Activity coefficient calculation model for NaAl(OH)4-NaOH-H2O system
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2005���4��
�������ߣ���С�� ������ ���� ��־�� ������ ��ܿ ������
����ҳ�룺908 - 912
Key words��Debye-H��ckel equation; activity coefficient; calculation model
Abstract: Resorting to Debye-H��ckel equation, the conception of the apparent dielectric constant �š� of sodium aluminate solution is introduced. By supposing that all the influences are attributed to it, �š� is successfully related to caustic ratio ��K, concentration mNaOH, T and temperature T. Then an activity coefficient calculation model for NaAl(OH)4-NaOH-H2O system from 25�� to 100�� is established, which can be used in much wider ��K and mNaOH, T ranges than those covered by the equilibrium solubility data.
LI Xiao-bin(��С��), L�� Wei-jun(������),LIU Gui-hua(����), PENG Zhi-hong(��־��),
ZHOU Qiu-sheng(������), MENG Yun(�� ܿ), REN Wan-neng(������)
(School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China)
Abstract: Resorting to Debye-H��ckel equation, the conception of the apparent dielectric constant �š� of sodium aluminate solution is introduced. By supposing that all the influences are attributed to it, �š� is successfully related to caustic ratio ��K, concentration mNaOH, T and temperature T. Then an activity coefficient calculation model for NaAl(OH)4-NaOH-H2O system from 25�� to 100�� is established, which can be used in much wider ��K and mNaOH, T ranges than those covered by the equilibrium solubility data.
Key words: Debye-H��ckel equation; activity coefficient; calculation model CLC number: TF801
Document code: A
1 INTRODUCTION
Concentrations of the sodium aluminate solutions in alumina industry are usually very high, and the diverges of precipitation mechanism and structure in sodium aluminate solution[1-4] also exist, which makes the study of activity coefficients extremely difficult. Therefore, though lots of work has been done on the thermodynamic properties of sodium aluminate solution[5-7] and many solubility data have been obtained, the researches of the activity coefficient are scarce. As Debye-H��ckel activity coefficients calculation equation has a simple formula and definite meanings of various physical parameters, it is convenient to be used in practical calculation. Meanwhile, it is also suitable for calculating the activity coefficients of NaAl(OH)4 in NaAl(OH)4-NaOH-H2O system at equilibrium[8].
In this paper, the idiographic issues of using Debye-H��ckel model in NaAl(OH)4-NaOH-H2O system are analyzed, and then the resolvents to break the ranges of equilibrium solubility data and to guide the establishment of activity coefficients calculation model are brought forward.
2 PRIMARY ISSUES AND CORRESPONDING RESOLVENTS
In order to discuss conveniently, the classical Debye-H��ckel model[9] is listed as follows:
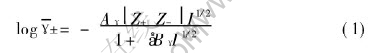
where
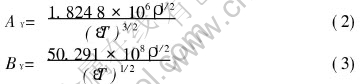
In Eqn.(1), I stands for the effective ionic intensity of the solution in unit of mol/kg; Z+ and Z- denote the charge of the cations and anions, respectively; �z refers to the ion size parameter and �zNaAl(OH)4 is 3.595�@[8]; A��, B�� represent the Debye-H��ckel parameters in units of (kg/mol)1/2 and kg1/2/(mol1/2��cm), respectively; T means temperature in K; and ��, �� stand for the dielectric constant and density(g/cm3) of H2O when H2O is supposed to own a continuous phase.
Debye-H��ckel model employs four basic hypotheses, which contributes the huge interaction among ions to the interaction between centric ions and ion spheres to make the model less complex. The discussion of the applicability of Debye-H��ckel model in NaAl(OH)4-NaOH-H2O system also proves that this model is really suitable for the system. For NaAl(OH)4-NaOH-H2O system, the primary issues and corresponding resolvents are as follows.
1) In Debye-H��ckel model, the key parameter to determine the values of activity coefficients is ionic intensity I. Supposing the aluminate ions existed in NaAl(OH)4-NaOH-H2O system are only Al(OH)-4, there is only one kind of cations. When total caustic concentration mNaOH, T(mol/kg) is known, the ionic intensity of the whole solution is decided. But obviously, the activity coefficients are various at different caustic ratios ��K(Na2O/Al2O3, molar ratio) even mNaOH, T is certain. So the problem of the influence of ��K on activity coefficients can not be settled only with the parameter I. In Debye-H��ckel model, �� is another key parameter to determine the interaction among ions. Because the water is supposed to own a continuous phase in the case of dilute solution, the original definition of �� is the dielectric constant of water. In this paper, the conception of the apparent dielectric constant �š� of sodium aluminate solution is introduced and all the influences are attributed to �š�. According to this, the ultimate calculation model, which provides the calculation method of the dielectric constant �š� relating to ��K, mNaOH, T(mol/kg), T(K), is given.
