����ϳɾ��ֶ��������������̵Ĵ�����
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2016���2��
�������ߣ���� ���Ӣ �¹���
����ҳ�룺557 - 564
�ؼ��ʣ��������֣��������������յ��ڣ��¶ȣ���ʼ��������
Key words��jarosite seed; jarosite process; induction period; temperature; initial precipitate rate
ժ Ҫ����ϸ���ϳɾ��ֺͻ�ѧ��(����ϸ��)�ϳɾ��ֵĴ����ܽ����о���ɨ�����������X�������������ʾ������ϳɵľ��ֱ���⻬����Ҫ�ɻƼ�������ɣ���ѧ���ϳɵľ��־߲˻��ṹ����Ҫ�ɲݻ�������ɡ���Ի�ѧ�����֣�������ִ����ܸ��ţ��յ���������20 min���¶Ƚ���75 ��C����ʼ���������������ﵽ3.933 %/min������������ֵij��������ᾧ�ȸߴ�97.22%������������ʱ�ᾧ�Ƚ�Ϊ12.89%�������������ֿ�ʹ��������ת������ȫ��
Abstract: The catalytic performance on jarosite process of jarosite seeds via biosynthesis and chemical processes were studied, respectively. The SEM and XRD results showed that biosynthetic jarosite seeds (BIO seeds) had smooth surface and mainly consisted of potassium jarosite. The chemical synthesis jarosite seeds (CHM seeds) had a loose cauliflower-like surface and mainly consisted of hydronium jarosite. The catalytic performance of BIO seeds was better than that of CHM seeds on the formation of final jarosite product. The induction time can be shortened to 20 min, the reaction temperature can be reduced to only 75 ��C, and the initial reaction speed was enhanced obviously, up to 3.933%/min. The crystallinity of final jarosite product using BIO seeds can achieve 97.22%, while it was only 12.89% without seeds. This indicates that the precipitation process of jarosite is more complete with BIO seeds.
Trans. Nonferrous Met. Soc. China 26(2016) 557-564
Hai-jun LI, Hong-ying YANG, Guo-bao CHEN
School of Materials and Metallurgy, North Eastern University, Shenyang 110819, China
Received 23 March 2015; accepted 13 October 2015
Abstract: The catalytic performance on jarosite process of jarosite seeds via biosynthesis and chemical processes were studied, respectively. The SEM and XRD results showed that biosynthetic jarosite seeds (BIO seeds) had smooth surface and mainly consisted of potassium jarosite. The chemical synthesis jarosite seeds (CHM seeds) had a loose cauliflower-like surface and mainly consisted of hydronium jarosite. The catalytic performance of BIO seeds was better than that of CHM seeds on the formation of final jarosite product. The induction time can be shortened to 20 min, the reaction temperature can be reduced to only 75 ��C, and the initial reaction speed was enhanced obviously, up to 3.933%/min. The crystallinity of final jarosite product using BIO seeds can achieve 97.22%, while it was only 12.89% without seeds. This indicates that the precipitation process of jarosite is more complete with BIO seeds.
Key words: jarosite seed; jarosite process; induction period; temperature; initial precipitate rate
1 Introduction
Ferric iron that commonly exists in leaching solution needs to be removed before the recovery of valuable metals. The jarosite process is an iron-removal method that has many advantages, such as easy operation, little entrainment of other metals, and maintenance of equilibrium between sulfuric acid and alkali ions [1-3]. However, the dynamics of this process is slow, and induction period in the initial stage is long [4-6]. Seed-addition method is widely used to shorten induction period and accelerate precipitation rate [7,8].
The relationship between the amount of crystal seed and the rate of jarosite precipitation is gradually becoming distinct. TEIXEIRA and TAVARES [9] studied the jarosite process of MnSO4-rich iron solutions and found that when the amount of seeds added is 0-10 g/L, the initial rate linearly increases with increased seeds added.
Excessive jarosite seeds can cause condensation in solution, and the seed undergoes so-called ��deactivation��. WON and PAIK [10] demonstrated that the initial rate of jarosite precipitation has a limit despite the initial rate increase along with the increase of seed addition. PAMMENTER and HAIGH [11] studied the rate of jarosite precipitation under seed addition of 0-300 g/L and determined that the optimum seed addition is about 100 g/L for hydrometallurgy zinc plant. DUTRIZAC [7] discovered that seeds can reduce the reliance of the jarosite process on pH and temperature.
