
Hydrogen sorption properties of Ti-Zr hydride doped NaAlH4
ZHUANG Peng-hui(庄鹏辉), LIU Xiao-peng(刘晓鹏), LI Zhi-nian(李志念),
WANG Shu-mao(王树茂), JIANG Li-jun(蒋利军), LI Hua-ling(李华玲)
General Research Institute for Nonferrous Metals, Beijing 100088, China
Received 15 July 2007; accepted 10 September 2007
Abstract: The as-prepared Ti-Zr hydride powder is used as dopant to improve hydrogen storage properties of NaAlH4 upon mechanical milling under argon atmosphere. The as-milled sample is investigated by X-ray diffraction(XRD), scanning electron microscopy(SEM) and Sievert’s technology test. It is observed that Ti-Zr hydride doped NaAlH4 discharges 2.7% and 4.0% (mass fraction) of hydrogen in 40 min and 11 h at 160 ℃, respectively, and keeps its reversible dehydrogenation capacity at 4.0% (mass fraction) after 10 hydrogenation/dehydrogenation cycles. These results show the Ti-Zr hydride doped NaAlH4 has good reversible hydrogen storage capacity and kinetics. XRD and SEM investigations also show that the doped Ti-Zr hydride uniformly distributes in NaAlH4 substrate and keeps stable during the hydrogenation/dehydrogenation cycle, indicating that Ti-Zr hydride plays the main surface-catalytic role on improving reversible hydrogen storage properties of NaAlH4.
Key words: hydrogen storage materials; sodium alanates; Ti-Zr hydride dopant
1 Introduction
Sodium alanate, NaAlH4, is a potential hydrogen storage candidate with a hydrogen content of 5.55% (mass fraction). This compound slowly decomposes in a two-step reaction:
NaAlH4→1/3Na3AlH6+2/3Al+H2 3.70% (mass fraction) H2 (1)
Na3AlH6→3NaH+Al+3/2H21.85% (mass fraction) H2 (2)
These reactions take place slowly at 210 ℃ and 250℃, respectively, moreover, they are irreversible. In 1997, Bogdanovic and Schwickardi reported that the elimination of hydrogen from solid NaAlH4 could be markedly accelerated and rendered reversion under moderate conditions of doping hydride with a few molar percent of Ti or/and Zr organ[1]. Following this breakthrough, many intensifying efforts were done to utilize the NaAlH4 as a practical hydrogen storage materials[2-4]. Further investigations showed that doped Ti and Zr compound are useful for increasing kinetics of reactions (1) and (2), respectively[5]. However, the doping process results in the formation of sodium by-product and some gas impurities, degrading the reversible hydrogen storage capacity and thermal cycling stability of NaAlH4[6]. To avoid forming the sodium by-product, WANG et al[7-8] bring the nano-titanium powder into NaAlH4 by mechanically ball-milling to get the Ti hydride powder doped NaAlH4 composite, which exhibits good hydrogenation/dehydrogenation capacity and kinetics .
In order to bring the advantages of Ti and Zr on improving kinetics of reactions (1) and (2) respectively and do not involve the by-products, gas impurities in the doping process, for the first time, the authors selected a Ti-Zr hydride as doping compound to improve hydrogen storage properties of NaAlH4, and used ball milling method to prepare it due to the Ti-Zr hydride is brittle and easy to be crushed into small powder and be uniformly distributed into NaAlH4 substrate. The aim of the present paper is to investigate the hydrogen storage properties of NaAlH4 doped by this novel Ti-Zr hydride, and also to study the catalytic mechanism of Ti-Zr hydride doped NaAlH4.
2 Experimental
Ti-Zr alloy was prepared by magnetic levitation melting under Ar protection, the sample was turned over and remelted for homogeneity. The purity of raw material Ti and Zr is >99.9% (mass fraction). Then alloy was crushed into 1.7 mm pats and sealed into stainless steel reactor. The alloy was vacuumed at 550 ℃ for 0.5 h, and then cooled to room temperature and bring into 3.5 MPa hydrogen to generate Ti-Zr hydride. Lastly, the Ti-Zr hydride and grinding balls with ratio of 1?10 were sealed into stainless steel vial to ball mill for 5 h by SPEX8000 mill. The average particle size of the as-milled powder is less than 5 μm.
