Corporation), Co(NO3)2��6H2O (99.0%, Aladdin Industrial Corporation), and Fe(NO3)3��9H2O (99.0%, Aladdin Industrial Corporation) were dissolved in 250 mL of DI water with constant stirring; the total concentration of metal ions was 0.08 mol/L. Then, PVP K30 (molecular weight 58000, purity 99.0%, Aladdin Industrial Corporation) was added to the solution, and the mass ratio of metal ions to PVP was controlled to 1:3. A transparent gel formed after the solution was agitated on a hot-plate at 80 ��C and dried in an oven at 250 ��C for 6 h to form an LSCF precursor powder. The as-synthesized precursor was calcined in a muffle furnace at 600 ��C for 4 h at a heating rate of 2 ��C/min. The obtained LSCF was represented as layered-LSCF. At the same time, the control group synthesized by sol-gel method employing the combination of EDTA (99.5%, Aladdin Industrial Corporation) and CA (99.0%, Aladdin Industrial Corporation) as complexing agents was set up for comparison. A stoichiometric ratio of metal nitrates according to LSCF was dissolved in DI water followed by continuous stirring. When completely dissolved, EDTA and CA were added to the mixed solution with the molar ratio of metal ions to EDTA to CA of 1:1:1.5. Then, NH3��H2O was slowly added to adjust the pH to approximately 7. The subsequent step was as well as that in the preparation of layered LSCF. The obtained LSCF was represented as bulk-LSCF.
2.2 Characterization
The crystal structures of the LSCF perovskite oxides were examined using an X-ray diffraction meter (XRD, D8-Advance, Bruker; Cu K�� radiation; operated at 40 kV, 40 mA; ��=0.15406 nm), with a 2�� angle from 20��to 80��and a step of 0.02��. The morphology and microstructures of the layered-LSCF and bulk-LSCF were observed using scanning electron microscope (SEM, MLA-650F, FEI) and transmission electron microscope (TEM, TECNAI-G20, FEI). Furthermore, the Brunauer�CEmmett�CTeller (BET) specific surface area and pore structure were characterized by a gas adsorption analyzer (TriStar II 3020, Micromeritics Instrument Corporation), with N2 gas as the adsorbent. The composition and chemical states of the catalysts were carried out on an X-ray photoelectron spectroscope (XPS, 250XI, Thermo Scientific).
2.3 Electrochemical measurement
At first, electrodes were prepared by a controlled drop-casting technique, involving an RRDE made of glassy carbon (GC, 0.196 cm2, Pine Research Instrumentation). The GC electrode was polished to a mirror finish by 0.05 ��m Al2O3 and cleaned with DI water and dried before being coated with active materials. The catalyst ink was prepared by ultrasonically dispersing 10 mg of LSCF and 2 mg of conductive carbon (Super P) in 1.19 mL ethanol and 0.01 mL Nafion solution (5 wt%, Sigma-Aldrich) for 1 h to form a homogeneous ink. Then, 10 ��L as-prepared catalyst ink was dropped on the surface of GC to form a working electrode after spin coating (300 r/min) and drying, yielding an approximate electrocatalyst loading of 0.4247 mg/cm2.
Electrochemical measurements for the ORR were carried on a standard three-electrode-cell (Pine Research Instrumentation) at room temperature in an RRDE configuration with a CHI 760E Bi-potentiostat. This Bi-potentiostat consisted of an as-prepared working electrode, a counter electrode (Pt wire), and a reference electrode (Ag/AgCl, 3.0 mol/L NaCl). The electrolyte was 0.1 mol/L potassium hydroxide (KOH) solution, which was saturated with O2 for at least 30 min prior to each test, and bubbling O2 was maintained throughout the test. Linear sweep voltammetry (LSV) curves were swept from 0.1 to -0.8 V (vs. Ag/AgCl) at a scan rate of 5 mV/s at various rotating speeds (400, 625, 900, 1225, 1600 and 2000 r/min) in 0.1 mol/L O2-saturated KOH solution. All potential values reported in this work were referenced to the reversible hydrogen electrode (RHE):
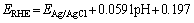
Meanwhile, potential values are iR-corrected to compensate for the influence of the solution resistance [22]. The formula used for this compensation is:
EiR-corrected=E-IR
where I is the current and R is the uncompensated ohmic electrolyte resistance (about 43 ��), measured by electrochemical impedance spectra (EIS) in O2-saturated 0.1 mol/L KOH. EIS was recorded at 0.702 V (vs. Ag/AgCl), with frequencies ranging from 100 kHz to 0.1 Hz under an AC voltage of 5 mV.
