�����»���Na2CO3���������ֱ��̼�ȷ���ԭ������
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2021���6��
�������ߣ���֮�� Ф���� �亣�� �ܹ���
����ҳ�룺1818 - 1827
�ؼ��ʣ�������������Na2CO3��̼�Ȼ�ԭ
Key words��ilmenite concentrates; microwave; Na2CO3; carbothermic reduction
ժ Ҫ�����һ�ֻ���ֱ��̼�Ȼ�ԭ�������ȵ���������ɫ��Ч�����¹��ա�ͨ�����Ӹ�����Na2CO3 ���ٻ�ԭ��Ӧ�Ľ��У�����Na2CO3����ѭ�����á���Ӧԭ��(������+ʯī+Na2CO3)�������¼�����1073~1123 K������20 min����Ҫ��ԭ����ΪNa2TiO3�ͽ��������������Ľ������ʿ��Դﵽ92.67%~93.21%����ԭ�������ξ���ˮϴ��CO2��������ĥ�ʹ�ѡ���룬���տ��Եõ������Ϻ����ϡ����и�������TiO2�������Դﵽ90.04% (��������)�����ɺ�����������TiCl4��TiO2�����ڽ������ʡ���ѡ����Ч�ʺͲ���������ԵȲ������Ż��õ��Ļ�ԭ�¶�Ϊ1123 K�����⣬���������Na2CO3����������������µĻ�ԭ������
Abstract: A clean and efficient route for the utilization of ilmenite concentrates was proposed by direct carbothermic reduction in microwave field. High dosage of Na2CO3, which can be recycled, was added to accelerate the reduction reaction of ilmenite concentrates. After microwave heating in the temperature range of 1073-1123 K for 20 min, the main products were Na2TiO3 and metallic Fe with the metallization ratios being as high as 92.67%-93.21%. The reduction products were processed by water leaching, ball-milling in CO2 atmosphere and magnetic separation in turn. The final products after magnetic separation were Fe-rich materials and Ti-rich materials (90.04 wt.% TiO2), which can be used to produce iron and TiCl4 or TiO2. The optimized heating temperature was 1123 K in terms of metallization ratios, magnetic separation and caking property of the reduction products. Besides, the reduction mechanism of ilmenite concentrates with the addition of Na2CO3 in microwave field was also proposed.
Trans. Nonferrous Met. Soc. China 31(2021) 1818-1827
Zhi-gang YU1,2,3, Jing-wu XIAO1,2,3, Hai-yan LENG1,2,3, Kuo-chih CHOU1,2,3
1. School of Materials Science and Engineering, Shanghai University, Shanghai 200072, China;
2. State Key Laboratory of Advanced Special Steels, Shanghai University, Shanghai 200072, China;
3. Shanghai Key Laboratory of Advanced Ferro-metallurgy, Shanghai University, Shanghai 200072, China
Received 28 June 2020; accepted 7 April 2021
Abstract: A clean and efficient route for the utilization of ilmenite concentrates was proposed by direct carbothermic reduction in microwave field. High dosage of Na2CO3, which can be recycled, was added to accelerate the reduction reaction of ilmenite concentrates. After microwave heating in the temperature range of 1073-1123 K for 20 min, the main products were Na2TiO3 and metallic Fe with the metallization ratios being as high as 92.67%-93.21%. The reduction products were processed by water leaching, ball-milling in CO2 atmosphere and magnetic separation in turn. The final products after magnetic separation were Fe-rich materials and Ti-rich materials (90.04 wt.% TiO2), which can be used to produce iron and TiCl4 or TiO2. The optimized heating temperature was 1123 K in terms of metallization ratios, magnetic separation and caking property of the reduction products. Besides, the reduction mechanism of ilmenite concentrates with the addition of Na2CO3 in microwave field was also proposed.
Key words: ilmenite concentrates; microwave; Na2CO3; carbothermic reduction
1 Introduction
Ilmenite is a kind of complex mineral containing both iron and titanium, which is of vital importance in metallurgical industry [1,2]. However, there is no clear and efficient route to produce iron and TiO2 pigment directly at present. Chlorination process is not a suitable method due to the low-grade TiO2 and high impurity of ilmenite concentrates [3,4]. Sulfate process seems to be an efficient route to produce TiO2 pigments [5], but it is restrained by the severe environmental challenge [6,7]. Conventional blast furnace route requires high temperature, which is difficult to extract TiO2 from the produced Ti-rich slags [8]. Many researchers [9-12] have investigated the gas- based reduction of ilmenite. However, there is still a long way for the industrialization of gas-based reduction of ilmenite due to the shortage of cheap gas. Solid-state reduction of ilmenite [13-19] is a promising method to obtain iron and TiO2 pigment at relatively low temperatures. However, it is time-consuming with the reduction time being around 2-3 h in general.