The starting material, NaAlH4 (purity of 93%, particle size of 43 μm) purchased from Acros Organics America was used without purification. The initial material, Ti-Zr hydride doped NaAlH4 was prepared by ball milling method. Ti-Zr hydride powder was weighted in the molar ratio of NaAlH4?Ti-Zr hydride=97?3 in an argon filled glove box. The powders were mixed by ball milling under 0.5 MPa Ar in a high-energy SPEX8000 mill for 5 h with ball-to-powder mass ratio of 4?1. After mixing, a sample of about 0.5 g was transferred to the argon atmosphere glove box of a stainless steel reaction vessel for hydrogen storage properties measurement.
Hydrogenation and dehydrogenation behaviors were measured with a Sivert’s type apparatus. Precise pressure measurement and temperature controlling as well were accomplished by using a high-precision pressure transducer and electric oven, respectively. A typical cyclic experiment entailed dehydrogenation at 160 ℃ and hydrogenation at 120 ℃ with an initial hydrogen pressure of 0.1 MPa and 9.5 MPa, respectively. Mass of Ti-Zr hydride was taken into calculation of hydrogen capacity of material.
The phase structure and microstructure of sample were characterized by powder X-ray diffraction(XRD, X-pert Pro, CuKα radiation) and scanning electron microscope (SEM, HITACHIS4800) equipped with an energy dispersive X-ray(EDX) analysis unit.
3 Results and discussion
3.1 Phase structure of sample
Fig.1 shows the XRD patterns for as-milled, dehydrogenated and rehydrogenated Ti-Zr hydride doped NaAlH4, respectively. For further analysis, the XRD pattern for as-milled Ti-Zr hydride is also shown in Fig.1(d). It is seen in Fig.1(a) that the main phase is NaAlH4, that means NaAlH4 almost keeps unchangeable during ball milling process. However, as shown in Fig.1(b), after heating to 160 ℃, the as-milled Ti-Zr hydride doped NaAlH4 begins to decompose into Al, NaH and a little of Na3AlH6, but no trace of NaAlH4 can be found, indicating the reaction(1) of NaAlH4 finished entirely and the reaction(2) is going partly. Subsequently,
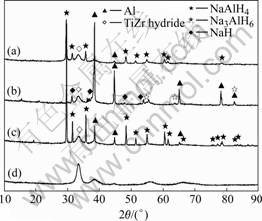
Fig.1 XRD patterns for as-milled state (a), dehydrogenated at 160 ℃, 0.1 MPa for 11 h (b), hydrogenated at 120 ℃, 9.5 MPa for 10 h (c) of 3% (mole fraciton) Ti-Zr hydride doped NaAlH4 and as-milled Ti-Zr hydride (d)
after rehydriding at 120 ℃ and 9.5 MPa, except existence of a little amount of Al, NaAlH4 phases are regenerated, as given in Fig.1(c), indicating a complete restoration of NaAlH4 from Al, NaH by effect of Ti-Zr hydride dopant, namely that the Ti-Zr hydride doped with NaAlH4 has a good reversible hydrogenation/ dehydrogenation properties.
It is seen from Figs.1(a) to (d), the phase structure of Ti-Zr hydride identified by XRD basically keeps unchangeable for the as-milled, dehydrogenated and rehydrogenated samples, respectively.
3.2 Hydrogen storage properties
Fig.2 shows the dehydrogenation kinetics curves for as-milled Ti-Zr hydride doped NaAlH4 at 160 ℃, 0.1 MPa condition. The sample can release about 2.7% and 4.0% (mass fraction) hydrogen in 40 min and 10 h, respectively. The characteristics feature of a two-step dehydrogenation reaction in Ti-NaAlH4 system[9-14] can also be found in the decomposition of Ti-Zr hydride doped NaAlH4 sample. Different decomposition rates of NaAlH4 cause its two different dehydrogenation kinetics region, as illustrated inⅠandⅡregions of Fig.2, which correspond to the reactions (1) and (2), respectively. It can also be found in Fig.3 that the hydrogen decomposition capacity of the sample remains at more 4.0% (mass fraction) after 10 hydrogenation and/or dehydrogenation cycles, indicating that Ti-Zr hydride doped NaAlH4 has a remarkable cycling stability. Differing to the traditional mental Ti[7], TiCl3 or TiH2 dopants[9], the Ti-Zr hydride dopant not only make Ti-Zr hydride doped NaAlH4 composite having good kinetic properties in reaction (1) but also enhancing the kinetic properties of reaction (2). That means the element Zr in Ti-Zr hydride dopant also performs important catalytic effect on improving the dehydrogenation kinetics of NaAlH4.
