
Influence of pH values on electroless Ni-P-SiC plating on
AZ91D magnesium alloy
GOU Yin-ning(������), HUANG Wei-jiu(��ΰ��), ZENG Rong-chang(���ٲ�), ZHU Yi(�� �)
School of Materials Science and Engineering, Chongqing University of Technology, Chongqing 400050, China
Received 23 September 2009; accepted 30 January 2010
Abstract: A novel Ni-P-SiC composite coating was prepared by electroless plating in order to improve the corrosion capacity and wear resistance of AZ91D magnesium alloy. The influence of pH values on deposition rates and properties of the coatings was studied. The microstructure and phase structure of the Ni-P-SiC coatings were analyzed by scanning electron microscopy (SEM) and X-ray diffractometry (XRD). The corrosion and wear resistance performances of the coatings were also investigated through electrochemical technique and pin-on-disk tribometer, respectively. The results indicate that the composite coating is composed of Ni, P and SiC. It exhibits an amorphous structure and good adhesion to the substrate. The coatings have higher open circuit potential than that of the substrate. The composite coating obtained at pH value of 5.2 possesses optimal integrated properties, which shows similar corrosion resistance and ascendant wear resistance properties to the substrate.
Key words: electroless plating; Ni-P-SiC composite coating; magnesium alloy; corrosion; wear resistance
1 Introduction
Magnesium and its alloys have low density and high specific strength, which make them possible for structural applications in the aerospace, electronic and automobile industries[1-3]. A relatively poor resistance to corrosion and wear of magnesium has, however, hindered their extensive utilization[4]. In order to improve the practical usage of magnesium alloys, many researchers attempted to develop coatings with high corrosion and wear resistance[5-8]. The electroless nickel plating attracts special interest due to the good corrosion and wear resistance, high electrical and thermal conductivity[9]. Some researches indicate that a co-deposit coating in the presence of fine ceramic particles, such as SiC, Al2O3 and diamond[10-13] or MoS2, PTFE and graphite[14-16] within an electroless nickel matrix, might effectively improve the properties of antiwear and lubricity of substrate material.
It is well know that SiC is a ceramic material with excellent corrosion and erosion resistance. The electro- less Ni-P-SiC composite coatings are thus attractive and have been investigated worldwide[11]. Several researches on steel substrates indicate that the corrosion resistance and wear resistance of Ni-P-SiC composite coatings are superior to that of Ni-P coatings[11, 17]. Therefore, the electroless Ni-P-SiC composite coating might provide a better wear resistance for magnesium alloys.
Nevertheless, little efforts could be found in the literatures dealing with corrosion and wear resistance of electroless Ni-P-SiC composite coatings on magnesium alloys. The AZ91D alloy is one of the most commercially used magnesium alloys[18]. The study aims to incorporate SiC particles into a Ni-P coating by electroless plating on magnesium alloy AZ91D substrates and study the effect of pH values on the deposition rates, micro-hardness, surface morphology and corrosion resistance as well as the wear resistance of electroless Ni-P-SiC composite coatings.
2 Experimental
The specimens were cut from a die-cast magnesium alloy AZ91D (8.5% Al, 0.55% Zn, 0.3%Mn and balance Mg) with a size of 20 mm��20 mm��2 mm. Before pre- treatment process, the surfaces of the specimens were polished with progressively finer silicon carbide papers (up to 800 grit) and cleaned in an ultrasonic bath.
The SiC solid particles with grain size of 1-2 ��m were firstly added to a composite electroless plating solution. Prior to the plating, the SiC particles were immersed in hydrochloric acid for 24 h in order to remove metal impurities that may exist. The particles were finally flushed with distilled water and then dried with a hot air.
The main components of solution included 20-25 g/L Ni2+, 25-30 g/L 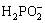 , 1-8 g/L SiC with pH value range of 4.8-5.2 at 80-90 ?C. The pH of the solutions were adjusted with NH3��H2O solution. Prior to each experiment, the alloy substrates were first degreased in a NaOH solution, then etched for 30 s with 600 mL/L
, 1-8 g/L SiC with pH value range of 4.8-5.2 at 80-90 ?C. The pH of the solutions were adjusted with NH3��H2O solution. Prior to each experiment, the alloy substrates were first degreased in a NaOH solution, then etched for 30 s with 600 mL/L 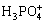 , 1 g/L NaF solution and subsequently activated in a 40% HF solution, and then cleaned with distilled water. The samples treated with pH value of 4.4, 4.8, 5.2, 5.6, 6.0 were designated as sample 1, 2, 3, 4, 5, respectively.