As a clean and efficient process for the preparation of materials, microwave techniques attracted considerable attentions in recent years [20-24]. Many researchers [18,25-29] have proven that ilmenite concentrates had excellent microwave-absorbing characteristics, which could be heated up quickly in microwave field. KELLY and ROWSON [29] investigated the utilization of microwave energy for the reduction of oxidized ilmenite concentrates in a time-saving and energy-saving way. LI et al [18] found that iron was formed in the reduction of ilmenite at 1173 K in microwave field, which is lower than the reduction temperature by conventional electric furnace heating. Therefore, microwave heating is benefit for the solid-state reduction of ilmenite.
Additives can intensify the reduction of ilmenite. However, the amount of additives is normally below 5 wt.% due to the restraint of caking problem [16-18,30,31]. In conventional heating process, the caking problem is difficult to be avoided due to the long heating-time and high heating-temperature. However, in microwave heating process, it is possible to control the caking problem of additives due to the fast and efficient heating characteristics of microwave.
In the present work, a direct carbothermic reduction method of ilmenite concentrates was investigated by adding high dosage of Na2CO3 in microwave field. The caking problems of the reduction products could be controlled by microwave irradiation due to the low heating temperature and short reduction time.
2 Experimental
2.1 Materials
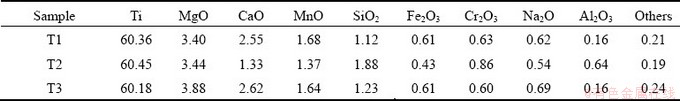
The ilmenite concentrates used in this investigation were supplied by Panzhihua Iron and Steel Company (Panzhihua, China). The main chemical composition is presented in Table 1. The graphite (��99.8 wt.%) and Na2CO3 (��99.8 wt.%) were supplied by Sino Pharm Chemical Reagent Co., Ltd.
Table 1 Chemical compositions of ilmenite concentrates (wt.%)

2.2 Instrument and procedures
The schematic diagrams of microwave heating apparatus (3 kW and 2.45 GHz) and conventional heating instrument are illustrated in Fig. 1.

Fig. 1 Schematic diagrams of heating apparatus
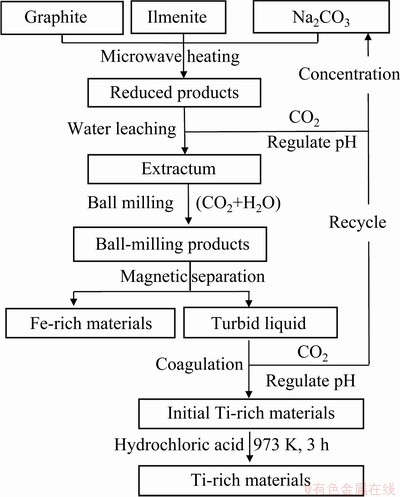
Fig. 2 Flowsheet of new route
The reduction reaction is given in the following equation:
FeTiO3+2C+Na2CO3��Fe+Na2TiO3+3CO�� (1)
The process flowsheet for direct carbothermic reduction of ilmenite concentrates is illustrated in Fig. 2. First, the ilmenite (FeTiO3) was mixed with graphite (C) and Na2CO3 with larger mole ratios than the stoichiometric ratios by ball-milling for 2 h. The chemical compositions of the samples are listed in Table 2. The molar ratios of Na2CO3 and C in Table 2 were determined by the molar ratio of FeTiO3, which were set as 1 mol according to Eq. (1). In order to investigate the effect of Na2CO3, two samples with more C (0Na2CO3-8C) and less Na2CO3 (0.5Na2CO3-2.8C) than the stoichiometric ratios were also prepared under the same condition for comparison.
Table 2 Chemical compositions of samples

All the samples were placed in a BN crucible and heated up to certain temperatures under Ar gas in a tube microwave furnace, and all the samples can be heated up to the target temperatures (1023-1173 K) within 10 min, which indicates the good microwave heating property of the mixtures. The purity of the gas used in the investigation was 99.999 wt.%. The reduction products were processed with the steps shown in Fig. 2 to produce Ti-rich and Fe-rich materials.
