Бт»ҜЙйФьөДјоРФҪюіцј°Ҫюіц¶ҜБҰС§
°Ч ГНЈ¬ЦЈСЕҪЬЈ¬БхНтУоЈ¬ХЕҙ«ёЈ
(ЦРДПҙуС§ ТұҪрҝЖС§Ул№ӨіМС§ФәЈ¬әюДП іӨЙіЈ¬410083)
ХӘ ТӘЈәІЙУГЗвСх»ҜДЖИЬТәҪюіцБт»ҜЙйФьЈ¬К№AsУлCuәНBiөИҪрКфУРР§·ЦАлЈ¬УРАыУЪБт»ҜЙйФьөДЧЫәПАыУГЎЈ¶ФЗвСх»ҜДЖҪюіцБт»ҜЙйФь¶ҜБҰС§ҪшРРМҪМЦЎЈСРҫҝҪб№ыұнГчЈәөұ·ҙУҰОВ¶ИОӘ90 ЎжЈ¬№МТәұИОӘ1?6Ј¬·ҙУҰКұјдОӘ1.5 hЈ¬NaOHУлAs2S3өДДҰ¶ыұИОӘ7.2?1КұЈ¬ЗвСх»ҜДЖҪюіцБт»ҜЙйФьЈ¬ЙйҪюіцВКҙпөҪ95.90%Ј¬НӯҪюіцВКҪцОӘ0.087%Ј»ҫӯ№эЗвСх»ҜДЖҪюіцЈ¬ФьЦРCuәНBiЦКБҝ·ЦКэ·ЦұрҙУ10.90%әН1.85%ФцјУөҪ50.00%әН10.63%Ј¬CuәНBiөГөҪёЯ¶Иё»јҜЈ»ИЬТәЦРAs2S3УлNaOH·ҙУҰОӘКХЛхОҙ·ҙУҰРҫА©ЙўҝШЦЖЈ¬Ждұн№Ы»о»ҜДЬОӘ3.682 kJ/molЎЈ
№ШјьҙКЈәБт»ҜЙйФьЈ»ЗвСх»ҜДЖЈ»ҪюіцЈ»¶ҜБҰС§
ЦРНј·ЦАаәЕЈәX705 ОДПЧұкК¶ВлЈәA ОДХВұаәЕЈә1672-7207(2008)02-0268-05
Alkaline leaching and leaching kinetics of arsenic sulfide residue
BAI Meng, ZHENG Ya-jie, LIU Wan-yu, ZHANG Chuan-fu
(School of Metallurgical Science and Technology, Central South University, Changsha 410083, China)
Abstract: In order to utilize arsenic sulfide residue from sulfide precipitation of contained arsenic wasted water, As was separated effectively from Cu and Bi after arsenic sulfide was leached in the sodium hydroxide solution. The leaching kinetics was studied. The results show that the leaching rate of As is up to 95.90% and the copper leaching rate is only 0.087% when the reaction temperature is 90 Ўж, the ratio of solid to liquid is 1:6, the reaction time is 1.5 h, the mole ratio of NaOH to As2S3 is 7.2:1. The mass fractions of Cu and Bi in the leached slag increase from 10.90% and 1.85% to 50.00% and 10.63%, respectively. Cu and Bi are highly concentrated. The reaction between NaOH and As2S3 in the solution is controlled by diffusion and the reaction is applicable to the shrinking core model. The active energy is 3.682 kJ/mol.