3 Results and discussion
To determine the crystalline structure of the synthesized LSCF perovskite oxides, the XRD pattern (Figure 1) was analyzed. Obvious characteristic peaks were identified, indicating that well-crystallized perovskite oxides were synthesized at 600��C. As can be seen, the diffraction patterns of the layered-LSCF and bulk-LSCF did not differ much. All the characteristic peaks of (110), (020), (202), (220), (222), (132), (224), and (332) appeared at 2��=22.83��, 32.37��, 40.13��, 46.63��, 52.43��, 57.67��, 67.76��and 77.39��, respectively, which is consistent with the standard peaks of La0.7Sr0.3Co0.3Fe0.7O3 (JCPDS:89-1268). These peaks were found to be characteristic of a single-phase crystalline perovskite oxide with an orthorhombic crystal and a space group of Pbnm, which is consistent with those in a previous report [23]. Moreover, the obtained lattice parameters are a=5.503  , b=5.512
, b=5.512 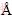 , and c=7.788
, and c=7.788 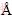 for the layered LSCF, and a=5.512
for the layered LSCF, and a=5.512 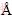 , b= 5.508
, b= 5.508 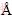 , and c=7.789
, and c=7.789 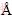 for the bulk LSCF. The detected XRD patterns demonstrate that well-crystallized layered LSCF and bulk-LSCF were successfully synthesized.
for the bulk LSCF. The detected XRD patterns demonstrate that well-crystallized layered LSCF and bulk-LSCF were successfully synthesized.
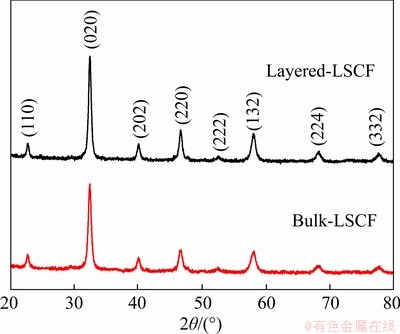
Figure 1 X-ray diffraction patterns of layered LSCF and bulk-LSCF
Figures 2(a) and (b) show the morphologies of the layered LSCF and bulk-LSCF, respectively. It can be observed from Figure 2(a) that the layered LSCF has a clear layered structure with a particle size of approximately 6 ��m. With further observation of the edge of the layered LSCF, some ordered pores can be seen inside. Layered LSCF does not easily agglomerate and is stacked in an orderly manner, which is conducive to the uniform distribution of active sites on the surface. For comparison, accounting for the agglomeration during the calcination process, the bulk-LSCF (Figure 2(b)) shows an irregular accumulation, that is not conducive to the distribution of active sites, which is consistent with the previously reported morphology [24, 25]. The TEM images are also shown in Figures 2(c) and (d) to further characterize the morphologies and microstructures of the layered LSCF and bulk-LSCF. From Figure 2(c), it can be clearly observed that layered LSCF has a decentralized network structure and a predictable rich porosity. Compared with the microstructure of bulk-LSCF shown in Figure 2(d), it can be intuitively observed that the small particles of bulk-LSCF tend to be agglomerated, which is similar to what is revealed by the SEM images. According to the high-resolution TEM (HR-TEM) images (Figures 2(e) and (f)), a lattice fringe with a plane distance of 0.276 nm can be seen in both the layered LSCF and bulk-LSCF, which is consistent with the (020) crystal plane orientation of the corresponding XRD results. In addition, the HR-TEM images show clear lattice planes, demonstrating that the synthesized LSCF perovskite oxides possessed high crystallinity.
To quantitatively determine the BET specific surface area of the synthesized LSCF and further analyze its porous structure, the N2 adsorption/ desorption isotherm and pore size distribution of the prepared catalyst were measured, as shown in Figures 3(a) and (b). The BET specific surface areas of the layered LSCF and bulk-LSCF are 23.74 and 5.08 m2/g, respectively. Besides, the pore volume calculated by the Barrett-Joyner-Halenda (BJH) method is 0.046 and 0.034 cm3/g for layered LSCF and bulk-LSCF, respectively. Owing to the layered porous structure of layered LSCF with an exposed surface and interconnected space, which serves to increase the specific surface area, the obtained specific surface area of layered LSCF is significantly higher than that of bulk-LSCF. According to the IUPAC isotherm classification, shown in Figures 3(a) and (b), all prepared materials are type IV isotherms, which is mainly related to the existence of mesopores in the materials [26]. Figure 3(a) shows that the isotherm hysteresis loops of the layered-LSCF are H3 type, and in the range of relative pressure (P/P0) from 0.5 to 0.8, the adsorption quantity of LSCF increases obviously. In addition, the pore size is mainly distributed between 2 and 20 nm and concentrated at about 8 nm, which suggests the formation of a great quantity of mesopores in the layered LSCF, consistent with the layered porous structure of the sample. Figure 3(b) shows that bulk-LSCF prepared by the traditional method has no clear hysteresis loop, indicating that it does not have a uniform porous structure. The above results demonstrate that layered LSCF has a porous structure with a large specific surface area. A layered porous structure not only helps to provide more defects, but also effectively buffers the volume change of the electrode and is less prone to the destruction of the structure of the catalyst material [27].