2.3 Analytical method
The phases of the reduction products were characterized by X-ray diffraction analysis (XRD, Bruker AXS, D8 Advance, Cu K�� radiation, 40 kV and 40 mA). Microscopic observation and analysis for the reduced samples were conducted by scanning electron microscope and energy dispersive spectroscopic (SEM and EDS, HITACHI SU-1500, HITACHI, Japan). The contents of Ti and Fe in Ti-rich and Fe-rich materials were determined by ICP-OES (Perkin-Elmer 7300DV, America). The chemical compositions of samples were determined by X-ray fluorescence (XRF, XRF-1800, Shimadzu, Kyoto, Japan). The total Fe (TFe) and metallic Fe (MFe) contents in the reduction products were determined by titrimetric analyses for four times, respectively. The metallization ratios (��) of reduction products were defined as
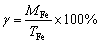 (2)
(2)
3 Results and discussion
3.1 Direct reduction in microwave field
3.1.1 Phase transitions during reduction
The XRD results of Sample 4 held for various time at 1073 K in microwave field are depicted in Fig. 3. The phases of graphite, metallic Fe and Na0.75Fe0.75Ti0.25O2 could be observed at this temperature, and Na2TiO3 phase emerged at 0 min (Fig. 3(a)). The peak intensities of C and Na0.75Fe0.75Ti0.25O2 phases gradually decreased, while those of metallic Fe and Na2TiO3 increased, as the reaction continued. It can be inferred that Na0.75Fe0.75Ti0.25O2 is an intermediate phase in the process. The peaks of Na0.75Fe0.75Ti0.25O2 phase almost disappeared when the sample was held for 20 min, which was chosen as an optimized holding time.
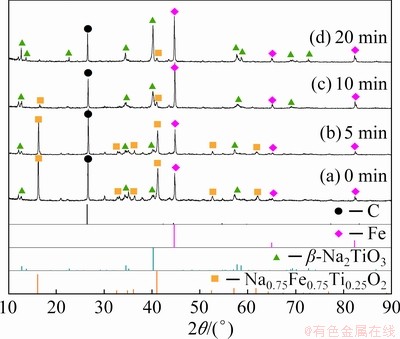
Fig. 3 XRD patterns of reduction products held for different time
The XRD patterns of Sample 4 held for 20 min at different temperatures (1023-1173 K) in microwave field are depicted in Fig. 4, where the main products were metallic Fe and Na2TiO3. As an intermediate product, the peak intensity of Na0.75Fe0.75Ti0.25O2 phase decreased gradually with the increase of heating temperature. Na2TiO3 was the target product and its peak intensity was enhanced with the increase of reduction temperature from 1023 to 1123 K. However, Na2TiO3 was reduced to Na0.99TiO2 when the heating temperature was increased to 1173 K, which can be obtained from the comparison between Figs. 4(c) and (d). As a result, 1123 K was selected as the optimized heating temperature in microwave field.
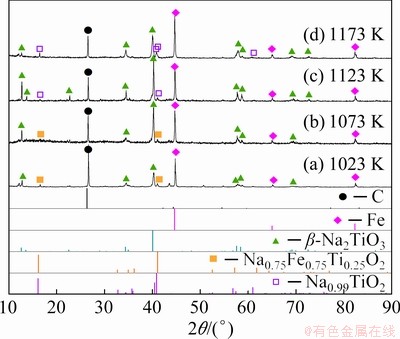
Fig. 4 XRD patterns of reduction products heated at different temperatures
The influence of Na2CO3 addition on the final products was also investigated through the XRD results. As illustrated in Fig. 5(a), ilmenite was reduced to metallic Fe, rutile and TiO when no Na2CO3 was added, indicating that the reduction was incomplete. In order to eliminate the influence of insufficient C, excessive C was added to the Sample 2 (Fig. 5(b)) for comparison. The phases of the reduction products in Fig. 5(b) didn��t change much compared with those in Fig. 5(a), which means the amount of C in Sample 1 is sufficient for the reduction reaction. As depicted in Fig. 5(c), ilmenite was reduced to metallic Fe, NaFeTiO4 and Na9.5Mg0.75Ti10.25O26 with the addition of 0.5Na2CO3 (0.5 reaction equivalent, Sample 3). In other words, the addition of Na2CO3 changed the reaction pathway, which makes the ilmenite reduced more easily. However, the reaction was incomplete with the addition of 0.5Na2CO3 for the reduction products containing NaFeTiO4 phase, which can be reduced to metallic Fe. After adding sufficient Na2CO3 in Sample 4, ilmenite was totally reduced to metallic Fe and Na2TiO3 (Fig. 5(d)).
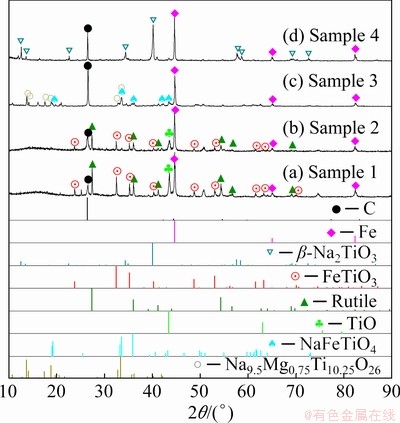
Fig. 5 XRD patterns of reduction products with different Na2CO3 additions
To confirm the effect of microwave heating, Sample 4 was reduced at 1073 K for 20 min by conventional electric heating for comparison. The XRD patterns of the reduction products are illustrated in Fig. 6, where XRD patterns of ilmenite and raw materials are also plotted. It is easy to find that Sample 4 was reduced to the intermediate phase of Na0.75Fe0.75Ti0.25O2 by electric heating (Fig. 6(c)), while it was reduced to the target products (Fe and Na2TiO3) by microwave heating (Fig. 6(d)). By combining the XRD results in Figs. 6(c) and (d), it can be concluded that microwave heating benefits for the reaction in the same condition of heating.
