A novel, environmentally friendly and facile method for oxidative energy storage in nickel hydroxide film electrodes
来源期刊:中国有色金属学报(英文版)2012年第2期
论文作者:方文斌 练惠勤 王建明 徐蕾 张丽莹 张鉴清 居春山
文章页码:335 - 341
关键词:氧化性能量储存;氢氧化镍膜;氧还原电极;氧化
Key words:oxidative energy storage; nickel hydroxide film; oxygen-reducing cathode; oxidation
摘 要:
采用多种电化学方法研究了一种由氢氧化镍膜电极和氧还原电极组成的电化学电池体系的氧化性能量储存行为。应用扫描电子显微镜(SEM)和X射线衍射(XRD)分析方法表征样品的结构和表面形貌。研究发现,当耦合体系中阴极电解液的氧含量较高或pH值较低时,可以明显改善氧化性能量在氢氧化镍膜电极中的储存。氧化?放电循环测试结果表明,在采用1.0 mol/L Na2SO4 (pH=2)作为阴极电解液的耦合体系中氧化处理600 s后,氢氧化镍膜电极在第1次和第12次循环后的放电容量分别为79.0和97.9 mC/cm2。这表明设计的氧化性能量储存和转换体系具有优异的电化学可逆性。
Abstract:
The oxidative energy storage behaviors of a designed novel system comprising a nickel hydroxide film electrode and an oxygen-reducing platinum cathode were investigated by various electrochemical techniques. The structure and morphology of samples were characterized by scanning electron microscopy (SEM) and X-ray diffraction (XRD) analysis. It is found that the oxidative energy storage in the Ni(OH)2 electrodes can be obviously enhanced in the coupling system containing the cathode electrolytes with higher oxygen content or lower pH value. The results of the oxidation?discharge cycle tests show that the Ni(OH)2 film electrode oxidized in the coupling system with 1.0 mol/L Na2SO4 (pH=2) as cathode electrolyte for 600 s presents discharge capacities of 79.0 mC/cm2 at the first cycle and 97.9 mC/cm2 at the 12th cycle, suggesting the excellent reversibility of the investigated oxidative energy storage and conversion system.

![]()

Trans. Nonferrous Met. Soc. China 22(2012) 335-341
FANG Wen-bin1, LIAN Hui-qin1, WANG Jian-ming1, XU Lei1, ZHANG Li-ying1, ZHANG Jian-qing1, JU Chun-shan2
1. Department of Chemistry, Zhejiang University, Hangzhou 310027, China;
2. Jiangsu Huafu Energy Sources Co. Ltd., Gaoyou 225600, China
Received 11 February 2011; accepted 28 October 2011
Abstract: The oxidative energy storage behaviors of a designed novel system comprising a nickel hydroxide film electrode and an oxygen-reducing platinum cathode were investigated by various electrochemical techniques. The structure and morphology of samples were characterized by scanning electron microscopy (SEM) and X-ray diffraction (XRD) analysis. It is found that the oxidative energy storage in the Ni(OH)2 electrodes can be obviously enhanced in the coupling system containing the cathode electrolytes with higher oxygen content or lower pH value. The results of the oxidation-discharge cycle tests show that the Ni(OH)2 film electrode oxidized in the coupling system with 1.0 mol/L Na2SO4 (pH=2) as cathode electrolyte for 600 s presents discharge capacities of 79.0 mC/cm2 at the first cycle and 97.9 mC/cm2 at the 12th cycle, suggesting the excellent reversibility of the investigated oxidative energy storage and conversion system.
Key words: oxidative energy storage; nickel hydroxide film; oxygen-reducing cathode; oxidation
1 Introduction
The searching for high-efficient, low cost, and environmentally friendly electrochemical energy conversion and storage systems has been attracted a great deal of attention [1-3]. Nickel hydroxide/nickel oxyhydroxide electrodes have aroused increasing attention in decades because of their extensive applications in various fields such as nickel-based alkaline batteries [4, 5], electrochemical capacitors [6], electrocatalysis in organic synthesis [7], sensors [8], and electrochromic devices [9]. Ni(OH)2/NiOOH has been identified as one of the most important redox couples due to its distinctive electrochemical properties [10]. The transition from Ni(OH)2 to NiOOH (oxidation of Ni(OH)2) implies the oxidative energy storage in nickel hydroxide [11, 12]. Generally, the oxidation of Ni(OH)2 may be achieved by an electrochemical charging process [13-15] or a chemical oxidation process [16, 17]. However, these processes need the supply of electrical energy or the oxidizing agents with detrimentally environmental consequences.