Figure 2 SEM images of layered LSCF (a) and bulk-LSCF (b); TEM images of layered LSCF (c) and bulk LSCF (d); High-resolution TEM images of layered LSCF (e) and bulk-LSCF (f)
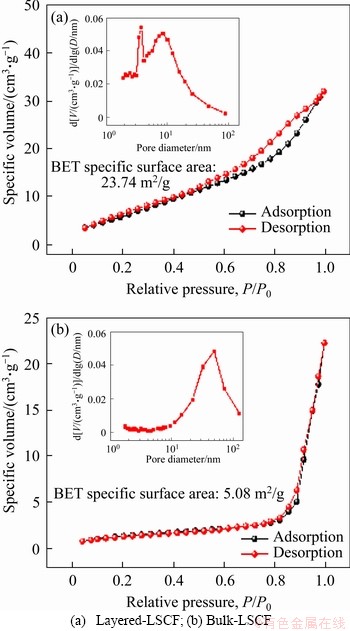
Figure 3 N2 adsorption-desorption isotherms and pore size distribution curves (insets):
The composition and chemical states of the layered LSCF and bulk-LSCF were analyzed by XPS. By the test, the existence of La, Sr, Co, Fe, O and adventitious C in both layered LSCF and bulk-LSCF were confirmed by the wide-scan survey spectra (Figure 4(a)). A previous study showed that replacing part of the La3+ with Sr2+ would lead to a charge imbalance in the structure. In order to neutralize the charge, the oxidation states of Fe or Co would change [28]. The high-resolution XPS results of Co 2p and Fe 2p for the layered LSCF and bulk-LSCF are shown in Figures 4(b) and (c). For Co 2p in Figure 4(b), the two main peaks are situated at about 780.2 eV (Co3+ 2p3/2) and about 794.7 eV (Co3+ 2p1/2); meanwhile, the satellite peaks of Co2+ are at about 785.3 eV (Co2+ 2p3/2) and about 795.7 eV (Co2+ 2p1/2), indicating that Co exhibited oxidation states of +2 and +3 [29]. Similarly, in Figure 4(c), the peaks of Fe 2p at about 709.9 eV (Fe3+ 2p3/2), about 712.0 eV (Fe3+ 2p1/2), about 723.3 eV (Fe2+ 2p3/2), and about 725.2 eV (Fe2+ 2p1/2) are associated with the +2 and +3 oxidation states of Fe [30]. The area fraction obtained by the spectra for the different oxidation states of Fe and Co are listed in Tables 1 and 2. The above analysis shows that Fe and Co tend to the multiple oxidation states in both the layered LSCF and bulk-LSCF. In addition, Figure 4(d) shows the spectra of the O1s peaks located at about 528.7, 529.4 and 531.6 eV, which could be distinguished as the lattice oxygen (Olatt) and adsorption oxygen (Oads). According to Table 3, the proportion of Oads/Olatt of layered LSCF (3.21) is higher than that of bulk-LSCF (2.39). We ascribed the high ratio of Oads/Olatt in layered LSCF to its high surface oxygen adsorption capacity, which is beneficial for improving the efficiency in the ORR [31].
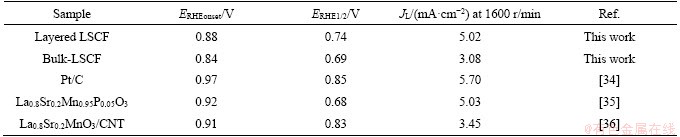
To determine the ORR catalytic activity of the Layered LSCF and bulk-LSCF, linear sweep voltammetry (LSV) measurements were performed at speeds of 400, 625, 900, 1225, 1600 and 2000 r/min in 0.1 mol/L O2-saturated KOH solution. The results are shown in Figures 5(a) and (b). Clearly, with the continuous increase in the rotation speed, the current density also increases regularly. The main reason for this is that high-speed rotation can accelerate the oxygen diffusion speed [32]. The LSV curves of the layered LSCF and bulk-LSCF at 1600 r/min are compared in Figure 5(c). The limiting currents of the layered LSCF and bulk-LSCF were 5.02 and 3.08 mA/cm2, respectively. The increase in the limiting current is mainly due to the increased defects of the layered LSCF, particularly the edge of the layered LSCF. Furthermore, the large number of pores in the layered LSCF facilitate the transmission of oxygen. The initial potentials of the layered LSCF and bulk-LSCF are 0.88 and 0.84 V, and the half-wave potentials are 0.74 and 0.69 V, respectively. In addition, Table 4 lists the electrochemical performance of the layered LSCF, bulk-LSCF, and other reported perovskite oxide catalysts. A more positive initial potential and half-wave potential measured by the layered LSCF are related not only to the porous structure but also to the oxygen adsorption capacity on the catalyst surface. According to the XPS results, layered LSCF has a stronger ability to adsorb oxygen, which can effectively improve the ORR efficiency. Figure 5(d) shows the Tafel curves of the layered LSCF and bulk-LSCF at 1600 r/min. The corresponding Tafel plots were obtained by the LSV curves near the onset potential with the Tafel equation [33]:
��=blg|j|+a
where �� is the potential; the absolute value of b is the Tafel slope; j is the current density. The Tafel slope of the layered-LSCF and bulk-LSCF are 86.42 and 93.57 mV/dec, respectively. A low Tafel slope indicates that a larger current density can be achieved with the same potential increase, further suggesting that layeredLSCF has a higher ORR catalytic activity.