Key words: arsenic sulfide residue; sodium hydroxide; leaching; kinetics
Ййј°Жд»ҜәПОпСПЦШОЈәҰИЛАаҪЎҝөЈ¬ТСұ»ГА№ъјІІЎҝШЦЖЦРРДәН№ъјК·А°©СРҫҝ»ъ№№И·¶ЁОӘөЪ1АаЦВ°© Оп[1]Ј¬ЦчТӘНЁ№эТыЛ®Нҫҫ¶¶ФИЛМеФміЙОЈәҰИЛМеҪЎҝө[2]ЎЈТтҙЛЈ¬ЦР№ъЎўГА№ъЎўОчЕ·ЎўИХұҫөИ°СЙйБРОӘУЕПИҝШЦЖөДЛ®ОЫИҫОпЦ®Т»ЎЈ»р·ЁИЫБ¶НӯІъЙъSO2СМЖшЈ¬ҫӯ№эПҙөУәуІъЙъЛбРФ·ПЛ®ә¬ЙйёЯҙп10 g/LЎЈҙҰАнә¬Йй·ПЛ®өД·Ҫ·ЁЦчТӘУРЭНИЎ·ЁЎўАлЧУҪ»»»·ЁЎўОьёҪ·ЁЎўКҜ»ТЦРәНМъСОРхДэіБөн·ЁәНБт»ҜіБөн·ЁөИ[3-5]Ј¬ЖдЦРЈ¬ЭНИЎ·ЁЎўАлЧУҪ»»»·ЁЎўОьёҪ·ЁЦчТӘУГУЪҙҰАнөНЕЁ¶Иә¬Йй·ПЛ®Ј¬ТұБ¶і§ә¬Йй·ПЛ®ЖХұйІЙУГРхДэіБөн·ЁәНБт»ҜіБөн·ЁҙҰАнЎЈІЙУГКҜ»ТЦРәНМъСОРхДэ·ЁҙҰАнФьБҝҙуКұЈ¬РиТӘҪшРР№М»ҜҙҰАнЈ¬·сФтІъЙъ¶юҙООЫИҫЎЈОӘБЛАыУГБт»ҜЙйФьЈ¬ИХұҫЧЎУС№«ЛҫНЁ№эБтЛбНӯЦГ»»әНҝХЖшСх»Ҝ·Ё·ЦАлНӯәНЙйЈ¬ІЙУГSO2»№ФӯВЛТәЦРЙйөГөҪAs2O3ЎЈёГјјКхіЙ КмЈ¬As2O3ҙҝ¶ИёЯЈ¬°ІИ«РФәГЈ¬ө«№ӨТХБчіМёҙФУЈ¬ЙъІъіЙұҫ№эёЯ[6-8]ЎЈЧЫәПАыУГБт»ҜЙйФьҫЯУРЦШҙуТвТе[9-10]Ј¬ФЪҙЛЈ¬ұҫОДЧчХЯІЙУГЗвСх»ҜДЖИЬТәҪюіцБт»ҜЙйФьЈ¬К№Бт»ҜЙйФьЦРЙйУлНӯәНойөИөГөҪУРР§·ЦАлЈ¬Іў¶ФҪюіц¶ҜБҰС§ҪшРРСРҫҝЎЈ
1 КөСйФӯБПј°ІҪЦи
1.1 КөСйФӯБП
ДіТұБ¶і§ә¬Йй·ПЛ®ҫӯ№эБт»ҜДЖіБөнҙҰАнәуөГөҪБт»ҜЙйФьЈ¬ЖдіЙ·ЦИзұн1ЛщКҫЈ¬БЈ¶И·ЦІјәНРОГІ·ЦұрИзНј1әННј2ЛщКҫЎЈ
ұн1 Бт»ҜЙйФь»ҜС§іЙ·Ц
Table 1 Chemical composition of arsenic sulfide residue w/%

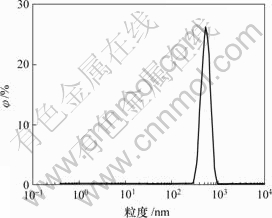
ҰХОӘМе»э·ЦКэ
Нј1 Бт»ҜЙйБЈ¶И·ЦІј
Fig.1 Particle size distribution of arsenic sulfide residue

Нј2 Бт»ҜЙйФьSEMХХЖ¬
Fig.2 SEM image of arsenic sulfide residue
УЙұн1ҝЙЦӘЈ¬Бт»ҜЙйФьЦРAsЈ¬CuәНSЦКБҝ·ЦКэ·ЦұрҙпөҪ18.17%Ј¬10.90%әН19.25%Ј¬ЛөГчЦчТӘіЙ·ЦОӘБт»ҜЙйәНБт»ҜНӯЎЈБт»ҜЙйФьМе»эЖҪҫщБЈҫ¶ОӘ786 nmЈ¬ЙЁГиөзҫө(SEM)КөСйЦӨКөБт»ҜЙйФьУЙПёРЎБЈЧУҫЫјҜОӘЛЙЙўМеЎЈ
