
Interaction between pyrite and cysteine
LIU Jian-she(柳建设), WANG Zhao-hui(王兆慧), LI Bang-mei(李邦梅), ZHANG Yan-hua(张艳华)
School of Resources Processing and Bioengineering, Central South University, Changsha 410083, China
Received 13 September 2005; accepted 22 November 2005
Abstract: The adsorption mechanism of cysteine on pyrite was studied by amounts adsorbed, FTIR and XRD measurements. The results obtained by adsorption experiment suggest that as the mass ratio of mineral to cysteine mp/mc is greater than 5, the amounts adsorbed on mineral is stable after adsorption for 15 min and cysteine adsorbing with mp/mc shows the same tendency. It can be inferred by its Langmuir-type adsorption isotherm that chemical interaction governs the entire adsorption process. The results from FTIR and XRD prove that the functional groups of cysteine appear with blue shift of their characteristic adsorption peak in FTIR spectrum; meanwhile, the lattice constant obviously decreases and the widening of crystal planes such as (210), (220) and (211) is found after cysteine adsorbing on mineral.
Key words: pyrite; cysteine; adsorption isotherm; adsorption mechanism; lattice constant
1 Introduction
L-cysteine, as an important sulfureous amino acid, has been widely used in medicine, food, cosmetic and feeding stuff industry[1]. Recently, L-cysteine is being focused in the field of microbial metallurgy[2, 3] as well due to its capacity to accelerate leaching rate[3, 4]. It was reported that the active group sulfydryl (―SH) of the molecule was involved in bio-leaching process[5-9]. A further research found that L-cysteine could interact spontaneously with pyrite, with bonds formation similar to those present in ferredoxin complexes of biological membrane[10]. Consequently, L-cysteine could disrupt chemical bond of ferrous sulfide and release iron sulfur clusters.
However, the present research was still in an initial step, and the method was limited in electrochemistry measuring techniques such as cyclic voltammetry[11]. Therefore, it will be very significant to study by means of all kinds of measurements available. In this study, the short-term interaction between cysteine and pyrite was studied by amounts adsorbed, FTIR and XRD measurement in order to analyse the interaction mechanism between cysteine and pyrite from different points of view.
2 Experimental 2.1 Materials
Mineral sample: Pyrite used in this experiment was well-crystallized mineral from domestic geological museum. Its chemical composition analysis by scanning electron probe is listed in Table 1. After ultrasonic treatment in order to remove the surface oxide resulting from exposure to air, mineral particles were well ground in agate mortar until their mesh size was smaller than 2 μm. The powder was sealed and reserved in wild-mouth bottle.
Agent: L-cysteine(biochemical reagent), cobalt chloride, aqueous ammonia and ammonium chloride were all analytical reagents. Water used in the experi- ment was double distilled.
2.2 Equipment and apparatus
The equipment and apparatus used in this experiment involved agate mortar, analytical balance (precision 0.1 mg), electric magnetic stirring apparatus, ultrasonic cleaner(CQ50), UV-9200 ultraviolet-visible light spectrometer(with 1cm quartz cuvette).
2.3 Method
Table 1 Chemical constitution of pyrite

2.3.1 Calibration curve of L-cysteine
The cysteine concentration in solution was determined by cobalt(Ⅱ) ion probe method[12] as follows: 1.5 mL of 100 μg/mL CoCl2 solution, 3.0 mL of NH4Cl-NH4OH buffer solution and 0.2-2.0 mL 0.1 mg/mL cysteine solution were progressively added to 10 mL of colorimetric cylinder. Then additional water was added till 10 mL volume was full. The absorbance of mixture was measured at 284 nm. The control experiment was performed with 2.0 mL water instead of cysteine. With A284 absorbance as X axis, the concentra- tion of cysteine solution as Y axis, the Calibration curve of L-cysteine was drawn finally.
2.3.2 Amounts adsorbed measurement
Mineral powder and cysteine(without it in control) were proportionally added before water was droped in until the mixtrue was up to the volume of 40 mL. After agitation with magnetic stirring apparatus for certain time, the mixtrue was set aside for 15 min. The absorbance was measured as the procedure above after 2 mL of supernate was taken.
2.3.3 FTIR measurement
After a series of procedures like blending, agitation and rinse, the mixture was filtered, dried by silica gel for 24 h. FTIR spectra of the sample in the absence and presence of cysteine were obtained on a NEXUS 470 FTIR spectrometer in the range of 400-4 000 cm-1, equipped with an IR source, KBr beam splitter. The transmission technique was applied and the samples were prepared as standard KBr pellets. For each sample 32 cans were recorded with a resolution of 4 cm-1.
2.3.4 XRD measurement
The raw samples were measured by Rigaku diffractometer in the 2θ range of 10? to 90?. Then the lattice constants of samples blended with a few high-purity Si powder, which was applied to assist calibration of measurement error in 2θ diffraction peak, were accurately determined.