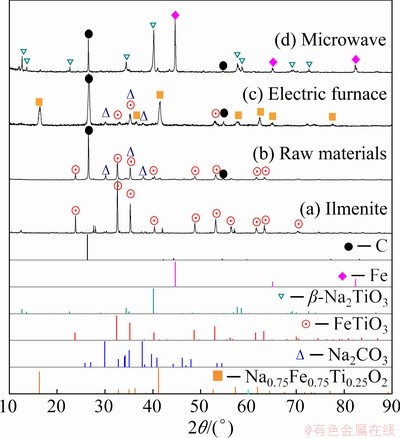
Fig. 6 Comparison of XRD patterns of reduction products by microwave and electric heating
3.1.2 Metallization ratios of reduction products
The metallization ratios of the reduction products heated at different temperatures for 20 min in microwave field are depicted Fig. 7. The metallization ratios increased first and then declined with the increase of reduction temperature. The reducibility of graphite is enhanced with the increase of reduction temperature [10], which may be the main contribution to the increased metallization ratios. When the heating temperature was increased to 1173 K, the metallization ratio decreased, which can be attributed to the formation of Na0.99TiO2. As shown in Fig. 4(d), Na2TiO3 was reduced to Na0.99TiO2 when the heating temperature was increased to 1173 K. The formation of Na0.99TiO2 phase may change the porosity of reduction system and harden the reduction products. In the process of titrimetric analyses, the metallic Fe cannot react completely due to the densification of the reduction products.
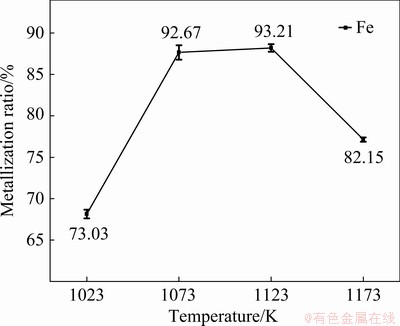
Fig. 7 Variation of metallization ratios with temperature
In order to confirm this assumption, the filtered residue of reduction products after testing the metallization ratio was examined by XRD. As shown in Fig. 8, Fe was observed in the residue after titrimetric analyses, which can support the assumption above.
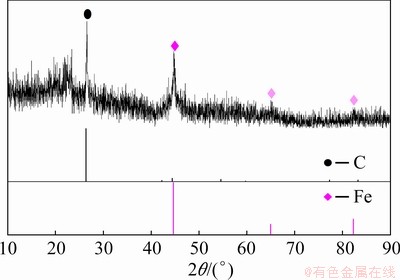
Fig. 8 XRD patterns of filtered residue after titrimetric analyses
3.1.3 Morphology of reduction products
The reduction products were analyzed by SEM and EDS to gain insight into the reduction mechanism. The SEM images of Sample 4 held for 20 min at 1073, 1123 and 1173 K are illustrated in Fig. 9. The main reduction products were metallic Fe and Na2TiO3 with many cracks and holes, which may be caused by the release of CO and/or CO2 at the reduction stage. The tendency that the structure of Na2TiO3 transformed from sheet to block with the increase of reduction temperature could also be found.

Fig. 9 SEM images of reduction products of Sample 4
As shown in Fig. 10, the distributions of Fe, Ti, Na and O were obtained at the selected areas of the products reduced at 1073, 1123 and 1173 K, respectively. Ti and Fe were gradually separated in the reduction products with the increase of reduction temperature, and the size of the metallic Fe tended to grow larger. Large size of metallic Fe is good for improving the efficiency of magnetic separation, which can be set as one of the factors for optimizing the heating temperature.

Fig. 10 SEM images and EDS results of reduction products of Sample 4
3.2 Preparation of Ti-rich materials
3.2.1 Magnetic separation
As shown in Fig. 11, the reduction products were processed with the steps of water leaching, ball-milling in CO2 atmosphere and magnetic separation to produce Ti-rich and Fe-rich materials, which were dried at 373 K for 12 h. The contents of Ti and Fe in Ti-rich and Fe-rich materials obtained from reduction products at different temperatures are illustrated in Fig. 11.
As depicted in Fig. 11(a), the content of Ti increased, while that of Fe decreased, with the increase of reduction temperature in the Ti-rich materials. Besides, the increasing tendency slowed down in the temperature range of 1123-1173K. However, the content of Fe increased slowly with the increase of reduction temperature in the Fe-rich materials (Fig. 11(b)), while the content of Ti increased first and then decreased. In the reduction process, the metallic Fe grew and gathered with the increase of reduction temperature. At this time, some Ti-containing species might be covered by metallic Fe at 1123 K, and then be uncovered when the temperature was increased to 1173 K, which may be the reason for the change of Ti content in Fe-rich materials.