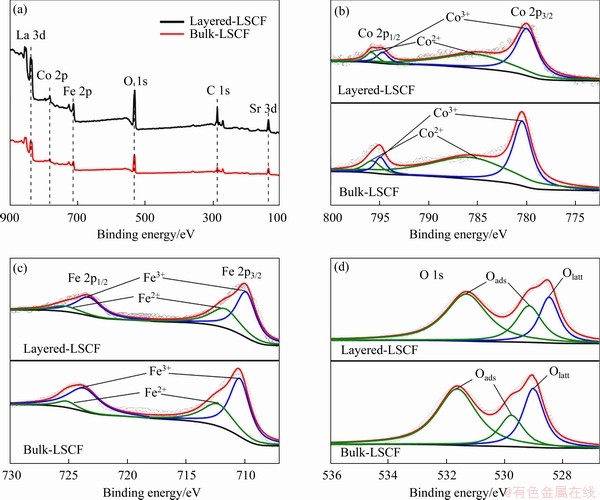
Figure 4 XPS wide-scan survey of layered LSCF and bulk-LSCF (a), and high-resolution XPS spectra of Co 2p (b),Fe 2p (c) and O 1s (d)
Table 1 Components of Co 2p peaks of LSCF by different treatments

Table 2 Components of Fe 2p peaks of LSCF by different treatments
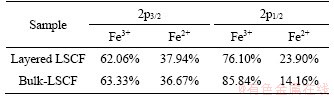
Table 3 Adsorption and lattice oxyen, and their ratio values in LSCF by different treatments
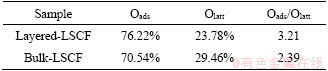
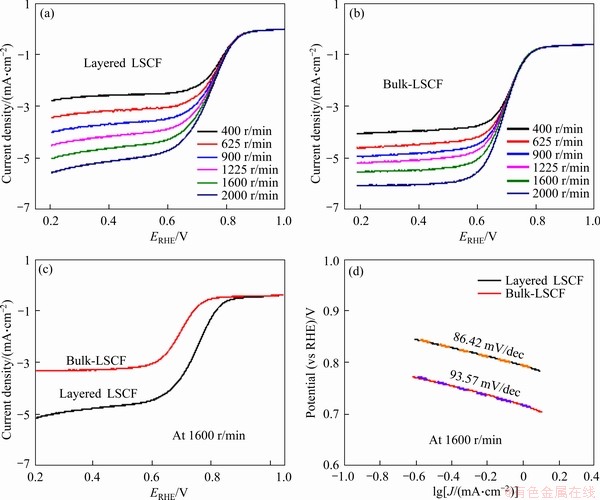
Figure 5 Linear sweep voltammetry curves of layered LSCF (a) and bulk-LSCF electrocatalysts (b) for ORR in 0.1 mol/L O2-saturated KOH solution at different rotating speeds; Comparison of LSV curves of layered LSCF and bulk-LSCF at 1600 r/min (c) and Tafel plots of layered LSCF and bulk-LSCF for ORR in 0.1 mol/L O2-saturated KOH solution at 1600 r/min (d)
Table 4 Comparison of initial potentials (Eonset), half-wave potentials (E1/2), and limiting currents (JL) of other reported ORR catalysts
To further investigate the ORR kinetics of layered LSCF, the calculated number of electron transfers (n) and yield of intermediate products HO2-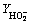 generated can be obtained by recording real-time ring and disk currents during linear sweep tests and performing further calculation using the following equations [37]:
generated can be obtained by recording real-time ring and disk currents during linear sweep tests and performing further calculation using the following equations [37]:
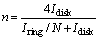
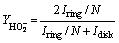
where Idisk is the disk current; Iring is the ring current; N (0.424) is the collection efficiency. The calculation results are shown in Figures 6(a) and (b), with electron transfer numbers for the layered LSCF and bulk-LSCF of about 3.86 and 3.82, and peroxide (HO2-) yields of about 11.52% and 13.07%, respectively. The electron transfer number of the layered-LSCF is very close to 4.0 and a lower yield of HO2- is generated, indicating that the ORR process of layered-LSCF is closer to the efficient four-electron transfer pathway, which is similar to the highest ORR active catalyst (Pt/C).
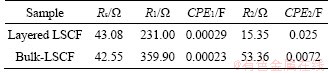
The impedance spectra of the layered LSCF and bulk-LSCF were recorded with a frequency range of 100 kHz to 0.1 Hz at 0.702 V (vs. RHE), as shown in Figure 6(c). The experimental data were fitted to an appropriate equivalent circuit using Zview software. The corner of Figure 6(c) shows the equivalent circuit diagram, in which Rs is the ohmic resistance of the electrolytes, R1 is the high-frequency resistance associated with the incorporation of an oxygen ion through the electrode and at the electrode/electrolyte interface, CPE1 is the constant phase element of the catalyst layer, R2 is the low-frequency resistance associated with the charge transfer at the electrode surface, and CPE2 is the constant phase element of the double layer [38]. The EIS features for both catalysts are quite similar, and the fitting results are listed in Table 5. By using the same test system, both catalysts have the same ohmic resistance (about 43 ��). Furthermore, layered LSCF has a lower charge transfer resistance (R2) (about 15 ��) than bulk-LSCF (about 53 ��), as shown in Table 5. The lower R2 value corresponds to a faster reaction rate, providing an explanation for the better ORR kinetics of layered-LSCF.