3 Results and discussion 3.1 Calibration curve of L-cysteine
The measured calibration curve of L-cysteine is presented in Fig.1. It is indicated that the cysteine concentration shows a good linear relationship with absorbance at the wavelength of 284 nm, with correlation coefficient of 0.996. It suggests that this method is applicable for the adsorbed amounts measurement in the experiment.
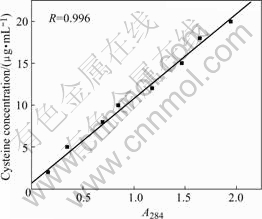
Fig.1 Calibration curve of cysteine
3.2 Measurement of adsorbed amounts
The experiment was done with different mass ratios of mineral to cysteine (mp/mc) of 5, 10, 15, 20, 25, 30, 40 and different time. Fig.2 shows a three-dimensional image presenting the correlation between the adsorbed amounts and adsorption time. As mp/mc is equal to 5, the adsorbed amounts increases with time increasing, however, the adsorbed amounts is stable in the higher lever of mp/mc. It may arise from the maximum probability of contact and adhesion due to the comparatively larger cysteine concentration and minimum probability of desorption caused by less chance of collision and confriction under the condition of minor pulp concentration.
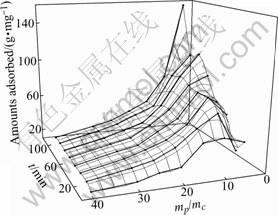
Fig.2 Correlation between adsorbed amounts and adsorption time
Fig.3 shows the correlation between the adsorbed amounts and mp/mc in different adsorption time. It can be seen that the curves show the same tendency except for mp/mc=5.
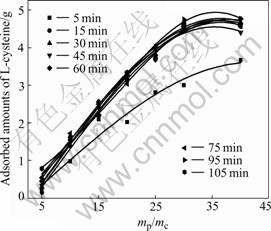
Fig.3 Correlation between adsorbed amounts and mp/mc
Several information about surface properties of adsorbent and interaction between adsorbent and adsorbate can be acquired from adsorption isotherm[13]. Hence, it seems to be critical to assay adsorption isotherm for cysteine adsorbing on mineral. Fig.4 shows the adsorption isotherm with fixed mineral mass and varied cysteine concentration. The shape of curve exhibits that the adsorbed amounts become saturated when the cysteine concentration is over 30 μg/mL, suggesting that it belongs to Langmuir-type adsorption isotherm. From the analysis above, it can be predicted that chemical adsorption occurs between cysteine and pyrite.
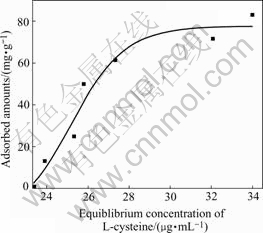
Fig.4 Adsorption isotherm for crysteine adsorbing on pyrite
3.3 Results of FTIR
Fig.5 presents the FTIR spectra before and after interaction between cysteine and pyrite. There are distinct differences in the range of wavenumber from 3 000-500 cm-1. The characteristic adsorption peak of cysteine appears as listed in Table 2.
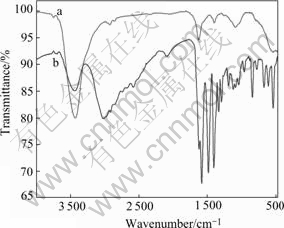
Fig.5 FTIR spectra before(a) and after(b) interaction between cysteine and pyrite
Table 2 Characteristic adsorption peaks in FTIR spectra of cysteine after interaction with pyrite

It has been reported in the literatures[14,15] that, the amino group is located at 3 300-3200 cm-1( ) and 2 962.86 cm-1(νN―H) where a broad adsorption peak appears; hydrosulfuryl turns up at 2 551.02 cm-1; severals of broad and scattered adsorption peaks of carboxylic acid appear at 1 423.78 cm-1 (δCOO―), 1 296.17 cm-1 (νC―O), 1 062.64 cm-1 and 941.14 cm-1 (δOH).
) and 2 962.86 cm-1(νN―H) where a broad adsorption peak appears; hydrosulfuryl turns up at 2 551.02 cm-1; severals of broad and scattered adsorption peaks of carboxylic acid appear at 1 423.78 cm-1 (δCOO―), 1 296.17 cm-1 (νC―O), 1 062.64 cm-1 and 941.14 cm-1 (δOH).
The blue shift of characteristic adsorption peak in FTIR spectra indicates the chemical interaction between cysteine and pyrite. Two possible complexation structures between cysteine and iron are presented in Fig.6. However, its accurate structure awaits further study.
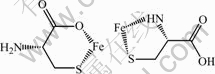
Fig.6 Possible structures of complexation between cysteine and iron
3.4 Results of XRD
Pyrite has a cubic structure with lattice constant a=5.419 ?. Its space group is Pa3 with crystal cell molecules Z=4. The lattice constant from refined XRD results before and after interaction between cysteine and pyrite is a1=5.420 81 ?, a2=5.419 04 ?, respectively. The gap between them is up to 0.011 7 ?, indicating the crystal structure of pyrite is badly destroyed when cysteine is added. The refined XRD results before and after interaction show that after interation, the interplane distances of (210), (220) and (211) are widened. It is suggested that cysteine has a distinct action with pyrite and is more powerful to break the crystal than water.