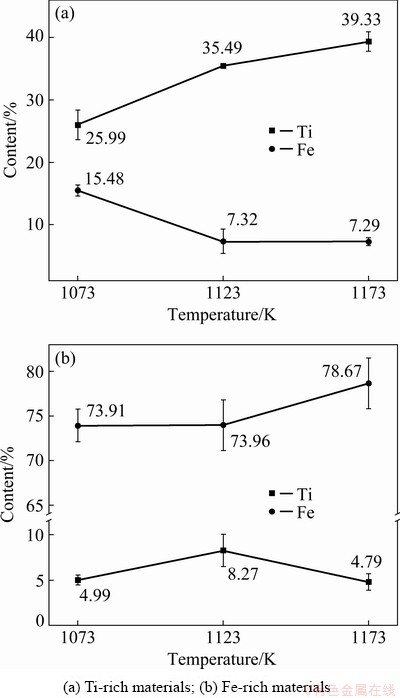
Fig. 11 Content variations of Ti and Fe in separated materials
3.2.2 Final products
It was demonstrated by experiment that the caking problems of the sample adhering to the BN crucible would occur at lower temperature (1123 and 1073 K), while that would not occur at higher temperature (1173 K). At 1173 K, Na2CO3 was melted, and the sample would adhere to the crucible, which may result in the corrosion of furnace. Considering the results of metallization ratio, magnetic separation and caking problems, 1123 K was optimized as a suitable reduction temperature.
The XRD patterns of the Ti-rich and Fe-rich materials are illustrated in Fig. 12, where the main products were TiO2 and Fe in Ti-rich and Fe-rich materials, respectively.
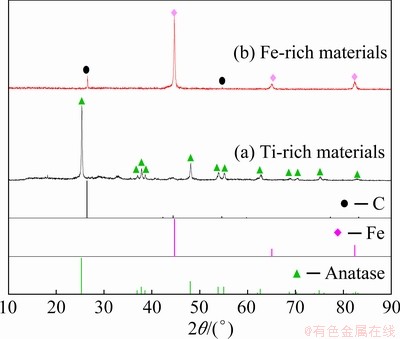
Fig. 12 XRD patterns of final products
The compositions of Ti-rich materials marked with T1, T2 and T3 obtained by the same procedures were analyzed by X-ray fluorescence (XRF) and the results are shown in Table 3.
Supposing that TiO2 was the only oxide of titanium, then the content of TiO2 in Ti-rich materials can be calculated according to the following equation:
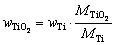 (3)
(3)
where 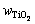 and wTi are the mass fractions of TiO2 and Ti;
and wTi are the mass fractions of TiO2 and Ti; 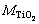 and
and 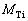 are the molar masses of TiO2 and Ti. However, the mass fractions of Samples T1, T2 and T3 will be 100.71%, 100.86% and 100.41%, if calculated by Eq. (3). The results (��100%) suggest the existence of other oxides of titanium besides TiO2, such as Ti2O3, whose content is too little to be detected by XRD. The real mass fraction of TiO2 (
are the molar masses of TiO2 and Ti. However, the mass fractions of Samples T1, T2 and T3 will be 100.71%, 100.86% and 100.41%, if calculated by Eq. (3). The results (��100%) suggest the existence of other oxides of titanium besides TiO2, such as Ti2O3, whose content is too little to be detected by XRD. The real mass fraction of TiO2 (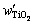 ) should be calculated by the following equation:
) should be calculated by the following equation:
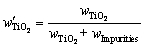 (4)
(4)
where wImpurities is mass fraction of impurities, such as CaO, MgO and SiO2, which can be obtained through Table 3.
Table 3 Chemical compositions of Ti-rich materials (wt.%)
According to Eq. (4), the real purity of TiO2 in Ti-rich materials was 90.16, 90.41 and 89.56 wt.% for Samples T1, T2 and T3, respectively. The average purity of the three samples was 90.04 wt.%, which demonstrates the feasibility of the above process. Fe-rich and Ti-rich materials obtained by this technique can be used as raw materials to produce iron by the electric furnace and to produce TiCl4 by chlorination process [7], respectively. In addition, Ti-rich materials can also be used to produce TiO2 in a series of processes. For the preparation of Ti-rich materials, the reduction temperature was prior to be considered in the work. However, other parameters, such as size of metallic Fe, ball-milling time, hydrolysis rate of Na2TiO3 and partial pressure of CO2, will also influence the efficiency of magnetic separation. It is expected to improve the efficiency of magnetic separation of Ti and Fe by adjusting these parameters to obtain a higher quality of Ti-rich materials.
3.3 Mechanism of reduction in microwave field
The reduction mechanism of the sample in microwave field is presented in Fig. 13.