The jarosite process changes when jarosite seeds are added to iron-rich solution. This solution accelerates the rate of jarosite precipitation and improves the operation and precipitation filtration performance. Jarosite precipitation was applied in most factories [1], and many factories have built systems for jarosite seed recovery and addition. However, seed dosage, temperature, and pH in the jarosite process are found to have a considerable number of problems. These problems occur because of the formation of iron hydroxide during jarosite precipitation that can destroy jarosite seed performance [12]. The use of synthetic jarosite seeds can solve the aforementioned problems [11].
Methods of producing synthetic jarosite seeds include chemical and biological technologies. Chemical method can produce synthetic jarosite seeds at high temperature [13,14], whereas biological method was carried out at room temperature [15,16]. The crystallinity of jarosite prepared by biological technology is better than that produced by the chemical process [17,18]. The biosynthetic technology of jarosite is given more attention and recognized as an important way to synthesize jarosite seeds because of its mild reaction conditions. Jarosite synthesized by biological technology was used for catalytic materials [19], yellow pigments [20], building materials [21,22], and adsorption materials [13,23] because of its distinctive performance.
Most studies on the effect of jarosite seeds on precipitate formation rate are based mostly on chemical synthesis [8-11], whereas research on the lower-cost biological method is limited. In this work, we studied the catalytic properties of jarosite seeds biologically synthesized from the jarosite process at low temperature and pH and attempted to increase the formation rate of jarosite. This new synthesis method of jarosite seeds can improve the efficiency of de-ironing process in hydrometallurgy, and reduce production cost.
2 Experimental
ZY101 strain of Acidithiobacillus ferrooxidans bacteria was separated and domesticated for a long time with 9K medium [15,24] in a biometallurgy laboratory. The 9K medium was composed of 3.00 g (NH4)2SO4, 0.10 g KCl, 0.50 g K2HPO4, 0.50 g MgSO4��7H2O, 0.01 g Ca(NO3)2, 44.48 g FeSO4��7H2O, and 1 L distilled water. The initial pH of this medium was adjusted to 1.8 with H2SO4.
2.1 Preparation of jarosite seeds by biological method
About 200 mL of 9K medium containing ZY101 bacteria was added to a 500 mL conical flask. The initial number of ZY101 bacteria in the medium was 6��107 cells/mL [25]. The initial pH of the medium was 2.5, and the medium temperature was 44 ��C, which was the suitable multiplication temperature for ZY101. A higher pH (2.50) could result in significant increase of jarosite yield [26,27]. A large amount of yellow precipitate formed from 9K medium after 48 h.
The precipitate was centrifuged at 2000 r/min for 5 min with a centrifuge (Model: 3-30K, Sigma Company). The precipitate was washed with sulfuric acid (pH 1.6) to avoid the phase transformation of jarosite and to remove impurities absorbed by the surface of grain sediment [28,29]. Biological seeds were obtained after precipitate dehydration by ethanol at room temperature. These seeds were named BIO seeds.
2.2 Preparation of jarosite seeds by chemical method
Ferric sulfate solution with 9.0 g/L Fe3+ was heated to 95 ��C [7]. The initial pH of jarosite generation was controlled at 2.0 and restored to the same value during the reaction process at 20 min intervals. About 100 g/L sodium carbonate was used as the adjusting agent. A considerable amount of yellow precipitate was produced at 95 ��C after 2 h [7]. The same method in Section 2.1 was used to separate the precipitate. Chemical crystal seeds named CHM seeds were then obtained.
2.3 Jarosite process
About 100 mL ferric sulfate solution with 28.63 g/L Fe3+ was added into a 75-95 ��C water bath. About 0.1 g jarosite seed was placed inside after pH was adjusted to the designated value. During reaction, pH was restored to the initial value at 20 min intervals. At the same time, ferric iron mass in liquid phase was determined. The efficiency (��) of jarosite process was calculated according to:
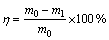 (1)
(1)
where m1 is Fe3+ mass at time t, and m0 is Fe3+ mass at t=0.
