J. Cent. South Univ. Technol. (2008) 15: 484-487
DOI: 10.1007/s11771-008-0091-y
Relationship between initial efficiency and structure parameters of carbon anode material for Li-ion battery
SHEN Jian-bin(申建斌), TANG You-gen(唐有根), LIANG Yi-zeng(梁逸曾), TAN Xin-xin(谭欣欣)
(School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China)
Abstract: The initial efficiency is a very important criterion for carbon anode material of Li-ion battery. The relationship between initial efficiency and structure parameters of carbon anode material of Li-ion battery was investigated by an artificial intelligence approach called Random Forests using D10, D50, D90, BET specific surface area and TP density as inputs, initial efficiency as output. The results give good classification performance with 91% accuracy. The variable importance analysis results show the impact of 5 variables on the initial efficiency descends in the order of D90, TP density, BET specific surface area, D50 and D10; smaller D90 and larger TP density have positive impact on initial efficiency. The contribution of BET specific surface area on classification is only 18.74%, which indicates the shortcoming of BET specific surface area as a widely used parameter for initial efficiency evaluation.
Key words: Li-ion battery; carbon anode material; initial efficiency; structure parameters
1 Introduction
Non-aqueous electrolyte is irreversibly reduced during the first charge process, which forms a thin passive film on the surface of carbon anode of Li-ion battery called solid electrolyte interphase (SEI) or SEI layer[1]. A stable and dense SEI layer can prevent further reaction between electrolyte and carbon anode, while an unstable and loose SEI layer cannot prevent solvation Li+ or organic solutions from passing through, finally leading to the intercalation of solvation Li+ or decomposition of electrolyte[2].
On one side, the initial efficiency can directly reflect the irreversible capacity loss of carbon anode material, and on the other side it can give some information about the SEI layer. Therefore, the initial efficiency is a very important criterion for carbon anode material. The particle distribution of carbon anode material can influence the charge-discharge efficiency of Li-ion battery[3]. Carbon anode material with certain particle distribution has higher discharge capacity. Defects at grain boundary of hexagonal and rhombic structure can hinder the intercalation of solvation Li+ and then influence the initial efficiency[4]. However, the reduction of solvents easily takes place on the defects at the grain boundary. No conclusion has been drawn which process would be dominant. And the number of fine grain edge has a great impact on the irreversible capacity of natural graphite, as stated by ZAGHIB[5]. The particle size and BET specific surface area should not be considered important parameters for initial efficiency evaluation. But their models are based on the assumption of flaky graphite which has apparent limitation. Also the initial efficiency of natural graphite is increased by surface modification which can reduce the first irreversible capacity loss[6]. To a great extent, the structure, the particle size and the BET specific surface area of carbon anode material can determine its initial efficiency in a certain electrolyte.
Little work has been done on the relationship between initial efficiency and structure parameters for anode carbon material. It is of importance to predict the initial efficiency of carbon anode material by its physical parameters. The purpose of present work is to classify different carbon anode materials using physical parameters such as D10, D50, D90, BET specific surface area and TP density as inputs while initial efficiency as output. The definition of D10 is equivalent diameter where 10% (mass of the particles) of the powder has a smaller diameter (and hence the remaining 90% is coarser); the definition of D50 and D90 can be derived similarly; TP means tap density.
2 Experimental
2.1 Particle size
After ultrasonic dispersion, the particle size of carbon anode material was measured by CILAS 1064 laser particle size analyzer at 25 ℃ using water as dispersion medium.
2.2 BET specific surface area
BET specific surface area of carbon anode material was measured by ASAP fast BET analyzer at 77.2 K using N2 as absorption medium.
2.3 TP density
TP density of carbon anode material was measured by PF-100 B intelligent analyzer at frequency of 150 times per minute and amplitude of 3 mm.
2.4 Coin cell
Electrodes were made by coating the mixture of 84%(mass fraction) carbon anode material, 8% PVDF binder and 8% super carbon black on 0.015 mm thick copper foil. Films were heated under vacuum circum- stances at 100 ℃ for 4 h. They were poured into a stainless steel pellet and a pressure of 3 kPa was applied to make a 1 mm thick electrode pellet (diameter=14 mm). Working electrode was prepared by heating pellet under vacuum circumstances at 80 ℃ for 2 h. Lithium foil was used as counter-electrode and Celgard 2400 was used as separator. Electrolyte was 1 mol/L LiPF6/EC+ DMC+EMC(volume ratio, 1?1?1). The coin cell was prepared in the argon-filled glove box. The charge and discharge tests were performed on a CHI660A electrochemical workstation in voltage range of 0-2.5 V. Totally 44 samples were tested. A 44×6 matrix was constructed by the carbon anode material parameters and their initial efficiency. Those carbon anode materials with initial efficiency higher than 90% were labeled with class 1 while the others were labeled with class 0, as illustrated in Table 1. In Table 1, efficiency is the ratio of discharge capacity to charge capacity.