2) The data used in this model are equilibrium solubility data, which causes the applicable regions of ��K and concentration limited. Resorting to the comparison of the activity data with other references, the possible correlative laws can be explored and the activity data can be extended to a broader range of ��K, mNaOH, T(mol/kg) . Then the calculation of activity coefficients of NaAl(OH)4-NaOH-H2O system in non-equilibrium conditions could be sought for.
3 ESTABLISHMENT OF CALCULATION MODEL
3.1 Relationship between ��NaAl(OH)4 and ��K, mNaOH, T, T at equilibrium
According to a large amount of studies on the sodium aluminate solution, the most possible structure of aluminate ions in sodium aluminate solution is tetrahedral Al(OH)-4[10, 11].In this paper, supposing all the aluminate ions are Al(OH)-4, the dissolution of gibbsite in NaOH solution can be defined as
Al(OH)3+NaOH��NaAl(OH)4(4)
The equilibrium constant K of Eqn.(4) can be expressed as
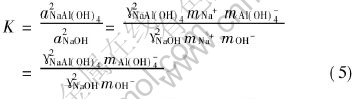
where aNaAl(OH)4, aNaOH, ��NaAl(OH)4, ��NaOH represent the activities and the activity coefficients of the subscripted species. mNa+, mAl(OH)-4, mOH- refer to the stoichiometric molar concentrations of the subscripted species in unit of mol/kg, respectively.
From Eqn.(5),
��NaAl(OH)4=(KmOH-/mAl(OH)-4)1/2��NaOH(6)
In order to know the influence of ��K, mNaOH, T(mol/kg) and T(K) on the activity coefficients in sodium aluminate solution, we ought to know the values of K and ��NaOH in Eqn.(6), as well as the corresponding relationship of mOH-, mAl(OH)-4 and ��K, mNaOH, T (mol/kg).
The values of K can be obtained by the following empirical equation reported by Wesolowski[12]:
logK=-96.5506+2374.88/T+16.07899lnT-0.0139828T(7)
For ��NaOH, the researches of activity coefficients of NaOH solutions have been presented completely[7, 12] at present and the satisfied results can be achieved either by extended Debye-H��ckel equation or by Pitzer equation. In this paper, the values of ��NaOH are calculated by Pitzer equation and parameters[13]:
ln��NaOH=-A��[m1/2/(1+bm1/2)+(2/b)��ln(1+bm1/2)]+2m��(0)NaOH+
(2��(1)NaOH/��2)[1-(1+��m1/2-��2m/2)��exp(-��m1/2)]+3m2C��NaOH/2(8)
In sodium aluminate solution, we have
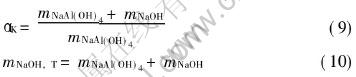
In order to discuss the relationship between the activity coefficients of sodium aluminate solution and temperature, caustic ratio ��K , total caustic concentration mNaOH, T, the corresponding function can be obtained from Eqn.(6) and solubility data[12, 14]:
��NaAl(OH)4=p1+p2��mNaOH, T+p3��m2NaOH, T+
p4����K+p5����2K+p6��ln(mNaOH, T)+p7��ln��K(11)
The corresponding parameters of Eqn.(11) at different temperatures can be referred to Ref.[8].
3.2 Extension of ��NaAl(OH)4 laws
Theoretically, the values of ��NaAl(OH)4 under equilibrium condition can be calculated by Eqn.(11). The calculation results also show that Eqn.(11) fits well with those of references in the range of mNaOH, T and ��K covered by the solubility data, while there are some deviations beyond the range. That is to say, Eqn.(11) can only be used in the temperature, ��K, mNaOH, T ranges of the solubility data. Among the equilibrium solubility data, the high temperature refers only to low ��K and vice versa, which means that it is difficult to calculate activity coefficients by Eqn.(11) under wide ��K range in which the alumina production requires. Therefore, the extension principle of ��NaAl(OH)4must be investigated in order to extrapolate the adaptability range.
There is no systematic research on the activity coefficients of this system, and the data are scarce. Only the data of ��NaAl(OH)4 at 40�� dealing with Pizer equation are comparatively systematic[15], so we take them for comparison. Meanwhile, considering the solubility data adopted in this paper, there are only two points of temperature: 100��(mNaOH, T=0.5-11mol/kg, ��K=1.8-2.4) and 70��(mNaOH, T=0.01-11mol/kg, ��K=1.8-4.5), which have a wider range of mNaOH, T and their ��K values are similar to those of Ref.[15]. So the data calculated by Eqn.(11) in 100��, ��K=1.5 and 70��, ��K=3 are compared with the data[12] calculated by Pizer model at 40�� and corresponding values of ��K. The results are shown in Fig.1.