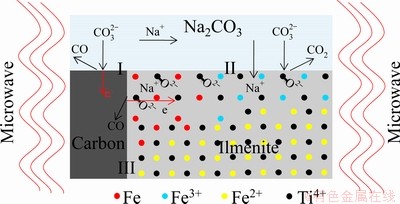
Fig. 13 Schematic diagram of reduction mechanism in microwave field
Reaction occurring at Interface II:
M+ ��MO2-+CO2�� (5)
��MO2-+CO2�� (5)
Reactions occurring at Interface III:
C+ MO2-��M+2e+CO�� (6)
CO+ MO2-��M+2e+CO2�� (7)
Fe3++3e��Fe (8)
Fe2++2e��Fe (9)
where M represents the species containing Ti and O.
As shown in Fig. 13, both graphite and ilmenite can absorb the microwave to improve the reaction activities. At Interface II,  could combine with ilmenite and generate CO2 and Na+, and Na+ would diffuse into the matrix of ilmenite. At Interface III, on one hand, C or CO lost 2e when combined with MO2- with the absorbing of microwave; on the other hand, Fe2+ obtained 2e and lost the bonds with oxygen and titanium, which altered the structure of the matrix of ilmenite. The alterations promoted the transferring capability of electrons in ilmenite, which intensified the reduction of ilmenite. Another possible mechanism is a direct route where
could combine with ilmenite and generate CO2 and Na+, and Na+ would diffuse into the matrix of ilmenite. At Interface III, on one hand, C or CO lost 2e when combined with MO2- with the absorbing of microwave; on the other hand, Fe2+ obtained 2e and lost the bonds with oxygen and titanium, which altered the structure of the matrix of ilmenite. The alterations promoted the transferring capability of electrons in ilmenite, which intensified the reduction of ilmenite. Another possible mechanism is a direct route where 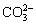 combined with graphite at the Interface I [32,33], with the reaction below, and Fe2+/Fe3+ was reduced to Fe in the reactions occurring at Interface III.
combined with graphite at the Interface I [32,33], with the reaction below, and Fe2+/Fe3+ was reduced to Fe in the reactions occurring at Interface III.
2C+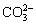 ��3CO��+2e (10)
��3CO��+2e (10)
However, under electrical heating, the reduction product was only Na0.75Fe0.75Ti0.25O2, without the formation of Fe (Fig. 6(c)), indicating that no reactions occurred at Interface III (Fig. 13). The differences in reaction activities of graphite and ilmenite under microwave heating and electrical heating might lead to those two reaction mechanisms.
4 Conclusions
(1) The optimized heating temperature was 1123 K in terms of metallization ratios, magnetic separation and caking property of the reduction products.
(2) With the addition of Na2CO3, the reduction temperature and time were significantly decreased.
(3) The improved reaction activities of graphite and ilmenite under microwave heating might contribute to the direct carbon-thermic reduction of ilmenite concentrates with Na2CO3 addition.
(4) After microwave heating in the temperature range of 1073-1123 K for 20 min, the main products of the sample were Na2TiO3 and metallic Fe with the metallization ratios being as high as 92.67%-93.21%.
(5) The purity of TiO2 in Ti-rich materials was 90.04 wt.%, which can be used as raw materials to produce TiCl4 or TiO2. It is expected to develop a clean and efficient route for the utilization of ilmenite by adding high dosage of Na2CO3 in microwave field.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Nos. 51734002, 51474141), China Postdoctoral Science Foundation (No. 2020M671071), and Independent Research and Development Project of State Key Laboratory of Advanced Special Steel, Shanghai Key Laboratory of Advanced Ferro- metallurgy, Shanghai University, China (No. SKLASS 2019-Z014).
References
[1] LIANG Bin, LI Chun, ZHANG Cheng-gang, ZHANG Yong-kui. Leaching kinetics of Panzhihua ilmenite in sulfuric acid [J]. Hydrometallurgy, 2005, 76(3-4): 173-179.
[2] WANG Shuai, GUO Yu-feng, JIANG Tao, CHEN Feng, ZHENG Fu-qiang, TANG Min-jun, YANG Ling-zhi, QIU Guan-zhou. Appropriate titanium slag composition during smelting of vanadium titanomagnetite metallized pellets [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(12): 2528-2537.
[3] LI Chun, LIANG Bin, GUO Ling-hong, WU Zi-bin. Effect of mechanical activation on the dissolution of Panzhihua ilmenite [J]. Minerals Engineering, 2006, 19(14): 1430-1438.
[4] BORDBAR H, ABEDINI H, YOUSEFI A A. Parameters affecting reaction rate and conversion of TiO2 chlorination in a fluidized bed reactor: Experimental and modeling approach [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(10): 2114-2124.