Oxygen is a very attractive oxidation agent for oxidative energy storage because of its strong oxidative nature, environmentally benign characteristics and almost unlimited reserves [18]. Nickel hydroxide only exists in alkaline or neutral medium. However, the relatively low oxidative ability of oxygen in the above media results in failure in the oxidation of Ni(OH)2. Although oxygen has much higher oxidative nature in acidic medium, the dissoluble characteristic of Ni(OH)2 limits its direct oxidation using oxygen. To the best of our knowledge, the oxidation of Ni(OH)2 utilizing a naturally green oxidizing agent (oxygen) has not been reported.
In this work, we investigate the oxidative energy storage in Ni(OH)2 films by a designed novel system comprising a nickel hydroxide film electrode and an oxygen-reducing platinum cathode, and the oxidative energy stored in the Ni(OH)2 electrodes is evaluated by galvanostatic discharge tests. The effects of some key factors on the oxidative energy storage are also discussed.
2 Experimental
2.1 Electrode preparation
Nickel hydroxide films were prepared by a cathodic electrodeposition from a 0.02 mol/L nickel nitrate aqueous solution [19]. The substrates used for deposition were an indium-tin oxide (ITO)-coated glass plate or nickel foil with a working area of 1.0 cm2. Prior to use, the ITO substrates were ultrasonically cleaned for 30 min. The nickel foils were first etched in 1.0 mol/L HCl solution for 10 min, and then they were successively rinsed in acetone and deionized water. After cleaning, the substrate was placed opposite a platinum foil counterelectrode in a rectangular acrylic vessel containing 0.02 mol/L nickel nitrate solution. Nickel hydroxide films were deposited by applying a cathodic current density of 1 mA/cm2 for 20 min at room temperature.
2.2 Energy storage device
The energy storage setup consisted of two cells: one for nickel hydroxide electrode and the other for an oxygen-reducing platinum cathode, as shown in Fig. 1. The two electrodes were galvanically coupled through an external circuit, and the two cells were connected by a salt bridge (glass tubing containing saturated KCl agar) to complete the circuit [20]. The anode cell included the Ni(OH)2 electrode, a Hg/HgO reference electrode, and 1.0 mol/L NaOH aqueous electrolyte with 1.0 mol/L NaF. The cathode cell was composed of a platinum foil (2 cm?2 cm) or a Pt-black coated Pt plate (1 cm?1 cm) and various aqueous electrolytes. The Pt-black coated Pt plate cathode was prepared from a Pt plate coated with Pt black by electrodeposition in a H2PtCl6 aqueous solution [21]. The coupling potentials were measured by a potentiostat (EG&G model 273A) controlled by a microcomputer. The coupling currents were recorded by an amperemeter.
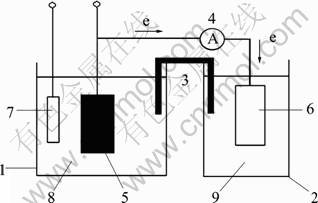
Fig. 1 Schematic diagram of energy storage system: 1―Anode cell; 2―Cathode cell; 3―Salt bridge; 4―Amperemeter; 5―Anode; 6―Cathode; 7―Reference electrode; 8―Anode electrolyte; 9―Cathode electrolyte
2.3 Electrochemical measurements
Electrochemical measurements were performed in a typical three-electrode glass cell. The oxidized nickel hydroxide films were used as the working electrode. The counter electrode was a platinum foil and the reference is a Hg/HgO electrode. The aqueous solution with 1.0 mol/L NaOH and 1.0 mol/L NaF was employed as electrolyte. Galvanostatic discharge tests were performed by a potentiostat (EG&G model 273A).