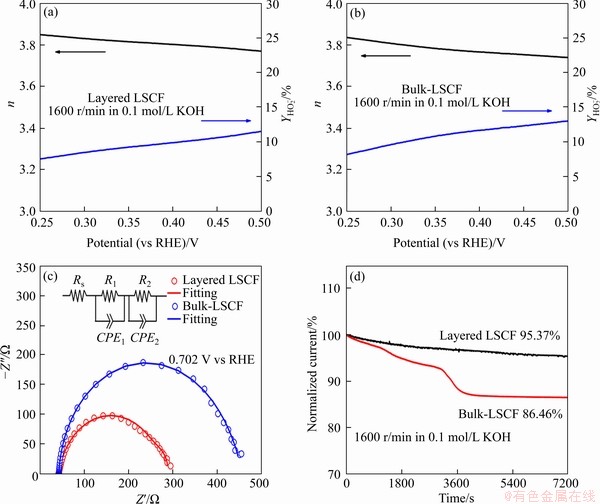
Figure 6 Electron transfer number and HO2- species yield of layered LSCF (a) and bulk-LSCF in 0.1 mol/L KOH (b), Nyquist plots and equivalent circuit (inset) of layered LSCF and bulk-LSCF at 0.702 V (vs RHE) for ORR in 0.1 mol/L KOH solution (c) and current-time curves of layered LSCF and bulk-LSCF in 0.1 mol/L O2-saturated KOH solution at 1600 r/min (d)
Table 5 Simulated elemental values of fitted equivalent circuit corresponding to electrochemical impedance spectra in Figure 6(c)
Stability is one of the most important factors in evaluating potential ORR catalysts. Based on the ORR tests in 0.1 mol/L O2-saturated KOH solution, the stabilities of layered LSCF and bulk-LSCF were studied by the chronoamperometric method. The potential was set at -0.6 V (vs. Ag/AgCl) and the rotation rate was set at 1600 r/min. Figure 6(d) shows the current-time curves. After calculation, the current density is reduced by 13.54% for bulk-LSCF throughout the test, but the current density of the layered LSCF shows a slower decreasing trend, decreasing by only 4.63% after 7200 s of continuous operation, suggesting the outstanding stability of layered LSCF.
4 Conclusions
In summary, the present study demonstrates an available sol-gel method using PVP as a complexing agent to synthesize layered LSCF. The obtained catalysts with a perfect perovskite structure possess higher specific surface area and have greater porosity than bulk-LSCF catalysts synthesized by the traditional sol-gel method. Furthermore, the results based on the electrochemical measurements in an alkaline solution indicate that compared with bulk-LSCF, layered LSCF has superior catalytic activity and excellent stability for the ORR. This result, combined with characterizations of the structure and morphology, suggests that electrochemical performance improvement of layered-LSCF could be attributed to the greater number of active sites provided by the high specific surface area and internal porous structures for O2 transport. This work provides a facile and efficient strategy to further enhance the activity of perovskite oxide as a promising cathode electrocatalyst for the metal-air batteries.
Contributors
The overarching research goals were developed by XU Zhi-feng, WANG Rui-xiang and LIU Jia-ming. XU Wei-lai and CHEN Tian-yu completed the synthesis and characterization of the catalysts. ZHONG Xiao-cong and XIE Yong-min provided the measured electrochemical performance data and analyzed the measured data. XIE Xiao-yun analyzed the calculation results. The initial draft of the manuscript was written by CHEN Zhe-qin. All authors replied to reviewers�� comments and revised the final version.
Conflict of interest
XU Wei-lai, XU Zhi-feng, CHEN Tian-yu, ZHONG Xiao-cong, XIE Yong-min, XIE Xiao-yun, CHEN Zhe-qin, LIU Jia-ming and WANG Rui-xiang declare that they have no conflict of interest.
References
[1] ZHANG Xin, WANG Xin-gai, XIE Zhao-jun, ZHOU Zhen. Recent progress in rechargeable alkali metal�Cair batteries [J]. Green Energy & Environment, 2016, 1(1): 4-17. DOI: 10.1016/j.gee.2016.04.004.
[2] LI Yan-guang, LU Jun. Metal�Cair batteries: Will they be the future electrochemical energy storage device of choice?[J]. ACS Energy Letters, 2017, 2(6): 1370-1377. DOI: 10.1021/acsenergylett.7b00119.
[3] CHENG Fang-yi, CHEN Jun. Metal�Cair batteries: From oxygen reduction electrochemistry to cathode catalysts [J]. Chemical Society Reviews, 2012, 41(6): 2172-2192. DOI: 10.1039/c1cs15228a.