2.4 Characterization methods
pH was determined with a pHS-3C meter after calibrating with standard buffer solution (pH 6.86 and 4.00). The concentration of ferric iron was determined by 1,10-phenanthroline spectrophotometry (when [Fe3+]< 0.1 g/L) or by titanium trichloride�Cpotassium dichromate titrimetry (when [Fe3+]>0.1 g/L). The concentration of ferric iron in precipitate was determined by trichloride�C potassium dichromate titrimetry and sulphur was determined by barium sulphate gravimetric method.
The samples were subjected to X-ray diffraction (XRD) analysis using a X-ray diffractometer (Model: PW3040/60, Panalytical Company, Netherlands). The scanning range was 10��-90��, and the scanning speed was 0.2 (��)/s. A scanning electron microscopy (SEM) system (Model: SSX-550; Shimadzu, Tokyo, Japan) was used to observe the morphology of seeds and precipitates of jarosite process at an accelerating voltage of 15 kV. The particles size (D50) was obtained on the particle size analyzer (Model: BT-9300HT, Bettersize Company, Dandong, China).
3 Results and discussion
3.1 Characterization of jarosite seeds
Figure 1 shows the morphologies of BIO and CHM seeds. BIO seed is sleek and smooth edge, with loose cauliflower-like morphology and size below 5 ��m. CHM seeds appear like a compact, stacked corolla, with uneven particle size and pointed edge. The average particle sizes (D50) of BIO and CHM seeds are 1.07 and 0.93 ��m, respectively.
Figure 2 shows the XRD patterns of BIO and CHM seeds. XRD shows amorphous impurities in seeds. The main composition of BIO seeds is potassium jarosite, whereas CHM seeds comprise hydronium jarosite. According to the diffraction peak (113) of the crystal plane of jarosite, the crystallinities are 57.63% for CHM seeds and 89.69% for BIO seeds.

Fig. 1 Secondary electron morphologies of jarosite seeds by different methods
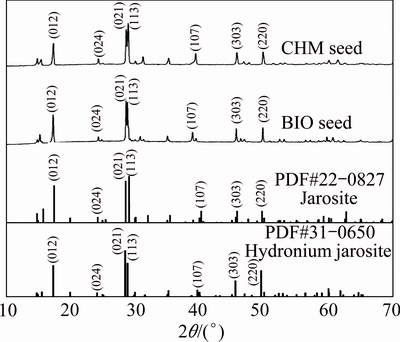
Fig. 2 XRD patterns of jarosite seed
3.2 Efficiency of jarosite process with different seeds
The morphology and crystallinity of the two seeds significantly differ; thus, the jarosite process with two kinds of seeds was studied at different pH values (1.5-2.5) and temperatures (75-95 ��C). Figure 3 describes the relationship between the temperature and efficiency of iron removal at different pH values.
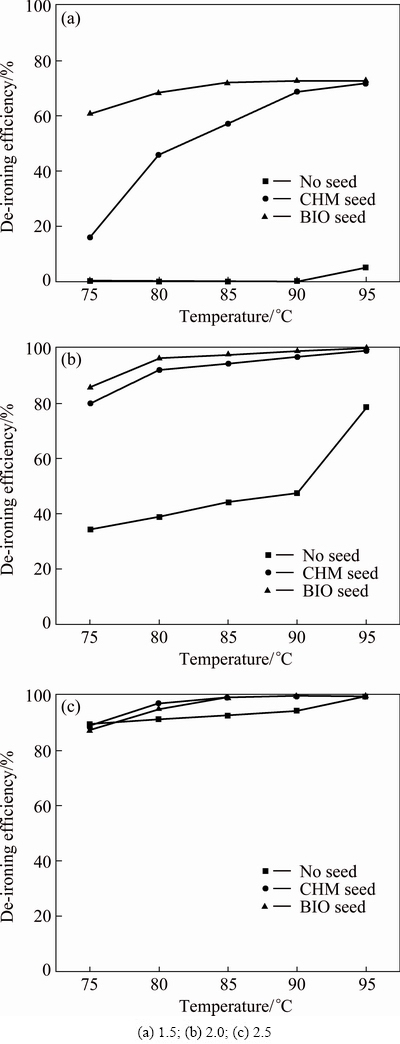
Fig. 3 Effect of different seeds on de-ironing efficiency in jarosite process at different pH values
Figure 3 shows that the jarosite process in sulfuric acid is endothermic. At different pH values, efficiency increases with increasing temperature. The increase tendency is exponential and the inflexion point is at 90 ��C in the absence of any seed in the jarosite process. In the presence of BIO or CHM seed, the increase tendency is logarithmic and the inflexion point is at 80 ��C. The change in increase tendency is ascribed to the decreased induction period.