, 1 g/L NaF solution and subsequently activated in a 40% HF solution, and then cleaned with distilled water. The samples treated with pH value of 4.4, 4.8, 5.2, 5.6, 6.0 were designated as sample 1, 2, 3, 4, 5, respectively.
The deposition rates were calculated according to mass addition of the samples. The electrochemical tests in 3.5% NaCl solutions were carried out by using an EG&G model 273 potentiostat to investigate the corrosion property. Three-electrode systems were applied: the working electrode exposed a surface area of approximately 1 cm2, a saturated calomel electrode (SCE) and a platinum plate were used as reference electrode and auxiliary electrode, respectively. The polarization measurements started after the samples were immersed in the solution for 5 min. The potential scanned from -300 to 300 mV (versus open current potential, OCP) at scanning rate of 0.5 mV/s.
The wear behaviors of the magnesium alloy and the coatings were studied using pin-on-disk tribometer tester (CSEM) at load of 0.49 N and speed of 3 cm/s at room temperature and 50% relative humidity. GCr15 steel balls with diameter of 6.0 mm were used as the friction partner for these tests.
A JEOL JSM-6460LV scanning electron microscope (SEM) was employed to inspect the surface morphology of the coatings, the wear tracks and the cross-section; and an EDX attachment was used for qualitative elemental chemical analysis. The structure of the coating was studied by BDX3300 type X-ray diffractometer (XRD). The hardness of the materials was evaluated using a HVS-1000 micro-hardness tester with a Vickers indenter at load of 1.96 N and duration time of 15 s. The final micro-hardness value quoted was an average of 10 replicate measurements.
3 Results and discussion
3.1 Effect of pH value on deposition rate of coatings
The influence of pH value on deposition rate of the coatings is shown in Fig.1. It is indicated that the pH values have a significant effect on deposition rates. The deposition rates of the Ni-P-SiC coating increase linearly with increasing pH values at two stages: the first stage at pH of 4.4-5.2 and the second stage at pH of 5.2-6.0. The influence of pH values on deposition rates is stronger in the first stage than in the second stage. The reason for above phenomenon might be attributed to the fact that different pH values might result in the change of surface charge on substrate and affect the deposition rate.
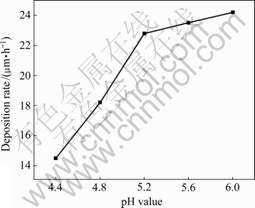
Fig.1 Effects of pH value on deposition rate
3.2 Effects of pH value on surface morphology of coatings
SEM observation reveals that Ni-P-SiC composite coatings obtained in different pH values display quiet different surface morphologies (shown in Fig.2). The surface obtained at pH of 4.4 is relatively smooth and has a variety of visible particles (see Fig.2(a)). An element analysis made on the surface (see Fig.2(b)) indicates that Ni, Mg and Al elements exist in the coating prepared at pH value of 4.8, which implies that the sample is not fully covered by the composite coating. These results demonstrate that it is difficult to plate at very low pH values. As the pH values increase, the composite coatings show a typical spherical nodular structure with good uniformity and dense coverage (Fig.2(c)). However, the delamination occurs at high deposition rate at pH value of 6.0 and thereby a very thin coating forms on the substrate (Fig.2(e)). By comparing these coatings with each other, a compact and defect-free composite coating is obtained at pH value of 5.2. As shown in Fig.2(f), SiC particles are evenly distributed and embedded within the coating. The coating has a good bonding to the substrate.
Fig.3 shows the EDS spectrum of electroless Ni-P-SiC composite coatings at the optimum process
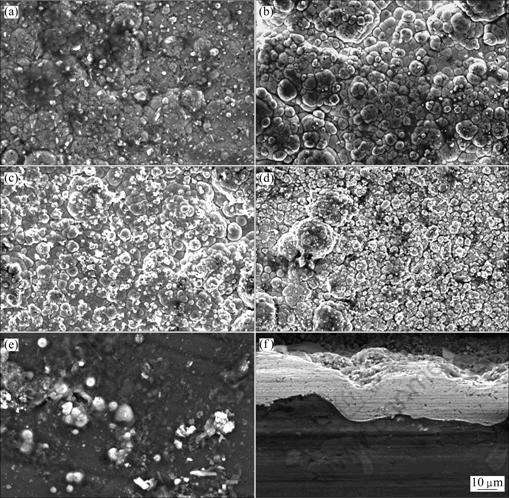
Fig.2 SEM images of Ni-P-SiC coatings obtained in different pH values: (a) 4.4; (b) 4.8; (c) 5.2; (d) 5.6; (e) 6.0; (f) Cross-sectional view of Ni-P-SiC coating at pH of 5.2
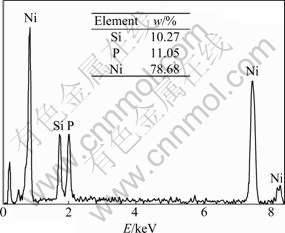
Fig.3 EDS spectrum of electroless Ni-P-SiC coating
parameters, which indicates that the coating contains 78.68% Ni, 11.05% P and 10.27% Si (mass fraction). The phosphorus content indicates that the coating is a kind of high phosphorus coating. The XRD pattern (shown in Fig.4) of electroless Ni-P-SiC composite

Fig.4 XRD pattern of electroless Ni-P-SiC coating
coatings shows a broad strong peak (at around 2��=45?), which is designated as the amorphous structure of the coating. The other several sharp small peaks correspond to the crystalline SiC, which demonstrates that SiC particles exist in solid state.