2.4 Physical characterization
The morphologies of thin film electrodes were examined by a Philips model XL30 scanning electron microscope (SEM). The XRD patterns of the film electrodes were recorded using a Rigaku D/Max 2550 X-ray diffractometer with Cu Ka radiation at 40 kV and 300 mA. A rate of 6 (°)/min over the scan range of 10°-80° (2q) was used to detect the phase structure of the films.
In the above experiments, the reagents were of A.R. grade, and the solutions were prepared by deionized water.
3 Results and discussion
3.1 Effect factors of oxidative energy storage
Figure 2 shows the SEM image of the as-prepared Ni(OH)2 film on ITO substrate. Obviously, the film is nano porous, with particle size of 50-200 nm. A porous structure may provide easy access of the surfaces to liquid electrolytes, thus increasing effective surface area for electrochemical reactions. The nanostructured porous nickel hydroxide film is very important for electrochemical energy storage systems because it has high specific surface area, fast redox reactions, and shortened diffusion path in solid phase [22].

Fig. 2 SEM image of as-prepared Ni(OH)2 film on ITO substrate
The open-circuit potentials of platinum foil in the selected cathode electrolytes are listed in Table 1. Note that the open-circuit potentials of the as-prepared Ni(OH)2 films on ITO in the solution with 1.0 mol/L NaOH and 1.0 mol/L NaF (-0.38 V to -0.25 V vs Hg/HgO) are much lower than those of Pt foil shown in Table 1. It can be seen from Fig. 1 that when the Ni(OH)2 film electrode is connected galvanically to the platinum electrode, the electrons from the Ni(OH)2 film can be transported to the cathode through the external circuit due to the potential difference between the two electrodes, and the Ni(OH)2 electrode and the platinum electrode are anodically and cathodically polarized, respectively. This finally results in the formation of the identical potential for the Ni(OH)2-platinum galvanic coupling system. At the platinum cathode, a reduction reaction occurs to consume the electrons from the external circuit. Simultaneously, the following oxidation reaction appears at the anode [9]:
Ni(OH)2+OH-=NiOOH+H2O+e (1)
Table 1 Open-circuit potentials (φoc) (vs Hg/HgO) of Pt foil in various cathode electrolytesa
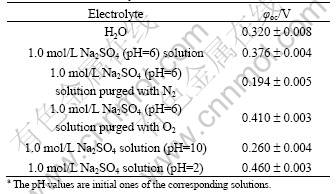
The potential variations of the Ni(OH)2-platinum coupling systems with various cathode electrolytes during the 1.0 h coupling and the galvanostatic discharge curves of the corresponding oxidized Ni(OH)2 film electrodes at a current density of 5.0 mA/cm2 are depicted in Fig. 3. As shown in Fig. 3(a), the potentials of the Ni(OH)2-platinum system first rise sharply, and then increase slowly during the coupling. The coupling potentials of the systems with various cathode electrolytes are in the order: 1.0 mol/L Na2SO4(O2)>1.0 mol/L Na2SO4>H2O>1.0 mol/L Na2SO4 (N2). This is completely consistent with the order in the discharge potentials and discharge capacities of the corresponding oxidized Ni(OH)2 film electrodes (Fig. 3(b)). The Ni(OH)2 film electrode oxidized in the coupling system with 1.0 mol/L Na2SO4 as cathode electrolyte presents a flat discharge plateau at relatively high potentials (250-350 mV) and a large discharge capacity, implying the effective oxidation of Ni(OH)2 [23]. As indicated by the corresponding discharge curves in Fig. 3(b), the oxidation of Ni(OH)2 is notably deteriorated by substituting H2O for 1.0 mol/L Na2SO4 as cathode electrolyte, probably resulting from the very low conductivity of water. The purge of oxygen in the cathode electrolyte (1.0 mol/L Na2SO4) greatly improves the oxidation of Ni(OH)2 by increasing the electrode potential of Pt, which leads to the significant enhancement in discharge performance of the oxidized Ni(OH)2 film electrode. In comparison, the aeration of the cathode electrolyte with nitrogen obviously decreases the potentials of both the platinum electrode (Table 1) and the coupling system, thus almost no charge is stored in the Ni(OH)2 film as indicated by the corresponding discharge curve. The above results suggest that the reduction reaction of oxygen at the platinum electrode has large contribution to the energy storage in the Ni(OH)2 film electrodes.