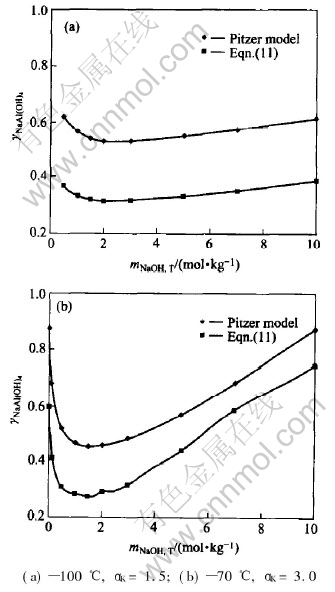
Fig.1 Comparison of ��NaAl(OH)4 calculated by Pitzer model[12] and by Eqn.(11)
Fig.1 shows that the change of ��NaAl(OH)4 with mNaOH, T is almost with the same trend and range when ��K is identical, which is so called the parallel law. The calculation results also show that in the range of mNaOH, T and ��K at any other temperature covered by solubility data, the similar parallel law is also maintained. Furthermore, the values of ��NaAl(OH)4 reduce 0.04 per 10�� with temperature increasing at the same mNaOH, T and ��K. In order to calculate the activity coefficients of the solution beyond the equilibrium solubility data, we ought to calculate the activity coefficients at other temperatures with the same ��K and concentration by Eqn.(11), and then utilize the law to get corresponding activity coefficients.
Combining the range of solubility data, the applicable range of the laws mentioned above is: T=298-373K, mNaOH, T=0.1-10mol/kg, ��K=1.2-15, which can meet the practical needs of alumina production.
3.3 Calculation model of ��NaAl(OH)4
According to the above discussion, the activity coefficients of broader sodium aluminate solution range can be calculated. But the above calculation has no definite physical meanings and is difficult to be testified. What��s more, the process is complicated and is hard to be taken into practical use. Therefore, supposing NaAl(OH)4 owns a continuous phase, the conception of the apparent dielectric constant �š� of sodium aluminate solution is introduced and all the influence is attributed to �š�. Then the extended ��NaAl(OH)4 data are used to Debye-H��ckel model to calculate the corresponding values of �š�. Accordingly, the activity coefficients can be calculated by a comparable simple formula with definite physical meanings so far as the values of �š� are ascertained.
Using Eqn.(11) and parallel law to calculate the activity coefficients of sodium aluminate solution of different temperatures, mNaOH, T, ��K, and then using Eqns.(1)-(3) to calculate corresponding values of �š�, the empirical fitting equation and corresponding parameters(listed in Table 1) of �š� related to mNaOH, T, ��K at different temperatures can be obtained:

Investigation of �š� under different conditions can obtain the following principles:
1) The values of �š� decrease with the increase of temperature in the case of a certain mNaOH, T and ��K, because the temperature increase leads to molecule reduce of unit volume for the solution expansion.
2) The values of �š� increase with the increase of ��K at a certain mNaOH, T and temperature, which may be explained in the way that the high ��K means the high solute concentration due to the increment of ion OH- with smaller size and decrement of ion Al(OH)-4 with bigger size.
3) The values of �š� increase with the increase of mNaOH, T at a certain temperature and ��K. It is
Table 1 Parameters at different temperatures in Eqn.(12)
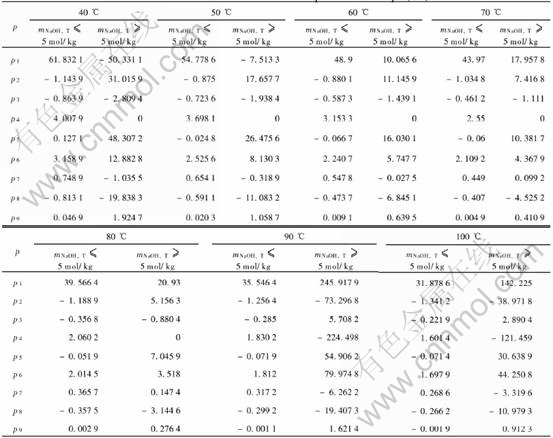

Fig.2 Comparison of equilibrium solubility data calculated by calculation model in this study(��) and reference(��)
maybe caused by the solute concentration increases along with the mNaOH, T.
All the principles mentioned above accord with the dielectric theories, proving the rationality of introducing �š�.
From the work mentioned above, combining parameters of Debye-H��ckel model[7], the activity coefficients calculation model for NaAl(OH)4-NaOH-H2O system is obtained:
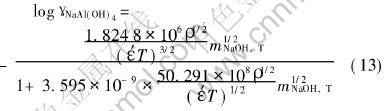
where �š� can be calculated by Eqn.(12).