3.3 Effects of pH value on hardness of coatings
Fig.5 shows the relationship between micro- hardness of coating and pH value. It can be seen from Fig.5 that the hardness of coating increases with increasing pH value, attains the highest value when the pH value is 5.2 and displays a remarkable reduction in the pH range of 5.6-6.0. This result is in good accord with the observation on surface morphology. The large hardness of composite coating obtained at pH of 5.2 may be attributed to the finer grains of the coatings.
3.4 Effects of pH value on corrosion resistance of coatings
The typical polarization curves of the composite coatings in 3.5% NaCl solutions are shown in Fig.6. The OCP of the composite coatings at different pH values is higher than that of the substrate. On the other hand, the composite coatings show higher corrosion current density than the substrate except for Sample 3. The reason
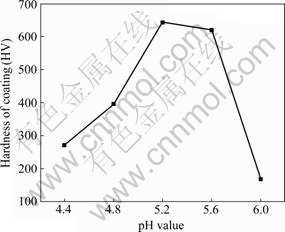
Fig.5 Relationship between micro-hardness of coating and pH value
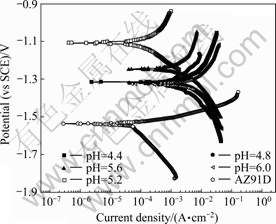
Fig.6 Polarization curves of AZ91D and its coatings at different pH values
for that is ascribed to the pores and defects between the matrix and the SiC particles, which leads to galvanic corrosion due to the difference in potentials. However, it is noticeable that the Sample 3 fabricated at pH of 5.2 shows relatively high OCP and low current density, which indicates that the pH value of 5.2 is the optimization pH value.
3.5 Wear resistance of coatings
Fig.7 shows the friction coefficient of AZ91D substrate, Ni-P coating and Ni-P-SiC coating against GCr15 steel ball at different cycle number under dry friction condition. The substrate displays a mean friction coefficient (0.3), while the Ni-P coating exhibits a high friction coefficient and has a tendency to increase gradually. It is apparent that the introduction of SiC particles into the Ni-P matrix decreases the friction coefficient significantly and maintains the stable state in the whole wear course as expected. The friction coefficient of the Ni-P-SiC composite coating obtained at the optimized pH value of 5.2 against GCr15 counter-face is 0.18. This indicates that SiC particles embedded in the coating can reduce the frictional force.
The SEM images of the worn surfaces of the substrate, Ni-P coating and Ni-P-SiC coating are shown in Fig.8. There are wide grooves on the worn surface of magnesium alloy, which indicates that the evident wear occurs (Fig.8(a)). The localized wide wear track, slight
grooves and peeled coating on the Ni-P worn surface are discerned in Fig.8(b), whereas there is slight friction occurred on the top surface of the Ni-P-SiC coating (Fig.8(c)) and the rest part keeps well. These results reveal that the optimized Ni-P-SiC composite coating has an excellent wear resistance.

Fig.7 Friction coefficient of AZ91D substrate, Ni-P-SiC and Ni-P coating after different cycles

Fig.8 SEM images of worn surfaces of substrate(a), Ni-P(b) and Ni-P-SiC coatings(c)
4 Conclusions
1) The deposition rates of Ni-P-SiC initially increase dramatically when pH value increases from 4.8 to 5.2, and then increase slowly when pH value continuously increases from 5.2 to 6.0. The hardness increases with increasing the pH value up to 5.2, and then abruptly falls down in the pH range of 5.2-6.0.
2) The coatings are composed of 78.68% Ni, 11.05% P and 10.27% Si and exhibit an amorphous structure and good adhesion to the substrate.
3) The OCP of the composite coating is higher than that of the substrate, and the composite coating obtained at the pH value of 5.2 has corrosion resistance equivalent to the substrate. The wear resistance of magnesium alloys with coatings is significantly improved. At the pH value of 5.2, the integrated property of coatings is optimal.