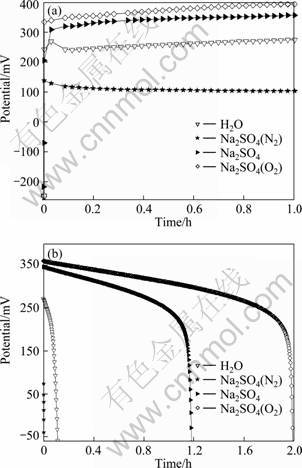
Fig. 3 Potential―time curves of Ni(OH)2-platinum coupling system with Pt foil in various cathode electrolytes (a), discharge curves of oxidized Ni(OH)2 film electrode in various cathode electrolytes at current density of 5.0 mA/cm2 (b) (Ni(OH)2 film electrodes used ITO as substrate)
Figure 4 shows the dependence of the oxidative energy storage on the pH value of the cathode electrolyte in the Ni(OH)2-platinum coupling system. It is noted in Fig. 4(b) that the temporal variations of the coupling currents for various systems have the same trend, that is, the coupling current has a relatively large initial value, followed by a sharp decrease, and then lowers slowly, and finally reaches a stable value. This is generally accordant with the temporal variations of the corresponding coupling potentials (Fig. 4(a)). The potentials of the coupling system with the alkaline Na2SO4 solution (pH=10) as cathode electrolyte during the coupling are below 100 mV (Fig. 4(a)), and no effective oxidation of Ni(OH)2 occurs as indicated by the very low coupling currents (Fig. 4(b)) and the poor discharge performance (Fig. 4(c)). While the high coupling potentials (>300 mV) and considerable large coupling currents of the systems with the acidic Na2SO4 solutions (pH=6 or 2) as cathode electrolyte show that the Ni(OH)2 film may be effectively oxidized, which is confirmed by the corresponding discharge curves in Fig. 4(c). It is noted that the cathode electrolyte with lower pH value may markedly enhance the energy storage in the Ni(OH)2 films, as suggested by higher coupling potentials (Fig. 4(a)), larger coupling currents (Fig. 4(b)) and better discharge performance (Fig. 4(c)).

Fig. 4 Dependences of temporal potential (a) and current (b) variations for Ni(OH)2-platinum coupling system with Pt foil on pH value of cathode electrolytes, and discharge curves of oxidized Ni(OH)2 film electrodes in various cathode electrolytes at current density of 5.0 mA/cm2 (c) (Ni(OH)2 film electrodes used ITO as substrate)
Since the oxidation of Ni(OH)2 is the predominant reaction of the Ni(OH)2 film electrode during the galvanic coupling, the charges transferring from the anode to the cathode equal the charges stored in the anode. It can be calculated from Figs. 4(b) and (c) that the charges stored in the Ni(OH)2 film electrodes in the coupling systems with the pH=2 and pH=6 Na2SO4 cathode electrolytes are 426.4 and 356.2 mC/cm2, respectively, and the discharge efficiencies of the two oxidized Ni(OH)2 electrodes are 19.5% for the former and 6.06% for the latter. The relatively low discharge efficiencies of the oxidized Ni(OH)2 film electrodes could result from high electrode polarization, which may be increased by improving the electrode composition and structure [22].
The photographs of the Ni(OH)2 films on ITO substrate before and after oxidation for 1.0 h in the Ni(OH)2-platinum coupling systems using 1.0 mol/L Na2SO4 solutions with various pH values as cathode electrolyte are depicted in Fig. 5. The as-prepared film displays shallow green, typically characteristic color of Ni(OH)2 [15]. The coupling in the system using the alkaline Na2SO4 solution (pH=10) as cathode electrolyte induces little change in the color of Ni(OH)2 film, suggesting no effective oxidation of Ni(OH)2. In comparison, the colors of the Ni(OH)2 films oxidized in the coupling systems containing the cathode electrolytes with pH=6 and 2 are brown and black, respectively. Considering that the characteristic color of NiOOH is black [11], it can be reasonably concluded that the coupling in the systems with the acidic cathode electrolytes may lead to the effective oxidation of Ni(OH)2 film, and the oxidation effect enhances with decrease in the pH value of cathode electrolyte. This is generally consistent with the results of the preceding electrochemical experiments.