1.2 Кө Сй
1.2.1 Бт»ҜЙйФьөДјоҪю
Ҫ«ИЭ»эОӘ2 LөДИэҫұЖҝЦГУЪDF-101BРНјҜИИКҪәгОВҙЕБҰҪБ°иЖчЙПЈ¬»т№М¶ЁУЪі¬ј¶әгОВЖчЦРЈ¬°ҙТ»¶Ё№МТәұИјУИлЗвСх»ҜДЖИЬТәЈ¬ҝмЛЩҪБ°иЈ¬СёЛЩјУИлБт»ҜЙйФьЈ¬·ҙУҰТ»¶ЁКұјдәу№эВЛөГөҪә¬ЙйјоҪюТәЎЈ
1.2.2 јоҪю¶ҜБҰС§КөСй
ФЪИЭ»эОӘ1 LҙшҝМ¶ИөДФІЦщРНІЈБ§·ҙУҰЖчЦРјУИлЗвСх»ҜДЖИЬТәәуЈ¬СёЛЩјУИл100 gБт»ҜЙйФьЈ¬JBV-IIIРНұдЖөөчЛЩҪБ°иЖчҝШЦЖЧӘЛЩОӘ400 r/minЈ¬ід·ЦҪБ°иЎЈҝШЦЖ№МТәұИОӘ1?6Ј¬ЗвСх»ҜДЖУГБҝОӘУлAs2S3·ҙУҰөДАнВЫУГБҝөД1.2ұ¶ЎЈФЪ80 ЎжТФПВНЁ№эі¬ј¶әгОВЛ®ФЎҝШЦЖ·ҙУҰОВ¶ИЈ¬ОВ¶Иұд»Ҝ·¶О§ОӘ-1~1 ЎжЈ»ФЪ80 ЎжТФЙПНЁ№э№иУНУНФЎҝШЦЖ·ҙУҰОВ¶ИЈ¬ОВ¶Иұд»Ҝ·¶О§ОӘ-2~2 ЎжЎЈФЪ·ҙУҰ№эіМЦРёщҫЭ·ҙУҰТәГжІ№јУХфБуЛ®Ј¬ұЈіЦ·ҙУҰИЬТәМе»эІ»ұдЎЈ
1.2.3 Бт»ҜЙйФьҪюіцВКөДІв¶Ё
ІЙУГдеЛбјШөО¶Ё·ЁІв¶ЁјоҪюТәЦРЧЬЙйЕЁ¶ИЈ¬ёщҫЭІв¶ЁҪб№ыјЖЛгЙйҪюіцВК(ҰЗ(As))ЎЈ

2 КөСйҪб№ыУлМЦВЫ
2.1 Бт»ҜЙйФьөДјоРФҪюіц
ТұБ¶СМЖш·ПЛ®ЦРә¬УР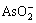 Ј¬
Ј¬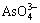 Ј¬Cu2+Ј¬Bi3+Ј¬Pb2+Ј¬Sb3+Ј¬Zn2+әНFe2+өИЈ¬јУИлБт»ҜДЖәу·ўЙъИзПВ ·ҙУҰЈә
Ј¬Cu2+Ј¬Bi3+Ј¬Pb2+Ј¬Sb3+Ј¬Zn2+әНFe2+өИЈ¬јУИлБт»ҜДЖәу·ўЙъИзПВ ·ҙУҰЈә

Ҫ«ә¬Йй·ПЛ®јУИлБт»ҜДЖәуІъЙъөДіБөнОпіЖОӘБт»ҜЙйФьЎЈКөСйЦРИЎ300 gБт»ҜЙйФьјУИлNaOHИЬТәЈ¬өұ·ҙУҰОВ¶ИОӘ26 ЎжЈ¬№МТәұИОӘ1?6Ј¬·ҙУҰКұјдОӘ1.5 hКұЈ¬NaOHУлAs2S3өДДҰ¶ыұИ¶ФЙйҪюіцВКөДУ°ПмИзНј3ЛщКҫЎЈ
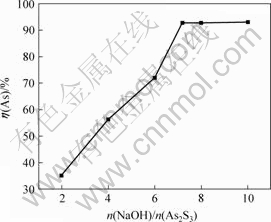
Нј3 NaOHУлAs2S3өДДҰ¶ыұИ¶ФЙйҪюіцВКҰЗ(As)өДУ°Пм
Fig.3 Influence of mole ratios of NaOH to As2S3 on leaching rate of As