The induction period can be expressed using Eqs. (2) and (3):
Fe3++3H2O��Fe(OH)3+3H+ (2)
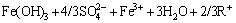 ��
��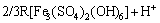 (3)
(3)
where R is Na+, K+, NH4+, or H3O+. Ferric ion can be separated as Fe(OH)3 after adding alkaline neutralizer. Fe(OH)3 is then converted into jarosite [12].
Equation (3) is the rate-determining step if neutralizers are sufficient. High temperature is suitable for jarosite production, and high active energy is needed for the new phase produced. Figure 3(b) shows that the efficiency is 34.15% at 75 ��C, pH 2.0, and without any seed. At 95 ��C, efficiency is 78.23%. This variation pattern at different temperatures is similar to the patterns in Figs. 3(a) and (c) without any seed.
Efficiency logarithmically increases when jarosite seeds are added, along with increasing temperature (Fig. 3). Efficiency with seeds is higher than that without seeds, as shown in Fig. 3(b), reaching 97.23% when BIO seeds are added and 93.95% when CHM seeds are added.
Change in efficiency from the exponential to logarithmic growth of iron removal proves that adding jarosite seeds can lower the dependence of separated jarosite from solution upon heating. Figure 3 shows that after adding jarosite seeds at 80-90 ��C, induction period is shortened, efficiency is increased, and reaction equilibrium is more rapidly reached.
Figure 3 also shows that the catalytic performance of BIO seeds is better than that of CHM seeds. Jarosite does not separate without jarosite seeds at 75 ��C after 2 h (Fig. 3(a)). Under the same conditions, efficiency increases after adding seeds, reaching 60.89% with BIO seeds and 16.00% with CHM seeds. These suggest that the jarosite process can occur at a lower temperature and in a broader pH range using BIO seeds than using CHM seeds.
The addition of jarosite seeds can reduce the dependence of the jarosite process on temperature and can promote jarosite growth. However, the jarosite process is significantly controlled by acidity. Equation (3) shows that H+ in the solution plays a significant role in the equilibrium of jarosite precipitate and solution. Efficiency remains stable when the initial iron concentration and equilibrated pH remain identical. Temperature and jarosite seeds control the reaction equilibrium time. Figure 3(a) shows that the equilibrium of jarosite process with BIO seeds is obtained at 85 ��C compared with 90-95 ��C using CHM seeds at pH 1.5.
Higher iron-removal efficiency is obtained with increasing pH. However, a high pH also means strong hydrolysis tendency of metallic ion. The addition of BIO seeds has a distinct advantage for jarosite formation compared with CHM jarosite seeds.
3.3 Generation rate of jarosite process with different seeds
Equation (3) shows no substantial change in final iron-removal efficiency at the same pH. Therefore, the efficiency difference in Fig. 3 is ascribed to the various generated velocities of jarosite. Figure 4 shows the rate of jarosite formation obtained from the jarosite process with BIO or CHM seeds at pH 2.0.