[4] AKHTAR N, AKHTAR W. Prospects, challenges, and latest developments in lithium�Cair batteries [J]. International Journal of Energy Research, 2015, 39(3): 303-316. DOI: 10.1002/er.3230.
[5] ZHAO Zheng-hang, LI Ming-tao, ZHANG Li-peng, DAI Li-ming, XIA Zhen-hai. Design principles for heteroatom-doped carbon nanomaterials as highly efficient catalysts for fuel cells and metal�Cair batteries [J]. Advanced Materials, 2015, 27(43): 6834-6840. DOI: 10.1002/adma.201503211.
[6] ZHANG Lei, HUANG Qiu-an, YAN Wei, SHAO Qin-si, ZHANG Jiu-jun. Design and fabrication of non-noble metal catalyst-based air-cathodes for metal-air battery [J]. The Canadian Journal of Chemical Engineering, 2019, 97(12): 2984-2993. DOI: 10.1002/cjce.23616.
[7] ZHUANG Shu-xin, HE Jia-yi, ZHANG Wei-peng, ZHOU Nan, LU Mi, LIAN Ji-qiong, SUN Jing-jing. Preparation and application of perovskite-type oxides for electrocatalysis in oxygen/air electrodes [J]. Journal of Central South University, 2019, 26(6): 1387-1401. DOI: 10.1007/s11771-019-4095-6.
[8] CHEN Fei-ming, XUE Li-chun, SHANG Zhen-xi, ZHANG Zhen-bao, CHEN Deng-jie. An enhanced non-noble perovskite-based oxygen electrocatalyst for efficient oxygen reduction and evolution reactions [J]. Journal of Solid State Chemistry, 2020, 282: 121119. DOI: 10.1016/j.jssc.2019. 121119.
[9] JIANG San-ping. Development of lanthanum strontium cobalt ferrite perovskite electrodes of solid oxide fuel cells-A review [J]. International Journal of Hydrogen Energy, 2019, 44(14): 7448-7493. DOI: 10.1016/j.ijhydene.2019.01.212.
[10] YU W, LEE Y, LEE Y, CHO G, PARK T, TANVEER W, KIM Y, CHA S. Performance enhancement of thin film LSCF cathodes by gold current collecting layer [J]. International Journal of Precision Engineering and Manufacturing-Green Technology, 2016, 3(2): 185-188. DOI: 10.1007/s40684-016-0024-4.
[11] GRABOWSKA E. Selected perovskite oxides: Characterization, preparation and photocatalytic properties-A review [J]. Applied Catalysis B: Environmental, 2016, 186: 97-126. DOI: 10.1016/j.apcatb.2015.12.035.
[12] KIM J, KIM G, KIM T, KWON S, BACK H, LEE J, LEE S, KANG H, LEE K. Efficient planar-heterojunction perovskite solar cells achieved via interfacial modification of a sol-gel ZnO electron collection layer [J]. Journal of Materials Chemistry A, 2014, 2(41): 17291-17296. DOI: 10.1039/ C4TA03954H.
[13] BIAN Liu-zhen, WANG Li-jun, CHEN Ning, LI Fu-shen, CHOU K. Enhanced performance of La0.7Sr0.3Fe0.9Ni0.1O3 cathode by partial substitution with Ce [J]. Ceramics International, 2017, 43(1): 982-987. DOI: 10.1016/j.ceramint. 2016.10.029.
[14] LI Shu-li, XIA Tian, LI Qiang, SUN Li-ping, HUO Li-hua, ZHAO Hui. A-site Ba-deficiency layered perovskite EuBa1-xCo2O6-�� cathodes for intermediate-temperature solid oxide fuel cells: Electrochemical properties and oxygen reduction reaction kinetics [J]. International Journal of Hydrogen Energy, 2017, 42(38): 24412-24425. DOI: 10.1016/j.ijhydene. 2017.08.049.
[15] JIN Chao, CAO Xue-cheng, ZHANG Li-ya, ZHANG Cong, YANG Rui-zhi. Preparation and electrochemical properties of urchin-like La0.8Sr0.2MnO3 perovskite oxide as a bifunctional catalyst for oxygen reduction and oxygen evolution reaction [J]. Journal of Power Sources, 2013, 241: 225-230. DOI: 10.1016/j.jpowsour.2013.04.116.
[16] ZHU Yin-long, ZHOU Wei, CHEN Yu-bo, YU Jie, XU Xiao-min, SU Chao, TADE M, SHAO Zong-ping. Boosting oxygen reduction reaction activity of palladium by stabilizing its unusual oxidation states in perovskite [J]. Chemistry of Materials, 2015, 27(8): 3048-3054. DOI: 10.1021/acs. chemmater.5b00450.
[17] LI Gang, FENG Ya-qing, GAO Pei, LI Xiang-gao. Preparation of mono-dispersed polyurea-urea formaldehyde double layered microcapsules [J]. Polymer Bulletin, 2008, 60(5): 725-731. DOI: 10.1007/s00289-008-0894-x.