4 Conclusions
1) The method of cysteine concentration measure- ment by cobalt (Ⅱ) ion probe is applicable for amounts adsorbed measurement in the experiment. As the mass ratio of mineral to cysteine mp/mc is larger than 5, the adsorbed amounts on mineral is stable after 15 min and cysteine adsorbing with mp/mc exhibits the similar behavior.
2) The adsorbed amounts is saturated when cysteine concentration is over 30 μg/mL with a fixed mass of pyrite. It can be preliminarily deduced that chemical adsorption occurs between cysteine and pyrite in terms of its Langmuir-type adsorption isotherm.
3) The functional groups of cysteine are present with blue shift of their characteristic adsorption peak in FTIR spectrum; meanwhile, the lattice constant is significantly reduced and widening of the crystal planes such as (210), (220) and (211) is found after cysteine adsorbing on mineral. The results of both FTIR and XRD suggest that chemical interaction governs the entire adsorption process.
References
[1] LIU Zhong, YANG Wen-bo, BAI Gang, TIAN Wang, JIN Yon-jie. Microbial enzyme conversion of L-cysteine and L-cystine [J]. Microbiology, 2003, 30(6): 16-21.(in Chinese)
[2] MIN Xiao-bo, CHAI Li-yuan, CHEN Wei-liang, ZHANG Chuan-fu, HUANG Bai-yun, KUANG Zhong. Study on bioleaching of refractory gold ore (Ⅰ)──Mechanism on bioleaching of pyrite by Thiobacillus ferrooxidans [J]. Trans Nonferrous Met Soc China, 2001, 11(5): 784-789.
[3] QIU Guan-zhou, LIU Jian-she, HU Yue-hua. Electrochemical behavior of chalcopyrite in presence of Thiobacillus ferrooxidans [J]. Trans Nonferrous Met Soc China, 2000, 10(S1): 23-25.
[4] ROJAS-CHAPANA J A, TRIBUTSCH H. Bio-leaching of pyrite accelerated by cysteine [J]. Process Biochemistry, 2000, 35: 815-824.
[5] HU Yue-hua, HE Zhi-guo, HU Wei-xin, PENG Hong, ZHONG Hui. Effect of two kinds of amino-acids on bioleaching metal sulfide [J]. Trans Nonferrous Met Soc China, 2004, 14(4): 794-797.
[6] ROJAS-CHAPANA J A, TRIBUTSCH H. Biochemistry of sulfur extraction in bio-corrosion of pyrite by Thiobacillus ferrooxidans [J]. Hydrometallurgy, 2001, 59: 291-300.
[7] ROJAS-CHAPANA J A, GIERSIG M, TRIBUTSCH H. The path of sulphur during the bio-oxidation of pyrite by Thiobacillus ferrooxidans [J]. Fuel, 1996, 75(8): 923-930.
[8] ROJAS-CHAPANA J A, BARTELS C C, POHLMANN L, TRIBUTSCH H. Co-operative leaching and chemotaxis of thiobacilli studied with spherical sulphurrsulphide substrates [J]. Process Biochem, 1998, 33(3): 239-248.
[9] TRIBUTSCH H, BENNET J C. Semiconductor-electrochemical aspects of bacterial leaching: I. Oxidation of metal sulphides with large energy gaps [J]. J Chem Technol Biotechnol, 1981, 31: 565-577.
[10] TRIBUTSCH H, ROJAS-CHAPANA J A. Metal sulfide semiconductor electrochemical mechanisms induced by bacterial activity [J]. Electrochimica Acta, 2000, 45: 4705-4716.
[11] ABD EI-HALIM A M, ALONSO-VANTE N, TRIBUTSCH H. Iron/sulphur centre mediated photoinduced charge transfer at (100) oriented pyrite surfaces [J]. Journal of Electroanalytical Chemistry, 1995, 399: 29-39.
[12] ZHANG Gui-zhu, WANG Yue-mei, ZHAO Pang. Cysteine determination using cobalt(Ⅱ) ion probe [J]. Chinese Journal of Rare Metals, 1998, 22(1): 55-59.(in Chinese)
[13] FU Xian-cai, SHEN Wen-xia, Yao Tian-yang. Physical Chemistry (4th version) [M]. Beijing: High Education Press, 1990.(in Chinese)
[14] LIU Jing-ping, LI Jin, GE Xing. Synthesis and free radical inhibition rate of copper(Ⅱ), iron(Ⅱ), manganese(Ⅱ) complexes with cysteine [J]. Chemical World, 2004(5): 235-238.(in Chinese)
[15] YUE Song. Preparations and structure characterition of the crystals of ferrous cysteine [J]. Chemical Research and Application, 2000, 12(4): 387-390.(in Chinese)
(Edited by YUAN Sai-qian)
Foundation item: Project(50374075) supported by the National Natural Science Foundation of China
Corresponding author: LIU Jian-she; Tel: +86-731-8836372; E-mail: ljscsu@263.net