4 EXAMPLES
Employing the calculation method provided above, the solubility data of Al2O3 in NaOH solution are obtained and compared with those reported by Russell et al[14]. Fig.2 shows the comparison results of solubility curves at 40�� and 70��.
The comparison in Fig.2 indicates that the current results derived from our method are in good agreement with those of literature. When the ionic intensity reaches 6mol/kg, the results are also satisfactory. This strongly supports the outstanding applicability of the method to solutions with high concentration. But the calculated data are a bit less than the literature data in the system with the concentration above 6mol/kg, which may be due to the inaccuracy of the equilibrium solubility data or the tetrahedral Al(OH)-4 ions being not dominative resulted from the structure change of aluminate ions at high concentration.
5 CONCLUSIONS
1) By introducing the conception of the apparent dielectric constant �š� of sodium aluminate solution and attributing all the influences to �š�, the classical Debye-H��ckel equation can be resorted to calculate the activity coefficients of sodium aluminate solution with a wider range of concentration.
2) It is a feasible way to calculate the activity coefficients of solutions basing on the equilibrium solubility data, and it also offers a new idea of the calculation and measurement of activity coefficients for similar systems.
REFERENCES
[1]YANG Z Y. The Technology of Alumina Production [M]. Beijing: Metallurgical Industry Press, 1982. 48-52.(in Chinese)
[2]LI Jie. The Study of the Structures and Precipitation Mechanism in Super Saturated Sodium Aluminate Solution [D]. Changsha: Central South University, 2001. 20-68.(in Chinese)
[3]ter Horst J H, Kramer H J M, van Rosmalen G M, et al. Molecular modeling of the crystallization of polymorphs. Part ��: the morphology of HMX polymorphs [J]. Journal of Crystal Growth, 2002, 237: 2215-2220.
[4]Gerson A R, Ralston J, Smart R S C. An investigation of the mechanism of gibbsite nucleation using molecular modelling [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 1996, 110: 105-117.
[5]App J A, Neil J M, Jun C H. Thermodynamic Properties of Gibbsite, Boehmite, Diaspore and the Aluminate Ion Between 0 and 350�� [R]. Lawrence Berkeley Laboratory Report 21482, 1988.
[6]App J A, Neil J M. Solubilities of aluminum hydroxides and oxyhydroxides in alkaline solution. Chemical Modeling of Aqueous Solutions �� [A]. Melchior D C and Bassett R L. American Chemical Society Symposium Series 416 [C]. Washington DC, 1990. 414-428.
[7]Pokrovskii V A, Helgeson H C. Thermodynamic properties of aqueous species and the solubilities of minerals at high pressures and temperatures: the system Al2O3-H2O-NaCl [J]. American Journal of Science, 1995, 295: 1255-1342.
[8]MENG Yun. The Optimization and Perfection of Thermodynamic Database in Alumina Production [D]. Changsha: Central South University, 2002. 36-46.(in Chinese)
[9]LI Y G. Thermodynamic of Solvent Extraction for Metal [M]. Beijing: Tsinghua University Press, 1985. 44-57.(in Chinese)
[10]Moolenaar R J, Evans J C, Mckeever L D. The structure of the aluminate ion in solutions at high pH [J]. Journal of Physical Chemistry, 1970, 74: 3629-3636.
[11]Barcza L, P��lfalvi-R��zsahegyi M. The aluminate lye as a system of equilibria [J], Materials Chemistry and Physicals, 1989, 21: 345-356.
[12]Wesolowski D J. Aluminum speciation and equilibria in aqueous solution(��): the solubility of gibbsite in the system Na-K-Cl-OH-Al(OH)4 from 0 to 100�� [J]. Geochimica et Cosmochimica Acta, 1992, 56: 1065-109.
[13]Pabalan R T, Pitzer K S. Thermodynamics of NaOH(aq) in hydrothermal solutions [J]. Geochimica et Cosmochimica Acta, 1987, 51: 829-8371.
[14]Russell A S, Edwards J D, Taylor C S. Solubility and density of hydrated aluminas in NaOH solutions [J]. Journal of Metals, 1955, 7: 1123-1128.
[15]ZHOU Jun, CHEN Qi-Yuan, LI Jie et al. Isopiestic determination of the osmotic and activity coefficients for NaOH-NaAl(OH)4-H2O system at 313.2K [J]. Geochimica et Cosmochimica Acta, 2003, 67: 3459-3472.
Foundation item: Project(50274076) supported by the National Natural Science Foundation of China; project(G1999064910) supported by the National Basic Research Program of China
Received date: 2004-10-12; Accepted date:2005-01-31
Correspondence: LI Xiao-bin, Professor, PhD; Tel: +86-731-8830453; E-mail: X.B.Li@mail.csu.edu.cn