[18] FAN Hai-long, WANG Le, FENG Xun-da, BU Ya-zhong, WU De-cheng, JIN Zhao-xia. Supramolecular hydrogel formation based on tannic acid [J]. Macromolecules, 2017, 50(2): 666-676. DOI: 10.1021/acs.macromol.6b02106.
[19] DU Y, YANG P, MOU Z, HUA N, JIANG L. Thermal decomposition behaviors of PVP coated on platinum nanoparticles [J]. Journal of Applied Polymer Science, 2006, 99(1): 23-26. DOI: 10.1002/app.21886.
[20] HAN M, NAM S. Thermodynamic and rheological variation in polysulfone solution by PVP and its effect in the preparation of phase inversion membrane [J]. Journal of Membrane Science, 2002, 202(1, 2): 55-61. DOI: 10.1016/S0376-7388(01)00718-9.
[21] JING Cheng-bin, HOU Jin-xia. Sol-gel-derived alumina/ polyvinylpyrrolidone hybrid nanocomposite film on metal for corrosion resistance [J]. Journal of Applied Polymer Science, 2007, 105(2): 697-705. DOI: 10.1002/app.26074.
[22] VANDERVLIET D, STRMCNIK D, WANG C, STAMENKOVIC V, MARKOVIC N, KOPER M. On the importance of correcting for the uncompensated ohmic resistance in model experiments of the oxygen reduction reaction [J]. Journal of Electroanalytical Chemistry, 2010, 647(1): 29-34. DOI: 10.1016/j.jelechem.2010.05.016.
[23] NIE Li-fang, LIU Ze, LIU Ming-fei, YANG Lei, ZHANG Yu-jun, LIU Mei-lin. Enhanced performance of La0.6Sr0.4Co0.2Fe0.8O3-�� (LSCF) cathodes with graded microstructure fabricated by tape casting [J]. Journal of Electrochemical Science and Technology, 2010, 1(1): 50-56. DOI: 10.5229/JECST.2010.1.1.050.
[24] ZHOU Wei, RAN Ran, SHAO Zong-ping, JIN Wan-qin, XU Nan-ping. Synthesis of nano-particle and highly porous conducting perovskites from simple in situ sol-gel derived carbon templating process [J]. Bulletin of Materials Science, 2010, 33(4): 371-376. DOI: 10.1007/s12034-010-0056-2.
[25] LI Geng, HE Bei-bei, LING Yi-han, XU Jian-mei, ZHAO Ling. Highly active YSB infiltrated LSCF cathode for proton conducting solid oxide fuel cells [J]. International Journal of Hydrogen Energy, 2015, 40(39): 13576-13582. DOI: 10.1016/j.ijhydene.2015.07.164.
[26] SHANG Zhen-xi, CHEN Zi-long, ZHANG Zhen-bao, YU Jing, TAN Shao-zao, CIUCCI F, SHAO Zong-ping, LEI Hao, CHEN Deng-jie. CoFe nanoalloy particles encapsulated in nitrogen-doped carbon layers as bifunctional oxygen catalyst derived from a prussian blue analogue [J]. Journal of Alloys and Compounds, 2018, 740: 743-753. DOI: 10.1016/j.jallcom.2018.01.019.
[27] XU Ji-jing, XU Dan, WANG Zhong-li, WANG Heng-guo, ZHANG Lei-lei, ZHANG Xin-bo. Synthesis of perovskite-based porous La0.75Sr0.25MnO3 nanotubes as a highly efficient electrocatalyst for rechargeable lithium�Coxygen batteries [J]. Angewandte Chemie International Edition, 2013, 52(14): 3887-3890. DOI: 10.1002/anie.201210057.
[28] MONTINI T, BEVILACQUA M, FONDA E, CASULA M, LEE S, TAVAGNACCO C, GORTE R, FORNASIERO P. Relationship between electrical behavior and structural characteristics in Sr-doped LaNi0.6Fe0.4O3 mixed oxides [J]. Chemistry of Materials, 2009, 21(8): 1768-1774. DOI: 10.1021/cm900467c.
[29] ZHOU Jun, SHIN T, NI Cheng-sheng, CHEN Gang, WU Kai, CHENG Yong-hong, IRVINE J. In situ growth of nanoparticles in layered perovskite La0.8Sr1.2Fe0.9Co0.1O4-�� as an active and stable electrode for symmetrical solid oxide fuel cells [J]. Chemistry of Materials, 2016, 28(9): 2981-2993. DOI: 10.1021/acs.chemmater.6b00071.
[30] ZHU Yin-long, ZHOU Wei, YU Jie, CHEN Yu-bo, LIU Mei-lin, SHAO Zong-ping. Enhancing electrocatalytic activity of perovskite oxides by tuning cation deficiency for oxygen reduction and evolution reactions [J]. Chemistry of Materials, 2016, 28(6): 1691-1697. DOI: 10.1021/acs.chemmater. 5b04457.