УЙНј3ҝЙЦӘЈ¬ЙйҪюіцВКЛжNaOHУлAs2S3өДДҰ¶ыұИФцјУ¶шФцјУЈ¬өұNaOHУлAs2S3өДДҰ¶ыұИОӘ7.2?1КұЈ¬ЙйҪюіцВКҙпөҪ92.72%Ј»јМРшФцҙуNaOHУГБҝЈ¬ЙйҪюіцВК»щұҫІ»ұдЈ¬ЖдККТЛөДДҰ¶ыұИОӘ7.2?1ЎЈ
Бт»ҜЙйФьЦРјУИлЗвСх»ҜДЖИЬТәЈ¬As2S3әНSb2S3іБөн·ўЙъИЬҪвЈ¬·ҙУҰИзПВЈә
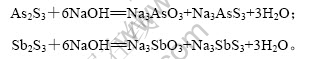
УЙ·ҙУҰФӯАнҝЙЦӘЈ¬As2S3НкИ«ИЬҪвЈ¬NaOH УлAs2S3АнВЫЙПөДДҰ¶ыұИОӘ6?1Ј¬іэЖдЛыОпЦКПыәДЗвСх»ҜДЖНвЈ¬КөјКЙПЈ¬Ц»УРЗвСх»ҜДЖ№эБҝКұЈ¬As2S3ІЕДЬід·ЦИЬҪвЈ¬Тт¶шNaOHУлAs2S3КөјКөДДҰ¶ыұИҙуУЪ6?1ЎЈ
ЙПКцЖдЛыМхјюІ»ұдЈ¬өұNaOH УлAs2S3АнВЫЙПөДДҰ¶ыұИОӘ7.2?1КұЈ¬·ҙУҰОВ¶И¶ФЙйҪюіцВКөДУ°ПмИзНј4ЛщКҫЎЈ
УЙНј4ҝЙЦӘЈ¬ЙйҪюіцВКЛж·ҙУҰОВ¶ИөДЙэёЯ¶ш»әВэФцјУЈ»өұ·ҙУҰОВ¶ИҙУ25 ЎжФцјУөҪ90 ЎжКұЈ¬ЙйҪюіцВКҙУ92.73%ФцјУөҪ96.40%ЎЈТтҙЛЈ¬·ҙУҰОВ¶И¶ФЙйҪюіцВКУ°ПмҪПРЎЎЈ
ФЪ·ҙУҰОВ¶ИОӘ90 ЎжЈ¬№МТәұИОӘ1?6Ј¬·ҙУҰКұјдОӘ1.5 hЈ¬NaOHУлAs2S3өДДҰ¶ыұИОӘ7.2?1МхјюПВЈ¬ИЎ1 kgБт»ҜЙйФьҪшРРА©ҙуКөСйЈ¬ЙйҪюіцВКҙпөҪ95.90%Ј¬Нӯ ҪюіцВКҪцОӘ0.087%ЎЈҪ«јоҪюФьҪшРР·ЦОцЈ¬AsЈ¬CuЈ¬BiЈ¬SЈ¬PbЈ¬ZnЈ¬NaәНFeөДЦКБҝ·ЦКэ·ЦұрОӘ2.616%Ј¬50.00%Ј¬10.63%Ј¬24.42%Ј¬0.61%Ј¬0.64%Ј¬0.95%әН1.22%ЎЈҝЙјыЈ¬НӯәНойөГөҪё»јҜЈ¬ЙйУлНӯәНой·ЦАлЎЈ

Нј4 ·ҙУҰОВ¶И¶ФЙйҪюіцВКҰЗ(As)өДУ°Пм
Fig.4 Influence of reaction temperature on leaching rate of As
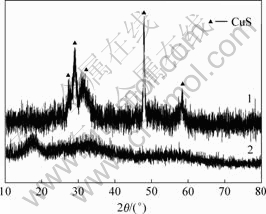
1ЎӘБт»ҜЙйФьЈ»2ЎӘјоҪюФь
Нј5 Бт»ҜЙйФьәНјоҪюФьXЙдПЯСЬЙдНјЖЧ
Fig.5 XRD patterns of As2S3 slag and basic leached slag
јоҪюФьУлБт»ҜЙйФьXЙдПЯСЬЙд(XRD)Ҫб№ыИзНј5ЛщКҫЎЈУЙНј5ҝЙЦӘЈ¬Бт»ҜЙйФьіКОЮ¶ЁРНМ¬Ј¬јоҪюФьЦРә¬УРБт »ҜНӯЎЈ
2.2 Бт»ҜЙйФьҪюіц¶ҜБҰС§
2.2.1 јоҪю№эіМЦРAsҪюіцВКУлКұјдөД№ШПө