[5] MENG Fan-cheng, XUE Tian-yan, LIU Ya-hui, ZHANG Guo-zhi, QI Tao. Recovery of titanium from undissolved residue (tionite) in titanium oxide industry via NaOH hydrothermal conversion and H2SO4 leaching [J]. Transactions of Nonferrous Metals Society of China, 2016, 26(6): 1696-1705.
[6] LI Chun, LIANG Bin, WANG Hai-yu. Preparation of synthetic rutile by hydrochloric acid leaching of mechanically activated Panzhihua ilmenite [J]. Hydrometallurgy, 2008, 91(1-4): 121-129.
[7] WU Fei-xiang, LI Xin-hai, WANG Zhi-xing, WU Ling, GUO Hua-jun, XIONG Xun-hui, ZHANG Xiao-ping, WANG Xiao-juan. Hydrogen peroxide leaching of hydrolyzed titania residue prepared from mechanically activated Panzhihua ilmenite leached by hydrochloric acid [J]. International Journal of Mineral Processing, 2011, 98(1-2): 106-112.
[8] OSTROVSKI O, ZHANG G, KONONOV R, DEWAN M A R, LI J. Carbothermal solid state reduction of stable metal oxides [J]. Steel Research International, 2010, 81(10): 841-846.
[9] GOU Hai-peng, ZHANG Guo-hua, HU Xiao-jun, CHOU Kuo-chih. Kinetic study on carbothermic reduction of ilmenite with activated carbon [J]. Transactions of Nonferrous Metals Society of China, 2017, 27(8): 1856-1861.
[10] GOU Hai-peng, ZHANG Guo-hua, YUAN Xin, CHOU Kuo-chih. Formation of titanium carbonitride via carbothermic reduction of ilmenite concentrate in nitrogen atmosphere [J]. ISIJ International, 2016, 56(5): 744-751.
[11] LU Chang-yuan, ZOU Xing-li, LU Xiong-gang, XIE Xue-liang, ZHENG Kai, XIAO Wei, CHENG Hong-wei, LI Guang-shi. Reductive kinetics of Panzhihua ilmenite with hydrogen [J]. Transactions of Nonferrous Metals Society of China, 2016, 26(12): 3266-3273.
[12] PERREAULT P, PATIENCE G S. Ilmenite�CCO reduction kinetics [J]. Fuel, 2016, 165: 166-172.
[13] LIU Shui-shi, GUO Yu-feng, QIU Guan-zhou, JIANG Tao, CHEN feng. Solid-state reduction kinetics and mechanism of pre-oxidized vanadium-titanium magnetite concentrate [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(10): 3372-3377.
[14] KUCUKKARAGOZ C S, ERIC R H. Solid state reduction of a natural ilmenite [J]. Minerals Engineering, 2006, 19(3): 334-337.
[15] WELHAM N J, WILLIAMS J S. Carbothermic reduction of llmenite (FeTiO3) and rutile (TiO2) [J]. Metallurgical and Materials Transactions B, 1999, 30(6): 1075-1081.
[16] GOU Hai-peng, ZHANG Guo-hua, CHOU Kuo-chih. Influence of pre-oxidation on carbothermic reduction process of ilmenite concentrate [J]. ISIJ International, 2015, 55(5): 928-933.
[17] GUPTA S K, RAJAKUMAR V, GRIEVESON P. The influence of weathering on the reduction of llmenite with carbon [J]. Metallurgical Transactions B, 1989, 20(5): 735-745.
[18] LI Wei, PENG Jin-hui, GUO Sheng-hui, ZHANG Li-bo, CHEN Guo, XIA Hong-ying, LIU Bing-guo. Carbothermic reduction kinetics of ilmenite concentrates catalyzed by sodium chloride and microwave-absorbing characteristics of reductive products [J]. Mining, Metallurgy & Exploration, 2013, 30(2): 108-116.
[19] CHEN Min, TANG Ai-tao, XIAO Xuan. Effect of milling time on carbothermic reduction of ilmenite [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(12): 4201-4206.
[20] LI Qian, PAN Yan-biao, LENG Hai-yan, CHOU Kuo-chih. Structures and properties of Mg�CLa�CNi ternary hydrogen storage alloys by microwave-assisted activation synthesis [J]. International Journal of Hydrogen Energy, 2014, 39(26): 14247-14254.
[21] IBRAHIM M K, HAMZAH E, SAUD S N, NAZIM E M, BAHADOR A. Parameter optimization of microwave sintering porous Ti-23%Nb shape memory alloys for biomedical applications [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(4): 700-710.
[22] DUAN Bo-hua, ZHANG Zhao, WANG De-zhi, ZHOU Tao. Microwave sintering of Mo nanopowder and its densification behavior [J]. Transactions of Nonferrous Metals Society of China, 2019, 29(8): 1705-1713.
[23] LI Yan, CHEN Li-li, ZHAO Fang-xian. Highly selective acetone sensor based on ternary Au/Fe2O3-ZnO synthesized via co-precipitation and microwave irradiation [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(1): 137-144.