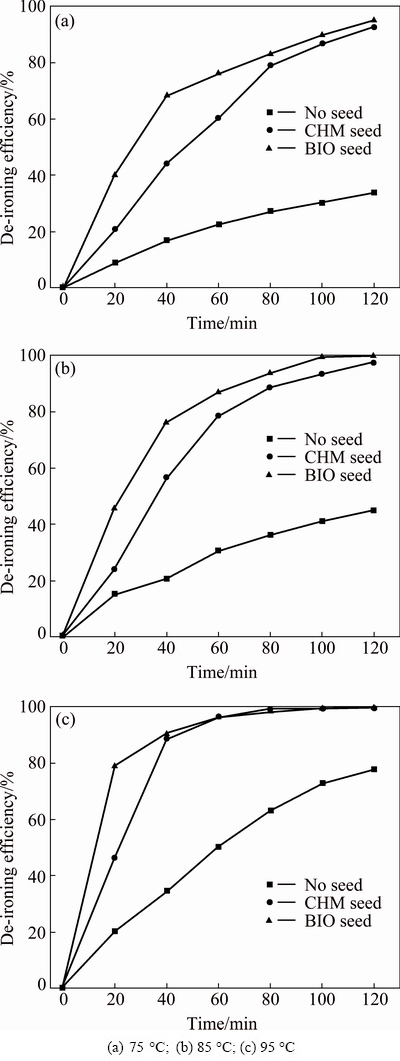
Fig. 4 Iron precipitation curves for jarosite process using different seeds at various temperatures and pH 2.0
Figure 4 shows that jarosite seeds can reduce the induction period of the jarosite process. A linear relationship exists between efficiency and reaction time without jarosite seeds. The increased variation in slopes of the curves of efficiency and reaction time proves that the induction period of jarosite process with seeds is shortened. Thus, the linear slope of the curves is considered as the initial rate of jarosite formation. Thus, the slopes of this linear part are fitted and then distinguished to display the catalytic performances of the synthetic jarosite. Equation (4) describes the fitting function of Fig. 4:
��=��+bt (4)
where b is the slope of curve that is firmly believed as the initial rate of jarosite process, a is a constant, t is reaction time, and �� is sedimentation rate of iron.
The fitting results are listed in Table 1. The initial rate significantly increases with increased amount of jarosite seed for both synthesis methods. The b value of the fitting curve is 0.2776 %/min for solution without jarosite seed at 75 ��C, 0.3590 %/min at 85 ��C increasing to 1.3371 %/min with CHM seeds, and 1.8907 %/min with BIO seeds at 85 ��C.
Fe3+ changes to Fe(OH)3 with the addition of Na2CO3 into solution, and the reaction is shown in Eq. (2). The change process is rapid, but the transition rate of Eq. (3) is very slow because the reaction depends on heat. The low b means that new jarosite must be activated and used during the induction period. After adding jarosite seeds, active seeds concentrates are high in the initial stage, which shortens the accumulated time of active seeds and increases b.
A higher slope of the fitting curve means a shorter reaction equilibrium time in the jarosite process. In Fig. 4, the iron-removal efficiency slowly increases with increasing holding time at constant temperature after the induction period ends, and an obvious inflection point exists. According to the difference in de-ironing efficiency around the inflection point, the time at the inflection point is defined as the equilibrium time of de-ironing. Table 1 shows that the initial rate of the jarosite process with BIO seeds is higher than that with CHM seeds, and the time to inflection-point appearance is shorter. At 85 ��C, b is 1.8907 %/min and equilibrium time is 40 min for BIO seeds, and the values are 1.3371 %/min and 60 min for CHM seeds, respectively. At 75 ��C, the corresponding values are 1.7157 %/min and 40 min for BIO seeds, and 0.9909 %/min and 80 min for BIO and CHM seeds, respectively. The induction period without seed is not suspended after 2 h, which means that it has no inflection point.
Research shows that an increase in the initial rate of the jarosite process with BIO seeds is higher than that with CHM seeds. However, both seeds have the same feature for the inferior limit of temperature. To compare the changes in initial rate among different jarosite seeds, the dip angles of fitting curves are calculated (Table 1). The dip angles are 1.3219 rad for BIO seeds and 1.1461 rad for CHM seeds at 95 ��C, as well as 1.0430 rad and 0.7808 rad at 75 ��C, respectively. The larger variation of angles of BIO seeds indicates their higher catalytic performance for the jarosite process at low temperature.
3.4 Morphology and phase of jarosite precipitate
Figure 5 shows the SEM images of jarosite precipitate using different jarosite seeds at 85 ��C, pH 2.0, and 2 h, as well as at constant temperature and time. The micrographs show the differences in grain size, surface smoothness, and assembly types of jarosite precipitates under different conditions. Element analysis results of the precipitate samples results are listed in Table 2.