ФЪІ»Н¬ОВ¶ИПВЈ¬ІвөГЙйҪюіцВКЛж·ҙУҰКұјдөД№ШПөИзНј6ЛщКҫЎЈ
ҙУНј6ҝЙЦӘЈ¬ЙйҪюіцВКЛж·ҙУҰКұјдСУіӨәН·ҙУҰОВ¶ИФцјУ¶шФцјУЈ¬БҪХЯіКПЯРФ№ШПөЈ»өұ·ҙУҰКұјдОӘ60 minЈ¬·ҙУҰОВ¶И·ЦұрОӘ25Ј¬40Ј¬55Ј¬70әН90 ЎжКұЈ¬ЙйҪюіцВК·ЦұрҙпөҪ92.72%Ј¬94.50%Ј¬95.00%Ј¬95.50%әН96.40%ЎЈ
2.2.2 Ҫюіц¶ҜБҰС§
Бт»ҜЙйФьЦР№ММеAs2S3УлNaOHИЬТә·ҙУҰЈ¬КфУЪТә№МПа·ҙУҰЈ¬¶шЗТAs2S3УлNaOH·ҙУҰОЮ№МПаІъОпЙъіЙЈ¬
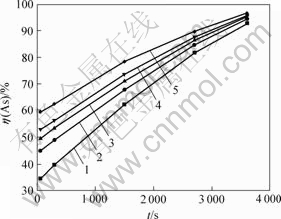
ҰИ/Ўж: 1ЎӘ25; 2ЎӘ40; 3ЎӘ55; 4ЎӘ70; 5ЎӘ90
Нј6 І»Н¬ОВ¶ИПВAsҪюіцВКҰЗ(As)Лж·ҙУҰКұјдөДұд»Ҝ№ШПө
Fig.6 Effect of reaction time on leaching rate of As at different temperatures
Жд·ҙУҰј°ПаұдұнКҫИзПВЈә
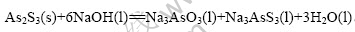
ЙПКц·ҙУҰҝЙИПОӘ·ҙУҰКЗФЪ№ММеҝЕБЈAs2S3ұнГжҪшРРөДТә-№МПа·ЗҙЯ»Ҝ·ҙУҰЎЈТә-№МПа·ЗҙЯ»Ҝ·ҙУҰЧоіЈјыөД·ҙУҰДЈРНОӘКХЛхОҙ·ҙУҰРҫДЈРН[11-14]Ј¬јтіЖОӘЛхРҫДЈРНЎЈЛхРҫДЈРНУЦ·ЦОӘБЈҫ¶І»ұдЛхРҫДЈРНәНҝЕБЈЛхРЎЛхРҫДЈРНЎЈ
БЈҫ¶І»ұдЛхРҫДЈРНөДМШөгКЗУР№МПаІъОпІгЙъіЙЈ¬·ҙУҰ№эіМЦРҝЕБЈБЈҫ¶І»ұдЎЈҝЕБЈЛхРЎЛхРҫДЈРНөДМШөгКЗЈәФЪ·ҙУҰ№эіМЦРЈ¬·ҙУҰОпҝЕБЈІ»¶ПЛхРЎЈ¬ОЮ№МПаІъОпІгЈ¬ІъОпИЬУЪИЬТәЦРЎЈ
Бт»ҜЙйУлЗвСх»ҜДЖ·ҙУҰЙъіЙј«ТЧИЬУЪЛ®өДСЗЙйЛбДЖәНБтҙъСЗЙйЛбДЖЈ¬ҝЙҙУҝЕБЈЛхРЎЛхРҫДЈРНСРҫҝЖд·ҙУҰ¶ҜБҰС§ЎЈөұОӘБчМеЦНБчДӨА©ЙўҝШЦЖКұЈ¬ҝЕБЈЛхРЎЛхРҫДЈРН¶ҜБҰС§·ҪіМОӘЈә
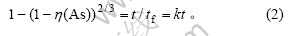
өұ·ҙУҰОӘ»ҜС§·ҙУҰҝШЦЖКұЈ¬¶ҜБҰС§·ҪіМОӘЈә

КҪЦРЈәҰЗ(As)ОӘ·ҙУҰОпҪюіцВКЈ»tОӘ·ҙУҰКұјдЈ»tfОӘНкИ«·ҙУҰКұјдЈ»ҰСBОӘ№ММе·ҙУҰОпГЬ¶ИЈ»RsОӘ№ММеҝЕБЈіхКј·ҙУҰ°лҫ¶Ј»bОӘ№ММе·ҙУҰОпјЖБҝПөКэЈ»MBОӘ№ММе·ҙУҰОпПа¶Ф·ЦЧУЦКБҝЈ»hОӘ·ҙУҰЛЩВКіЈКэЈ»CAОӘТәМе·ҙУҰОпЕЁ¶ИЎЈ¶ФУЪДіТ»№М¶ЁМеПөЈ¬ЗТБчМе·ҙУҰОпЕЁ¶ИCAҪьЛЖІ»ұдКұЈ¬tfҝЙК¶ОӘіЈКэЈ¬1/tfФтҝЙұнКҫОӘk(ЖдЦРkОӘұн№Ы·ҙУҰЛЩВКіЈКэ)ЎЈ