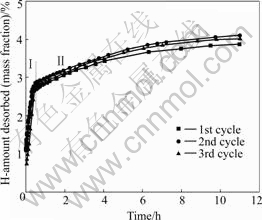
Fig.2 Dehydrogenation kinetics curves for 3% (mole fraction) Ti-Zr hydride doped NaAlH4 at 160 ℃ and 0.1 MPa

Fig.3 First ten cycles of dehydrogenation kinetics curves for 3% (mole fraction) Ti-Zr hydride doped NaAlH4 at 160 ℃ and 0.1 MPa
3.3 Catalyzing mechanism
The nature of the active Ti species that catalyzes the reversible hydrogenation/dehydrogenation of NaAlH4 has been a subject of great interest, speculation, and controversy. Several Ti-containing species, including Al-Ti alloy[15-16], Ti0[1,17] and Ti cation with variable valance[11,18] have been speculated as possible candidate of the catalytically active species. However, the direct and convincing identification of this active species has been greatly hindered by the invisibility of Ti-containing species in conventional analytical the techniques. However, the analysis of XRD patterns from Figs.1(a)-(c) shows that the phase structure of Ti-Zr hydride basically keeps unchangeable at as-milled, dehydrogenated and rehydrogenated samples states, respectively. To further explore the possible role of Ti-Zr hydride on improving the kinetic properties of Ti-Zr hydride doped NaAlH4 composite, the as-milled sample was subjected to SEM and EDS examination. As shown in Fig.4, Ti and Zr are all highly dispersed in as-milled sample matrix. These findings suggest that Ti-Zr hydride should act as surface-localized active species to catalyze the reversible dehydrogenation of NaAlH4. Moreover, the dispersion degree of the Ti-Zr hydride in NaAlH4 may greatly influence its decomposition kinetics. Further investigations on this prospect are underway to check the possibility.

Fig.4 SEM and EDAX maps of Ti, Zr in Ti-Zr hydride doped NaAlH4: (a) Electronic map; (b) Ti Kα1; (c) Zr Lα1
4 Conclusions
1) Mechanical milling of NaAlH4 with the prepared Ti-Zr hydride under argon atmosphere has been found to have good reversible hydrogenation/dehydrogenation properties, the sample releases 2.7% and 4.0% (mass fraction) hydrogen in 40 min and 11 h at 160 ℃, 0.1 MPa, respectively.
2) After 10 hydrogenation/dehydrogenation cycles, the hydrogen decomposition capacity of the sample remains at more 4.0% (mass fraction).
3) The dopant Ti-Zr hydride uniformly distributes in the matrix of NaAlH4 and keeps its phase unchangeable during the hydrogenation/dehydrogenation cycles, indicating that the surface-catalytic effect of the dopant plays the main role in the reversible hydrogen storage properties of NaAlH4.
References
[1] BOGDANOVIC B, SCHWICKARDI M. Ti-doped alkali metal aluminium hydrides as potential novel reversible hydrogen storage material [J]. J Alloys Compd, 1997, 253: 1-9.
[2] GROSS K J, THOMAS G J, JENSEN C M. Catalyzed alanates for hydrogen storage [J]. J Alloys Compd, 2002, 330/332: 683-690.
[3] ANTON D L. Hydrogen desorption kinetics in transition metal modified NaAlH4 [J]. J Alloys Compd, 2003, 356/357: 400-404.