References
[1] ALTUN H, SEN S. The effect of PVD coatings on the wear behaviour of magnesium alloys [J]. Materials Characterization, 2007, 58(10): 917-921.
[2] GRAY J E, LUAN B. Protective coatings on magnesium and its alloys��A critical review [J]. Journal of Alloys and Compounds, 2002, 336(1/2): 88-113.
[3] LIU Zhen-min, GAO Wei. Electroless nickel plating on AZ91 Mg alloy substrate [J]. Surf Coat Technol, 2006, 200(16/17): 5087-5093.
[4] HOCHE H, SCHEERER H, PROBST D, BROSZEIT E, BERGER C. Development of a plasma surface treatment for magnesium alloys to ensure sufficient wear and corrosion resistance [J]. Surf Coat Technol, 2003, 174/175: 1018-1023.
[5] ALTUN H, SEN S. The effect of PVD coatings on the corrosion behaviour of AZ91 magnesium alloy [J]. Materials & Design, 2006, 27(10): 1174�C1179.
[6] MONTEMOR M F, SIM?ES A M, CARMEZIM M J. Characterization of rare-earth conversion films formed on the AZ31 magnesium alloy and its relation with corrosion protection [J]. Appl Surf Sci, 2007, 253(16): 6922-6931.
[7] WU C S, ZHANG Z, CAO F H, ZHANG L J, ZHANG J Q, CAO C N. Study on the anodizing of AZ31 magnesium alloys in alkaline borate solutions [J]. Appl Surf Sci, 2007, 253(8): 3893-3898.
[8] KOUISNI L, AZZI M, ZERTOUBI M, DALARD F, MAXIMOVITCH S. Phosphate coatings on magnesium alloy AM60 (Part 1): Study of the formation and the growth of zinc phosphate films [J]. Surf & Coat Technol, 2004,185(1): 58-67.
[9] LI Li-min, HU Wen-bin, LUO Shou-fu, SHEN Bin. Electroless nickel plating on difficult-to-plate substrates [J]. Electroplating & Pollution Control, 2002, 22(3): 13-17. (in Chinese)
[10] MALLORY G., HAJDU J B. Electroless plating: Fundamentals and applications [M]. Orlando: American Electroplaters and Surface Finishers Society, 1996: 261-265.
[11] ZHANG Shu-sheng, HAN Ke-jiang, CHENG Lin. The effect of SiC particles added in electroless Ni-P plating solution on the properties of composite coatings [J]. Surf & Coat Technol, 2008, 202(12): 2807-2812.
[12] BALARAJU J N, KALAVATI, RAJAM K S. Influence of particle size on the microstructure, hardness and corrosion resistance of electroless Ni-P-Al2O3 composite coatings [J]. Surf & Coat Technol, 2006, 200(12/13): 3933-3941.
[13] REDDY V V N, RAMAMOORTHY B, KESAVAN NAIRP. A study on the wear resistance of electroless Ni-P/diamond composite coatings [J]. Wear, 2000, 239(1): 111-116.
[14] HU X G., CAI W J, XU Y F,WAN J C,SUN X J. Electroless Ni-P-(nano-MoS2) composite coatings and their corrosion properties [J]. Surf Eng, 2009, 25(5): 361-366.
[15] ZHAO Q, LIU Y, M?LLER-STEINHAGEN H, LIU G. Graded Ni-P-PTFE coatings and their potential applications [J]. Surf & Coat Technol, 2002, 155(2/3): 279-284.
[16] KUMAR M A, AGARWALA R C, AGARWALA V. Synthesis and characterization of electroless Ni-P coated graphite particles [J]. Bull Mater Sci, 2008, 31(5): 819-824.
[17] WU Ya-ting, SHEN Bin, LIU Lei, HU Wen-bin. The tribological behaviour of electroless Ni-P-Gr-SiC composite [J]. Wear, 2006, 261(2): 201-207.
[18] CELOTTO S. TEM study of continuous precipitation of Mg-9%wt Al-1%wt Zn alloy [J]. Acta Mater, 2000, 48(20): 1775-1787.
(Edited by LI Yan-hong)
Foundation item: Project(KJ070602) supported by Program of Applied Science Foundation of Chongqing Education Committee, China; Project(KF0604) supported by the Open Foundation of Key Laboratory of Low Dimensional Materials & Application Technology (Xiangtan University), Ministry of Education, China
Corresponding author: HUANG Wei-jiu; Tel: +86-23-68666425; E-mail: huangweijiu@cqut.edu.cn