Table 1 Fitting results of precipitation curves in Fig. 4 by linear function
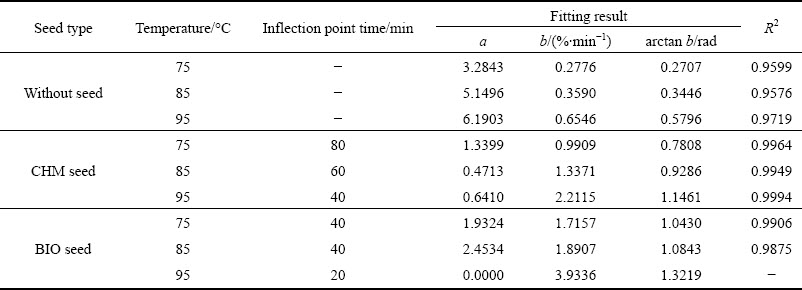

Fig. 5 Secondary electron micrographs of precipitates with different seeds
Table 2 Elemental analysis of precipitate with different seeds in Fig. 5

Precipitates with BIO seeds comprise well- developed crystal faces, with 2�C5 ��m angular grain size that is close to that of CHM seeds. These grains are formed by the growth-disengagement process of BIO seeds in the jarosite process [4]. In this experiment, fewer secondary nuclei are possible with CHM seeds because precipitates are covered by some amorphous materials and the size of particles is similar to that of CHM seeds.
The activated Fe(OH)3 which can adsorb a large number of sulfate radical in solution by electrostatic effect energy is colloid. In the process of Eq. (3), the ratio of Fe to S in jarosite crystals is 3:2. Elemental composition analysis results of grain sediments show that the mole ratio of Fe to S is 3:2. Precipitate with CHM seeds is similar to that with BIO seeds (Fig. 5(a)) but with many amorphous substances. The mole ratio of Fe to S is 5:4, which proves that not all Fe(OH)3 transformed into jarosite.
Figure 5(c) shows precipitate without seeds. Amorphous grains with non-uniform size and many viscous substances are observed. Elemental analysis results show that the mole ratio of Fe to S is 1:1, suggesting that only a small portion of Fe(OH)3 transformed into jarosite at the end of de-ironing. This result agrees with the predicted phenomenon in this work. The induction period is more than 2 h for the jarosite process without seeds.
The evolution process of the crystal structure of precipitate was investigated by XRD analysis, and the results are shown in Fig. 6. All precipitates are confirmed to correspond with jarosite (PDF# 31-0650), but the crystallinity of these precipitates differs according to diffraction peak (021) of the crystal plane. Precipitate obtained without seeds shows a significant amorphous noise in the background with crystallinity of 12.89%. The crystallinity of precipitate obtained with BIO seeds is 97.22%, and that with CHM seeds is 88.11%. The position of diffraction peak is resolved by the distance of crystal plane.
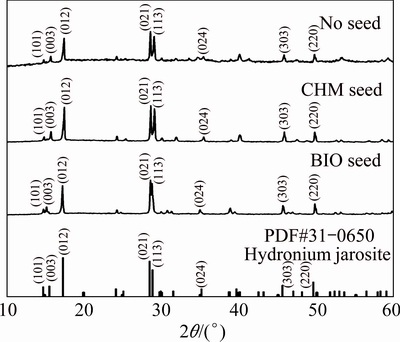
Fig. 6 XRD patterns of precipitates with different seeds
These results are consistent with SEM findings, confirming that the seeds can improve the crystallinity of jarosite precipitate and that BIO seeds are better.
4 Conclusions
1) Leaching bacteria ZY101 are successfully applied to synthesizing jarosite seeds with higher crystallinity. Compared with CHM seeds, BIO seeds can shorten induction period, weaken dependence on temperature, and reduce temperature limit in the jarosite process.
2) The initial rate of the jarosite process with BIO seeds is enhanced, and its equilibrium time becomes shorter, particularly at a low temperature.
3) Jarosite precipitate with BIO seeds has angular grains at 85 ��C, pH 2.0 and 2 h. The mole ratio of Fe to S is close to 3:2, which is the theoretical composition of the jarosite molecule. The mole ratios of Fe to S in precipitate are 5:4 with CHM seeds and 1:1 without seeds. Not all Fe(OH)3 molecules transform into jarosite; hence, amorphous substances increase, as shown in the morphology and XRD patterns of the precipitate.