[4] BOGDANOVIC B, FELDERHOFF M, KASKEL S, POMMERIN A, SCHLICHTE K, SCHUTH F. Improved hydrogen storage properties of Ti-doped sodium alanate using titanium nanoparticles as doping agents [J]. Adv Mater, 2003, 15(12): 1012-1015.
[5] ZIDAN R A, TAKARA S, HEE A G., JENSEN C M. Hydrogen cycling behavior of zirconium and titanium-zirconium-doped sodium aluminum hydride [J]. J Alloys Compd, 1999, 285: 119-122.
[6] GROSS K J, MAJZOUB E H, SPANGLER S W. The effects of titanium precursors on hydriding properties of alanates [J]. J Alloys Compd, 2003, 356/357: 423-428.
[7] WANG P, JENSEN C M. Method for preparing Ti-doped NaAlH4 using Ti powder: Observation of an unusual reversible dehydrogenation behavior [J]. J Alloys Compd, 2004, 379: 99-102.
[8] WANG P, KANG X D, CHENG H M. Exploration of the nature of active Ti species in metallic Ti-doped NaAlH4 [J]. Phys Chem B, 2005, 109: 20131-20136.
[9] SRINIVASAN S S, BRINKS H W, HAUBACK B C, SUN D, JENSEN C M. Long term cycling behavior of titanium doped NaAlH4 prepared through solvent mediated milling of NaH and Al with titanium dopant precursors [J]. J Alloys Compd, 2004, 377: 283-289.
[10] KANG X D, WANG P, SONG X P, YAO X D, LU G Q, CHENG H M. Catalytic effect of Al3Ti on the reversible dehydrogenation of NaAlH4 [J]. J Alloys Compd, 2006, 424: 365-369.
[11] SUN D, KIYOBAYASHI T, TAKESHITA H T, KURIYAMAET N, CHENG H M. X-ray diffraction studies of titanium and zirconium doped NaAlH4: Elucidation of doping induced structural changes and their relationship to enhanced hydrogen storage properties [J]. J Alloys Compd, 2002, 337: L8-L11.
[12] SANDROCK G, GROSS K, THOMAS G. Effect of Ti-catalyst content on the reversible hydrogen storage properties of the sodium alanates [J]. J Alloys Compd, 2002, 339: 299-308.
[13] THOMAS G J, GROSS K J, YANG N C Y, JENSEN C M. Microstructural characterization of catalyzed NaAlH4 [J]. J Alloys Compd, 2002, 330/332: 702-707.
[14] GROSS K J, GUTHRIE S, TAKARA S, TOMAS G. In-situ X-ray diffraction study of the decomposition of NaAlH4 [J]. J Alloys Compd, 2000, 297: 270-281.
[15] BOGDANOVIC B, FELDERHOFF M, GERMANN M, HA¨RTEL M, POMMERIN A, SCH?TH F, WEIDENTHALER C, ZIBROWIUS B. Investigation of hydrogen discharging and recharging processes of Ti-doped NaAlH4 by X-ray diffraction analysis (XRD) and solid-state NMR spectroscopy [J]. J Alloys Compd, 2003, 350: 246-255.
[16] BRINKS H W, JENSEN C M, SRINIVASAN S S, HAUBACK B C, BLANCHARD D, MURPHY K. Synchrotron X-ray and neutron diffraction studies of NaAlH4 containing Ti additives [J]. J Alloys Compd, 2004, 376: 215-221.
[17] BOGDANOVIC B, BRAND R A, MARJANOVI?A, SCHWICKARDI M, T?LLE J. Metal-doped sodium aluminium hydrides as potential new hydrogen storage materials [J]. J Alloys Compd, 2000, 302: 236-258.
[18] INIGUEZ J, YILDIRIM T, UDOVIC T J, SULIC M, JENSEN C M. Structure and hydrogen dynamics of pure and Ti-doped sodium alanate [J]. Phys Rev B, 2004, 70: 060101.
(Edited by LAI Hai-hui)
Foundation item: Projects(2006AA05Z131; 2006AA05Z144) supported by the National High-Tech Research and Development Program of China
Corresponding author: LIU Xiao-peng; Tel: +86-10-82241238; E-mail: xpgliu@yahoo.com.cn