[24] LIU Chen-hui, ZHANG Li-bo, PENG Jin-hui, LIU Bing-guo, XIA Hong-ying, GU Xiao-chun, SHI Yi-feng. Effect of temperature on dielectric property and microwave heating behavior of low grade Panzhihua ilmenite ore [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(11): 3462-3469.
[25] HUANG Meng-yang, PENG Jin-hui, LEI Ying, HUANG Ming, ZHANG Shi-min. The temperature rise behavior and microwave-absorbing characteristics of ilmenite concentrate in microwave field [J]. Journal of Sichuan University (Engineering Science Edition), 2007, 39(2): 111-115. (in Chinese)
[26] HUANG Meng-yang, PENG Jin-hui, HUANG Ming, ZHANG Shi-min, LI Min, LEI Ying. Microwave-absorbing characteristics of mixtures about different proportions of carbonaceous reducer and ilmenite in microwave field [J]. The Chinese Journal of Nonferrous Metals, 2007, 17(3): 476-480. (in Chinese)
[27] WANG Xin-ying, LI Wei, ZHANG Li-bo, PENG Jin-hui. Microwave-absorbing characteristics and XRD characterization of magnetic separation products of reductive products of ilmenite concentrate [J]. Minerals, 2016, 6(4): 99.
[28] LI Wei, PENG Jin-hui, GUO Sheng-hui, ZHANG Li-bo, CHEN Guo, XIA Hong-ying. Carbothermic reduction kinetics of ilmenite concentrates catalyzed by sodium silicate and microwave-absorbing characteristics of reductive products [J]. Chemical Industry and Chemical Engineering Quarterly, 2013, 19(3): 423-433.
[29] KELLY R M, ROWSON N A. Microwave reduction of oxidised ilmenite concentrates [J]. Minerals Engineering, 1995, 8(11): 1427-1438.
[30] EL-TAWIL S Z, MORSI I M, YEHIA A, FRANCIS A A. Alkali reductive roasting of ilmenite ore [J]. Canadian Metallurgical Quarterly, 1996, 35(1): 31-37.
[31] LI Xiao-ming, WEN Zhen-yu, LI Yi, YANG Hai-bo, XING Xiang-dong. Improvement of carbothermic reduction of nickel slag by addition of CaCO3 [J]. Transactions of Nonferrous Metals Society of China, 2019, 29(12): 2658-2666.
[32] GIDDEY S, BADWAL S P S, KULKARNI A, MUNNINGS C. A comprehensive review of direct carbon fuel cell technology [J]. Progress in Energy and Combustion Science, 2012, 38(3): 360-399.
[33] LI Shuang-bin, JIANG Cai-rong, LIU Juan, TAO Hao-liang, MENG Xie, CONNOR P, HUI Jia-ning, WANG Shao-rong, MA Jian-jun, IRVINE J T S. Mechanism of enhanced performance on a hybrid direct carbon fuel cell using sawdust biofuels [J]. Journal of Power Sources, 2018, 383: 10-16.
��֮��1,2,3��Ф����1,2,3���亣��1,2,3���ܹ���1,2,3
1. �Ϻ���ѧ ���Ͽ�ѧ�빤��ѧԺ���Ϻ� 200072��
2. �Ϻ���ѧ ʡ��������Ʒ�������ұ�����Ʊ������ص�ʵ���ң��Ϻ� 200072��
3. �Ϻ���ѧ �Ϻ��и���ұ���¼�������Ӧ���ص�ʵ���ң��Ϻ� 200072
ժ Ҫ�����һ�ֻ���ֱ��̼�Ȼ�ԭ�������ȵ���������ɫ��Ч�����¹��ա�ͨ�����Ӹ�����Na2CO3 ���ٻ�ԭ��Ӧ�Ľ��У�����Na2CO3����ѭ�����á���Ӧԭ��(������+ʯī+Na2CO3)�������¼�����1073~1123 K������20 min����Ҫ��ԭ����ΪNa2TiO3�ͽ��������������Ľ������ʿ��Դﵽ92.67%~93.21%����ԭ�������ξ���ˮϴ��CO2��������ĥ�ʹ�ѡ���룬���տ��Եõ������Ϻ����ϡ����и�������TiO2�������Դﵽ90.04% (��������)�����ɺ�����������TiCl4��TiO2�����ڽ������ʡ���ѡ����Ч�ʺͲ���������ԵȲ������Ż��õ��Ļ�ԭ�¶�Ϊ1123 K�����⣬���������Na2CO3����������������µĻ�ԭ������
�ؼ��ʣ�������������Na2CO3��̼�Ȼ�ԭ
(Edited by Bing YANG)
Corresponding author: Hai-yan LENG, Tel: +86-21-56338065, E-mail: lenghaiyan@shu.edu.cn
DOI: 10.1016/S1003-6326(21)65619-4
1003-6326/ 2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press
2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press