The oxygen reduction reactions at Pt electrode in the acidic or alkaline solutions may be generally expressed in the following equations [23]:
O2+4H++4e=2H2O (pH<7) (2)
O2+2H2O+4e=4OH-(pH>7) (3)
It is well known that the standard electrode potential of the oxygen reduction reaction in the acidic solutions (Eq. (2)) is far higher than that in the alkaline solutions (Eq. (3)). With respect to the coupling system with the alkaline Na2SO4 solution (pH=10) as cathode electrolyte, the open-circuit potential of Pt electrode (Table 1) is much lower than the reversible potential of nickel hydroxide/nickel oxyhydroxide couple [24], thus no effective oxidation of nickel hydroxide occurs during the coupling. The open-circuit potential of Pt in the acidic Na2SO4 solutions (e.g., pH=2 or 6) is higher than the reversible potential of nickel hydroxide/nickel oxyhydroxide couple, resulting in significant oxidation of the Ni(OH)2 film in the corresponding coupling systems. From the view of electrochemical equilibrium, it can be concluded from equation (2) that the lower pH value in the cathode electrolyte may increase the equilibrium potential of the platinum electrode, thereby enhancing the energy storage in the Ni(OH)2 films. On the other hand, it should be also noted from equation (2) that the equilibrium potential of Pt rises with the increase of oxygen content in the cathode electrolyte. This is responsible for enhancement in the energy storage of the Ni(OH)2 film in the coupling system with the cathode electrolyte purged with O2 and the absence in the effective oxidation of the Ni(OH)2 film in the coupling system with the cathode electrolyte purged with N2.

Fig. 5 Photographs of Ni(OH)2 films on ITO substrate before (a) and after oxidation for 1.0 h in Ni(OH)2-platinum coupling system with Pt foil using 1.0 mol/L Na2SO4 solutions with pH=2 (b), pH=6 (c) and pH=10 (d) as cathode electrolyte
3.2 Reversibility of oxidative energy storage and conversion
In order to further investigate the reversibility of the oxidative energy storage and conversion, the oxidation and discharge cycles of the nickel hydroxide film electrodes using nickel foil substrate in the coupling system with the Pt-black coated Pt plate cathode using 1.0 mol/L Na2SO4 (pH=2) solution as cathode electrolyte were tested, and the results are presented in Fig. 6. It can be seen from Fig. 6(a) that the first temporal potential variation of the coupling system with the Ni(OH)2 film electrode on nickel foil is very similar to that for the coupling system with the Ni(OH)2 film electrode on ITO and the acidic (pH=2) Na2SO4 cathode electrolyte (Fig. 4), and the temporal potential variations in the following cycles almost copy the first variation except for much higher initial potentials (>300 mV). Figure 6(b) shows the galvanostatic discharge curves of the corresponding oxidized Ni(OH)2 film electrode at a current density of 100.0 mA/cm2. In the investigated cycles the oxidized Ni(OH)2 film electrode presents higher discharge plateaus and larger discharge capacities. It is noted in Fig. 6(b) that the discharge performance of the oxidized Ni(OH)2 film electrode almost enhances with the increase of cycle number, resulting from its gradual activation [5, 6]. It is calculated from Fig. 6 that the oxidized Ni(OH)2 film electrode on nickel foil presents discharge capacities of 79.0 mC/cm2 at the first cycle and 97.9 mC/cm2 at the 12th cycle, which is comparable to the discharge capacity (83.1 mC/cm2) of the Ni(OH)2 film electrode on ITO oxidized under similar conditions (Fig. 4). Note that the discharge current density (100.0 mA/cm2) of the oxidized Ni(OH)2 film electrodes using nickel foil substrate (Fig. 6) is much larger than that (5.0 mA/cm2) of the corresponding Ni(OH)2 electrode on ITO (Fig. 4) and that the oxidation time (10 min) for the former is much shorter than that (60 min) for the latter. It is reasonable to conclude that the oxidized Ni(OH)2 film electrode using nickel foil substrate has higher charge storage capability and better rate discharge performance. The substitution of the Pt-black coated Pt plate cathode with much larger specific surface area for the platinum foil in the coupling system could be mainly responsible for the markedly enhanced performance of the oxidized Ni(OH)2 film electrode using nickel foil substrate. In addition, the increase in electronic conductivity resulting from the substitution of nickel foil for ITO also has certain contribution to the improved rate discharge capability of the oxidized Ni(OH)2 film electrode.