3 Random Forests
The classification was performed by Random Forests (RF) on a PC (Pentium4 1.4 GHz, 512M RAM). Essentially, Random Forests belongs to decision tree. Fig.1 shows a simple example of decision tree[7]. The circle represents internal node labeled with variable and threshold. The peak represents root, while the bottom represents the leave node of tree, which gives the class. Supposing that a classification is performed on samples (5, 4, 6, 2, 2, 3), the process is going on along with the right sub-tree because x6 is larger than threshold 2. Then the process is going on along with the left sub-tree because x5 is smaller than threshold 5. The sample is labeled as class 3 since node containing threshold 5 directly goes to the end node containing class 3. Fig.1 demonstrates procedure of dividing a complicated decision process to a series of relatively simple node decisions, which has the advantage of using a relatively clear rule for classification. However, it is difficult to build an optimized decision tree and the poor generalization capability is in the most cases.
Table 1 Carbon material properties vs initial efficiency class
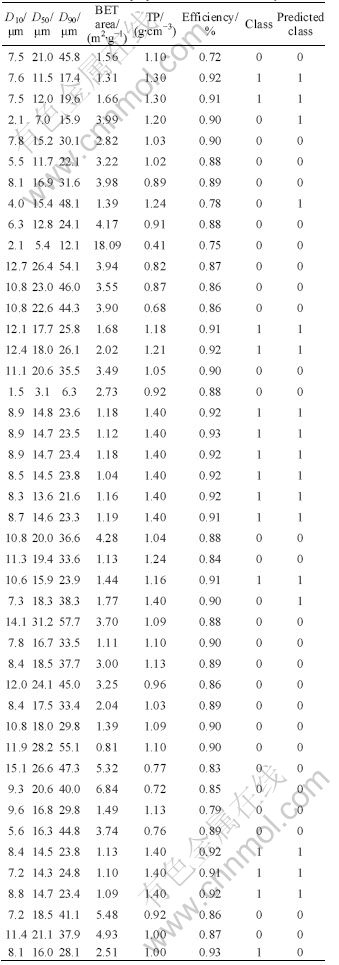
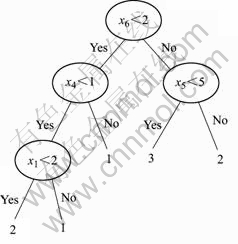
Fig.1 An example of decision tree
Bootstrap is a method for modeling by selecting a data set with n cases from the original data set with N cases for M times and building M data sets independently. Random Forests is a method using bootstrap to produce multi-decision trees for classification. Each node is split using the best in a subset of predictors randomly chosen at that node. The number of selected predictors is usually the square root of total variable numbers. The classification result is obtained by voting result of each classifier. The projection is conducted by projecting the matrix constructed by the proximities between cases to a lower dimensional space with the same distance proximities as those in the original space[8]. Random Forests has the advantages of fast operation, no pre- selection, importance estimation and robustness. So it is widely used in various fields including chemistry[9-11]. Here we used RF for classification.
4 Results and discussion
In this work, the number of sub-tree is 500 and the number of predictors randomly chosen at internal node is 2. The regression results show that 40 of 44 cases have been correctly classified (91% accuracy). Matrix constructed by the proximities between cases has been projected on a 3-dimension space with the same proximities as those in the original space to understand well the structure of each class as shown in Fig.2.
It is clear from Fig.2 that there is obvious boundary between two classes in the 3-dimension space (The black for class 0 and white for class 1). 4 cases with white color among the black are fall into the wrong classes and they are on the edge of the separation plane. Fig.3 was obtained by deleting the four outliers.
Fig.2 Data projection in 3-dimension space
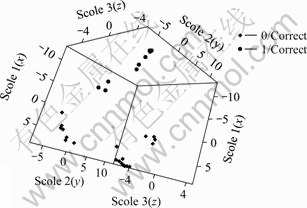
Fig.3 Data projection in 3-dimension space without incorrect sample
The separation plane is very clear, indicating that the classification via 5 parameters is effective.
The boundary between two classes is still very clear in the 2-dimension space, as shown in Fig.4.
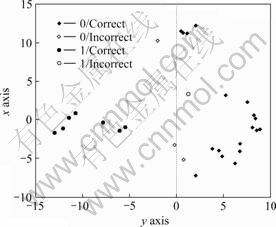
Fig.4 Data projection in 2-dimension space
One of the most important characteristics of RF lies in its variable importance analysis which summarizes the contribution of each variable on the classification. The result of variable importance analysis in Table 2 demonstrates that D90 and TP density contribute 65% to the classification, which indicates the distribution of big particle and TP density have a great impact on the initial efficiency. To some degree, bigger particle means slower solid diffusion[12] that would decrease initial efficiency, while higher TP density means smaller porosity for material and poorer electrolyte wetting ability that would increase initial efficiency. On the contrary, BET specific surface area has small contribution (18.74%) on the classification, which indicates BET specific surface area cannot represent the active reaction area during the first cycle. This result accords with that in Ref.[5]. However, from the original data set, the trends of initial efficiency decreasing with increasing BET specific surface area can be observed. And usually, smaller particle size means bigger BET specific surface area, so D50 is an important parameter for the initial efficiency[13]. Clearly, a compromise in the particle size and surface area as well as other property of carbon anode material is needed by process control to obtain the higher initial efficiency[14-15].