References
[1] ARREGUI V, GORDON A R, STEINTVEIT G. The jarosite process ��Past, present and future [C]//Lead-Zinc-Tin'80, TMS-AIME World Symposium on Metallurgy and Environment Control. Pittsburgh: The Metallurgical Society of AIME, 1979: 97-123.
[2] DUTRIZAC J E. The physical chemistry of iron precipitate in the zinc industry [C]//Lead-Zinc-Tin'80, TMS-AIME World Symposium on Metallurgy and Environment Control. Pittsburgh: The Metallurgical Society of AIME, 1979: 532-564.
[3] BARON D, PALMER C D. Solubility of jarosite at 4-35 ��C [J]. Geochimica et Cosmochimica Acta, 1996, 60(2): 185-195.
[4] HUTCHISON R F S, PHIPPS P J. Formation and particle size of jarosite [C]//Austr. I.M.M. Conference. Parkville: Australasian Institute of Mining and Metallurgy, 1977: 319-327.
[5] HARTIG C, BRAND P, BOHMHAMMEL K. On the mechanism of alunite and jarosite precipitate [J]. Neue H��tte, 1990, 35: 205-209. (in German)
[6] MARGULIS E V, GETSKIN L S, KRAVETS M V, ALKATSEV M L, YAROSLAVTSEV A S. Induction period of jarosite formation in zinc sulphate solutions [J]. Izv Vyssh Uchebn Zaved Tsvetn Metall, 1976, 4: 145-147. (in Russia)
[7] DUTRIZAC J E. The effect of seeding on the rate of precipitate of ammonium jarosite and sodium jarosite [J]. Hydrometallurgy, 1996, 42(3): 293-312.
[8] DUTRIZAC J E. The effectiveness of jarosite species for precipitating sodium jarosite [J]. The Journal of the Minerals, Metals & Materials Society, 1999, 51(12): 30-32.
[9] TEIXEIRA L A, TAVARES L Y. Precipitation of jarosite from manganese sulphate solutions [C]//Iron Control in Hydrometallury. Chichester: Ellis Horwood Limited Publisher, 1986: 431-453.
[10] WON C W, PAIK Y H. Precipitation kinetics of hydronium jarosite at elevated temperatures [J]. Taehan Kumsok Hakhoe Chi, 1982, 20(7): 594-602. (in Korean)
[11] PAMMENTER R V, HAIGH C J. Improved metal recovery with the low-contaminant jarosite process [C]//Extraction Metallurgy '81. London: Inst Mining Metall, 1981: 379-392.
[12] CORNELL R M, SCHWERTMANN U. The iron oxides [M]. Weinheim: WILEY-VCH Verlag GmbH & Co. KGaA, 2003: 118-119.
[13] DUTRIZAC J E, CHEN T T. The behaviour of phosphate during jarosite precipitation [J]. Hydrometallurgy, 2010, 102(1-4): 55-65.
[14] DUTRIZAC J E. Comparative rates of precipitation of ammonium jarosite and sodium jarosite in ferric sulphate-sulphuric acid media [J]. Canadian Metallurgical Quarterly, 2010, 49(2): 121-130.
[15] LIU J, XIU X, CAI P. Study of formation of jarosite mediated by Thiobacillus ferrooxidans in 9K medium [J]. Procedia Earth and Planetary Science, 2009, 1(1): 706-712.
[16] OBA N, MICHIOKA C, KATO M, YOSHIMURA K, MIBU K. Physical properties of the novel Jarosite-type compound NaFe3(SeO4)2(OH)6 [J]. Journal of Physics and Chemistry of Solids, 2005, 66(8-9): 1438-1441.
[17] SASAKI K, KONNO H. Morphology of jarosite-group compounds precipitated from biologically and chemically oxidized Fe ions [J]. The Canadian Mineralogist, 2000, 38(3): 45-56.
[18] ZHU C J, LU J J, LU X C, WANG R C, LI Q. SEM study on jarosite mediated by Thiobacillus ferrooxidans [J]. Geological Journal of China Universities, 2005, 11(2): 234-238.
[19] XU Z, LIANG J, ZHOU L. Photo-fenton-like degradation of azo dye methyl orange using synthetic ammonium and hydronium jarosite [J]. Journal of Alloys and Compounds, 2013, 546: 112-118.