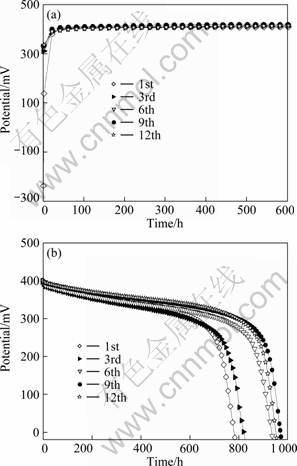
Fig. 6 Potential―time curves of Ni(OH)2-platinum coupling system with Pt-black coated Pt plate cathode using 1.0 mol/L Na2SO4 (pH=2) as cathode electrolyte at various cycles (a) and discharge curves of oxidized Ni(OH)2 film electrode at 100.0 mA/cm2 (b) (Ni(OH)2 film electrodes used nickel foil as substrate)
The XRD patterns of the as-prepared Ni(OH)2 film on nickel foil and the corresponding film oxidized for 10 min in the coupling system with 1.0 mol/L Na2SO4 (pH=2) as cathode electrolyte are illustrated in Figs. 7 and 8, respectively. As shown in Fig. 7, the two peaks at 2q=22° and 34° can be indexed to the a-Ni(OH)2 phase according to JCPDS card No. 38―715, and the peaks at 2q=44°, 52° and 77° result from metallic nickel substrate according to JCPDS card No. 04―850, indicating that the as-prepared Ni(OH)2 film has the alpha phase structure. The two peaks at 2q=12° and 38° in Fig. 8 may be indexed to the (003) and (102) planes of g-NiOOH in terms of JCPDS card No. 06―075, respectively. This suggests that a-Ni(OH)2 is partially transferred to g-NiOOH by the oxidation in the galvanic coupling system, consistent with the results of electrochemical tests in Fig. 6.

Fig. 7 XRD pattern of as-prepared Ni(OH)2 film on nickel foil
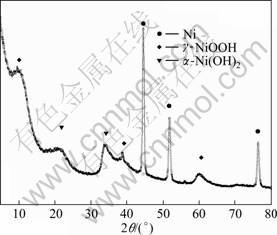
Fig. 8 XRD pattern of Ni(OH)2 film on nickel foil oxidized for 10 min in coupling system with Pt-black coated Pt plate cathode using 1.0 mol/L Na2SO4 (pH=2) as cathode electrolyte
4 Conclusions
1) The oxidative energy storage in the Ni(OH)2 film electrodes has been successfully achieved by a designed novel energy storage system comprising a nickel hydroxide film electrode and an oxygen-reducing Pt cathode. The oxidative energy stored in the Ni(OH)2 electrodes can be electrochemically discharged. The increase of oxygen content and the decrease of pH value in the cathode electrolytes of the coupling system may enhance the oxidative energy storage in the Ni(OH)2 film electrodes.
2) The nickel hydroxide film electrode using nickel foil substrate in the coupling system with the Pt-black coated Pt plate cathode using 1.0 mol/L Na2SO4 (pH=2) solution as cathode electrolyte shows higher oxidation-discharge cycling stability and better rate discharge capability. This suggests the excellent reversibility of the investigated oxidative energy storage and conversion system.