Table 2 Importance of variables

5 Conclusions
1) Regression is performed by Random Forests using D10, D50, D90, BET specific surface area and TP density of carbon anode material as inputs, and initial efficiency of carbon anode material as output. 91% accuracy is achieved and a clear boundary between classes can be observed in a low dimension space, which proves the effectiveness of regression method.
2) Further variable importance analysis shows D90 and TP density have great contributions on the classification, while BET specific surface area has a small contribution.
3) Based on the conclusion above, it is able to build a database of relationship between initial efficiency and structure parameters for carbon anode material. Also carbon anode material with high initial efficiency could be developed by analyzing the impact factor of historical data.
References
[1] PELED E. The electrochemical behavior of alkali and alkaline earth metals in nonaqueous battery systems: The solid electrolyte interphase model[J]. Journal of the Electrochemical Society, 1979, 126(12): 2047-2051.
[2] WU Yu-ping, DAI Xiao-bin, MA Jun-qi, CHEN Yu-jiang. Li-ion battery―Application and practice[M]. Beijing: Chemical Industry Press, 2004. (in Chinese)
[3] YUICHI S, TAKESHI N, KOICHI K, TAKANOBU K, AKIRA Y. Particle-size effect of carbon powders on the discharge capacity of lithium ion batteries[J]. Journal of Power Sources, 1998, 75(2): 271-277.
[4] KATIA G, ANNIE F, SERGE F, MICHEL C, BERNARD S, PHILIPPE B. Effect of graphite crystal structure on lithium electrochemical intercalation[J]. Journal of Electrochemical Society, 1999, 146(10): 3660-3665.
[5] ZAGHIB K, NADEAU G, KINOSHITAB K. Effect of graphite particle size on irreversible capacity loss[J]. Journal of Electrochemical Society, 2000, 147(6): 2110-2115.
[6] ZHOU You-yuan, LI Xin-hai, GUO Hua-jun, WANG Zhi-xing, YANG Yong, XIE Qiao-ling. Modification of natural graphite using pitch through dynamical melt-carbonization[J]. Journal of Central South University of Technology, 2007, 14(5): 651-655.
[7] ANDREW R W. Statistical pattern recognition[M]. 2nd ed. WANG Ping, YANG Pei-long, LUO Yong-xin. Beijing: Publishing House of Electronic Industry, 2004: 178-182.
[8] BREIMAN L. Random Forests[J]. Machine Learning, 2001, 45(1): 5-32.
[9] RAJARSHI G, PETER C J. Development of linear, ensemble, and nonlinear models for the prediction and interpretation of the biological activity of a set of PDGFR Inhibitors[J]. J Chem Inf Comput Sci, 2004, 44(6): 2179-2189.
[10] LI S Q, FEDOROWICZ A, SINGH H, SODERHOLM S C. Application of the Random Forest method in studies of local lymph node assay based skin sensitization data[J]. J Chem Inf Model, 2005, 45(4): 952-964.
[11] SVETNIK V, WANG T, TONG C, ANDY L, ROBERT P S, SONG Q H. Boosting: An ensemble learning tool for compound classification and QSAR modeling[J]. J Chem Inf Model, 2005, 45(4): 786-799.
[12] NOGARAJAN G S, VAN Z J W, SPOTNITZ R M. A mathematical model for intercalation electrode behavior I: Effect of particle-size distribution on discharge capacity[J]. Journal of Electrochemical Society, 1998, 145(3): 771-779.
[13] ZAGHIB K, SONG X, GUERFI A, KOSTECHKI R, KINOSHIT K. Effect of particle morphology on lithium intercalation rates in natural graphite[J]. Journal of Power Sources, 2003, 124(2): 505-512.
[14] YANG Yong, PENG Wen-jie, GUO Hua-jun, WANG Zhi-xing, LI Xin-hai, ZHOU You-yuan, LIU Yun-jian. Effects of modification on performance of natural graphite coated by SiO2 for anode of lithium ion butteries[J]. Trans Nonferrous Met Soc China, 2007, 17(6): 1339-1342.
[15] L? Chun-ping, ZHAO Xin-bing, CAO Gao-shao, ZHU Tie-jun. Effects of graphite on Zn-Sb alloys as anode materials for lithium ion batteries [J]. Trans Nonferrous Met Soc China, 2000, 10(2): 204-208.
Foundation item: Project(2001AA501433) supported by the National High-Tech Research and Development Program of China
Received date: 2008-01-09; Accepted date: 2008-04-08
Corresponding author: TANG You-gen, Professor; Tel: +86-731-8836618; Fax: +86-731-8830886; E-mail: ygtang@263.net
(Edited by YANG Hua)