[31] PENG Xiang-feng, WANG Zhen-hai, WANG Zhao, PAN Yun-xiang. Multivalent manganese oxides with high electrocatalytic activity for oxygen reduction reaction [J]. Frontiers of Chemical Science and Engineering, 2018, 12(4): 790-797. DOI: https://doi.org/10.1007/s11705-018-1706-y.
[32] HUANG Jian-jian, ZHU Neng-wu, YANG Ting-ting, ZHANG Tai-ping, WU Ping-xiao, DANG Zhi. Nickel oxide and carbon nanotube composite (NiO/CNT) as a novel cathode non-precious metal catalyst in microbial fuel cells [J]. Biosensors and Bioelectronics, 2015, 72: 332-339. DOI: 10.1016/j.bios.2015.05.035.
[33] WU Heng-bo, WANG Jie, YAN Ji, WU Ze-xing, JIN Wei. MOF-derived two-dimensional N-doped carbon nanosheets coupled with Co�CFe�CP�CSe as efficient bifunctional OER/ORR catalysts [J]. Nanoscale, 2019, 11(42): 20144-20150. DOI: 10.1039/C9NR05744G.
[34] CHEN Xiao-dong, WANG Ning, SHEN Kui, XIE Yang-kai, TAN Yong-peng, LI Ying-wei. MOF-derived isolated Fe atoms implanted in N-Doped 3D hierarchical carbon as an efficient ORR electrocatalyst in both alkaline and acidic media [J]. ACS Applied Materials & Interfaces, 2019, 11(29): 25976-25985. DOI: 10.1021/acsami.9b07436.
[35] SHEN Yu-juan, ZHU Yin-long, SUNARSO J, GUAN Da-qin, LIU Bo, LIU Hong, ZHOU Wei, SHAO Zong-ping. New phosphorus-doped perovskite oxide as an oxygen reduction reaction electrocatalyst in an alkaline solution [J]. Chemistry�CA European Journal, 2018, 24(27): 6950-6957. DOI: 10.1002/chem.201705675.
[36] MIYAZAKI K, KAWAKITA K, ABE T, FUKUTSUKA T, KOJIMA K, OGUMI Z. Single-step synthesis of nano-sized perovskite-type oxide/carbon nanotube composites and their electrocatalytic oxygen-reduction activities [J]. Journal of Materials Chemistry, 2011, 21(6): 1913-1917. DOI: 10.1039/c0jm02600j.
[37] PAULUS U, SCHMIDT T, GASTEIGER H, BEHM R. Oxygen reduction on a high-surface area Pt/Vulcan carbon catalyst: A thin-film rotating ring-disk electrode study [J]. Journal of Electroanalytical Chemistry, 2001, 495(2): 134-145. DOI: 10.1016/S0022-0728(00)00407-1.
[38] XU Yu-jiao, TSOU A, FU Yue, WANG Jin, TIAN Jing-hua, YANG Rui-zhi. Carbon-coated perovskite BaMnO3 porous nanorods with enhanced electrocatalytic perporites for oxygen reduction and oxygen evolution [J]. Electrochimica Acta, 2015, 174: 551-556. DOI: 10.1016/j.electacta.2015.05.184.
(Edited by FANG Jing-hua)
���ĵ���
��ײ�״���ѿ���������La0.6Sr0.4Co0.2Fe0.8O3��������ԭ������
ժҪ������ת��װ�õķ�չ����ͳɱ������ԵĴ��������о��Ծ���ϩ������ͪΪ��ϼ����ϳ��˶�ײ�״���ѿ���������La0.6Sr0.4Co0.2Fe0.8O3(LSCF)���Ʊ��Ķ�ײ�״LSCF�ȱ����Ϊ23.74 m2/g���ȴ�ͳ�ܽ�-�������Ʊ���LSCF(5.08 m2/g)�߳���4���������������������ԭ��Ӧ������0.1 mol/L KOH��Һ��������ת���̵缫���в��ԣ���1600 r/minת�����ܴﵽ0.88 V(��ת��Ϊ������缫)����ʼ��λ��5.02 mA/cm2�ļ������ܶȡ��봫ͳ�����Ʊ���LSCF���ѿ�����������ȣ����о����Ƶõ���������и��ߵĴ����Ժ��ȶ��ԡ�
�ؼ��ʣ�����ԭ��Ӧ������������ѿ�״�ṹ
Foundation item: Project(20192BAB216015) supported by the Science and Technology Program of Jiangxi Province, China; Projects(GJJ180464, GJJ171499) supported by the Science and Technology Program of Education Department of Jiangxi Province, China; Project(jxxjbs17057) supported by the Scientific Research Foundation of Jiangxi University of Science and Technology, China; Project([2018] 50) supported by the Key R&D Programs of Science and Technology Project of Ganzhou City, China; Project([2017] 179) supported by the Science and Technology Project of Ganzhou City, China
Received date: 2020-04-21; Accepted date: 2020-10-12
Corresponding author: CHEN Zhe-qin, Master, Laboratory Technician; Tel: +86-15970833528; E-mail: jxuchen@foxmail.com; ORCID: https://orcid.org/0000-0002-3322-4620