3) From the view of the oxidation of Ni(OH)2, the present work develops a facile method utilizing a naturally green oxidizing agent (oxygen) replacing the supply of electrical energy or other oxidizing agents with detrimentally environmental consequences. Further, the present method can be easily extended to the oxidative energy storage in other film electrodes at reductive states.
References
[1] MANTHIRAM A, MURUGAN A V, SARKAR A, MURALIGANTH T. Nanostructured electrode materials for electrochemical energy storage and conversion [J]. Energy Environ Sci, 2008, 1(6): 621-638.
[2] YANG Shun-yi, WANG Xian-you, WANG Ying, CHEN Quan-qi, LI Jiao-jiao, YANG Xiu-kang. Effects of Na content on structure and electrochemical performances of NaxMnO2+δ cathode material [J]. Trans Nonferrous Met Soc China, 2010, 20(10): 1892-1898.
[3] GUO Hua-jun, LI Xiang-qun, HE Fang-yong, LI Xin-hai, WANG Zhi-xing, PENG Wen-jie. Effects of sodium substitution on properties of LiMn2O4 cathode for lithium ion batteries [J]. Trans Nonferrous Met Soc China, 2010, 20(6): 1043-1048.
[4] CHEN Hui, WANG Jian-ming, PAN Tao, XIAO Hui-ming, ZHANG Jian-qing. Structure and electrochemical performance of Al and Zn co-substituted a-Ni(OH)2 [J]. The Chinese Journal of Nonferrous Metals, 2003, 13(1): 85-90. (in Chinese)
[5] CHEN H, WANG J M, PAN T, ZHAO Y L, ZHANG J Q, CAO C N. The structure and electrochemical performance of spherical Al-substituted alpha-Ni(OH)2 for alkaline rechargeable batteries [J]. J Power Sources, 2005, 143(1-2): 243-255.
[6] WU M S, WANG M J. Electrochemical preparation of highly regulated nickel oxide nanoflakes on carbon nanofiber for electrochemical capacitor [J]. Electrochem Solid State Lett, 2010, 13(1): A1-A3.
[7] PAN Tao, WANG Jian-ming, CHEN Hui, ZHAO Yan-lin, XIAO Hui-ming, ZHANG Jian-qing. An improved analysis method of constant-current charge/discharge curves and its application to nickel hydroxide electrode [J]. Trans Nonferrous Met Soc China, 2003, 13(2): 462-466.
[8] TAN Y W, SRINIVASAN S, CHOI K S. Electrochemical deposition of mesoporous nickel hydroxide films from dilute surfactant solutions [J]. J Am Chem Soc, 2005, 127(10): 3596-3604.
[9] ZHAO D D, ZHOU W J, LI H L. Effects of deposition potential and anneal temperature on the hexagonal nanoporous nickel hydroxide films [J]. Chem Mater, 2007, 19(16): 3882-3891.
[10] WU Mei-yin, WANG Jian-ming, ZHANG Jian-qing, CAO Chu-nan. Structure and electrochemical performance of Mn-substituted nickel hydroxide [J]. Acta Phys-Chim Sin, 2005, 21(5): 523-527. (in Chinese)
[11] KOSTECKI R, RICHARDSON T, MCLARMON D. Photochemical and photoelectrochemical behavior of a novel TiO2/Ni(OH)2 electrode [J]. J Electrochem Soc, 1998, 145(7): 2380-2385.
[12] TAKAHASHI Y, TATSUMA T. Remote energy storage in Ni(OH)2 with TiO2 photocatalyst [J]. Phys Chem Chem Phys, 2006, 8(23): 2716-2719.
[13] HUANG W, ZHENG J F, LI Z L. New oscillatory electrocatalytic oxidation of amino compounds on a nanoporous film electrode of electrodeposited nickel hydroxide nanoflakes [J]. J Phys Chem C, 2007, 111(45): 16902-16908.
[14] SHANGGUAN E B, CHANG Z R, TANG H W, YUANN X Z, WANG H J. Preparation of nickel oxyhydroxide by a new electrolysis method using spherical beta-Ni(OH)2 [J]. Int J Hydrog Energy, 2010, 35(8): 3214-3220.