ёщҫЭНј6Ј¬Чч1-(1-ҰЗ(As))2/3Ул·ҙУҰКұјдөДЗъПЯЈ¬Ҫб№ыИзНј7ЛщКҫЈ»ёщҫЭНј7ЗуіцІ»Н¬ОВ¶ИПВёчЦұПЯРұВКјҙkЈ¬Ччln kУл1/T№ШПөНјЈ¬ИзНј8ЛщКҫЎЈ

ҰИ/Ўж: 1ЎӘ25; 2ЎӘ40; 3ЎӘ55; 4ЎӘ70; 5ЎӘ90
Нј7 І»Н¬ОВ¶ИПВ1-(1-ҰЗ(As))2/3Ул·ҙУҰКұјдөД№ШПө
Fig.7 Relationship between 1-(1-ҰЗ(As))2/3 and reaction time at different temperatures

Нј8 ln kУл1/T№ШПөНј
Fig.8 Relationship between ln k and 1/T
ҙУНј7ҝЙЦӘЈ¬1-(1-ҰЗ(As))2/3Ул·ҙУҰКұјдіКБјәГПЯРФ№ШПөЈ¬Па№ШПөКэrҫщҙуУЪ0.999 8Ј¬ЛөГчБт»ҜЙйФьјоҪю№эіМЦРAs2S3ФЪNaOHИЬТәЦРөД·ҙУҰОӘКХЛхОҙ·ҙУҰРҫА©ЙўҝШЦЖЎЈ·ҙУҰОӘА©ЙўҝШЦЖКұЈ¬Т»°г·ҙУҰОВ¶И¶ФЙйҪюіцВКУ°ПмҪПРЎЈ¬ХвУл·ҙУҰОВ¶И¶ФҪюіцВКУ°ПмКөСйҪб№ыТ»ЦВЎЈ
ФЪ»ҜС§·ҙУҰЦРЈ¬·ҙУҰЛЩ¶ИіЈКэkКЗОВ¶ИөДәҜКэЈ¬ОВ¶И¶Ф·ҙУҰЛЩ¶ИіЈКэөДУ°ПмҝЙУГ°ўАЫДбОЪЛ№№«КҪ ұнКҫЈә
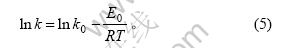
КҪЦРЈәkОӘ·ҙУҰЛЩ¶ИіЈКэЈ»k0ОӘЖөВКТтЧУЈ»E0ОӘ»о»ҜДЬЈ»TОӘИИБҰС§ОВ¶ИЈ»RОӘЖшМеіЈКэЎЈёщҫЭНј7ЗуРұВКөГұн№Ы»о»ҜДЬE0ОӘ3.682 kJ/molЈ¬ЗуҪШҫаөГЖөВКТтЧУk0ОӘ1.868ЎБ103Ј¬јҙ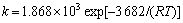 ЎЈ
ЎЈ
Т»°гөШЈ¬өұ·ҙУҰ»о»ҜДЬРЎУЪ62.78 kJ/molКұЈ¬·ҙУҰЛЩ¶ИәЬҝм[15]ЎЈБт»ҜЙйФьЦРAs2S3УлNaOH·ҙУҰ»о»ҜДЬөНЈ¬ЛөГчБт»ҜЙйФьЦРAs2S3ТЧИЬУЪЗвСх»ҜДЖЎЈө«КЗЈ¬УЙУЪ·ҙУҰОпЦРCuSөИІ»ИЬОпөД°ь№ьЧчУГәНЙъіЙОпөДА©ЙўЧи°ӯЈ¬К№Жд·ҙУҰЛЩ¶ИјхВэЎЈ
3 Ҫб ВЫ
a. ІЙУГЗвСх»ҜДЖҪюіцБт»ҜЙйФьЈ¬өұ·ҙУҰОВ¶ИОӘ 90 ЎжЈ¬№МТәұИОӘ1?6Ј¬·ҙУҰКұјдОӘ1.5 hЈ¬NaOHУлAs2S3өДДҰ¶ыұИОӘ7.2?1КұЈ¬ЙйҪюіцВКҙпөҪ95.90%Ј¬НӯҪюіцВКОӘ0.087%ЎЈ
b. ҫӯ№эЗвСх»ҜДЖҪюЈ¬ФьЦРCuәНBiЦКБҝ·ЦКэ·ЦұрҙУ10.90%әН1.85%ФцјУөҪ50.00%әН10.63%Ј¬НӯәНойөГөҪёЯ¶Иё»јҜЎЈ