[20] SHI Qi-li. Study on composition of jarosite crystal by Acidithiobacillus ferrooxidans [D]. Shanghai: Donghua University, 2010. (in Chinese)
[21] ASOKAN P, SAXENA M, ASOLEKAR S R. Hazardous jarosite use in developing non-hazardous product for engineering application [J]. Journal of Hazardous Materials, 2006, 137(3): 1589-1599.
[22] VU H,  J, HRON T. Recovery of pigment-quality magnetite from jarosite precipitates [J]. Hydrometallurgy, 2010, 101(1-2): 1-6.
J, HRON T. Recovery of pigment-quality magnetite from jarosite precipitates [J]. Hydrometallurgy, 2010, 101(1-2): 1-6.
[23] ASOKAN P, SAXENA M, ASOLEKAR S R. Recycling hazardous jarosite waste using coal combustion residues [J]. Materials Characterization, 2010, 61(12): 1342-1355.
[24] SILVERMAN M P, LUNDGREN D G. Studies on the chemoautotrophic iron bacterium Ferrobacillus ferrooxidans. I. An improved medium and a harvesting procedure for securing high cell yields [J]. J Bacteriol, 1959, 77(5): 642-647.
[25] WANG S M, ZHOU L. A renovated approach for increasing colony count efficiency of Thiobacillus ferrooxidans and Thiobacillus thiooxidans: Double-layer plates [J]. Acta Scientiae Circumstantiae, 2005, 25(1): 1418-1420. (in Chinese)
[26] WANG M, ZHOU L. Simultaneous oxidation and precipitation of iron using jarosite immobilized Acidithiobacillus ferrooxidans and its relevance to acid mine drainage [J]. Hydrometallurgy, 2012, 125: 152-156.
[27] NURMI P,  B, SASAKI K, KAKSONEN A H, RIEKKOLA-VANHANEN M, TUOVINEN O H, PUHAKKA J A. Biooxidation and precipitation for iron and sulfate removal from heap bioleaching effluent stream [J]. Hydrometallurgy, 2010, 101(3-4): 7-14.
B, SASAKI K, KAKSONEN A H, RIEKKOLA-VANHANEN M, TUOVINEN O H, PUHAKKA J A. Biooxidation and precipitation for iron and sulfate removal from heap bioleaching effluent stream [J]. Hydrometallurgy, 2010, 101(3-4): 7-14.
[28] SMITH A M L, HUDSON-EDWARDS K A, DUBBIN W E, WRIGHT K. Dissolution of jarosite [KFe3(SO4)2(OH)6] at pH 2 and 8: Insights from batch experiments and computational modeling [J]. Geochimicaet Cosmochimica Acta, 2006, 70(3): 608-621.
[29] WANG H, BIGHAM J M, TUOVINEN O H. Formation of Schwertmannite and its transformation to jarosite in the presence of acidophilic iron-oxidizing microorganisms [J]. Materials Science and Engineering C, 2006, 26(4): 588-592.
��������Ӣ���¹���
������ѧ ������ұ��ѧԺ������ 110819
ժ Ҫ����ϸ���ϳɾ��ֺͻ�ѧ��(����ϸ��)�ϳɾ��ֵĴ����ܽ����о���ɨ�����������X�������������ʾ������ϳɵľ��ֱ���⻬����Ҫ�ɻƼ�������ɣ���ѧ���ϳɵľ��־߲˻��ṹ����Ҫ�ɲݻ�������ɡ���Ի�ѧ�����֣�������ִ����ܸ��ţ��յ���������20 min���¶Ƚ���75 ��C����ʼ���������������ﵽ3.933 %/min������������ֵij��������ᾧ�ȸߴ�97.22%������������ʱ�ᾧ�Ƚ�Ϊ12.89%�������������ֿ�ʹ��������ת������ȫ��
�ؼ��ʣ��������֣��������������յ��ڣ��¶ȣ���ʼ��������
(Edited by Xiang-qun LI)
Foundation item: Projects (51104036, 51374066, 51304047) supported by the National Natural Science Foundation of China
Corresponding author: Hong-ying YANG; Tel/Fax: +86-24-83673932; E-mail: yanghy@smm.neu.edu.cn
DOI: 10.1016/S1003-6326(16)64144-4