[15] TAKASHI Y, TATSUMA T. Oxidative energy storage ability of a TiO2/Ni(OH)2 bilayer photocatalyst [J]. Langmuir, 2005, 21(26): 12357-12361.
[16] PAN J Q, SUN Y Z, WANG Z H, WAN P Y, YANG Y S, FAN M H. Nano-NiOOH prepared by splitting method as super high-speed charge/discharge cathode material for rechargeable alkaline batteries [J]. J Power Sources, 2009, 188(1): 308-312.
[17] FU X Z, ZHU Y J, XU Q C, LI J, PAN J H, XU J Q, LIN J D, LIAO D W. Nickel oxyhydroxides with various oxidation states prepared by chemical oxidation of spherical b-Ni(OH)2 [J]. Solid State Ions, 2007, 178(13-14): 987-993.
[18] BEMIER E, HAMELIN J, AGBOSSOU K, BOSE T K. Electric round-trip efficiency of hydrogen and oxygen-based energy storage [J]. Int J Hydrog Energy, 2005, 30(2): 105-111.
[19] CORRIGAN D A, BENDERT R M. Effect of coprecipitated metal-ions on the electrochemistry of nickel-hydroxide thin-films- cyclic voltammetry in 1 M KOH [J]. J Electrochem Soc, 1989, 136(3): 723-728.
[20] PARK H, KIM K Y, CHOI W. Photoelectrochemical approach for metal corrosion prevention using a semiconductor photoanode [J]. J Phys Chem B, 2002, 106(18): 4775-4781.
[21] KANEKO M, UENO H, SAITO R, NEMOTO J. Highly efficient photoelectrocatalytic decomposition of biomass compounds using a nanoporous semiconductor photoanode and an O2-reducing cathode with quantum efficiency over 100 [J]. Catal Lett, 2009, 131(1-2): 184-188.
[22] WU M S, HUANG Y A, JOW J J, YANG W D. HSIEH C Y, TSAI H M. Anodically potentiostatic deposition of flaky nickel oxide nanostructures and their electrochemical performances [J]. Int J Hydrog Energy, 2008, 33(12): 2921-2926.
[23] SAITO R, UENO H, NEMOTO J, FUJII Y, IZUOKA A, KANEKO M. Photoelectrochemical conversion of NO3- to N2 by using a photoelectrochemical cell composed of a nanoporous TiO2 film photoanode and an O2 reducing cathode [J]. Chem Commun, 2009, 22: 3231-3233.
[24] BARNARD R, RANDELL C F, TYE F L. Studies concerning charged nickel-hydroxides 2. Thermodynamic considerations of the reversible potential [J]. J Appl Electrochem, 1980, 10(1): 127-141.
方文斌1, 练惠勤1, 王建明1, 徐 蕾1, 张丽莹1, 张鉴清1, 居春山2
1. 浙江大学 化学系, 杭州 310027; 2. 江苏华富能源有限公司, 高邮 225600
摘 要:采用多种电化学方法研究了一种由氢氧化镍膜电极和氧还原电极组成的电化学电池体系的氧化性能量储存行为。应用扫描电子显微镜(SEM)和X射线衍射(XRD)分析方法表征样品的结构和表面形貌。研究发现,当耦合体系中阴极电解液的氧含量较高或pH值较低时,可以明显改善氧化性能量在氢氧化镍膜电极中的储存。氧化-放电循环测试结果表明,在采用1.0 mol/L Na2SO4 (pH=2)作为阴极电解液的耦合体系中氧化处理600 s后,氢氧化镍膜电极在第1次和第12次循环后的放电容量分别为79.0和97.9 mC/cm2。这表明设计的氧化性能量储存和转换体系具有优异的电化学可逆性。
关键词:氧化性能量储存;氢氧化镍膜;氧还原电极;氧化
(Edited by YANG Hua)
Foundation item: Projects (50972128, 51174176) supported by the National Natural Science Foundation of China
Corresponding author: WANG Jian-ming; Tel: +86-571-87951513; Fax: +86-571-87951895; E-mail: wjm@zju.edu.cn
DOI: 10.1016/S1003-6326(11)61180-1