c. Бт»ҜЙйФьјоҪю№эіМЦРAs2S3ФЪNaOHИЬТәЦРөД·ҙУҰОӘКХЛхОҙ·ҙУҰРҫА©ЙўҝШЦЖЈ¬Жд¶ҜБҰС§·ҪіМКҪОӘЈә
1-(1-ҰЗ(As))2/3=ktЎЈ
ЖдЦРЈә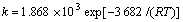 ЎЈ
ЎЈ
ІОҝјОДПЧЈә
[1] іВҫҙҫь, ҪҜ°ШИӘ, Нх О°. іэЙйјјКхПЦЧҙУлҪшХ№[J]. ҪӯОч»Ҝ№Ө, 2004(2): 1-4.
CHEN Jing-jun, JIANG Bo-quan, WANG Wei. Status and progress of arsenic removal[J]. Jiangxi Chemical Engineering, 2004(2): 1-4.
[2] Katsoyiannis I A, Zouboulis A I. Application of biological processes for the removal of arsenic from groundwater[J]. Water Research, 2004, 38(1): 17-26.
[3] Dutre V, Andecasteele C. Solidification/stabilization of hazardous arsenic containing waste from a copper refining process[J]. Journal of Hazardous Materials, 1995, 40(1): 55-68.
[4] ЦмТеДк, ХЕ »Ә, БәСУЕф, өИ. ЙйЛбёЖ»ҜәПОпөДИЬҪв¶Иј°ЖдОИ¶ЁРФЛжpHЦөөДұд»Ҝ[J]. »·ҫіҝЖѧѧұЁ, 2005, 25(12): 1652-1660.
ZHU Yi-nian, ZHANG Hua, LIANG Yan-peng, et al. Dependence of solubility and stability of calcium arsenates on pH value[J]. Acta Scientiae Circumstantiae, 2005, 25(12): 1652-1660.
[5] ХЕИЩБј, ЗрҝЛЗҝ, Р»УАҪр, өИ. НӯТұБ¶ЙБЛЩВҜСМіҫСх»ҜҪюіцУлЦРәННСЙй[J]. ЦРДПҙуѧѧұЁ: ЧФИ»ҝЖС§°ж, 2006, 37(1): 73-78.
ZHANG Rong-liang, QIU Ke-qiang, XIE Yong-jin, et al. Treatment process of dust from flash smelting furnace at copper smelter by oxidative leaching and neutralization process from leaching solution[J]. Journal of Central South University: Science and Technology, 2006, 37(1): 73-78.
[6] БхІэУВ. №уПӘТұБ¶і§СЗЙйЛбЙъІъ№ӨТХ[J]. УРЙ«ТұБ¶, 1998(2): 8-10.
LIU Chang-yong. Arsenous acid production in Guixi Smelter[J]. Non-Ferrous Smelting, 1998(2): 8-10.
[7] іВО¬ЖҪ, АоЦЩУў, ұЯҝЙҫэ, өИ. КӘКҪМбЙй·ЁФЪҙҰАн№ӨТө·ПЛ®ј°·ПФьЦРөДУҰУГ[J]. ЦР№ъ»·ҫіҝЖС§, 1999, 19(4): 310-312.
CHEN Wei-ping, LI Zhong-ying, BIAN Ke-jun, et al. Application of wet-method for extracting arsenic in treating industrial wastewater and residues[J]. China Environmental Science, 1999, 19(4): 310-312.
[8] МпОДФц, іВ°ЧХд, ірУВәЈ. УРЙ«ТұҪр№ӨТөә¬ЙйОпБПөДҙҰАнј°АыУГПЦЧҙ[J]. әюДПУРЙ«ҪрКф, 2004, 20(6): 11-15.
TIAN Wen-zeng, CHEN Bai-zhen, QIU Yong-hai. Review of As-containing materials treatment and utilization in nonferrous metallurgy industry[J]. Hunan Nonferrous Metals, 2004, 20(6): 11-15.
[9] ЦЈСЕҪЬ, Нх УВ, ХФЕК·е. Т»ЦЦАыУГә¬Йй·ПЛ®ЦЖұёСЗЙйЛбНӯәНЙйЛбНӯөД·Ҫ·Ё: ЦР№ъ, 200610032456.1[P]. 2006-10-25.
ZHENG Ya-jie, WANG Yong, ZHAO Pan-feng. A method of producing arsenite copper and arsenate copper from waste acid contained As: CN 200610032456.7[P]. 2006-10-25.
[10] іВ°ЧХд, №ЁЦсЗа. БтЛбНӯҪбҫ§ДёТәЦЖұёЙйЛбНӯөД№ӨТХјјКх[J]. ЦРДП№ӨТөҙуѧѧұЁ: ЧФИ»ҝЖС§°ж, 2000, 31(4): 300-302.
CHEN Bai-zhen, GONG Zhu-qing. A technology of preparing arsenate copper from the mother liquid of copper sulfide[J]. Journal of Central South University of Technology: Natural Science, 2000, 31(4): 300-302.
[11] Tkacovo K, Balaz P. Selective leaching of zinc from mechanically activated complex Cu-Pb-Zn concentrate[J]. Hydrometallurgy, 1993, 33(3): 291-300.
[12] ЦмұюіҪ. »ҜС§·ҙУҰ№ӨіМ[M]. ұұҫ©: »ҜС§№ӨТөіц°жЙз, 1993.
ZHU Bing-chen. Chemical reaction engineering[M]. Beijing: Chemical Industry Press, 1993.
[13] Ekinci Z, Colak S, Cakici A. Technical note leaching kinetics of sphalerite with pyrite in chloride saturated water[J]. Minerals Engineering, 1998, 11(3): 279-283.
[14] Breed A W, Hansford G S. Studies on the mechanism and Kinetics of bioleaching[J]. Minerals Engineering, 1999, 12(4): 383-392.
[15] ёөПЧІК, іВИр»Ә. ОпАн»ҜС§: ПВІб[M]. ұұҫ©: ИЛГсҪМУэіц°жЙз, 1979.
FU Xian-cai, CHEN Rui-hua. Physical chemistry: 2nd Volume[M]. Beijing: People Education Press, 1979.
КХёеИХЖЪЈә2007-04-23Ј»РЮ»ШИХЖЪЈә2007-06-25
»щҪрПоДҝЈә№г¶«КЎҙҙРВ»щҪрЧКЦъПоДҝ(200501045)
НЁРЕЧчХЯЈәЦЈСЕҪЬ(1959-)Ј¬ДРЈ¬әюДПіЈөВИЛЈ¬ҪМКЪЈ¬І©КҝЙъөјКҰЈ¬ҙУКВТұҪрЎўІДБПәН»·ҫіұЈ»ӨСРҫҝЈ»өз»°Јә0731-8836285Ј»E-mail: zzyyjj01@yahoo.com.cn